Metabolismo ii fotossintese
-
Upload
education-for-all -
Category
Education
-
view
387 -
download
3
Transcript of Metabolismo ii fotossintese

Other Other
Metabolic Metabolic
PathwaysPathways
Fats Glycogen Protein


The body fat is our major source of stored energy
• Our adipose tissue is made of fat cells adipocytes.
• A typical 70 kg person has about 135 000 kcal of energy stored as fat, 24 000 kcal as protein, 720 kcal as glycogen reserves, and 80 kcal as blood reserves, and 80 kcal as blood glucose.
• The energy available from stored fats is about 85 % of the total energy available in the body.

Lipolysis via β-oxidation
Lipases

Beta-oxidation of fatty acids
• β-oxidation of FA produces acetyl CoA
and NADH and FADH2, which are
sources of energy (ATP)
• First, FA are converted to acyl CoA in the
cytoplasm:

Carnitine shuttle
• For transport into mitochondria, CoA is replaced with carnitineby acylcarnitine transferase I
• Inside mitochondria a corresponding enzyme (II) forms acyl CoA
• Malonyl CoA inhibits acylcarnitine transferase I
• So, when FA synthesis is active, FA are not transported into mitochondria
• Defects in FA transport (including carnitine deficiency) are known

Beta-Oxidation of Fatty Acids
In reaction 1, oxidation:
• Removes H atoms from
the αααα and ββββ carbons.
ββββ αααα
the αααα and ββββ carbons.
• Forms a trans C=C bond.
• Reduces FAD to FADH2.

Beta-Oxidation of Fatty Acids
In reaction 2, hydration:
• Adds water across the
trans C=C bond.
ββββ αααα
trans C=C bond.
• Forms a hydroxyl group
(—OH) on the ββββ carbon.

Beta (ββββ)-Oxidation of Fatty Acids
In reaction 3, a
second oxidation:
• Oxidizes the hydroxyl
ββββ αααα
• Oxidizes the hydroxyl
group.
• Forms a keto group
on the ββββ carbon.

Beta (ββββ)-Oxidation of Fatty Acids
In Reaction 4, acetyl CoA is cleaved:
• By splitting the bond • By splitting the bond between the αααα and ββββcarbons.
• To form a shortened fatty acyl CoA that repeats steps 1 - 4 of ββββ-oxidation.

Beta (ββββ)-Oxidation of Myristic (C14) Acid

Beta (ββββ)-Oxidation of Myristic (C14) Acid
(continued)
7 Acetyl
CoA6 cycles

ββββ-oxidação dos ácidos gordos – uma via em espiral

Cycles of β-Oxidation
The length of a fatty acid:
• Determines the number of oxidations and
• The total number of acetyl CoA groups.
Carbons in Acetyl CoA ββββ-Oxidation CyclesCarbons in Acetyl CoA ββββ-Oxidation Cycles
Fatty Acid (C/2) (C/2 –1)
12 6 5
14 7 6
16 8 7
18 9 8

β-Oxidation and ATP
Activation of a fatty acid requires:
• 2 ATP
One cycle of oxidation of a fatty acid produces:
• 1 NADH 3 ATP
• 1 FADH 2 ATP• 1 FADH2 2 ATP
Acetyl CoA entering the citric acid cycle produces:
• 1 Acetyl CoA 12 ATP

ATP for Lauric Acid C12
ATP production for lauric acid (12 carbons):
Activation of lauric acid -2 ATP
6 Acetyl CoA6 acetyl CoA x 12 ATP/acetyl CoA 72 ATP
5 Oxidation cycles5 NADH x 3ATP/NADH 15 ATP
5 FADH2 x 2ATP/FADH2 10 ATP
Total 95 ATP

Oxidation of Unsaturated Fatty Acids.
• Oxidation of monounsaturated fatty acyl-CoA requires additional reaction performed with the help of the enzyme isomerase.
• Double bonds in the unsaturated fatty acids are in the cis configuration and cannot be acted upon by enoyl-CoA hydratase (the enzyme in the cis configuration and cannot be acted upon by enoyl-CoA hydratase (the enzyme catalyzing the addition of water to the trans double bond generated during β-oxidation.
• Enoyl-CoA isomerase repositions the double bond, converting the cis isomer to trans isomer, a normal intermediate in β-oxidation.

