New Way Chemistry for Hong Kong A-Level Book 11 Chapter 8 Covalent Bonding 8.1 Formation of Covalent...
-
Upload
corey-strickland -
Category
Documents
-
view
241 -
download
5
Transcript of New Way Chemistry for Hong Kong A-Level Book 11 Chapter 8 Covalent Bonding 8.1 Formation of Covalent...

New Way Chemistry for Hong Kong A-Level Book 11
Chapter 8Chapter 8Covalent BondingCovalent Bonding
8.18.1 Formation of Covalent Bonds Formation of Covalent Bonds
8.2 8.2 Dative Covalent Bonds Dative Covalent Bonds
8.3 8.3 Bond Enthalpies Bond Enthalpies
8.48.4 Estimation of Average Bond Estimation of Average Bond
Enthalpies using Data from EnergeticsEnthalpies using Data from Energetics

New Way Chemistry for Hong Kong A-Level Book 12
8.58.5 Use of Average Bond Enthalpies to Use of Average Bond Enthalpies to
Estimate Enthalpy Changes of ReactionsEstimate Enthalpy Changes of Reactions
8.6 8.6 Relationship between Bond Enthalpies Relationship between Bond Enthalpies
and Bond Lengthsand Bond Lengths
8.7 8.7 Shapes of Covalent Molecules and Shapes of Covalent Molecules and
Polyatomic IonsPolyatomic Ions
8.88.8 Multiple Bonds Multiple Bonds
8.98.9 Covalent Crystals Covalent Crystals

New Way Chemistry for Hong Kong A-Level Book 13
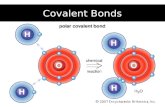
Electron Sharing in Covalent Bonds
H H
Shared electrons
The shared electron pair spends most of the time between the two nuclei.
e-
e-
Attraction between oppositely charged nuclei and shared electrons ( _____________ in nature)
electrostatic
Overlapping of atomic orbitals covalent bond formationOverlapping of atomic orbitals covalent bond formation
8.1 Formation of Covalent Bonds (SB p.203)

New Way Chemistry for Hong Kong A-Level Book 14
8.1 Formation of Covalent Bonds (SB p.203)
A hydrogen molecule is achieved by partial overlapping of 1s orbitals

New Way Chemistry for Hong Kong A-Level Book 15
Thus electrons are shared between the two atoms.
Compare electron-density-map for ionic compounds:
8.1 Formation of Covalent Bonds (SB p.204)
Electron density map for covalent compoundsThere is substantial electron density at all points along the internuclear axis.

New Way Chemistry for Hong Kong A-Level Book 16
8.1 Formation of Covalent Bonds (SB p.204)
Electron density map for ionic compounds

New Way Chemistry for Hong Kong A-Level Book 17
Covalent Bonds in ElementsSome Examples
Dot and cross diagramDot and cross diagram
8.1 Formation of Covalent Bonds (SB p.205)

New Way Chemistry for Hong Kong A-Level Book 18
Covalent Bonds in CompoundsSome Examples
Carbon
1s 2s 2p
All the above examples obey ____________.All the above examples obey ____________.Octet rule
8.1 Formation of Covalent Bonds (SB p.205)

New Way Chemistry for Hong Kong A-Level Book 19
Some Examples All the above examples obey ____________.All the above examples obey ____________.
Carbon
1s 2s 2p
electrons from H
Octet rule
octet
8.1 Formation of Covalent Bonds (SB p.205)
Covalent Bonds in Compounds

New Way Chemistry for Hong Kong A-Level Book 110
Octet Rule and its limitations
BF3
Why doesn’t B form ionic compounds with F?Why doesn’t B form ionic compounds with F?
B: small atomic size
high I.E.’s required to become a cation.
B: small atomic size
high I.E.’s required to become a cation.
electrons from F
8.1 Formation of Covalent Bonds (SB p.206)
not fullfilling octect (electron deficient)

New Way Chemistry for Hong Kong A-Level Book 111
Octet Rule and its limitations
PCl5
Why Phosphorus can expand its octet to form PCl5?
Why Phosphorus can expand its octet to form PCl5?
There is low-lying vacant d-orbital in P.
There is low-lying vacant d-orbital in P.
electrons from Cl
8.1 Formation of Covalent Bonds (SB p.207)

New Way Chemistry for Hong Kong A-Level Book 112
NH3BF3 Molecule8.2 Dative Covalent Bonds (SB p.208)

