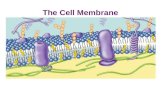
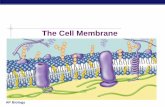
The plasma membrane of a cell is the boundary that separates the living cell from its surroundings. ...
-
Upload
emmeline-blair -
Category
Documents
-
view
225 -
download
0
Transcript of The plasma membrane of a cell is the boundary that separates the living cell from its surroundings. ...


The plasma membrane of a cell is the boundary that separates the living cell from its surroundings.
It is only about 8nm thick—it would take over 8,000 of them to equal the thickness of a piece of paper.
It is essential to maintain the internal environment of the cell.

The plasma membrane exhibits selective permeability—it allows some substances to pass through more easily than others.
What needs to come into a cell? (oxygen, water, nutrients, ions)
What needs to leave a cell?(carbon dioxide, water, wastes, proteins)

The most abundant molecules in cell membranes are phospholipids and proteins. Phospholipids are amphipathic. What does this mean?
Phospholipids make a bilayer around the cell with water inside the cell kept separate from water outside the cell.

The membrane is a fluid structure with a “mosaic” of various proteins embedded in or attached to the phospholipids.
See video: http://www.youtube.com/watch?v=LKN5sq5dtW4&feature=related

Fig. 7-7
Fibers ofextracellularmatrix (ECM)
Glyco-protein
Microfilamentsof cytoskeleton
Cholesterol
Peripheralproteins
Integralprotein
CYTOPLASMIC SIDEOF MEMBRANE
GlycolipidEXTRACELLULARSIDE OFMEMBRANE
Carbohydrate

Membranes are not locked into place. Most of the lipids and some of the
proteins can shift laterally side-to-side. It is rare for them to flip-flop transversely. Adjacent phospholipids move within the
membrane rapidly—107 times/second
Unsaturated fatty acid chains in the phospholipids makes membranes more fluid since the “kinks” in the “tails” don’t pack together tightly. This keeps it fluid even in cold temperatures.

The steroid cholesterol is wedged between phospholipids in the plasma membrane of animal cells.
At higher temperatures, cholesterol makes the membrane less fluid by restraining phospholipid movement.
Because cholesterol keeps the membrane from packing tightly, it lowers the temperature required for the membrane to solidify (keeping animals from freezing)
Membranes must be fluid to work properly; they are usually about as fluid as salad oil.


A mosaic of different proteins is embedded in the plasma membrane. Proteins determine most of the membrane’s functions.
Active transport of materials
Enzymes
Signal transduction

Cell-Cell RecognitionInter-cellular Joining Attachment to the
cytoskeleton and the extra-cellular matrix

Integral Proteins: penetrate the hydrophobic core of the lipid bilayer
Peripheral Proteins: Not embedded in
the lipid bilayer at all
They are appendages

N-terminus
C-terminus
HelixCYTOPLASMICSIDE
EXTRACELLULARSIDE
The Structure of a trans-membrane protein:

A cell’s ability to distinguish one type of neighboring cell from another is crucial.
It helps sort cells into different tissues and helps the organism recognize foreign cells.
Membrane carbohydrates may be bonded to proteins (Glyco-proteins) or to lipids (Glyco-lipids)
Cells recognize other cells by binding to surface molecules, often to carbohydrates.

Membranes have distinctive inside and outside faces.
When a vesicle fuses with the plasma membrane, the OUTSIDE of the vesicle membrane fuses with the INSIDE of the plasma membrane.
Therefore, molecules that start on the INSIDE of the ER end up on the OUTSIDE of the plasma membrane.

Synthesis of membrane components and their orientation in the resulting membrane.
The plasma membrane has distinct cytoplasmic (orange) and extracellular (aqua) faces, with the extracellular face arising from the inside face of the ER, Golgi, and vesicle membranes.
ER1
Transmembraneglycoproteins
Secretoryprotein
Glycolipid
2Golgiapparatus
Vesicle
3
4
Secretedprotein
Transmembraneglycoprotein
Plasma membrane:
Cytoplasmic face
Extracellular face
Membrane glycolipid

http://www.wisc-online.com/objects/ViewObject.aspx?ID=ap1101

Most important function of the cell membrane: to regulate what goes in/out of the cell
Form fits function: the fluid mosaic model helps explain how membranes regulate the cell’s molecular traffic

Nonpolar molecules such as hydrocarbons, CO2 and O2 are hydrophobic and can pass easily through the lipid bilayer. Why?

Ions and polar molecules cannot pass easily across the lipid bilayer. Why not?
Polar molecules such as glucose and other sugars pass only slowly across the membrane.
Even water passes more slowly than expected, since it is such a small molecule. Why?

Cell membranes are permeable to certain ions and other polar substances such as water.
These hydrophilic substances can avoid contact with the “Hydrophobic Zone” by passing through transport proteins that span the membrane.

Channel proteins function by having a hydrophilic channel that certain molecules can use as a tunnel through the membrane
Aquaporins help water molecules move through the membrane easily. Each one allows up to 3 billion water molecules/second!

Carrier proteins hold onto their “passengers” and change shape in such a way that shuttles them across the membrane.
A transport protein is specific for the substance it moves across.


