hZK /E K W, ZD >/D/d U hE/d r /y · Aurobindo Pharma Limited. Unit IX Executive Summary E-2 Team...
Transcript of hZK /E K W, ZD >/D/d U hE/d r /y · Aurobindo Pharma Limited. Unit IX Executive Summary E-2 Team...

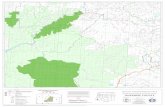
AUROBINDO PHARMA LIMITED, UNIT - IX SY.NO. 305, 369 to 371, 373, 374 AND 377,
GUNDLAMACHANOOR VILLAGE, HATNOOR MANDAL, SANGAREDDY DISTRICT, TELANGANA
EXECUTIVE SUMMARY
SUBMITTED TO TELANGANA STATE POLLUTION CONTROL BOARD,
REGIONAL OFFICE, SANGAREDDY

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-1
EXECUTIVE SUMMARY
Introduction
Among the largest ' Vertically Integrated ' pharmaceutical companies in India, Aurobindo
has robust product portfolio spread over major product areas encompassing CVS, CNS,
Anti-Retroviral, Antibiotics, Gastroenterological, Anti-Diabetics and Anti-Allergic with
approved manufacturing facilities by USFDA, UKMHRA, WHO, MCC-SA, ANVISA-
Brazil for both APIs & Formulations.
Aurobindo Pharma Limited established in 1986 with a single unit manufacturing Semi-
Synthetic Penicillin (SSP) and now a well-integrated pharma company, Aurobindo
Pharma features among the top 10 companies in India in terms of consolidated revenues.
Aurobindo exports to over 125 countries across the globe with more than 70% of its
revenues derived out of international operations.
M/s. Aurobindo Pharma Limited, Unit IX obtained Environment Clearance Vide file no.
F. No. J-11011/83/2004-IA II (I) dated 21.06.2005. The unit had renewed consent for
operation vide order no. TSPCB/ SRD/ CFO & HWA/HO/ 2019 dated 10.06.2019 valid
till 31.03.2024. It is proposed to expand the API manufacturing capacity from 34.7 TPM to
173 TPM in existing site area of 25.04 acres. The capital cost for the proposed expansion
project is Rs. 50 crores. The cost estimate form environment management is 6.12 crores
and annual recurring expenditure is 11.8 crores. The expansion involves additional
production blocks, utilities and enhancement of treatment facilities, storages and
additional equipment to enhance the capacity, at the existing site in Sy.Nos. 305, 369 to
371, 373, 374 and 377 Gundlamachanoor village, Hatnoor mandal, Sangareddy district,
Telangana. Prior environmental clearance is mandated by Ministry of Environment and
Forests, vide SO 1533, dated September 14, 2006, for synthetic organic chemicals
manufacturing activity. The terms of reference for the environmental impact assessment
studies was obtained from MoEF&CC vide letter no F.No. J-11010/198/2017-IA II (I)
dated 31.07.2017 as part of environmental clearance process. The certified compliance
letter from the regional office of MoEFCC, Bangalore is obtained vide letter no. F. No. EP
/12.1/346/AP/1135 dated 18.07.2018.