New Way Chemistry for Hong Kong A-Level Book 113
Dative Covalent Bonds
The dative covalent bond (also known as the coordinate bond) is a type of covalent bond in which the shared pair of electrons is supplied by only one of the bonded atoms.
The dative covalent bond (also known as the coordinate bond) is a type of covalent bond in which the shared pair of electrons is supplied by only one of the bonded atoms.
Remarks(1) The atom that supplies the shared pair of electrons is known as the donor while the other atom involved in the dative covalent bond is known as the acceptor.
(2) Once formed, a dative covalent bond cannot be distinguished from a ‘normal’ covalent bond.
8.2 Dative Covalent Bonds (SB p.208)

New Way Chemistry for Hong Kong A-Level Book 114
Ammonium Ion (NH4+)
8.2 Dative Covalent Bonds (SB p.209)

New Way Chemistry for Hong Kong A-Level Book 115
Aluminium Chloride Dimer (Al2Cl6)
AlCl3
Why doesn’t Al form ionic compounds with Cl?Why doesn’t Al form ionic compounds with Cl?
Al: relative small atomic size; high I.E.’s required to become a cation of +3 charge.
Al: relative small atomic size; high I.E.’s required to become a cation of +3 charge.
(a dimer of AlCl3)
8.2 Dative Covalent Bonds (SB p.209)

New Way Chemistry for Hong Kong A-Level Book 116
Bond Enthalpies
Why do successive B.D.E. of C-H differ?Why do successive B.D.E. of C-H differ?
(Average) bond enthalpy; E(C-H)
4
335)( 425)( 480)( 422)(
= +415.5 kJ mol-1
8.3 Bond Enthalpies (SB p.210)
CH4(g) CH3(g) + H(g) H = +422 kJ mol-1
CH3(g) CH2(g) + H(g) H = +480 kJ mol-1
CH2(g) CH(g) + H(g) H = +425 kJ mol-1
CH(g) C(g) + H(g) H = +335 kJ mol-1
øø
øø
Bond Dissociation EnthalpiesB.D.E of a certain bond is the amount of energy required to break one mole of that bond under standard conditions.e.g. H-H(g) 2H(g) H (H-H) = +431 kJ mol-1ø

New Way Chemistry for Hong Kong A-Level Book 117
Bond Enthalpies
Why is this value of E(C-H) still different from the previously calculated one (+415.5 kJ mol-1)?
Why is this value of E(C-H) still different from the previously calculated one (+415.5 kJ mol-1)?
8.3 Bond Enthalpies (SB p.211)
Bond Average bond enthalpy (kJ mol-1)
H-H
C-C
C═ C
C≡ C
C-H
O-H
+436
+348
+612
+837
+412
+463

New Way Chemistry for Hong Kong A-Level Book 118
Bond Enthalpies
Remarks
• Bond enthalpies refer to the energy required to break bonds (+ve)
• For the formation of a certain bond, the enthalpy change is represented by “- bond enthalpy” (-ve)
• The bond enthalpy is a measure of bond strength. How?
8.3 Bond Enthalpies (SB p.211)

New Way Chemistry for Hong Kong A-Level Book 119
From the Enthalpy Change of Atomization of a Compound
The enthalpy change of atomization of methane (CH4) is +1662 kJ mol-1. Find a value for the bond enthalpy of C-H based on the above data.
E(C-H) = +415.5 kJ mol-1E(C-H) = +415.5 kJ mol-1
8.4 Estimation of Average Bond Enthalpies using Data from Energetics (SB p.212)
The atomization of methane involves the breaking of a four C-H bonds. Assume that all four C-H bonds are equal in strength.
The average bond enthalpy of C-H bonds
= ¼ x (+1 662) kJ mol-1 = +415.5 kJ mol-1
C(g) + 4H(g) ΔH = +1 662 kJ mol-1

New Way Chemistry for Hong Kong A-Level Book 120
The standard enthalpy change of atomization of a compound is the enthalpy change when one mole of gaseous compound is broken down into its constituent atoms in the gaseous state under standard conditions, e.g.
CH4(g) C(g) + 4H(g) 1 mole
The standard enthalpy change of atomization of a compound is the enthalpy change when one mole of gaseous compound is broken down into its constituent atoms in the gaseous state under standard conditions, e.g.
CH4(g) C(g) + 4H(g) 1 mole
The standard enthalpy change of atomization of a element is the enthalpy change when one mole of gaseous atoms is formed into its constituent atoms in the gaseous state under standard conditions, e.g.
½Cl2(g) Cl(g) 1 mole
The standard enthalpy change of atomization of a element is the enthalpy change when one mole of gaseous atoms is formed into its constituent atoms in the gaseous state under standard conditions, e.g.
½Cl2(g) Cl(g) 1 mole
8.4 Estimation of Average Bond Enthalpies using Data from Energetics (SB p.212)

