Glycogen metabolism ppt
-
Upload
phari-dajangju -
Category
Health & Medicine
-
view
127 -
download
9
Transcript of Glycogen metabolism ppt

GLYCOGEN METABOLISM
Dr. Phari DajangjuJNMC, AMU

IntroductionStorage form of GlucosePolymer held by glycosidic linkagesStored in Liver and MusclesSource of fuel for brain

StructureIn cytoplasm as granulesinner linear chainOuter branched Central Glycogenin protein


Function
Storage formEnergy for brainReserve fuel for muscle contraction

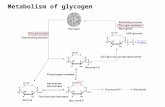
GLYCOGENOLYSIS


Enzymes of Glycogenolysis
PhosphorylaseBifunctional-Debranching
enzyme
Phospho- glucomutase
Glucose-6-Phosphatase


Step 1:Depolymerization (Release of Glu-1-P)
Enzyme:PhosphorylaseCo Enzyme:Pyridoxal phosphate Orthophosphate splits between C1 and C4 of
adjacent glucoseStops 4 units before branching point


Step 2: Remodelling & Debranching
Bifunctional enzymeTransferaseAlpha-1,6-glucosidaseRelease of free glucose residueLinear Glycogen

Step 3:Conversion of Glu-1-P to Glu-6-
PEnzyme: PhosphoglucomutaseThe active site of Mutase has
phosphorylated serineGlu-1,6-bisP intermediate formed


Step 4: Fate of Glu-6-P

Reaction catalysed by Phosphotase

Regulation of Glycogenolysis
Regulation of enzyme Glycogen Phosphorylase
Hormonal action of Glucagon/Epinephrine
Mechanism is similar in Liver and MuscleEpinephrine in Muscle

Glycogen Phosphorylase
2 Isozyme Glycogen Phosphorylase a and bEach exists in R and T stateDimer with Serine residue Phosphorylase kinase promotes
Phosphorylase b—>a


Phosphorylase kinase
Structure-4 subunits, 4 of eachActivation-1.Phosphorylation of beta -partial activation2.Calcium attaches to delta-full activation

Regulation of glucose breakdown
in musclesLow energy state-1.Epinephrine through Phosphorylase
kinase Phosphorylase b—->a2.AMP from degradation of ATP as allosteric
activator of Phosphorylase b from T—>R stateResting state-1.ATP shift Phophorylase b R—>T state2.High Glu-6-P favour T state

Regulation of glycogen breakdown
in Liver
Liver Phosphorylase sensitive to free GluFree Glu binds to active site, causes
covalent modification R—>T stateLow Glu..activate Phosphorylase aInsulin promotes uptake of Glu and
phosphorylation to Glu-6-P

Glucagon signal pathway
Alpha cells of pancreas secrete GlucagonEpinephrine by adrenal medulla Epinephrine bind to alpha adrenergic
receptorSimilar cascade of reactions


NAMRATA CHHABRA, M.D.
Role of Insulin in Glycogen degradation
Both Phosphorylase and Phosphorylase kinase are dephosphorylated and inactivated by protein phosphatase.
Protein phosphatase is stimulated by Insulin, Therefore Insulin by inhibiting the
activation of these enzymes inhibits the overall process of glycogenolysis.
14-Jan-17

GLYCOGENESIS

GLYCOGENESIS
Mainly in Liver and MusclesLiver Glycogen functions as storage and export
of glucose for maintaining levelMuscle glycogen as readily available source of
glucose


Phases of Glycogenesis
Activation Initiation
ElongationGlycogen Branching


ACTIVATION OF GLUCOSE

Step 1:Phosphorylation of Glucose

Step 2:Conversion of Gluc-6-P to Gluc-1-P

Enzyme UDP Glu Pyrophosphorylase
Step-3- Conversion of Glucose-1-P to UDP-Glucose

INITIATION

InitiationGlycosidic bond between the C1 of the glucose moiety of UDP-glucose and the hydroxyl oxygen of a tyrosine side-chain of Glycogenin.
Glucosyl Transferase
UDP is released as a product.
Each subunit of glycogenin catalyzes the addition of eight glucose units to its partner in the glycogenin dimer.
At this point, glycogen synthase takes over to extend the glycogen molecule.



ElongationCatalyzed by Glycogen SynthaseNew glucosyl units added to nonreducing
terminal residues of glycogen. Formation of α-1,4-glycosidic linkage. .

