Shapes of molecules The shapes of molecules and ions can be described by valence shell electron...
-
Upload
pauline-curtis -
Category
Documents
-
view
218 -
download
0
Transcript of Shapes of molecules The shapes of molecules and ions can be described by valence shell electron...


Shapes of molecules

Shapes of molecules The shapes of
molecules and ions can be described by valence shell electron pair repulsion
VSEPR predicts the shapes and bond angles of simple molecules
Valence Shell Electron Pair Repulsion

It’s all about the pairs All of the electrons in an ion or molecule are in
organised into pairs There are two types of electron pairs:
Bonding pairs (the two shared electrons in a covalent bond)
Lone pairs (two electrons in a pair not involved in bonding – also known
as non-bonding pairs).
Electron pairs will repel each other as far as possible. Like charges repel
This causes the shape of any molecule or ion

It’s all about the pairs Chemists sometimes use lines to show bonds
becomes H — Cl
Draw out the following molecules just using a line to show the bonds O2 , H2O, CH4 , NH3 , SF6 , BF3 and PF5
They are in 2D not 3Dlike real life
XO ClH
X X
X X
XX
What is the problem when you draw these molecules onto a piece of paper?

Beryllium Fluoride
Simple rules to work it out:
1. Draw a stick diagram. (displayed formula technically!)
2. Add in any lone pairs onto the stick diagram.
3. Count the total number of bonds AND lone pairs to work out the shape.
LINEAR
2 Bonding pairs 0 Lone pairs
The furthest these pairs of electrons can spread apart is 180o
XO
OXF B
eF
O O O O
OO
OO
O OO O

Boron Trifluoride
Simple rules to work it out:
1. Draw a stick diagram. (displayed formula technically!)
2. Add in any lone pairs onto the stick diagram.
3. Count the total number of bonds AND lone pairs to work out the shape.
TRIGONAL PLANAR
3 Bonding pairs 0 Lone pairs
The furthest these pairs of electrons can spread apart is 120o
1200 provides the greatest distance EVEN IN 3-dimensional space
XO
OX
F
B
FO O O O
OO
OO
O OO O
X O
O O
FOO
OO

Further examples
TETRAHEDRAL 4 Bonding pairs 0 Lone pairs
HEXAGONAL 6 Bonding pairs 0 Lone pairs

Double and triple bonds
TETRAHEDRAL 4 bonding pairs
(even though one is double)
0 Lone pairs
The furthest these pairs of electrons can spread apart is 109.5o
Double and triple bonds do not effect the shape (well not enough
that we have to worry!)
This phosphate ion PO43- has a
tetrahedral shape

5 Bonding pairs 5 bonding pairs and 0 lone pairs is called:
TRIGONAL BI-PYRAMIDAL
It is a combination of the 1st and 2nd
examples: linear and
trigonal planar
So what would the TWO
different bond angles be?

Practice to fluency Decide the shape and
bond angle(s) of the following molecules on the right
Prepare an explanation for the rest of the class explaining the shape and bond
The fact that some of these molecules are ions is irrelevant to the shape!
CO2NO3-
PF5 CH4
SO42-SF6

Lone pairs vs. bonding pairs Lone pairs are more compact as they are
closer to the nucleus than bonding pairs This causes lone pairs to provide more
repulsion LP-LP
•The greatest repulsion is between two lone pairs
LP-BP
•Followed by the repulsion between one lone pair and one bonding pair
BP-BP
•The weakest repulsion is between two bonding pairs

Ammonia
PYRAMIDAL
Ammonia has 3 bonding pairs and 1 lone pair With 4 pairs in total it is similar to tetrahedral However the LP repels the bonding pairs and reduces
the bond angle by 2.5o from 109.5o to 107o

Water
NON-LINEAR (BENT)
Water has 2 bonding pairs and 2 lone pair With 4 pairs in total it is similar to tetrahedral However the TWO LP’s repels the bonding pairs and reduces
the bond angle by 2.5o and 2.5o from 109.5o to 104.5o

3D diagrams To draw 3D diagrams
on 2D paper chemists draw the bonds slightly differently
Solid wedges show a bond that comes ‘up’ from the paper or screen (towards you)
Hashed wedges show a bond that goes ‘down’ from the paper (away from you)
methane TETRAHEDRAL

XY6
Bond angles?
“Real” shape?
Name?
Examples?

XY5
Bond angles?
“Real” shape?
Name?
Examples?

XY4
Bond angles?
“Real” shape?
Name?
Examples?

XY3
Bond angles?
“Real” shape?
Name?
Examples?

XY2
Bond angles?
“Real” shape?
Name?
Examples?

