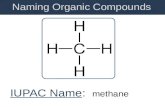
NAMING COMPOUNDS Naming compounds from their chemical symbol is similar to what youve already done ...
Click here to load reader
-
Upload
georgina-horton -
Category
Documents
-
view
216 -
download
0
description
Transcript of NAMING COMPOUNDS Naming compounds from their chemical symbol is similar to what youve already done ...

NAMING COMPOUNDS
• Naming compounds from their chemical symbol is similar to what you’ve already done – just opposite

Ca3P2
Ca P 2
i.) separate the two elementsii.) add 1’s with the element that has no numberiii.) cross-over from bottom to topiv.) add the charges – REMEMBER that first one (metal) is positive and the second (non-metal) is negativev.) make sure the charges match those found in the Periodic Table*** If they do not, MULTIPLY both charges, with the same number to find their true charge
vi.) write the name – the metal stays the same but the non-metal changes it’s ending to “ide”
3
32+ -
calcium phosphide

AlBr3
Al Br 3
i.) separate the two elementsii.) add 1’s with the element that has no numberiii.) cross-over from bottom to topiv.) add the charges – REMEMBER that first one (metal) is positive and the second (non-metal) is negativev.) make sure the charges match those found in the Periodic Table*** If they do not, MULTIPLY both charges, with the same number to find their true charge
vi.) write the name – the metal stays the same but the non-metal changes it’s ending to “ide”
1
13+ -
aluminum bromide

MgO
Mg O 1
i.) separate the two elementsii.) add 1’s with the element that has no numberiii.) cross-over from bottom to topiv.) add the charges – REMEMBER that first one (metal) is positive and the second (non-metal) is negativev.) make sure the charges match those found in the Periodic Table*** If they do not, MULTIPLY both charges, with the same number to find the non-metals true charge
vi.) write the name – the metal stays the same but the non-metal changes it’s ending to “ide”
1
11+ -
magnesium oxide
×2
Oxygen should be a -2
×22 2

PRACTICE QUESTIONSLiI Ca3N2
CaBr2 Al2O3
Ag2O BaCl2
BeS ScP
lithium iodide
calcium bromide
silver oxide
beryllium sulfide
calcium nitride
aluminum oxidebarium chloridescandium phosphide

LiI• i.) separate the two elements Li I• ii.) add 1’s with the element that has no number Li1
I1
• iii.) cross-over from bottom to top Li11 I11
• iv.) add the charges – REMEMBER that first one (metal) is positive and the second (non-metal) is negative
Li+11 I1-1
• v.) make sure the charges match those found in the Periodic Table
• *** If they do not, MULTIPLY both charges, with the same number to find their true charge
• vi.) write the name – the metal stays the same but the non-metal changes it’s ending to “ide”
• lithium iodide

CaBr2• i.) separate the two elements Ca Br• ii.) add 1’s with the element that has no number Ca1
Br2
• iii.) cross-over from bottom to top Ca21 Br2
1
• iv.) add the charges – REMEMBER that first one (metal) is positive and the second (non-metal) is negative
Ca+21 Br2
-1
• v.) make sure the charges match those found in the Periodic Table
• *** If they do not, MULTIPLY both charges, with the same number to find their true charge
• vi.) write the name – the metal stays the same but the non-metal changes it’s ending to “ide”
• calcium bromide

Ca3N2• i.) separate the two elements Ca N• ii.) add 1’s with the element that has no number Ca3
N2
• iii.) cross-over from bottom to top Ca23 N2
3
• iv.) add the charges – REMEMBER that first one (metal) is positive and the second (non-metal) is negative
Ca+23 N2
-3
• v.) make sure the charges match those found in the Periodic Table
• *** If they do not, MULTIPLY both charges, with the same number to find their true charge
• vi.) write the name – the metal stays the same but the non-metal changes it’s ending to “ide”
• calcium nitride

Al2O3• i.) separate the two elements Al O• ii.) add 1’s with the element that has no number Al2
O3
• iii.) cross-over from bottom to top Al32 O32
• iv.) add the charges – REMEMBER that first one (metal) is positive and the second (non-metal) is negative
Al+32 O3
-2
• v.) make sure the charges match those found in the Periodic Table
• *** If they do not, MULTIPLY both charges, with the same number to find their true charge
• vi.) write the name – the metal stays the same but the non-metal changes it’s ending to “ide”
• aluminum oxide