Nanoscale hybrid objects: from 3D SANS study to tunable 2D...
Transcript of Nanoscale hybrid objects: from 3D SANS study to tunable 2D...

Nanoscale hybrid objects: from 3D SANS study to tunable 2D arrays
formationGéraldine Carrot
Laboratoire Léon Brillouin, CEA-Saclay.

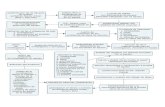
10 nm Polymerization in organic or aqueous medium Study of the structure via SANS (3-D) Use of Contrast MatchingOrganization in 2D and neutron reflectivity
SiO2
Platinum nanoparticles
Pt
Nano-objects based on polymer-graftedinorganic nanoparticles
Bio-application as bio-sensor (glucose probe)
3 nm
Short chains Long chains
dd
Silica nanoparticles

Density of chainsChain lengths
Colloidal suspension in water or in organic solvent
Necessary to avoid aggregation
Control of the surface state
By grafting polymer chains:
Dormant specie
Active specie K = ka / kd
ka
kdCH2 C X
R1
R2
CH2 C
R1
R2
+ Cu(I)X / Ligand + Cu(II)X2 / Ligand
Controlled polymerization principle:

SiO2SiO2SiO2SiO2 SiO2SiO2
O
OO O
O
Br
OO
(CH2)3
CH3
OO(CH2)3
CH3
OO(CH2)3
CH3
Br
OO
(CH2)3
H3C
OO
(CH2)3
H3C
OO
(CH2)3
H3C
Br
O
O
(CH2)3
H3C
O
O
(CH2)3H3C
O
O
(CH2)3H3C
OOO
O
O
Br
O
(CH2)3
CH3
O
O
(CH2)3
CH3
O
O
(CH2)3
CH3
Br
OO(H2C)3
OO(H2C)3
OO(H2C)3
Br
O
O
(H2C)3
CH3
O
O
(H2C)3
CH3
O
O
(H2C)3
CH3
Br
O O
(CH2)3
O O
(CH2)3
CH3
O O
(CH2)3
CH3CH3
Br
O
O
(CH2)3
CH3
O
O
CH2)3(
CH3
O
OCH3
(CH2)3
H3C
CH3
H3CO
OHOH
OH
OH
OH
OH
HOHO
HO
HO
HOHO
HOHO
OHOH
S
O Br
OSi
SO
Br
OSi
S O
Br
OSi
S O
Br
OSi
S
OBr
OSi
SO
Br
O
O
OO
O
OSi
SO
Br
OH
OH
OH
OH
HO
HO
HO
O
OO O
O
OOO
O
O
HS
OSi
SH
OSi
SH
O Si
SH
OSi
SH
OSi
HS
O
O
OO
O
OSi
HSOH
OH
OH
OH
HO
HO
HO
Br
O
Br MABu
n
n
n
n
n
n
n
n
Cu/PMDETADMAP
Controlled radical polymerization from silica nanoparticles
0
0.05
0.1
0.15
0.2
0 100 200 300 400 500
Conversion
Conversion
temps (min)0
0.05
0.1
0.15
0.2
0.25
0 10 20 30 40 50 60
Cinétique
ln([M]0/[M])
t 2/3
0
2000
4000
6000
8000
1 104
1.2 104
1
1.1
1.2
1.3
1.4
1.5
0 0.05 0.1 0.15 0.2
Mn
Mw/M
n
Conv. (%)

Kinetic study of the polymerization via SANS :dispersion state of particles
Desaggregation of the particles as the polymerization proceed
D solvent
Polymer
SiO2
“Polymer matching”
q (Å-1)
0
50
100
150
200
0.01 0.1
I (cm-1)
1st kinetic point
Finalpoint
H solvent
0.0001
0.001
0.01
0.1
1
10
100
0.001 0.01 0.1 1
I (cm-1)
q (Å-1)
After purification: back tothe same state of aggregationthan the initial particles
El Harrak A., Carrot G.,Oberdisse J., Boué F. Macromolecules 2004, 37, 6376-6384.

of the signal at low q
Kinetic study of the polymerization via SANS :Corona growth
0.001
0.01
0.1
1
10
100
0.01 0.1
q (Å -1)
I (cm-1)
Initial point
Final point“Silica matching”
30 min
60 min
230 min
420 min
Polymerization time
Oscillation at the intermediate q with a shift toward low qIncrease of the corona size
of the quantity of polymer