New Way Chemistry for Hong Kong A-Level Book 121
The enthalpy change of atomization of butane (C4H10) and pentane (C5H12) are +5165 kJ mol-1 and +6337 kJ mol-1 respectively. Find a values for the bond enthalpies of C-H and C-C based on the above data.
8.4 Estimation of Average Bond Enthalpies using Data from Energetics (SB p.214)From the Enthalpy Changes of Atomization of Two Compounds
For butane,
3 E(C-C) + 10 E(C-H) = +5 165 kJ mol-1 …….(1)
For pentane,
4 E(C-C) + 12 E(C-H) = +6 337 kJ mol-1 ……..(2)
Solving simultaneous equations (1) and (2), we obtain the following bond enthalpy values.
E (C-H) = +412.25 kJ mol-1
E (C-C) = +347.5 kJ mol-1

New Way Chemistry for Hong Kong A-Level Book 122
8.5 Use of Average Bond Enthalpies to Estimate Enthalpy Changes of Reactions (SB p.214)
Reaction of ethene and hydrogen
Sum of bond enthalpies of products
Enthalpy change
of reaction =
Sum of bond enthalpies of reactants
-

New Way Chemistry for Hong Kong A-Level Book 123
8.5 Use of Average Bond Enthalpies to Estimate Enthalpy Changes of Reactions (SB p.215)
Enthalpy profile for the reaction of ethene and hydrogen

New Way Chemistry for Hong Kong A-Level Book 124
?
Sum of bond enthalpies of reactants= E(C=C) + 4E(C-H) + E(H-H)= (612) + 4(412) + (436)= +2696 kJmol-1
Sum of bond enthalpies of products= E(C-C) + 6E(C-H)= (348) + 6(412) = +2820 kJmol-1
Hr = 2696 - (+2820)
= -124 kJ mol-1
Only an estimated value for H
r .Why?Only an estimated value for H
r .Why?
8.5 Use of Average Bond Enthalpies to Estimate Enthalpy Changes of Reactions (SB p.214)
Reaction of ethene and hydrogen

New Way Chemistry for Hong Kong A-Level Book 125
Consider C-C, C=C and CC
bond order = 1
bond order = 2
bond order = 3
As the bond order increases, the bond strength also increases.As the bond order increases, the bond strength also increases.
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.217)
Bond Average bond
enthalpy (kJ mol-1)
H-H
C-C
C═ C
C≡ C
C-H
O-H
+436
+348
+612
+837
+412
+463
Bond Enthalpies as an Indication of the Strength of Covalent Bonds

New Way Chemistry for Hong Kong A-Level Book 126
Bond length (for covalent bond)Bond length (for covalent bond)
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.217)
Bond Lengths

New Way Chemistry for Hong Kong A-Level Book 127
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.217)
Bond lengths of some covalent bondsBond Molecule Bond lengths (nm)O-H H2O
H2O2
CH3OHHCOOH
0.0960.0970.0960.096
C-C DiamondC2H6
C3H8
C2H5OH
0.1540.1540.1540.155
C-H CH4
C2H4
0.1100.109
By what technique can these bond lengths be measured?
Bond lengths of some covalent bonds
X-ray diffraction

New Way Chemistry for Hong Kong A-Level Book 128
Any conclusion for the relationship between bond length & bond enthalpy?
Any conclusion for the relationship between bond length & bond enthalpy?
Usually a longer bond length corresponds to a lower value of bond enthalpy (weaker bond).
Usually a longer bond length corresponds to a lower value of bond enthalpy (weaker bond).
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.217)
Bond Bond length (nm)
Bond enthalpy
(kJ mol-1)
H-H
Cl-Cl
Br-Br
I-I
H-F
H-Cl
H-Br
H-I
0.074
0.199
0.228
0.266
0.092
0.127
0.141
0.161
436
242
193
151
565
431
364
299
Bond enthalpies and bond lengths

New Way Chemistry for Hong Kong A-Level Book 129
Explain why the bond enthalpy of F-F is smaller than that of Cl-Cl even though the bond length of F-F is the shortest among the halogens.
As the size of fluorine atom is very small, the repulsion between the non-bonding pairs of electrons on the fluorine atoms weaken the F-F bond.
Bond Bond Length /nm Bond Enthalpy / kJ mol-1
Cl-Cl 0.199 242Br-Br 0.228 193 I-I 0.266 151
FF
Non-bonding e-
/ lone pair of e-
F-F 0.142 158
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.217)
Special Situation for F2