Protein Catabolism
Proteins are degraded into amino acids
Ammonium ion is converted
to urea in most mammals
The liver is the major site of protein degradation in mammals
First step in protein
degradation is the
removal of the nitrogen

Catabolism of proteins:
-amino acids undergo deamination to remove the amino
group
-remainder of the amino acid is converted to a molecule
that enters glycolysis or the Krebs cycle
Protein Catabolism
that enters glycolysis or the Krebs cycle
-for example:
alanine is converted to pyruvate
aspartate is converted to oxaloacetate

Amino acid Catabolism

• Almost all plants are photosynthetic autotrophs, as
are some bacteria and protists– Autotrophs generate their own organic matter through
photosynthesis
– Sunlight energy is transformed to energy stored in the form of
chemical bonds
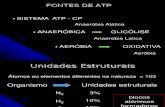
THE BASICS OF PHOTOSYNTHESIS
(a) Mosses, ferns, andflowering plants
(b) Kelp
(c) Euglena (d) Cyanobacteria

Light Energy Harvested by Plants &
Other Photosynthetic Autotrophs
6 CO2 + 6 H2O + light energy → C6H12O6 + 6 O2
Photosynthesis is the process by which autotrophic organisms use light
energy to make sugar and oxygen gas from carbon dioxide and water

6CO2 6H2O C6H12O6 6O2lightenergy
→→→→+ ++
Experimental evidence
• Where did the O2 come from?
– radioactive tracer = O18
6CO2 6H2OC6H12O6 6O2
lightenergy
→→→→+ ++
Experiment 1
energyenergy
6CO2 6H2OC6H12O6 6O2
lightenergy
→→→→+ ++
Experiment 2
Proved O2 came from H2O not CO2 = plants split H2O
Water is oxidized
Carbon dioxide is reduced

• Chloroplasts
absorb light
energy and
convert it to
LightReflected
light
THE COLOR OF LIGHT SEEN IS THE
COLOR NOT ABSORBED
convert it to
chemical energy
Absorbedlight
Transmittedlight
Chloroplast
Blue and red
wavelengths are
absorved

WHYWHY AREARE PLAPLANTS NTS GREGREEN?EN?
Plant Cells
have Green
Chloroplasts
The thylakoid
membrane of the
chloroplast is
impregnated with
photosynthetic
pigments (i.e.,
chlorophylls,
carotenoids).

Chloroplasts: Sites of Photosynthesis
• Photosynthesis
– Occurs in chloroplasts, organelles in certain
plants
– All green plant parts have chloroplasts and
carry out photosynthesiscarry out photosynthesis
• The leaves have the most chloroplasts
• The green color comes from chlorophyll in the
chloroplasts
• The pigments absorb light energy

• In most plants, photosynthesis occurs
primarily in the leaves, in the chloroplasts
• A chloroplast contains:
– stroma, a fluid
Photosynthesis occurs in chloroplasts
– stroma, a fluid
– grana, stacks of thylakoids
• The thylakoids contain chlorophyll
– Chlorophyll is the green pigment that captures
light for photosynthesis

The location and structure of chloroplasts
LEAF CROSS SECTION MESOPHYLL CELL
LEAF
Chloroplast
Mesophyll
CHLOROPLAST Intermembrane space
Outermembrane
Innermembrane
ThylakoidcompartmentThylakoidStroma
Granum
StromaGrana

Chloroplast Pigments• Chloroplasts contain several pigments
– Chlorophyll a
– Chlorophyll b
– Carotenoids
(carotenes and xanthophylls)
Chlorophyll a and b
CHO in chlorophyll b
Phyto tail

Different pigments absorb light differently
Violet
Blue
Red
Orange
Green
Yellow
• Is only pigment that directly participates in the light reactions
• Other pigments add energy to chlorophyll a or dissipate excessive light
energy
• Absorption of light elevates an electron to a higher energy orbital
(increased potential energy)
Chlorophyll a


• The Calvin cycle makes
sugar from carbon dioxide
Light
Chloroplast
Lightreactions
Calvincycle
NADP++++
ADP+ P
• The light reactions
convert solar energy to
chemical energy
– Produce ATP & NADPH
AN OVERVIEW OF PHOTOSYNTHESIS
sugar from carbon dioxide
– ATP generated by the light
reactions provides the energy
for sugar synthesis
– The NADPH produced by the
light reactions provides the
electrons for the reduction of
carbon dioxide to glucose
reactionscycle

• Two connected photosystems collect photons of
light and transfer the energy to chlorophyll
electrons
In the light reactions, electron transport In the light reactions, electron transport
chains generate ATP, NADPH, & Ochains generate ATP, NADPH, & O22
• The excited electrons are passed from the primary
electron acceptor to electron transport chains
– Their energy ends up in ATP and NADPH

Photosystems
• Collection of pigments and
proteins found associated
with the thylakoid
membrane that harness
the energy of an excited
electron to do work
• Captured energy is
transferred between
photosystem molecules
until it reaches the
chlorophyll molecule at the
reaction center