Corona model
R2
R1
0
1 1 0 - 6
2 1 0 - 6
3 1 0 - 6
4 1 0 - 6
5 1 0 - 6
0 0 .0 2 0 .0 4 0 .0 6 0 .0 8 0 .1
I.q4
q (Å-1)
Polymer-grafted silica nanoparticles after purification: corona characterization
q-2Nagg = 5e ≈ 70 Å with 30% of polymerρcore = ρ solvant
Vcorona = 1.28 .10-17 cm-3
Mcorona = 4.0.10-18 gWith Mw=12000 g/mol
198 chains per particle
G. Carrot, A. El Harrak, J. Oberdisse, J. Jestin, F. Boué SoftMatter, 2006, 2, 1043.
“Silica matching”
0.001
0.01
0.1
1
10
100
1000
0.001 0.01 0.1 1q(Å-1)

0.01 0.11E-4
1E-3
0.01
0.1
1
I(q) (
cm-1)
q (Å-1)
Grafted silica nanoparticles in aqueous phase*
0.01 0.10.01
0.1
1
10
q-2
I(q) (
cm-1)
q(Å-1)
R = 20,3 Åσ= 0.28
*Thesis of Jérôme Vinas, CROPS, Marseille, D. Bertin, D.Gigmes
Am Si(OR)3 HO OSiAmO
OSilice+
hydrolyse
condensation
HO
HO
Silice
Same signal asthe bare particles
Polymer signal inSilica matching Si
Polymer matching

ATRP from platinum nanoparticles
-80.7PtPMAB810.540100000PtMAB8-2
36.283.6PtPMAB815.4334100000PtMAB8-1
29.583.1PtPMAB715.3113100000PtMAB7-2
34.288.0PtPMAB719.2455100000PtMAB7-1
Tg (°C)%Polymer
Polymerization batch
Conv.%
Reaction time
Theoretical Mn
Sample
(1) H. Perez et al. Chem. Mater. 1999, 11, 3460-3463. (3) G. Carrot, H. Perez Polym. Prep. 2006, 827(2) H. Perez et al. Chem. Mater. 2001, 13, 1512. (4) G. Carrot, F. Gal, H. Perez, Langmuir, in Press.
S
N H 2
S
N H 2
2 )
1) NaBH 4
S
N H 2
S N H 2
S
H 2 N
S
H 2 N
S
N H 2
SH 2 N PtS N H 2
2
PtCl
C lCl
ClS
N H
S
N H
S
N H
S N H
S
H N
S
H N
S
N H
SHN Pt
O
B r
O B r
O
B r
O
B rO
B r
O
B r
B r
Br
(CH3)2BrCCOB r
DMAP

0 200 400 600 800 1000 12000.0
0.1
0.2
0.3
Con
v%
time (min)0 20 40
0.0
0.1
0.2
0.3
Ln(M
o/M)
t2/3
Surface polymerizations from platinum nanoparticles *
PtPt PtPt
PtLangmuir FilmsPt
Pt Pt Pt
PtPt Pt
compressioncompression
G. Carrot, F. Gal, C. Cremona, H. Perez, Langmuir, in Press.

30 40 50 60 70 80 900
20
40
60
80
100
(b)
(311),(222)(220)
(200)
(111)
Inte
nsity
(arb
. uni
ts)
Two Theta in degree
0.01 0.1
1E-3
0.01
0.1
1
I(q)
q (Å-1)
“Polymer matching”
“Platinum matching”
Study of the nano-objects in solution via SANS
57PtPMA8-(2) p =10.5 %
68PtPMA8-(1) p = 15.4 %
75PtPMA7-(2) p = 15.4 %
95PtPMA7-(1) p=20 %
Rg (Å) Samples
Radius of giration obtained by the fit of the signal in the Guinier regime
0.01 0.11E-5
1E-4
PtPMABu8-1 PtPMABu8-2
I(q)(q
2 )
q (Å-1)
Pt Pt

0.01 0.1
0.01
0.1
1
I(q) (
cm-1)
Wavevector q (Å-1)0.01 0.1
0.01
0.1
1
I(q)(c
m-1)
Wavevector q (Å-1)
Fit of the spectra with a Star model
Zimm-Stockmayer-Benoît model:
)13()()()(
2222
−==
ffqRfqRqR E
gBg
Lg=x
“Platinum matching”
Pt Pt
⎥⎦
⎤⎢⎣
⎡−++−−−−−= ))3(
2)exp()2()2exp()1(
2(2)(
2ffx
fxff
fxff
xxF

