SHAPES OF MOLECULES
-
Upload
kasimir-swanson -
Category
Documents
-
view
33 -
download
0
description
Transcript of SHAPES OF MOLECULES

SHAPES OF MOLECULES

REMINDER ABOUT ELECTRONS
Electrons have negative chargesNegative charges “repel” each other In molecules, electrons want to get as
far away from each other as possibleAs a result, this repulsion of electrons
leads to the shape of the molecule

SHAPE OF MOLECULES There is a simple model used to
determine the shape of molecules. VSEPR (Valence Shell Electron Pair
Repulsion) A simple model that predicts the general
shape of a molecule based on the repulsion between both the bonding and nonbonding electron clouds
Always based on the CENTRAL atom

DEFINITIONElectron cloud: Any type of bond (single, double or triple) or any set of unshared pairs of electrons.
Unshared pair of electron: any pair of electrons not involved in a covalent bond.

2 ELECTRON CLOUDS Example: CO2 Count the number of electron clouds
surrounding the central atom. . . .O=C=O· · ··
There are 2 double bonds around the central Carbon (C) Thus, there are 2 electron clouds

2 ELECTRON CLOUDSElectrons have a negative charge
and repel each otherThus, the electrons that maintain
the bond will move as far away from each other as possible
If the central atom has only two electron clouds, it will have a linear shape

LINEARView the clip
NOTE: That the attached atoms are 180° apart from each other

3 ELECTRON CLOUDS Again count the number of electron
clouds around the central atom (SO3) ..
: O: .. | ..
:0-S=O ·· ··

3 ELECTRON CLOUDSAgain the electron clouds
want to move as far from each other as possible
When the central atom has 3 electron clouds surrounding it, the molecule has a trigonal planar shape

TRIGONAL PLANARView the clip
NOTE: The attached atoms are 120° apart from each other

3 ELECTRON CLOUDSWhat happens if one of the electron clouds is an unshared pair of electrons
O3
Do the Lewis dot structure for this in your notes

3 ELECTRON CLOUDS Notice the following:
You have one double bond You have one single bond You have one unshared pair of
electrons When you view a molecule, you can’t
see the unshared pair of electrons This creates a bent shape

BENT View the clip
NOTE: That the attached atoms and unshared pair are 120° apart from each other Since you can’t see the unshared pair, the
molecule looks bent

4 ELECTRON CLOUDS
Again count the number of electron clouds
CCl4Draw the Lewis dot structure in your notes

4 ELECTRON CLOUDSSame as before, the electron
clouds want to get as far from each other as possible
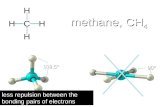
When the central atom has 4 electron clouds surrounding it, you get a tetrahedral

TETRAHEDRALView the clip
NOTE: That the attached atoms are 109.5° apart from each other

4 ELECTRON CLOUDSWhat happens if you have 3
atoms bound to a central atom with one unshared pair
NH3
Do the Lewis dot structure for this in your notes

4 ELECTRON CLOUDSNotice the following:
You have three single bondsYou have one unshared pair of
electronsWhen you view a molecule, you
can’t see the unshared pair of electrons
This creates a pyramidal shape

PYRAMIDALView the clip
NOTE: That the attached atoms and unshared pair are 109.5° apart from each otherSince you can’t see the unshared
pair, the molecule looks like a pyramid

4 ELECTRON CLOUDSWhat happens if you have 2
atoms bound to a central atom with two unshared pairs
H2ODo the Lewis dot structure for
this in your notes

4 ELECTRON CLOUDSNotice the following:
You have two single bondsYou have two unshared pairs of
electronsWhen you view a molecule, you
can’t see the unshared pair of electrons
This creates a bent shape

BENTView the clip
NOTE: That the attached atoms and unshared pair are 109.5° apart from each otherSince you can’t see the unshared
pair, the molecule looks like a pyramid

TRY THESE HANDOUT For each of the following, write the
Lewis structure and indicate the shape:
1.CBr4
2.CS2

POLARITY OF MOLECULES We’ve already discussed the difference
between nonpolar, polar and ionic bonds (electronegativity difference)
Molecular shape is important for determining the polarity of a molecule.
Covalently bonded molecules can be polar or nonpolar based on the shape of the molecule

EXAMPLE Let’s look at the shapes of
H2O
CF4
First of all: H-O bond of water has an electronegativity
difference of 1.4 (polar covalent) C-F bond of CF4 has an electronegativity
difference of 1.5 (polar covalent)

EXAMPLE Since both H2O and CF4 have polar
covalent bonds, we would expect both molecules to be polar covalent
This is not the case IN YOUR NOTES: Draw the molecular
shape of both: H2O
CF4

ANSWER – THINK OF TUG-O-WAR Water is a bent molecule
As a result water is a polar molecule You have a partial charge of σ- for O and
σ+ for H CF4 is a tetrahedral molecule
Because of the shape you have a nonpolar molecule
Even though you have partial charges, the charges cancel out because of the shape

WRITE WHICH SHAPES ARE POLAR/NONPOLAR?
Take a look at your handout that shows the shape of different molecules.
Which shapes do you think are polar?
Which shapes do you think are nonpolar?

ANSWERPolar shapes (have lone pairs):
BentPyramidal
Nonpolar shapes (do not have lone pairs):LinearTrigonal planarTetrahedral

TRY THE FOLLOWING Determine the Lewis structure of the following.
Are the molecules polar or nonpolar?
1.Cl2O
2.CO2
3.NF3

ANSWER
1. Bent, polar molecule2. Linear, nonpolar
molecule3. Pyramidal, polar
molecule