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-2
Location of the Project:
The plant site is located at Sy.Nos. 305, 369 to 371, 373, 374 and 377 Gundlamachanoor
village, Hatnoor mandal, Sangareddy district, Telangana. The site is situated at the
intersection of 170 38’53.03” (N) latitude and 780 10’ 24.11” (E) longitudes. The site is
surrounded by Narsapur – Sangareddy state highway in west direction, Covalent
laboratories Pvt. ltd., in East and south directions and Cirex Pharmaceuticals Ltd., in north
direction. The main approach road is NH 9 (Hyderabad – Mumbai) passing at a distance
of 9.5 Km in south direction. The nearest habitation from the plant site is Gundlamachnoor
village at a distance of 0.74 kms in southeast direction. The nearest town Sangareddy is at
a distance of 8.7 km in southwest direction. The nearest railway station of Patancheru is at
a distance of 16.7 km in southeast direction and the nearest airport is located at a distance
of 52 km in southeast direction. Nakkavagu stream is at a distance of 1.2 km in west
direction, flowing from south to north. Manjra river stream is at a distance of 2.25 km in
west direction, flowing from southwest to northeast. There is no reserve forest, national
park, sanctuary, biosphere reserves and critically polluted area within 10 km from the site.
Product Profile
The manufacturing capacity after expansion is presented in following tables;
Manufacturing Capacity - After Expansion S.No Name of Product Capacity
TPM Kg/Day
1 Ciprofloxacin Hydrochloride 12 400
2 Metropolol (base) 11.25 375
3 N-Isopropyl-2-methyl-2-n-propyl-3-hydroxypropyl carbamate 6 200
4 Losartan Potassium 7.5 250
5 Aminocarbinol Tartate 6.75 225
6 N-[2-Amino-4,6-dichloro-5-Pyrimidinyl] formamide 6.75 225
7 (2S,3S,5S)-2-Amino -3-Hydroxy-5-[2S-(1-Tetrahydropyrimid-2-Oxly)-3-Methyl butanoyl} Amino-1,6-Diphenylhexane, (S)-Pyroglutamic acid Salt
6.75 225
8 Trityl losartan 6 200
9 [R-(R*,S*[[2-Methyl-1-(1-oxopropoxy) propoxy] (4-phenyl butyl) phosphinyl] acetic acid Cinchonidine salt (MOPPA)
6.75 225
10 Prenyl Half Ester 6 200
11 Metaprolol Tartrate 6.75 225
12 Candesartan Methyl Ester 4.5 150
13 N-Methyl Paroxetine 3.75 125
14 N,N-Dimethyl -3-Chloropropyl Amine (Citalopram – int –A) 3 100
15 N-[2-Amino-4,6-Dichloro-5-Pyrimidinyl] Foramamide (Dichloro 3 100

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-3
Compound of Famicyclovir)
16 Iron sucrose 3 100
17 2,6-Dimethyl Phenoxy acetic acid 3.3 110
18 Lamivudine 3 100
19 Simavastatin 3 100
20 Zidovudine 3 100
21 Poly Phosphate Ester 3 100
22 4-(Dimethylamino) Butanal Diethyl Acetal 3 100
23 Cilexetil Chloride 3 100
24 O Acetyl Thio Ester 3 100
25 Bis Methyl Silyl Urea (BSU) 3 100
26 4-Amino-2-Hydroxymethyl-1-Butanol Hydrochloride 3 100
27 (2S,3S,5S)-5-amino)-2-[N-[5-Thiazolyl) Methoxy Carbonyl] Amino]-1,6-Diphenyl-3-Hydroxyhexane
2.63 87.67
28 Indinavir Sulfate 1.5 50
29 N- {[N-Methyl -N{(2-Isopropyl-4-Thiazolyl) Methyl] Amino} Carbonyl}-L-Valine
1.5 50
30 L-Valine Methyl Ester 1.5 50
31 N-(Trifluoro Acetyl)-L-Lysine (Lisinopropril) 1.5 50
32 2-Ethoxy-1-Naphthoic Acid 1.5 50
33 Sevelamer Hydrochloride 1.5 50
34 AR Modafinil 1.5 50
35 (Bromomethyl)Biphenyl Methyl Ester (Telmisartan Stage II) 1.13 37.67
36 1-Bromo-4a,5,9,10-Tetrahydro-3-Methoxy-6-Oxo-6H-Benzofuro{3a,3,2-ef}[2] Benzazepin-11-(12H)-Carboxaldehyde (Bromoformylnarnorvedine)
1.05 35
37 Rabeprazole Sodium 0.75 25
38 Methyl (2E)-3-[4-(4-Florophenyl)-6-Isopropyl-2-[N-methyl (N-Methyl sulfonyl) amino]Pyrimidine-5-yl]acrylate ([E] Pyrimidine Alkene) (substituted HydroxyPentanoic Acid
0.75 25
39 Pitavastatin Intermediate 0.75 25
40 Benzyl(2S,3aR,7aS) Octahydro-1H-Indole-2-Carboxylate Hydrochloride (BOHI HCl)
0.6 20
41 1-[4-(Pyridin-2-yl) phenyl]-5(s)-2,5-Bis-[(tert-Butyloxycarbonyl)-Amino]-4(S) Hydroxy-6-Phenyl-2-Azahexane (DIBOC Intermediate)
0.45 15
42 N-[(S)-Ethoxy Carboxyl-1-Butyl]-(S)-Alanine-(ECBA) 0.38 12.67
43 Benzyl(2S,3aS,7aS) Octahydro-1H-Indole-2-Carboxylate P-Toluenesulfonic acid (BOHI ESTER)
0.38 12.67
44 6-Hydroxy-2-(4-hydroxyphenyl) benzo[b]thiophene (Dihydroxybenzothiophene)
0.38 12.67
45 Nelfinavir Mesylate 0.3 10
46 (3S,4aS,8aS )-2-(2R)-2-{(4S)-2-(3-Hydroxy-2-methyl phenyl-4,5-dihydrooxazol-4-yl]-2-hydroxyethyl} decahyro isoquinoline-3-carboxyic acid-tert-butylamide
0.3 10
47 Trans-4-Cyclohexyl-L-Proline 0.3 10
48 3-Chloro-trans-Octahydro-1H-Quindin-2-One (Bohi) 0.3 10
49 2-(2-Ethoxy Phenoxy) Ethyl Methane Sulfonate (EPE Mesylate) 0.3 10
50 8-Methoxy Quinoline borondifluroride Chelate 0.3 10
51 N-(Methoxy Carbonyl)-(L)-TertLeucine (MOC Leucine) 0.3 10