Incorporation of UDP-Glucose into-glycogen

Glycogen Branching
Amylo-(1,4)—>(1,6)-transglucosidaseIncreases the solubilityIncreases terminal residue which are
sites for action of Synthase and Phosphorylase

Regulation of glycogen synthesis
Glycogen SynthaseGS 1 in muscles, GS2 in LiverPhosphorylation inactivates a-->bGlu-6-P is allosteric effector


NAMRATA CHHABRA, M.D.
Role of Insulin in Glycogenesis
Promotes Glycogenesis
Causes activation of Phosphoprotein Phosphatase resulting dephosphorylation of Glycogen Synthase
In liver insulin increases the activity of phosphodiesterase, promoting hydrolysis of cAMP
Insulin thus antagonizes effects of the cAMP cascade induced by glucagon & epinephrine.
14-Jan-17

REGULATION

NAMRATA CHHABRA, M.D.
General mechanisms involved in the regulation of enzyme activities
Regulation of enzyme activity
Induction/Repression
Covalent modification
Allosteric modification
Substrate/product
concentration14-Jan-17

NAMRATA CHHABRA, M.D.
Key enzymes involved in the regulation of glycogen
metabolism
Glycogen synthase- For Glycogenesis
Glycogen Phosphorylase
Both these enzymes are reciprocally regulated.

NAMRATA CHHABRA, M.D.
Reciprocal regulation of Enzymes
Glycogen Synthase & Phosphorylase activity are reciprocally regulated
At the same time as phosphorylase is activated by a rise in concentration of cAMP (via phosphorylase kinase), glycogen synthase is converted to the inactive form.
Thus, inhibition of glycogenolysis enhances net glycogenesis, and inhibition of glycogenesis enhances net glycogenolysis
Both processes do not occur at the same time.
14-Jan-17


NAMRATA CHHABRA, M.D.
Substrate concentration and allosteric modification
Substrate Glucose-6-PGlycogen Synthase is allosterically
activated by glucose-6-P. High blood glucose concentration leads to
elevated intracellular glucose-6-P. When glycolytic pathway is saturated,
excess glucose-6-P activates Glycogen synthase14-Jan-17

NAMRATA CHHABRA, M.D.
Covalent modification- General concepts Reversible phosphorylation and dephosphorylation
Hormone mediated cAMP mediated cascade
Phosphorylation is mediated by Protein kinase A
Dephosphorylation is carried out by Phosphatase
Insulin causes dephosphorylation by stimulating Phosphatase and Phosphodiesterase (enzyme that breaks down cAMP)
Glucagon causes phosphorylation by stimulating Protein kinase A
14-Jan-17

NAMRATA CHHABRA, M.D.
Regulation of glycogen synthase by covalent
modificationGlycogen synthase exists in both
phosphorylated or dephosphorylated statesActive glycogen synthase a is
dephosphorylated and inactive glycogen synthase b is phosphorylated
14-Jan-17

NAMRATA CHHABRA, M.D.
Covalent modification of glycogen synthase
Glycogen synthase
a
Glycogen synthaseb
Phosphatase Protein kinase A
ATPPi
H2O ADP
Active
Inactive14-Jan-17
p

NAMRATA CHHABRA, M.D.
Regulation of Glycogenolysis by Covalent Modification The cAMP cascade results in phosphorylation of a serine hydroxyl of Glycogen Phosphorylase, which promotes transition to the active state. The phosphorylated enzyme is less sensitive to allosteric inhibitors. Thus, even if cellular ATP and glucose-6-phosphate are high, Phosphorylase will be active.
14-Jan-17

NAMRATA CHHABRA, M.D.
Role of cAMP In Glycogen degradationcAMP activates cAMP dependent Protein Kinase
Phosphorylation of inactive phosphorylase kinase b to a
Phosphorylation of inactive Glycogen Phosphorylase b to a
In the liver, cAMP is formed in response to glucagon, muscle is insensitive to glucagon.
In muscle, increased cAMP formation is the action of norepinephrine
14-Jan-17

NAMRATA CHHABRA, M.D.
Role of calcium in muscle degradation
Phosphorylase Kinase is partly activated by binding of Ca++
Further activation is brought by phosphorylation.
Phosphorylase KinaseDephosphorylated
(inactive)
Phosphorylase kinase- Ca++
Partly active
Phosphorylase kinase- Ca++
Phosphorylated- active
Ca++
ATP
14-Jan-17

THANK YOU


![9[1]. Glycogen Metabolism](https://static.fdocuments.in/doc/165x107/577d346f1a28ab3a6b8e0213/91-glycogen-metabolism.jpg)