New Way Chemistry for Hong Kong A-Level Book 130
(often referred as ‘Atomic radius’)
Where b is the bond length r is the covalent radius
The space occupied by an atom in a covalently bonded molecule in the direction of the covalent bond (generally taken as half of the bond length of homoatomic molecules)
The space occupied by an atom in a covalently bonded molecule in the direction of the covalent bond (generally taken as half of the bond length of homoatomic molecules)
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.218)
Covalent Radius

New Way Chemistry for Hong Kong A-Level Book 131
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.218)
The covalent radii (in nm) of some elements

New Way Chemistry for Hong Kong A-Level Book 132
Predicting bond length of A-B if rA & rB are knownPredicting bond length of A-B if rA & rB are known
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.218)
Bond length of a covalent bond A-B
=Covalent radius of atom A
+Covalent radius of atom B
Bond lengths of atom A and atom B

New Way Chemistry for Hong Kong A-Level Book 133
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.219)
Bond Calculated bond length (nm)
Experimentally determined bond length (nm)
C-O
C-F
C-Cl
C-Br
C-C
H-Cl
C-H
N-Cl
0.150
0.149
0.176
0.191
0.154
0.136
0.114
0.173
0.143
0.138
0.177
0.193
0.154
0.128
0.109
0.174
Calculated and experimentally determined bond length
By what technique can the bond lengths be determined experimentally?
By what technique can the bond lengths be determined experimentally?
Similarelectronegativity

New Way Chemistry for Hong Kong A-Level Book 134
Bond Calculated bond length (nm) Experimentally determined bond length (nm)
C-O
C-F
C-Cl
C-Br
C-C
H-Cl
C-H
N-Cl
0.150
0.149
0.176
0.191
0.154
0.136
0.114
0.173
0.143
0.138
0.177
0.193
0.154
0.128
0.109
0.174
8.6 Relationship between Bond Enthalpies and Bond Lengths (SB p.219)
Calculated and experimentally determined bond length
Quite differentelectronegativity

New Way Chemistry for Hong Kong A-Level Book 135
Shape of a moleculeShape of a molecule
= geometric arrangement of atoms within the molecules
Valence Shell Electron Pair Repulsion Theory (VSEPR)Valence Shell Electron Pair Repulsion Theory (VSEPR)
The electron pairs in the outermost shell of the central atom in a molecule will stay as far apart as possible to minimize electronic repulsion.
The electron pairs are oriented at a maximum separation in space so as to minimize the coulombic repulsion of electron clouds.
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.220)
Shapes of Molecules and Polyatomic Ions

New Way Chemistry for Hong Kong A-Level Book 136
Beryllium chloride molecule (BeCl2)
BeClCl
Electronic Diagram
Shape in word linear
Bond angle= angle between
2 bonds
Bond angle= angle between
2 bonds
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.220)
Shape in Diagram

New Way Chemistry for Hong Kong A-Level Book 137
Boron trifluoride molecule(BF3)
Electronic DiagramShape in Diagram
Shape in word
trigonal planar
B
F F
F
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.220)

New Way Chemistry for Hong Kong A-Level Book 138
Methane (CH4) Molecule
Electronic Diagram Shape in Diagram
Shape in word
tetrahedral
CH H
H
H
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.221)

New Way Chemistry for Hong Kong A-Level Book 139
Electronic DiagramShape in Diagram
Shape in word
tetrahedral
CH H
H
H
C
H
H HH
bond in front of paper
bond behind paper
bond in the plane of paper
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.221)
Methane (CH4) Molecule

New Way Chemistry for Hong Kong A-Level Book 140
Phosphorus Pentachloride (PCl5) Molecule
Electronic DiagramShape in Diagram
Shape in word
trigonal bipyramidal
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.221)

New Way Chemistry for Hong Kong A-Level Book 141
Electronic Diagram Shape in Diagram
Shape in word
trigonal bipyramidal
P
Cl
Cl
Cl
Cl
Cl
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.221)
Phosphorus Pentachloride (PCl5) Molecule

New Way Chemistry for Hong Kong A-Level Book 142
Sulphur Hexafluoride (SF
6) Electronic Diagram Shape in Diagram
Shape in word
octahedral
S
F
F
F
F
F
F
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.221)

New Way Chemistry for Hong Kong A-Level Book 143
Ammonia (NH3) Molecule
Electronic Diagram Shape in Diagram
Shape in word
Trigonal pyramidal
H H
H
N
lp-lp repulsion > lp-bp repulsion > bp-bp repulsion
lp-lp repulsion > lp-bp repulsion > bp-bp repulsion
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.223)