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-4
52 3-(Methylamino)Propionitrile 0.3 10
53 N-(4-Aminobenzoyl)-β-Alanine (4ABBA) 0.15 5
54 (6S)-(-)2,6-Diamino -4,5,6,7-tertahydrobenzothiazole (Pramipexoldiamine)
0.15 5
55 (2S,4S)-FMOC-4-Cyclohexyl-Pyrrolidine-2-Carboxylate (FMOC-ChxPro-OH)
0.03 1
56 BT Hydrazine Sulfonic acid 0.02 0.67
57 Silodosin (Intermediate) 0.38 12.67
58 Fosaprepitant Dimeglumine 0.38 12.67
59 Mirabregon 0.75 25.00
60 Colesevelam 1.13 37.67
61 3-Hexyn-2-ol 0.08 2.67
62 Methoxitil 0.08 2.67
63 Rivastigimine 0.08 2.67
64 Bezafibrate 0.08 2.67
65 2-Isopentyl-2-Isopropy-1,3-Dimethoxypropane 0.38 12.67
66 Efavirenz 1.50 50
67 Tramadol HCl 3.75 125
68 Lorcaserin Hydrochloride 0.30 10
69 Teriflunomide 0.15 5
70 Apixaban 0.08 2.67
71 Canagliflozin 0.03 1
72 Atovaquone 0.75 25
73 Sodium ferric gluconate 2.25 75
74 Fluvastatin Sodium 2.25 75
75 Diprotected Rosuvastatin 2.25 75
76 Lamivudine Coupled Ester 1.13 37.67
77 Oxocompound Free Base 0.08 2.67
78 Sofosbuvir 0.08 2.67
79 Raltegravir Potasium 1.01 33.67
80 Flecanide acetate 0.38 12.67
81 Nebivilol 0.38 12.67
82 Clobazam 0.12 4
83 Nefopam HCl 0.08 2.67
84 Carbimazole 0.08 2.67
85 Ledipasvir 0.08 2.67
86 R & D Pilot Plant Trial Run Products (Bulk Drugs and Intermediates)
0.5 16.67
Total 173 5766.7

