The Cell Membrane & Passive Transport. The cell membrane is the boundary that separates the living...
-
Upload
olivia-kelly -
Category
Documents
-
view
214 -
download
0
Transcript of The Cell Membrane & Passive Transport. The cell membrane is the boundary that separates the living...

The Cell Membrane & Passive Transport

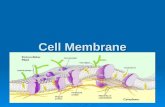
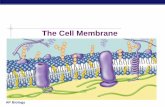
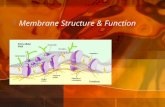
The cell membrane is the boundary that separates the living cell from its surroundings
Life has an inside and an outside

The Phospholipid BilayerPhospholipids are
the most abundant molecule in the cell membrane
Hydrophobic tails & a hydrophilic head

The Fluid Mosaic Model
• The membrane is a fluid structure with a mosaic of proteins, glycolipids and sterols embedded in it

The Fluid Mosaic Model
• Phospholipids in the plasma membrane can move within the bilayer

Selective Permeability
• A cell must exchange materials with its surroundings, a process controlled by the plasma membrane
• Plasma membranes are selectively permeable, they only allow some substances to cross the membrane.

Hydrophobic (nonpolar) molecules, can dissolve in the lipid bilayer and pass through the membrane rapidly

Polar molecules, such as sugars, do not cross the membrane easily

Passive Transport
Passive transport is diffusion of a substance across a membrane with no energy investment

Concentration Gradient
• A difference in the number per unit volume of ions or molecules of a substance between adjoining regions

DiffusionThe net movement of like molecules or ions
down a concentration gradient

(b) Diffusion of two solutes
Fig. 7-11b
Net diffusion
Net diffusion
Net diffusion
Net diffusion
Equilibrium
Equilibrium

Diffusion Rate
• Determined by steepness of gradient, molecular size, temperature & electric or pressure gradients

Osmosis
• Osmosis is the diffusion of water across a selectively permeable membrane
• Water diffuses across a membrane from the region of lower solute concentration to the region of higher solute concentration

Lowerconcentrationof solute (sugar)
Fig. 7-12
H2O
Higher concentrationof sugar
Selectivelypermeablemembrane
Same concentrationof sugar
Osmosis

Tonicity
• Tonicity is the ability of a solution to cause a cell to gain or lose water

Hypertonic Solution
Solute concentration is greater than that inside the cell; cell loses water

Hypotonic Solution
Solute concentration is less than that inside the cell; cell gains water

Isotonic Solution
Solute concentration is the same as that inside the cell; no net water movement across the plasma membrane

• Osmoregulation, the control of water balance, is a necessary adaptation for life in such environments
• The protist Paramecium, which is hypertonic to its pond water environment, has a contractile vacuole that acts as a pump
Video: Chlamydomonas Video: Paramecium Vacuole

Fig. 7-14
Filling vacuole 50 µm
(a) A contractile vacuole fills with fluid that enters from a system of canals radiating throughout the cytoplasm.
Contracting vacuole
(b) When full, the vacuole and canals contract, expelling fluid from the cell.

Water Balance of Cells with Walls
• Cell walls help maintain water balance• A plant cell in a hypotonic solution swells
until the wall opposes uptake; the cell is now turgid (firm)
• If a plant cell and its surroundings are isotonic, there is no net movement of water into the cell; the cell becomes flaccid (limp), and the plant may wilt
Copyright © 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings

Video: Plasmolysis
Video: Turgid Elodea
Animation: Osmosis
• In a hypertonic environment, plant cells lose water; eventually, the membrane pulls away from the wall, a usually lethal effect called plasmolysis