Tissue inhomogeneities in Monte Carlo treatment planning for proton therapy L. Beaulieu 1, M....
-
Upload
mabel-gregory -
Category
Documents
-
view
213 -
download
0
Transcript of Tissue inhomogeneities in Monte Carlo treatment planning for proton therapy L. Beaulieu 1, M....

Tissue inhomogeneities in Monte Carlo treatment planning for proton therapy
L. Beaulieu1, M. Bazalova2,3, C. Furstoss4, F. Verhaegen2,5
(1) Centre Hospitalier Univ de Quebec, Quebec, QC, CA, (2) McGill University, Montreal, QC, CA, (3) Stanford University, Stanford, CA, (4) Hopital Maisonneuve-Rosemont, Montreal, QC, CA, (5) Maastro Clinic, Maastricht, NL
Proton therapy is gaining popularity in the treatment of cancer and the
need for an accurate treatment planning system is obvious. Monte Carlo
(MC) dose calculation, despite the relatively long computation time, is the
most accurate way to determine the dose delivered to the patient during
radiation therapy. Whereas MC dose calculations for conventional
photon and electron radiotherapy have been studied extensively, proton
beam MC dose calculations have only recently received attention. In this
work, the importance of tissue segmentation in proton therapy is
investigated using dual-energy CT (DECT) imaging. Another challenge in
MC dose calculation treatment planning is metal streaking CT artifacts
with the associated tissue and mass density miss-assignment. Their
effect on MC proton beam dose calculations is studied for a prostate
patient with bilateral hip prostheses.
IntroductionIntroduction
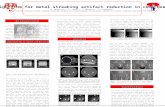
Tissue segmentation with dual-energy CT: CT images of a 30 cm
diameter cylindrical phantom with 9 tissue equivalent inserts (table1, fig
1a) were segmented into material and mass density maps using single-
energy CT (fig 1b) and DECT (fig 1c) material extraction. DECT tissue
segmentation can distinguish materials with similar relative electron
densities ρe having different effective atomic numbers Zeff. The effect of
inaccurate material segmentation for the two soft bone equivalent
materials (B200 and CB2-10) and an adipose-equivalent material (PE)
with the commonly used single energy CT material segmentation was
studied. A left lateral 16×16 cm2 200 MeV proton beam was simulated in
the MCNPX code. The mass densities for (1.9×1.9×20) mm3 voxels were
binned into 0.1 g/cm3 bins. The dose was calculated for the exact
geometry (Dexact), the single energy CT geometry (Dsingle) and the dual-
energy CT geometry (Ddual).
Materials and MethodsMaterials and Methods
Metal streaking artifacts: The effect of metal streaking artifacts and their
correction based on sinogram interpolation on MC proton beam dose
calculations was studied on a patient with bilateral hip prostheses. Dose
calculations were performed for three different simulation geometries:
considering only tissue of uniform density 1 g/cm3 (the water-only
geometry), and using a CT number to material and mass density
calibration curve with original CT images containing streaking artifacts
and with artifact corrected CT images.
A treatment plan with two 147 MeV proton beams (45° and 315°) was
simulated in the MCNPX code. First, the spread-out Bragg peak (SOBP)
was designed using simulations in a uniform water phantom (fig 2a). It is
impossible to model a modulator wheel in the MCNPX code, and
therefore the steps of the modulator wheel were approximated by 5 mm
thick PMMA blocks. In order to calculate the dose distribution of the
SOBP in the patient in a single MC simulation, 11 PMMA blocks were
inserted in the path of the (6×6) cm2 beams and the source particles (147
MeV protons) were sampled with their respective weights from the
volume between the blocks (fig 2b). The patient CT images with
(1.9×1.9×20) mm3 voxels were segmented into 4 materials (air, tissue,
bone and steel) using 0.1 g/cm3 mass density bins.
Tissue segmentation with dual-energy CT: Fig 3 presents the results of
the phantom study. The exact dose distribution is shown in fig 4a and the
differences from Dsingle and Ddual are presented in fig 3b and 3c,
respectively. In both Dsingle and Ddual, the position of the Bragg peak is
shifted with respect to the true position of the Bragg peak. The shift is 0.7
cm for Dsingle and 0.7 cm or less for Ddual. This is possibly due to mass
density differences in the single energy CT and DECT geometry from the
exact geometry. Fig 3d demonstrates the dose calculation error in the
miss-assigned B200 soft bone tissue equivalent insert. The dose in the
B200 insert was by 2% lower than in the exact and DECT geometry.
Metal streaking artifacts: The dose distributions calculated based on the
water-only geometry, on the original CT geometry and the artifact
corrected geometry are presented in fig 4. The shape of the 80% isodose
line conforms to the prostate in the water-only dose calculation (fig 4a).
ResultsResults
The dose distribution is significantly distorted in the original CT geometry
due to the artifacts (fig 4b). The apparent air between the prostheses
results in inaccurate doses with large statistical errors. Additionally due to
the air, the 20% and 30% isodose lines extend by 1.5 cm in the healthy
tissue. This might cause problems in treatment planning and its
optimization. The artifact corrected geometry produced a dose
distribution similar to the water-only dose distribution (fig 4b). The true
dose distribution is not known.
Figure 3: The exact dose distribution (Dexact) using a 200 MeV proton beam (a). PDD with two inhomogeneities (SB3 and B200) (b), the 2% dose calculation error is indicated by the arrow. The dose differences from Dexact for Dsingle (c) and Ddual (d).
The shift in the Bragg peak demonstrates the need for careful mass
density assignment in MC dose calculations for proton beams. The dose
calculation errors using the conventional single-energy CT tissue
segmentation below 2% suggest that the use of DECT for proton dose
calculations might only have a small added benefit. The patient study
shows that a metal artifact correction is necessary for patients with
bilateral hip prostheses.
ConclusionsConclusions
Figure 4: Dose distribution for a prostate patient calculated on the basis of homogeneous water geometry (a), on the basis on the geometry with metal artifacts (b) and using the artifact correct images (c). The arrows indicate the apparent range of protons due to artifacts.
MATERIAL ρe Zeff
lung (LN300) 0.292 7.864
lung (LN450) 0.438 7.835
polyethylene (PE) 0.945 5.740
CT Solid Water (SW) 0.986 8.111
B200 bone mineral 1.097 10.897
CB2 - 10% CaCO3 1.142 8.905
CB2 - 30% CaCO3 1.286 11.393
CB2 - 50% CaCO3 1.470 12.978
SB3 cortical bone 1.692 14.141
Figure 1: Figure 1: The exact geometry (a), the single-energy material segmentation (b) and the dual-energy CT material segmentation (c).
Table 1:Relative electron densities ρe and effective atomic numbers Zeff for materials used in the tissue inhomogeneity study.
a) b)
All CT geometries were converted into lattices and the dose was scored
using the *F8:H,P,E energy deposition tally. Protons, photons and
electrons were transported using the la150u cross section library with
energy cutoffs of 10 keV. In all simulations, 107 particles were simulated
in approximately 15 hours on a 3 GHz machine.
Figure2: The spread-out Bragg peak for patient dose calculations (a) and the MCNP geometry showing 0.5 cm PMMA blocks for modulation of the 147 MeV proton beam (b).
c)
a) b)
d)
a)
b) c)