6.2 Covalent Bonds
-
Upload
japiboy4638 -
Category
Documents
-
view
117 -
download
1
Transcript of 6.2 Covalent Bonds

6.2 Covalent Bonding
Covalent Bonding
You Will Need:iNotebookAt least 2pgs of CN
Agenda:Covalent Bonding LectureChemical Bonding VideoChemical Bonding QuizCovalent Bonding Wksht
Opener:
What is the difference between an anion and a cation?

6.2 Covalent Bonding
How are atoms held together in a covalent bond?
Covalent Bonds
The attractions between the shared electrons and the protons in each nucleus hold the atoms together in a covalent bond.
A covalent bond is a chemical bond in which two atoms share a pair of valence electrons.

6.2 Covalent Bonding
Sharing Electrons
When two atoms share one pair of electrons, the bond is called a single bond.
Covalent Bonds

6.2 Covalent Bonding
Molecules of Elements
Two hydrogen atoms bonded together form a unit called a molecule.
A molecule is a neutral group of atoms that are joined together by one or more covalent bonds.
Covalent Bonds

6.2 Covalent Bonding
Multiple Covalent Bonds
When two atoms share three pairs of electrons, the bond is called a triple bond. When two atoms share two pairs of electrons, the bond is called a double bond.
Each pair of shared electrons is represented by a long dash in the structural formula NN.
Covalent Bonds

6.2 Covalent Bonding
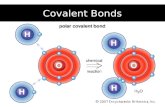
When atoms form a polar covalent bond, the atom with the greater attraction for electrons has a partial negative charge. The other atom has a partial positive charge.
Unequal Sharing of Electrons
The type of atoms in a molecule and its shape are factors that determine whether a molecule is polar or nonpolar.

6.2 Covalent Bonding
A covalent bond in which electrons are not shared equally is called a polar covalent bond.
Unequal Sharing of Electrons

6.2 Covalent Bonding
Shared electrons in a hydrogen chloride molecule spend less time near the hydrogen atom than near the chlorine atom.
Unequal Sharing of Electrons

6.2 Covalent Bonding
How do attractions between polar molecules compare to attractions between nonpolar molecules?
Attraction Between Molecules
Attractions between polar molecules are stronger than attractions between nonpolar molecules.