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-5
List of By-products – After Expansion S.No Name of Product Stage Name of By-Product Quantity
(Kg/day)
1 N-Isopropyl-2-methyl-2-n-propyl-3-hydroxypropyl carbamate
I Dimethylaniline HCl 182.8
Isopropylamine HCl 88
2 Losartan Potassium IV Trityl alcohol 172.8
3 Candesartan Methyl Ester I Tri n-Butyl tin Azide 109.7
4 N-Methyl Paroxetine I Triethylamine HCl 62.6
5 Lamivudine III Acetic acid 63.3
6 Simavastatin II Imidozole hydrochloride 85.8
7 Zidovudine I Trimethyl ether 101.9
8 4-Amino-2-Hydroxymethyl-1-Butanol HCl
I Sodium Diethyl Phosphate 129.2
9 Indinavir Sulfate VIII Spent Sulfuric acid 300.6
10 Deprotected Rosuvastatin II Spent Acetic Acid (20%) 2500
Manufacturing Process
Chemical synthesis produces majority of bulk drugs and intermediates currently in the
market. Chemical synthesis consists of four process steps - reaction, separation,
purification, and drying. Large volumes of solvents are used during chemical syntheses as
medium, extractions, and solvent interchanges. The manufacturing process of all the
compounds, reactions involved, material balance are presented at separate annexure of
EIA report.
Utilities
The proposed expansion requires additional steam for both process and effluent treatment
system. It is proposed to establish coal fired boiler of 1 x 12 TPH capacity in addition to
existing 1 x 12 TPH coal fired boiler, while keeping the existing 1 x 8 TPH coal fired boiler
as standby after expansion. The DG sets required for emergency power during load shut
down is estimated at 2140 kVA and accordingly 2 x 380 DG sets are proposed in addition
to exiting 2 x 500 kVA and 1 x 380 kVA DG sets.
List of Utilities
S.No Utility Permitted Proposed After Expansion
1 Coal Fired Boilers (TPH) 12 8
12 2 x 12 1 x 8*
2 DG Sets (kVA)** 2 x 500 1 x 380
2 x 380 2 x 500 3 x 380
* Shall be kept as standby **DG sets will be used during load shut down by TRANSCO

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-6
Water Requirement
Water is required for process, scrubbers, washing, cooling tower makeup, steam
generation and domestic purposes. The total water requirement after expansion increased
from 43.1 KLD to 726.4 KLD. The fresh water consumption is optimized by reusing treated
wastewater to an extent of 292.5 KLD, thus reducing fresh water consumption to 433.9
KLD during operation. The fresh water shall be drawn from ground water in addition to
reuse of treated wastewater. The water balance for daily consumption is presented in
below table.
Water Balance Purpose INPUT(KLD) OUTPUT (KLD)
Fresh Water Recycled Water Loss Effluent
Process & Washings 134.9 144.5*
Scrubber 20 20
QC and R&D 15 15
SRS System 35 35
RO/DM regeneration 20 20
Boiler 75 57.5 17.5
Cooling Tower 79 265 329 15
RO Back wash 20 20
Domestic 25 2.5 22.5
Garment Washings 5 5
Gardening 5 27.5 32.5 Gross Total 433.9 292.5 421.5 314.5
Total 726.4 736 * Effluents contains unreacted raw materials, water formed in reaction, soluble solvents, by-products etc.
Baseline Environmental Data
The baseline data was collected in the study area during March -May 2018. The baseline
data includes collection of Samples of ground water, surface water and soil, monitoring of
ambient air quality, noise levels, ecological status and meteorological parameters. The
analytical results show that the values are within the prescribed limits for air quality. The
ground water quality is observed to be above the limits for potable purpose when
compared to the prescribed standards of IS: 10500 – 2012 at few locations.
Identification and Quantification of Impacts
The impact assessment report has identified various sources of pollution and quantified
the pollution loads due to proposed project. The sources of pollution are air emissions
from utilities and process; liquid effluents from process, utilities and domestic usage; solid

