8.1 Covalent Bonds
-
Upload
ahmed-johnson -
Category
Documents
-
view
36 -
download
4
description
Transcript of 8.1 Covalent Bonds

8.1 Covalent Bonds

Molecular compounds = 2 non metals that share 1 or more
pairs of electrons…
group is electrically neutral.

Draw the dot structure for CF4

A dash shows a molecular bond and represents 2 electrons

How is water (H2O) different from sodium chloride (NaCl)?

Review Bond Types:Copy into powerpoint notes.
IONIC: Metal and Non Metal ions. Electrons jump from metal to non metal
to make “magnets”.METALLIC: Metal and Metal ions.
Electron sea floats between positive ions.CoVALENT: Non Metal and Non Metal
atoms (Not ions). Share electrons.

IONIC review: NaCl=Formula Unit=lowest ratio of ions in an ionic compound -atoms gained or lost e- to
form ions that are electrostatically bonded to each other (metal)

H2O =Molecular Formula= (Nonmetals) molecule
atoms share electrons to form stable noble gas configurations (atoms
don’t form ions but still follow the octet rule) H=2 electrons

Notice the octet rule H = He 2e-O=Ne 8e-

Molecular formula = shows the number and types of atoms in a
molecule
Discuss VESPER Theory and shapes of these molecules (electron pairs make 4 point pyramid). Discuss the Lewis Structures.

How is the molecular formula different from a formula unit?
Covalent Bond Ionic Bond

Sharing 2 or 3 pair of electrons

Sharing 2 pairs of electrons

Draw the compound CO2


Bond dissociation
energy = amount of
energy needed to
break a single covalent bond

The higher the BDE the
stronger and more stable the bond.

7 diatomic elements found on earth
H2, N2, O2, F2, Cl2, Br2, I2
Diatomic elements: 7,

VSEPR = valence-shell electron-pair repulsion theory. Causes
shape of molecules. repulsion between electron pairs causes molecular shapes to adjust so that the valence-electrons pairs
stay as far apart as possible(SEE p242)

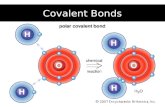
Polar bonds : electron pair is shared unevenly. Causes the molecule to act
like a magnet. (the element with higher
electronegativity (p.xxx) has the electron more often and become
slightly negative)

Subtract the electronegativity of the two atoms to find the type of bond. (pg 248) Bond Type0-0.39 Nonpolar covalent0.4-1.0 Moderately polar1.0-1.9 Very Polar2.0 + Ionic or Metallic

Hydrogen Bonds – hydrogen bonded to an atom with a high
electronegativity is weakly bonded to a nearby molecules
unshared electrons

Bond Typep.238 table 8.3
Electronegativity Difference0.0-0.39 nonpolar covalent
0.4-1.9 polar covalent metal = Ionic


Polar molecules – a molecule that has two poles (positive and negative,
only happens with polar bonds) based on VSEPR shape

Label bond type on worksheetIdentify VSEPR shape
Identify polar molecules

POLYATOMIC IONS:Sharing too many electrons…
Polyatomic ions (molecules with a charge)

Review

Draw the compound SCl2

Draw the compound SCl2

Molecules:Covalent Sharing of Electrons