Passive transport is diffusion of a substance across a membrane with no energy investment required by the cell. Materials move by themselves! How?
Diffusion = the movement of a substance from where it is more concentrated to where it is less concentrated. Molecules move DOWN a concentration gradient. They move from HIGH concentration to LOW.

Molecules move by themselves due to kinetic energy, the energy of motion. This is also called thermal energy.
Each molecule moves randomly, in all directions.
Diffusion occurs as molecules move randomly, but the NET movement of a population of molecules is to move AWAY from high concentration to lower concentration until it reaches equilibrium.

Osmosis is a special kind of diffusion. It is the diffusion of water across a selectively permeable membrane.
Water moves from where it is MORE concentrated to where it is LESS concentrated.

If there is a difference in the concentration of solutes in the water on either side of a membrane, the water level may change as it diffuses across the membrane.
It is the difference in free water concentration that makes the difference.
Why does the level of water go up on the right-hand side of the tube?

Isotonic Solution– a solution where the concentration of solutes is EQUAL inside and outside the cell
Hypertonic—a solution where the concentration of solutes is GREATER in the water outside the cell. (More solutes = less water)
Hypotonic—a solution where the concentration of solutes is LESS in the water outside the cell. (Less solutes=
more water)
Hypertonic Hypotonic

“Iso” = equal “Hyper” = Greater than
“Hypo” = Less than

The cells of plants, prokaryotes, fungi and some protists have cell walls.
When such a cell is placed in a hypotonic solution, the wall helps maintain the cell’s water balance.

Facilitated diffusion occurs when transport proteins embedded in the membrane aid the movement of substances. (Note: it is still moving DOWN the concentration gradient!)
Two types of transport proteins:
Channel protein Carrier Proteins

http://www.northland.cc.mn.us/biology/biology1111/animations/passive1.swf

Active transport uses the cell’s energy (ATP) to move materials across the membrane.
Materials that must move AGAINST the concentration gradient must be moved with active transport.

To pump a solute across a membrane against its gradient requires work; the cell must expend energy.
Carrier proteins embedded in the membrane can move these solutes.

The sodium-potassium pump exchanges sodium ions (Na+) for potassium (K+) across the cell membrane.
Each ion is in high concentration on one side of the membrane. The cell needs to maintain that higher concentration.

http://highered.mcgraw-hill.com/sites/0072495855/student_view0/chapter2/animation__how_the_sodium_potassium_pump_works.html
http://www.brookscole.com/chemistry_d/templates/student_resources/shared_resources/animations/ion_pump/ionpump.html

All cells have voltages across their plasma membranes.
Voltage is electrical potential energy—a separation of opposite charges.
The cytoplasm is negative (-); the extracellular fluid is positive (+).

The voltage across a membrane is called membrane potential. It ranges from about –50 to –200 millivolts.
The membrane potential acts like a battery, an energy source that affects the traffic of all charged substances (either + or -).
The membrane potential favors + charged ions. Why?

Two forces drive the diffusion of ions across the membrane:
1)a chemical force (the concentration gradient)
2)an electrical force (the membrane potential)
Together, these are called the electrochemical gradient
In the above example, there are more negative charges inside the cell than outside. Ions such as Na+ are more likely to move inside. Why? (2 reasons!)

Some membrane proteins that actively pump ions contribute to the membrane potential.
The Sodium-Potassium pump is an example because it pumps out 3 Na+ ions for every 2 K+ ions, creating a difference in charge.
An electrogenic pump creates voltage across a membrane. With each “crank” of the pump, there is a net transfer of one + charge, a process that stores energy as voltage.

The main electrogenic pump of plants, fungi, and bacteria is a proton pump.
A proton pump actively pumps out hydrogen ions (protons, H+).
The pumping of H+ transfers + charge from the cytoplasm, generating voltage.

A single ATP-powered pump that transports a specific solute can indirectly drive the active transport of other solutes as well.
A substance that has been pumped across the membrane can do work as it moves back across by diffusion.
An example of cotransport: A carrier protein such as this sucrose-H+ transporter is able to use the diffusion of H+ down its electrochemical gradient to drive the uptake of sucrose.

Exocytosis = the cell secretes (transports out of the cell) certain biological molecules by fusing vesicles with the plasma membrane.
The contents of the vesicle then spill to the outside of the cell and the vesicle membrane becomes part of the cell membrane.

In endocytosis, the cell takes in biological molecules (such as food) by forming new vesicles from the plasma membrane.
A small area of the plasma membrane sinks inward to form a pocket.
As the pocket deepens, it pinches in, forming a vesicle.

In Phagocytosis, a cell engulfs a particle by wrapping pseudopodia around it and packaging it within a membrane-enclosed sac that can be large enough to be called a vacuole.
The particle is then digested by a lysosome.

In pinocytosis, a cell “gulps” in droplets of extracellular fluid into tiny vesicles.
It is not the fluid itself that is needed by the cell, but the molecules dissolved in the droplets.

Receptor-mediated endocytosis enables the cell to bring in bulk quantities of specific substances, even if they are not in high concentrations outside the cell.

Embedded in the membrane are proteins with specific receptor sites exposed to the extracellular fluid.
The receptor proteins are usually clustered in regions of the membrane called coated pits.
These coated pits are lined on the inside of the cell with fuzzy proteins.