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-7
wastes from process, treatment systems and utilities; and noise pollution from utilities,
and process equipment. A detailed impact assessment was made using rapid impact
assessment matrix method. The salient features are presented as follows.
Impacts on Air quality: The impacts on air quality shall be due to the emissions from,
coal fired boilers, and standby DG sets. The incremental concentrations are quantified
using ISC-AERMOD model based on ISCST3 Algorithm for proposed utilities of 2 co-
located API manufacturing units, namely; Aurobindo Pharma Limited, Unit IX and Cirex
Pharmaceuticals Ltd., located at Gundlamachanoor village, Hatnoora mandal, Sangareddy
District, Telangana. The results indicate marginal increase in ambient air quality
concentration. The predicted values for SPM, PM10, PM2.5, SO2 and NOx are 1.95, 0.77, 0.34,
2.23 and 2.35 g/m3 respectively and the maximum values are observed at a distance of
0.5 km in northwest direction, and the cumulative values of baseline air quality combined
with predicted values are found to be within the prescribed limits of National Ambient
Air Quality Standards. The mitigative and control measures of air pollution shall ensure
that the impact on air quality is local – within the site area and its surroundings. The
fugitive and diffuse emissions were quantified and a box model was used to predict air
borne concentrations, and the results indicate work room concentrations, less than
threshold limit values (TLV) for various solvents.
Impacts on Water: Water is essentially used for process and utilities and domestic
purposes. The required water is drawn from ground water/reuse of stored runoff, in the
order of 433.9 KLD in addition to reuse of treated wastewater in the order of 292.5 KLD.
No impact on water quality is expected due to discharge of effluents as zero liquid
discharge is envisaged, which ensures reuse of treated wastewater for cooling towers
makeup.
Impacts on Noise quality: The noise levels may increase due to compressors, DG set and
other activities which emit noise levels of above 85 dB (A) at a reference distance of 1m
from the source. The predicted cumulative noise levels (as calculated by the logarithmic
model without noise attenuation) ranged between 47 and 65 dB (A). The increase in noise
levels shall have impact restricted to the site area.

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-8
Impacts on Soil: The solid wastes generated from process, utilities and effluent treatment
plant may have significant negative impacts if disposed indiscriminately. The total solid
waste will be stored separately in Hazardous waste storage area. Solid waste will be sent
to cements plants for co-incineration based on calorific value or sent to TSDF. The
operational phase impacts observed to be negative, shall be neutral after mitigation due to
effective implementation of management measures in handling, storing and transferring
of solid waste, effluents and chemicals, and development of green belt.
Impacts on Flora and Fauna: There are no endangered species of flora and fauna in the
impact area. The impact on biotic environment is neutral with the effect confined mainly
to the site area.
Environmental Monitoring Programme
It is proposed to monitor Ambient Air Quality (AAQ) for PM10, PM2.5, SO2 and NOX,
work room for VOC concentrations, stack emissions of boiler and DG sets for PM, SO2
and NOx, noise levels on quarterly basis. Water, treated wastewater are monitored on
daily basis, while Soil analysis is done once in a year.
Alternatives
The proposal is for expanding the existing unit, and the land area available for expansion
is sufficient and hence there is no requirement of alternative site or additional site area.
Additional Studies - Risk Assessment
Risk assessment was conducted and the heat radiation damage distances of pool fire in the
tank farm was limited to 31 m for a heat radiation of 4 KW/m2, and the same was within
the plant premises.
Project Benefits
The proposed project will provide employment to 200 people. The proposed project will
also generate indirect employment to the locals during construction phase in the order of
100 people for a period of 18 – 24 months. The proposed expansion shall have positive
impact on socioeconomic environment due to provision of employment both direct and
indirect and proposed CER activities.

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-9
Environment Management Plan
The management plan is drawn in consultation with project proponents and technical
consultants after evaluating various mitigation and control measures to address the
impacts identified, predicted and monitored. The impacts during construction stage are
temporary and less significant, the management plan for impacts identified during
operation stage is described as follows;
Liquid Effluents
The effluent generated from the proposed expansion is mainly from process & washings,
SRS system, scrubbing, QC and R&D, utility blow downs, RO back wash and RO rejects
and domestic wastewater. The effluents from process & washings, scrubbers, SRS system,
QC and R&D wastewater and RO/DM rejects are considered as HTDS stream, while
wastewater from utility blow downs, RO back wash are considered LTDS stream. HTDS
effluents will treat in stripper followed by MEE and ATFD, and MEE and ATFD in the
case of effluents from RO/DM rejects and scrubber. All LTDS effluent along with
condensate from MEE and ATFD shall be treated in biological treatment followed by RO
system. RO Rejects are sent to MEE and permeate is used for cooling towers make up.
Salts from ATFD are disposed to TSDF. Sludge from various units of Biological treatment
are thickened in sludge handling system and sent to TSDF. Wastewater from domestic
usage and garment washings are treated in sewage treatment plant and treated
wastewater is reused for on land irrigation.
Effluent Generated and Mode of Treatment Description Quantity (KLD) Mode of Treatment
Permitted After Expansion
HTDS Effluents
Process and washings
6.4 144.5 Sent to stripper. Stripper condensate shall be disposed to cement industries for co-processing/TSDF. Stripper bottom is sent to MEE followed by AFTD. Condensate from MEE and ATFD shall be sent to biological treatment plant followed by RO. RO rejects are sent to MEE and permeate is reused in cooling towers boiler make-up.
Scrubber 20
SRS System 35
RO/DM Rejects 20
QC and R&D 1.5 15
Total - I 7.9 234.5
LTDS Effluents
Boiler Blow downs 2.8 17.5 Sent to biological treatment system followed by