Sample Number of
branche, f
Rg gaussienne
RgL
(Å)
Rg branche
RgB
(Å)
Rg Star,Rg
E
(Å)
Number of chains per nm2
Mn of 1 branche,
MB (g.mol-1)
Mn from TGA
(g.mol-1)
PtPMA7-1
5.3 122 53 85.8 0.42 12055 13222
PtPMA7-2
5.2 102 44.3 72.33 0.42 8600 11000
PtPMA8-1
8 119 42.4 70.35 0.63 7723 8558
PtPMA8-2
7 97 36.9 60.8 0.55 5844 7940
Determination of the number of chains and molecular weight
0.01 0.1
1E-3
0.01
0.1
1
I(q) (
cm-1)
q (Å-1)
Pt
Star model

0 50000 1000000
10
20
30
40
P(m
N/m
)
S/Object (Ų)
Langmuir Films-Compression Isotherms
PtPMAB 7-2 (◊) Conv. 15%PtPMAB 7-1 (▲) Conv. 20%Compression
Reversibility
Compression at 2 mN/m 26 mN/m Decompression at 2mN/m

3D-Organization of particles in 2D films
cross-linked
multilayers(with or without Pt)
Alternating H and D
Dispersion of PS-g-Pt NP in cross-linkedfilms via spin-coating and N3 reactions
ANR 2007: Multiclick
Neutron reflectivity

« 3D » cross-linked hybrid multilayer filmsANR 2007 Multiclick Neutron reflectivity
No interdiffusionbetween layers
Alternated polymerlayers using H and D
S. Al-Akhrass, F. Gal, D. Damiron, P. Alcouffe, C. Hawker, F. Cousin, G. Carrot,E. Drockenmuller, Soft Matter (in press).

« 3D » multilayers filmsANR 2007 Multiclick
0.00 0.02 0.04 0.06 0.08 0.10
10-9
10-8
10-7
Particles/PS : 1/5 Particles/PS : 4/5
RQ
4 [Å-4]
Q [Å-1]
S. Al-Akhrass, F. Gal, D. Damiron, P. Alcouffe, C. Hawker, F. Cousin, G. Carrot,E. Drockenmuller, Soft Matter (in press)
Dispersed Pt NP
Neutron reflectivity

Thanks
J. Oberdisse, Université de MontpellierA. El Harrak, PhD student LLB, J. Jestin, CNRS LLB, F. Boué, CNRS LLB
H. Perez, CEA, SPAM-DRECAMFrançois Gal, PhD student LLB
F. Cousin, CEA LLBE. Drockenmuller, Université de LyonS. Al-Akhrass, Université de Lyon

![[XLS] · Web view100000 100000. 100000 100000. 100000 100000. 200000 100000. 500000 200000. 100000 3000. 500000 200000. 500000 700. 500000 70.](https://static.fdocuments.in/doc/165x107/5ab577a37f8b9a2f438c946c/xls-view100000-100000-100000-100000-100000-100000-200000-100000-500000-200000.jpg)
![LONG TERM PRECESSION MODELservo.aob.rs/eeditions/CDS/Srpsko bugarska konferencija/8... · 2013-08-30 · LONG TERM PRECESSION MODEL X ["]-100000 -50000 0 50000 100000 Y ["]-200000-150000-100000-50000](https://static.fdocuments.in/doc/165x107/5f0451417e708231d40d616e/long-term-precession-bugarska-konferencija8-2013-08-30-long-term-precession.jpg)




![[XLS] · Web view9/4/2014 100000000 100000 0 9/11/2014 500000 500000 0 9/23/2014 1900000000 5000000 0 9/27/2014 5000000 100000 0 9/30/2014 1750000000 50000000 0 9/1/2014 100000 100000](https://static.fdocuments.in/doc/165x107/5aa500977f8b9ab4788c9998/xls-view942014-100000000-100000-0-9112014-500000-500000-0-9232014-1900000000.jpg)

![[XLS]reports.mca.gov.inreports.mca.gov.in/Reports/MasterDataExcels/company... · Web view3800000 3800000 700000 530340 100000 100000 5000000 1200000 600000 509600 30000000 9377400](https://static.fdocuments.in/doc/165x107/5ad0382a7f8b9a71028d9ead/xls-view3800000-3800000-700000-530340-100000-100000-5000000-1200000-600000-509600.jpg)