RECAP
Molecules, or ions, possessing ONLY BOND PAIRS of electrons fit into a set of standard shapes. All the bond pair-bond pair repulsions are equal.
All you need to do is to count up the number of bond pairs and chose one of the following examples...
C
2 LINEAR 180º BeCl2
3 TRIGONAL PLANAR 120º AlCl3
4 TETRAHEDRAL 109.5º CH4
5 TRIGONAL BIPYRAMIDAL 90º & 120º PCl5
6 OCTAHEDRAL 90º SF6
BOND BONDPAIRS SHAPE ANGLE(S) EXAMPLE
A covalent bond will repel another covalent bond

IRREGULAR SHAPES
If a molecule, or ion, has lone pairs on the central atom, the shapes are slightly distorted away from the regular shapes. This is because of the extra repulsion caused by the lone pairs.
BOND PAIR - BOND PAIR < LONE PAIR - BOND PAIR < LONE PAIR - LONE PAIR
OO O
As a result of the extra repulsion, bond angles tend to be slightly less as the bonds are squeezed together.

Practice to fluency Decide the shape and
bond angle(s) of the following molecules on the right
For each example include: Name Bond angle(s) 3-D diagram The number of bonding
pairs The number of lone pairs
AlCl3BeCl2
CH4 NH4+

BERYLLIUM CHLORIDE
ClBe Be ClCl
Beryllium - has two electrons to pair up
Chlorine - needs 1 electron for ‘octet’
Two covalent bonds are formed
Beryllium still has an incomplete shell

BERYLLIUM CHLORIDE
Cl ClBe180°
BOND PAIRS 2
LONE PAIRS 0
BOND ANGLE...
SHAPE...
180°
LINEAR
ClBe Be ClCl
Beryllium - has two electrons to pair up
Chlorine - needs 1 electron for ‘octet’
Two covalent bonds are formed
Beryllium still has an incomplete shell

Al
ALUMINIUM CHLORIDE
Cl
Cl
Al120°
Cl
ClAl
Cl
Cl
Cl
BOND PAIRS 3
LONE PAIRS 0
BOND ANGLE...
SHAPE...
120°
TRIGONAL PLANAR
Aluminium - has three electrons to pair up
Chlorine - needs 1 electron to complete ‘octet’
Three covalent bonds are formed; aluminium still has an incomplete outer shell.

Al
ALUMINIUM CHLORIDE
Cl
Cl
Al120°
Cl
ClAl
Cl
Cl
Cl
BOND PAIRS 3
LONE PAIRS 0
BOND ANGLE...
SHAPE...
120°
TRIGONAL PLANAR
Aluminium - has three electrons to pair up
Chlorine - needs 1 electron to complete ‘octet’
Three covalent bonds are formed; aluminium still has an incomplete outer shell.

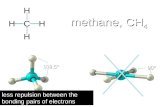
METHANE
C H CH
H
H
H
Carbon - has four electrons to pair up
Hydrogen - 1 electron to complete shell
Four covalent bonds are formed
C and H now have complete shells

METHANE
BOND PAIRS 4
LONE PAIRS 0
BOND ANGLE...
SHAPE...
109.5°
TETRAHEDRAL
C H CH
H
H
H
109.5°
H H
C
H
H
Carbon - has four electrons to pair up
Hydrogen - 1 electron to complete shell
Four covalent bonds are formed
C and H now have complete shells

METHANE
BOND PAIRS 4
LONE PAIRS 0
BOND ANGLE...
SHAPE...
109.5°
TETRAHEDRAL
C H CH
H
H
H
Carbon - has four electrons to pair up
Hydrogen - 1 electron to complete shell
Four covalent bonds are formed
C and H now have complete shells

AMMONIA
HN NH H
HBOND PAIRS 3
LONE PAIRS 1
TOTAL PAIRS 4
• Nitrogen has five electrons in its outer shell
• It cannot pair up all five - it is restricted to eight electrons in its outer shell
• It pairs up only three of its five electrons
• 3 covalent bonds are formed and a pair of non-bonded electrons is left
• As the total number of electron pairs is 4, the shape is BASED on a tetrahedron

AMMONIA
ANGLE... 107°
SHAPE... PYRAMIDAL
HN NH H
HBOND PAIRS 3
LONE PAIRS 1
TOTAL PAIRS 4
H
H
N
H
H
H
N
H
107°H
H
N
H
• The shape is based on a tetrahedron but not all the repulsions are the same
• LP-BP REPULSIONS > BP-BP REPULSIONS
• The N-H bonds are pushed closer together
• Lone pairs are not included in the shape

AMMONIA
HN NH H
HBOND PAIRS 3
LONE PAIRS 1
TOTAL PAIRS 4

Spot the errors Read this set of instructions and write it out
again correcting any mistakes as you go To predict the shape of different
molecules: A. Draw a dot and cross diagram for the molecule B. Count the total number of electrons around
the central atom (include electrons in the lone pairs)
C. Decide on the starting shape based on the total number of lone pairs (eg. 3 lone pairs = trigonal planar)
D. If lone pairs are present just ignore themE. Decide on the name