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-10
Cooling Towers Blow downs
15 RO. RO permeate reused for cooling towers makeup. RO rejects are sent to MEE.
RO Back Wash 0.5 20
Total – II 3.3 52.5
Domestic 1 22.5 Sent to sewage treatment plant and treated wastewater is reused for on land irrigation to develop green belt.
Garment Washings 5
Total – III 1 27.5
Grand Total (I+II+III)
12.2 314.5
Air Pollution
The sources of air pollution are proposed 1 x 12 TPH and existing 1 x 12 TPH and 1 x 8
TPH coal fired boilers and backup DG sets of 2 x 380 kVA in addition to existing 2 x 500
and 1 x 380 kVA capacity. Existing 1 x 12 TPH coal fired boiler will be kept as standby
after expansion. The proposed air pollution control equipment for 1 x 12 TPH coal fired
boiler is bag filter. DG sets shall be provided with effective stack height based on the
CPCB formula.
The process emissions contain ammonia, carbon dioxide, carbon monoxide, hydrogen,
nitrogen, oxygen, hydrogen bromide, hydrogen chloride and sulfur dioxide. Ammonia,
hydrogen chloride, hydrogen bromide, and Sulphur dioxide are sent to scrubber in series.
The resultant solutions after scrubbing are sent to ETP. Carbon dioxide, carbon monoxide,
Nitrogen, and Oxygen are let out into atmosphere following a standard operating
procedure, while Hydrogen gas is let out into atmosphere through a water column.
Emissions are also released from various operations of manufacturing like centrifuge,
drying, distillation, extraction etc. These emissions mainly contain volatile contents of the
material used for processing. It is proposed to provide vent condensers in series to
reactors, distillation columns, driers and centrifuge etc. to mitigate VOC emissions release.
Other vents are connected to common headers and scrubbers.
Solvent Use and Recycle
Solvents are used for extraction of products and as reaction medium. Used solvents are
recovered by distillation for reuse. Residues from distillation columns and mixed solvents
shall be sent to TSDF for incineration or cement plants for co-incineration. If any of the

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-11
distilled spent solvents are not reused due to statutory reasons the same shall be sold to
end users.
Solid Waste
Solid wastes are generated from process, solvent distillation, effluent treatment system,
DG sets and boilers. Stripper distillate, process residue and solvent residue are sent to
cement plants for co-incineration based on acceptability as the same contain significant
calorific value and are predominantly organic in nature. If these wastes are not suitable
for co-incineration, the same are sent to TSDF facility. The evaporation salts from ATFD,
and sludge from ETP are sent to TSDF for landfill. Waste oil and used batteries from the
DG sets are sent to authorized recyclers. Other solid wastes expected from the unit are
containers, empty drums which are returned to the product seller or sold to authorized
buyers after detoxification. Coal ash from boiler is sold to brick manufacturers.
Noise Pollution
Noise is anticipated from compressors and DG sets. DG sets shall be provided with
acoustic enclosure. Engineering controls like acoustic enclosures, barriers, shields, and
anti-vibrating pads will be provided to ensure reduction of noise levels and vibration.
Employees working in noise generating areas shall be provided with appropriate
personnel protective equipment.
Occupational Safety and Health
Direct exposure to chemicals or its raw materials may affect health of employees. Direct
exposure to hazardous materials is eliminated by providing closed handling facilities.
Personal Protective Equipment (PPE)i.e., hand gloves, safety goggles, safety shoes, safety
helmets, respiratory masks etc. are provided to all the employees working in the plant.
Company has a policy of providing PPEs to all personnel including contract workers.
Periodic medical checkup in addition to checkup during recruitment is adopted to
monitor health status of employees.
Prevention, maintenance and operation of Environment Control Systems
The pollution control equipment, and the effluent treatment system is monitored
periodically to estimate their efficiency and performance potential as part of adoptive