What Next?
• At the reaction center are
2 molecules
– Chlorophyll a
– Primary electron
acceptor
• The reaction-center • The reaction-center
chlorophyll is oxidized as
the excited electron is
removed through the
reduction of the primary
electron acceptor
• Photosystem I and II

Electron Flow• Two routes for the path of electrons stored in the primary
electron acceptors
• Both pathways
– begin with the capturing of photon energy
– utilize an electron transport chain with cytochromes for
chemiosmosis
• Noncyclic electron flow
– uses both photosystem II and I
– electrons from photosystem II are removed and replaced by
electrons donated from water
– synthesizes ATP and NADPH
– electron donation converts water into ½ O2 and 2H+
• Cyclic electron flow
– Uses photosystem I only
– electrons from photosystem I are recycled
– synthesizes ATP only

Photophosphorylation
cyclic
photophosphorylation
noncyclic
photophosphorylation

Noncyclic Photophosphorylation• Photosystem II regains electrons by splitting water, leaving
O2 gas as a by-product
Primaryelectron acceptor
Primaryelectron acceptor
Photons
PHOTOSYSTEM I
PHOTOSYSTEM II
Energy forsynthesis of
by chemiosmosis

Noncyclic Electron FlowElectrons at reaction-center
are energized
H2O split via enzyme
catalysed reaction forming 2H+,
2e-, and 1/2 O2. Electrons move
to fill orbital vacated by removed
electrons
Each excited electron is passed
along an electron transport chain
fueling the chemiosmotic
synthesis of ATP
The electrons are now lower in
energy and enters photosystem I
via plastocyanin (PC) where they
are re-energized
The electrons are then passed
to a different electron transport
system that includes the iron
containing protein ferridoxin.
The enzyme NADP+ reductase
assists in the oxidation of ferridoxin
and subsequent reduction of
NADP+ to NADPH

• The electron transport chains are arranged
with the photosystems in the thylakoid
membranes and pump H+ through that
membrane
Chemiosmosis powers ATP synthesis
in the light reactions
membrane
– The flow of H+ back through the membrane is
harnessed by ATP synthase to make ATP
– In the stroma, the H+ ions combine with NADP+
to form NADPH

• The production of ATP by chemiosmosis in
photosynthesis
Thylakoidcompartment(high H+)
Thylakoid
Light Light
Thylakoidmembrane
Stroma(low H+)
Antennamolecules
ELECTRON TRANSPORT
CHAIN
PHOTOSYSTEM II PHOTOSYSTEM I ATP SYNTHASE

Where are the photosystems found on
the thylakoid membrane?

Chemiosmosis in 2 Organelles
Both the Mitochondria and Chloroplast
generate ATP via a proton motive force
resulting from an electrochemical
imbalance across a membrane
Both utilize an electron transport chain
primarily composed of cytochromes to
pump H+ across a membrane.
Both use a similar ATP synthase
complex
Source of “fuel” for the process differs

Calvin Cycle
• Starts with CO2 and produces Glyceraldehyde 3-phosphate
• Three turns of Calvin cycle generates one molecule of product
• Three phases to the process: I) Carbon Fixation; II) Reduction of CO2; III)
Regeneration of RuBP

A molecule of CO2 is converted
from its inorganic form to an
organic molecule (fixation)
through the attachment to a 5C
sugar (ribulose bisphosphate or
RuBP).
Phase 1
Calvin Cycle
–Catalysed by the enzyme
RuBP carboxylase (Rubisco).
The formed 6C sugar
immediately cleaves into 3-
phosphoglycerate

Each 3-phosphoglycerate
molecule receives an
additional phosphate group
forming 1,3-
Bisphosphoglycerate (ATP
phosphorylation)
Phase 2
Calvin Cycle
phosphorylation)
•NADPH is oxidized and the
electrons transferred to
1,3-Bisphosphoglycerate
cleaving the molecule as it
is reduced forming
Glyceraldehyde 3-
phosphate

Phase 3
The final phase of the
cycle is to regenerate
RuBP
•Glyceraldehyde 3-
phosphate is converted
Calvin Cycle
phosphate is converted
to RuBP through a series
of reactions that involve
the phosphorylation of
the molecule by ATP

Variations Anyone?
• In hot/arid regions plants may run
short of CO2 as a result of water
conservation mechanisms
• C4 Photosynthesis
CO2 may be captured by
conversion of PEP
(Phosphoenolpyruvate) into (Phosphoenolpyruvate) into
oxaloacetate and ultimately malate
that is exported to cells where the
Calvin cycle is active
• CAM Photosynthesis
CO2 may be captured as inorganic
acids that my liberate CO2 during
times of reduced availability