New Way Chemistry for Hong Kong A-Level Book 144
Electronic DiagramShape in Diagram
Shape in word
Trigonal pyramidal
H H
H
N
bond in front of paper
bond behind paper
bond in the plane of paper
N
H HH
lone pair of e-
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.223)
Ammonia (NH3) Molecule

New Way Chemistry for Hong Kong A-Level Book 145
Water (H2O) Molecule
Electronic Diagram Shape in Diagram
Shape in word
V-shaped / bent
H HO
lp-lp repulsion > lp-bp repulsion > bp-bp repulsion
lp-lp repulsion > lp-bp repulsion > bp-bp repulsion
8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.223)

New Way Chemistry for Hong Kong A-Level Book 146
Electronic Diagram Shape in Diagram
Shape in word
V-shaped / bent
H H
lone pair of e-
O
O
H H
Water (H2O) Molecule8.7 Shapes of Covalent Molecules and Polyatomic Ions (SB p.224)

New Way Chemistry for Hong Kong A-Level Book 147
8.8 Multiple Bonds (SB p.227)
Comparison of bond lengths and bond enthalpies between single and multiple bonds
Bond Bond order Bond length
(nm)
Bond enthalpy
(kJ mol-1)
C-C
C═ C
C≡ C
1
2
3
0.154
0.134
0.120
+348
+612
+837
N-N
N═ N
N≡ N
1
2
3
0.146
0.120
0.110
+163
+409
+944
C-O
C ═O
1
2
0.143
0.122
+360
+743

New Way Chemistry for Hong Kong A-Level Book 148
Ethene (CH2=CH2)
Electronic DiagramShape in Diagram
Shape in word
(planar)
C C
H
HH
H
8.8 Multiple Bonds (SB p.227)
Effect of Multiple Bonding on Shapes of Molecules

New Way Chemistry for Hong Kong A-Level Book 149
Ethyne (CHCH)
Shape in Diagram Shape in word
linear
8.8 Multiple Bonds (SB p.227)

New Way Chemistry for Hong Kong A-Level Book 150
Carbon dioxide (CO2)
Shape in Diagram Shape in word
linear
8.8 Multiple Bonds (SB p.227)

New Way Chemistry for Hong Kong A-Level Book 151
Shape in Diagram Shape in word
Angular / (bent)
Sulphur dioxide (SO2)
8.8 Multiple Bonds (SB p.227)

New Way Chemistry for Hong Kong A-Level Book 152
Structures consist of millions of atoms covalently bonded together in a structural network and there is no simple molecule present.
Examples
diamond, graphite and quartz/silicon(IV) oxide
8.9 Covalent Crystals (SB p.228)
Giant Covalent Structures

New Way Chemistry for Hong Kong A-Level Book 153
Diamond
• Each C atom is covalently bonded to 4 other C atoms to form a three-dimensional network
• The C-C bonding pattern accounts for the high m.p., stability and extreme hardness
• Applications: scratch proof cookware, watch crystals, ball bearings and razor blade
8.9 Covalent Crystals (SB p.228)

New Way Chemistry for Hong Kong A-Level Book 154
Graphite
• Each C atom is covalently bonded to 3 other C atoms in the same layer. A network of coplanar hexagons is formed (C-C bond length: 0.142 nm)
• Weak van der Waals’ forces hold the layers together (distance between adjacent layers: 0.335 nm)
• Delocalized e- free to move within layers
• Properties: soft and slippery (used as pencil ‘lead’), conductor
8.9 Covalent Crystals (SB p.229)

New Way Chemistry for Hong Kong A-Level Book 155
Why graphite has a high m.p. than that of diamond?Why graphite has a high m.p. than that of diamond?
8.9 Covalent Crystals (SB p.230)
Comparison of the properties of diamond and graphite
Property Diamond Graphite
Density (g cm-3)
Hardness
Melting point (oC)
Colour
Electrical conductivity
3.51
10 (hardest)
3 827
Colourless transparent
None
2.27
< 1 (very soft)
3 652 (sublime)
Shiny black
High (along sheet)

New Way Chemistry for Hong Kong A-Level Book 156
Each Si atom is bonded tetrahedrally to 4 neighbouring O atomsEach O atom is bonded to 2 Si atoms, one at the centre of each of two adjacent tetrahedral
Quartz /Silicon(IV) oxide8.9 Covalent Crystals (SB p.230)

New Way Chemistry for Hong Kong A-Level Book 157
The END