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-12
management. Proactive maintenance and monitoring program for all equipment and
machinery is adopted to identify the problems/under performance of the equipment.
Necessary measures will be adopted to rectify the identified problems/defects. The
management agrees that the results of monitoring will be reviewed periodically to adopt
new measures if necessary, for efficient pollution control.
Transport systems
All the raw materials and finished products are transported by road. Dedicated parking
facility is provided for transport vehicles. There will be 10 addition truck trip per day to
the factory for transporting raw materials and products due to proposed expansion.
Traffic signs are placed in the battery limit. The drivers of vehicles will be provided with
TREM cards of chemicals and materials to be transported, and will be explained the
measure to be adopted during various emergencies
Reduce, Recycle and Reuse
A number of measures are proposed to achieve high yields and reduce generation of
wastes. It shall be endeavor of the R&D team to improve yields through constant research
and development activities. The solvents shall be recycled for reuse in the process after
distillation. Mother liquors from the first crop shall be reused for process. Treated
wastewater from ZLD system is reused for cooling towers make-up. Treated wastewater
from sewage treatment plant is reused for greenbelt development. It is also proposed to
explore recovery of various salts from MEE salts, and from process effluents to reduce
effluent loads, and quantity of solid waste. Proposed rainwater harvesting system shall
have a storage capacity of 1040 KL, and the stored run off will be used for plant operation
for approximately 28 days.
Green Belt Development
The management developed green belt in a total area of 8.27 acres and proposed to
increase density to enhance environmental quality through mitigation of fugitive
emissions, attenuation of noise levels, balancing eco-environment, prevention of soil
erosion, and creation of aesthetic environment

Aurobindo Pharma Limited. Unit IX Executive Summary
Team Labs and consultants E-13
Corporate Environment Responsibility
The ministry of Environment, Forest and Climate Change issued an office memorandum
mandating corporate environment responsibility expenditure contingent on the capital
cost of the project, in addition to the expenditure on environmental management. The
proposed project is a brown field project, the management proposed to spent 1% of the
proposed expansion capital cost i.e., Rs. 50 lakhs towards corporate environment
responsibility. Activities are identified to be implemented by the project proponent in
consultation with local public representatives and revenue authorities.
Environment Management Department
The Environment Management Cell of the project is headed by the President, Senior vice
president, vice president operations, DGM- EHS, followed by manager, assistant manager,
Dy. Manager and technicians/fitters.


![hE/s Z^/d [ '>/ ^dh / / dZ/ ^d - arts.units.it · Kdd X D hZK >h^^/ EEK D/ K î ì í ð l î ì í ñ , Z/ ^^hEdK ] } v } ] ¶ ] í ...](https://static.fdocuments.in/doc/165x107/5c6a77bb09d3f20f7f8cb050/hes-zd-dh-dz-d-artsunitsit-kdd-x-d-hzk-h-eek-d-k-i.jpg)
![P 4E Q 9EQ - Wing On Travel....EqL hjo>Lqk^ hj+G?hzk^ WHL \][^ZKNIJKILJKP WTXGXGXURGKLO LXSEhXpxzxy yyyx bkvQ/hOslC=l9p BiNjTjNfHb{cIf?8tTR5111a|U?V+TROfbKQf H?C:Gkg/{biN=+?}CeSCDQr1HKnI?c]OSCD](https://static.fdocuments.in/doc/165x107/613ce4344c23507cb635abf9/p-4e-q-9eq-wing-on-travel-eql-hjolqk-hjghzk-whl-zknijkiljkp-wtxgxgxurgklo.jpg)






![V.62I.sdNnywfm. · Magazines Classroom Posters Projectable Magazine Bgm^kZ\mbo^Pabm^[hZk]E^llhg M^Z\a^kl@nb]^ Bgm^kZ\mbo^>]bmbhg (additional subscription required) Nationl GegrphcporeYn](https://static.fdocuments.in/doc/165x107/603bbb58dcc63828e230e663/v62i-magazines-classroom-posters-projectable-magazine-bgmkzmbopabmhzkellhg.jpg)