To Do Make the Macromolecule Build Structure: Monomers and building polymers Important functions ...
-
Upload
aubrie-sutton -
Category
Documents
-
view
223 -
download
2
Transcript of To Do Make the Macromolecule Build Structure: Monomers and building polymers Important functions ...

To Do
Make the Macromolecule Build Structure: Monomers and building polymers Important functions Add: Why is the shape important? (Importance of
structure)
Venn Diagram instructionsStation instructionTrackers

CHEMISTRY OF LIFE
Unit 2

Getting Organized
Unit 2: Need to have your Table of Contents Need to have your Vocabulary Sheet
It is REQUIRED!!!!!! This is one of the ways we are all getting 3 and 4’s!

Umm…why are we learning about chemistry in biology?

Why are we learning about Chemistry???
5 elements make up over 90% of your body!
C, H, N, O, P
(Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorous)

BECAUSE…..
Every biological process is part of a chemical reaction!
All of the energy we need depends on chemical reactions!

Basic Chemistry REMINDERS…
Chemical symbols are used to represent each element. Found on the Periodic Table

Basic Chemistry REMINDERS…
Compounds are combinations of 2 or more elements.

Biochemistry
Study of the chemicals necessary for living things.
Also called Organic Chemistry.Involves the element carbon (C)

Organic Molecules Organic Molecules Inorganic MoleculesInorganic Molecules
Contain carbon, hydrogen and oxygen atoms together
Make up living things
Molecules that play an important role in living things
Don’t contain carbon, hydrogen and oxygen together
Make up nonliving things
Molecules that also play an important role in living things
Organic vs. Inorganic

Biochemistry• Macromolecules or Polymers are large
molecules that are made up of many repeating subunits or building block molecules known as monomers.
POLYMER
MONOMER
Definitions on NEXT Slide!

Biochemistry
Terms to Know: Monomer – the smallest unit of a substance (the
building block) Example: like one Lego block
Polymer – many monomers linked together to make a large structure; also called macromolecules (many repeating subunits linked together) Example: Lego blocks put together to make a Lego
house

Biochemistry
Types of Organic Molecules (Macromolecules)
1. Carbohydrates2. Lipids3. Proteins4. Nucleic Acids

Check Yourself!
1. What are the 4 “Core Organic Molecules”?2. What are the 5 essential elements? (Write
out the full names and the memory trick).3. What is the most important inorganic
compound in life?4. Compare and contrast Organic and
Inorganic molecules.

Properties of Water - Observations
What is the bond between hydrogen and oxygen that make the molecule hard to break? What is the bond that allows water to “stick” together?
Using the medicine dropper, place as many drops of water on the penny as you can with out spilling. Make sure to keep count and record

Observations
After dissolving the sodium hydroxide, write your observations
What do you think happened? (Remember the bonds)
What is adhesion? What is cohesion?

Test Tracking
Every time we have a test we will track our progress towards our BIG GOAL of 85% of HIGHER!
Congratulations to all of you that achieved our BIG GOAL on our first Unit Test
How to do this…. Our tests will be kept in our Tracking Folders (in your
mailboxes) We will each have individual mailbox numbers
You will get these today! For each objective on the test, you will create a bar chart of
your progress…. This will help us keep track of our progress towards our goal
and getting a 3 or 4 on the EOC!!!

Unit 1 Tracking KEY:
GREEN: 85% and higher
YELLOW: 70% - 84%
RED: 69% and below

Quick Write
1. Do you play any sports? If so, list one sport that you play. If not, list any activity you do that requires energy.
2. Why is it important that you eat before an activity?
3. List any foods you can think of that are high in one or more of the following compounds:
– Sugars– Fats– Proteins

Carbohydrates

FUNCTION
Function: to store and release QUICK energy If the energy is not used, it gets stored as FAT (or
LIPIDS)

SUBUNITS
Sugars and StarchesMonomer = monosaccharide (one unit of
sugar) Examples: Glucose & Fructose
Polymer = Polysaccharide (many units of sugar) Example: Starches

The ATOMS & RATIO
Made of CARBON (C), HYDROGEN (H), and OXYGEN (O)
These atoms are found in a special ration:
1:2:1 ratio (Example: C6H12O6)

It LOOKS Like… PicturesMonomer
Polymer

It is IMPORTANT because…
It is made by PRODUCERS in PHOTOSYNTHESIS
GLUCOSE is the sugar made by plants during photosynthesis.
Animals use this glucose to help create cellular energy (ATP)

TESTS
FOR SUGAR: Benedict’s Solution To help remember this: BEN LIKES SUGAR!
FOR STARCH: Iodine To help remember this: I DINE ON STARCH!

Monosaccharides
Monomer = 1 Unit of SUGARExamples:
Glucose Fructose
Monomers are SIMPLE carbohydratesThey give you quick energy and are typically
not very healthy Example: sugars found in candy bars!

