VESPR Model Pi and Sigma Buckyballs et al Molecular Shapes Periodic Table Potpourri $ 200 $ 200$200...
Transcript of VESPR Model Pi and Sigma Buckyballs et al Molecular Shapes Periodic Table Potpourri $ 200 $ 200$200...
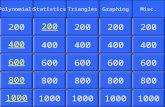
VESPR Model
Pi and Sigma
Buckyballs et al
Molecular Shapes Periodic
TablePotpourri
$200 $200 $200 $200 $200 $200
$400 $400 $400 $400 $400 $400
$600 $600 $600 $600 $600 $600
$800 $800 $800 $800 $800 $800
$1000 $1000 $1000 $1000 $1000 $1000
The Atom Bonding Reactions MatterMath of Chemistry
Solubility
$400 $400 $400 $400 $400 $400
$800 $800 $800 $800 $800 $800
$1200 $1200 $1200 $1200 $1200 $1200
$1600 $1600 $1600 $1600 $1600 $1600
$2000 $2000 $2000 $2000 $2000 $2000
Final Jeopardy Ionic X > 1.7
Polar Covalent 1.7 X .5 Pure Covalent X < .5
Write down the 3 divisions and numbers that you must use after you subtract the
Electronegativity numbers between 2 atoms in a
molecule.
$200Properties of the elements that repeat
consistently across the periods, determined by electron configuration
What is the definition of Periodic Properties?
$400
Amount of pull the valence electrons feel from protons in the nucleus
What is the effective Nuclear Charge (Z)?
$800
Goes up, because protons increase while shielding electrons stays the same
Going across a period, the effective nuclear charge...
$200
Effective nuclear charge and energy levels
What are factors which affect the size of an atom and how they affect it ?
$600
Trend of atomic size going across a period
What Decreases due to energy level staying the same, while effective
nuclear charge increases (more pull on valence electrons)?
$1000
The effective nuclear charge and atom size
What are factors which affect ionization energy and how they affect it ?
$200
Trend of ionization energy going down a family
What decreases (easier to remove) because size is
bigger, so valence electrons are further away ?
$400
Trend of ionization energy going across a period
What increases (harder to remove) due to effective nuclear charge
increasing (more pull - more likely to attract electrons) ?
$800
effective nuclear charge and atom size
What factors which affect electronegativity and how
they affect it ?
$1000
Decreases.. if size is bigger, less pull from nucleus
•What is the trend of electronegativity going down
a family
$200
Increases due to effective nuclear charge being greater
Trend of electronegativity going across a period
$800
Mendeleev - increasing molar mass, while today’s is increasing atomic number
How was Medeleevs scheme for listing elements different from that used in the modern
periodic table?
$1000
elements in same group have same number of valence electrons
What relationship can be seen between an elements
placement within a group and its electron configuration?
$200
Noble gases had not been discovered yet due to their inactivity
Why did Group 8 not appear on the Mendeleev periodic
table?
$400
increasing tendency to form neg ions therefore use less energy (minus -:
Exothermic
Trend of electron affinity across left to right
$600farther from nucleus therefore uses less
energy to add an electron also shielding can add electrons
Trend of electron affinity down top to bottom
$200
both negative ions (right side) give off energy to gain an electron + ions use
energyElectron affinity energy can
be what: + or -?
$400
1) shielding 2) Effective (Strong) nuclear charge 3) add another orbit of electrons
increases distance from nucleus
Name 3 concepts that can affect periodicity
$600Protons and neutrons in element 114
What exactly do these two pictures represent and where
are the from?
$800
The equal the levels from the nucleus out.
How do the periods on the periodic table relate to the electron orbitals and the
nucleus.
Daily Double
Dalton, Thompson, Rutherford and Bohr…Schrödinger, and Heisenberg
Name the 1st five scientists in “The Atom Song