POLYSACCHARIDES
Polymer = Many Units of SUGARExamples:
Starch sugars in plants (made of many repeating glucose molecules)
Glycogen energy storage in animal muscle Cellulose found in plant cell walls; animals can not
digest
Polysaccharides are COMPLEX carbohydratesThey are better for you than SIMPLE
carbohydrates Examples: brown rice, potatoes, pasta, bread

They typically END in…
-OSE
Example: glucOSE, fructOSE, lactOSE

What happens when they are not used?
They turn into FAT

Stations
Station 1: Organic vs. InorganicStation 2: Carb QuestionsStation 3: Build the Macromolecule-Label as a Carb-Glue the pieces down-Describe the FUNCTION-Describe the IMPORTANCE

Do Now
1. What are the subunits of a Carbohydrate?2. What is the test for a sugar?3. What is the test for a starch?4. What are the atoms of a Carb?5. What is the special ration of the atoms?6. What is the FUNCTION of a carb?

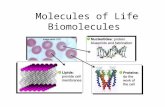
Lipids

FUNCTION of LIPIDSFunction:
Stored, long-term energy Very concentrated Twice as much energy as carbohydrates
Insulation Keeps you warm (think of the blubber on a whale!)
Body Padding Cushions body organs
Cell membranes PhosphoLIPID Bilayer Fat surrounds all of your cells and helps support the cell
Commonly called fats, oils and waxes

SUBUNITS
1 GLYCEROL and 3 FATTY ACIDSMonomer = long chains of 1 glycerol and 3
fatty acidsGLYCEROL
3 FATTY ACIDS

The ATOMS & RATIO
Made of CARBON (C), HYDROGEN (H), and OXYGEN (O)
RATION?THERE IS NO SPECIFIC RATION!! Lipids have the same ATOMS as carbohydrates, but lipids do not have a specific ration like carbohydrates do.

It LOOKS Like…
Pictures

2003-2004
Molecular Structure of Fat
not a chain (polymer) = just a “big fat molecule”
glycerol
fatty acid
fatty acid
fatty acid

TESTS
BROWN PAPER BAG TEST!If there IS a grease stain, then lipids are
present!

Saturated vs. Unsaturated
SATURATEDSATURATED
• Saturated – bonds in molecule are UNBENDABLE
• Tend to clog arteries• Typically from animals
(fats, butter, lard)
UNSATURATEDUNSATURATED
• Unsaturated – some bonds in molecule BENDABLE
• Better, but can still clog arteries
• Typically from plants (oils)


2003-2004
It’s IMPORTANT for your CELLS…
Cell membranes are made out of lipids phospholipids heads are on the outside touching water
“like” water (hydroPHILIC) tails are on inside away from water
“scared” of water (hydroPHOBIC) forms a barrier
between the cell & the outside

Stations
Station 1: Lipids QuestionsStation 2: Carbs vs. LipidsStation 3: Lipid molecule build-Label as a LIPID-Glue the monomers together on your paper-Describe the FUNCTION-Describe the IMPORTANCE

Lipids = FatsFunction
Stored Energy, support, insulation
Atoms
C, H, O
Important
Make up the CELL membrane (phospholipids)
Subunits
3 Fatty Acids & Glycerol Pictures
Test
Brown Paper Bag Test

Brown Paper Bag Test

Building A LIPID Macromolecule
Building our own Monomers and Polymers!

CarbsCarbs LipidsLipids
Carbs or Lipids?!
Cheese Bread Chocolate Cream PastaButter Fruit Cake Sausage Oil

Whiteboard Challenge
Quickly record your answer to the questions
Do not shout out or share answers
When I say “Boards Up,” flip answers to the front

1
What are the subunits of a Carbohydrate?

2
Draw the structure of Glucose

3
What are the 3 subunits of a Lipid

4
What is a Lipid?

5
What is one of the functions of a Carb?

6
What is one of the functions of a Lipid?

7
What are the 5 essential elements?

8
What is the range for an Acid?

9
What is the range for a Base?

10
What is the role of a buffer?

11
What is the test for a SUGAR?

12
What is the test for a STARCH?

13
What is the TEST for a LIPID?

14
What are the 3 elements in both CARBS and LIPIDS?

15
What is the RATION of atoms in a CARB?

Carb vs. Lipid
Compare and Contrast Carbs and Lipids

Exit Ticket
A student performed a food test on her lunch. The Iodine test and Brown paper bag test came back positive. What organic molecules were in her lunch?
A. Starches onlyB. Sugars and starchesC. Starches and lipidsD. Lipids and sugars

1. Carbohydrates are made of ________ & ___________.
2. Lipids are made of _________ & _______.
1. Lipids are tested using the __________.
1. Sugars are tested using the ____________.
1. Starches are tested using the ___________.