Gaps in the Treatment of Psoriasis and PsA for Systemic ... in the Treatment of Psoriasis and PsA...
Transcript of Gaps in the Treatment of Psoriasis and PsA for Systemic ... in the Treatment of Psoriasis and PsA...
Gaps in the Treatment of
Psoriasis and PsA for Systemic
Therapy
Mark Lebwohl, MD Sol and Clara Kest Professor
And Chairman
Department of Dermatology
Icahn School of Medicine at Mount Sinai
Mark Lebwohl is an employee of Mount Sinai which receives research
funds from: Abbvie, Amgen, Boehringer Ingelheim, Celgene, Eli Lilly,
Janssen / Johnson & Johnson, Kadmon, Medimmune/Astra Zeneca,
Novartis, Pfizer, Valeant and ViDac.
Dr. Lebwohl is also a consultant for Allergan, Aqua Leo-pharma,and
Promius.
Disclosure
Research gaps in psoriasis: opportunities for future studies
Ryan C, Korman NJ, Gelfand JM, Lim HW, Elmets CA, Feldman SR, Gottlieb AB, Koo JY, Lebwohl M, Leonardi CL, Van Voorhees AS, Bhushan R, Menter A.
J Am Acad Dermatol. 2014;70:146-67
Psoriasis outcome measures: a report from the GRAPPA 2012 annual meeting
Gottlieb AB, Armstrong AW.
J Rheumatol. 2013;40:1428-33
Research Gaps
• Can we predict who’ll develop PsA and can we prevent it?
• Can we predict who’ll develop other comorbidities (cardiac, renal, etc) and can we prevent them?
• Can we predict who’ll respond to which therapy?
Patient perspectives in the management of
psoriasis: Results from the population-
based Multinational Assessment of
Psoriasis and Psoriatic Arthritis Survey.
Lebwohl MG, Bachelez H, Barker J, Girolomoni
G, Kavanaugh A, Langley RG, Paul CF, Puig L,
Reich K, van de Kerkhof PC.
J Am Acad Dermatol. 2014 Feb 24. [Epub ahead
of print]
• 139,948 households were screened and
3426 patients
• prevalence of psoriasis/PsA ranged from
1.4% to 3.3%
• 79% had psoriasis alone and 21% had
PsA
• 27% (psoriasis) and 53% (PsA ± psoriasis)
of patients rated it as severe
• 45% had not seen a physician in a year;
Psoriasis and Psoriatic Arthritis: Timing of
Onset
Psoriasis 1st
Arthritis 1st
concurrent
72%
Psoriasis 1st
21%
Arthritis 1st
7% concurrent
Subclinical Joint Involvement in Patients With Psoriasis
• Joint structural damage can occur before the appearance of clinical symptoms of PsA
• In a study by Offidani and colleagues, which used MRI rather than conventional radiography to assess joint involvement, 68% of patients with psoriasis were found to have 1 or more arthritic signs
– Appeared before patients experienced clinically evident joint symptoms
– Joint damage was only detected by X-ray imaging in 32% of these patients
Source: Offidani A, et al. Acta Derm Venereol (Stockh). 1998;78:463-465.
Biannual radiographic assessments of hands and feet
in a three-year prospective followup of patients with early
rheumatoid arthritis.
van der Heijde DM, et al
Arthritis Rheum. 1992 Jan;35(1):26-34.
Sharp score or van der Heijde
score measures joint damage
on x-ray: narrowing and erosions
12†
**
Etanercept for Psoriatic Arthritis: Radiographic Improvement
*X-rays of hands and wrists (includes DIP joints); **P = .0001 vs placebo (stratified rank test). †Start of OLE.
Source: Mease P, et al. Arth Rheum. 2004;50:2264-2272.
Lebwohl M, et al. Presented at: 63rd Annual Meeting of AAD; February 18-22, 2005; New Orleans, La. (Abstract P2753).
Placebo (n=104)
Etanercept
25 mg biw (n=101)
OLE completers
originally randomized
to etanercept (n=71)
OLE completers
originally randomized
to placebo (n=70) -0.6
-0.4
-0.2
0
0.2
0.4
0.6
0.8
1
1.2
6 18 24
Study Month
Me
an
Ch
an
ge
in
mT
SS
*
Fro
m B
ase
lin
e
Adalimumab for Psoriatic Arthritis: Radiographic Results
Weeks
24
N Baseline 24 Wk Mean Change 48 Wk Mean Change
Placebo 152 21.8 0.9 1.0
Adalimumab 144 23.7 – 0.1* 0.1
*P≤.001 vs placebo for ranked ANCOVA.
Sources: Mease P, et al. Arth Rheum. 2005;52:3279-3289.
Mease P, et al. Presented at: EULAR Annual Meeting; June 8-11, 2005; Vienna, Austria.
Me
an
Ch
an
ge
in
mT
SS
*
Mean Change in mTSS Through Week 48
48
−0.5
0
0.5
1.0
1.5 Placebo Adalimumab
Total vdH-S Score – Mean Change from Baseline at Week 24*
*Median Change in
both groups was 0.0
-1.0
-0.5
0.0
0.5
1.0
Placebo (n=100) Infliximab 5 mg/kg (n=100)
To
tal vd
H-S
Sco
re
p<0.001
-0.70
0.82
Van der Heijde D, et al. Ann Rheum Dis. 2005;64 (Supplement III):109.
Treatment of psoriatic arthritis with
tumor necrosis factor inhibitors: longer-
term outcomes including enthesitis and
dactylitis with golimumab treatment in
the Long term Extension of a
Randomized, Placebo-controlled Study
(GO-REVEAL).
Kavanaugh A, Mease P.
J Rheumatol Suppl. 2012 Jul;89:90-3.
Effect of certolizumab pegol on signs
and symptoms in patients with
psoriatic arthritis: 24-week results of a
Phase 3 double-blind randomised
placebo-controlled study (RAPID-PsA)
Mease PJ, Fleischmann R, Deodhar AA,
Wollenhaupt J, Khraishi M, Kielar D,
Woltering F, Stach C, Hoepken B, Arledge
T, van der Heijde D.
Ann Rheum Dis. 2014;73:48-55.
*
PALACE 1: Apremilast in PsA (Phase 3)
• Apremilast, oral phosphodiesterase 4 (PDE4) inhibitor
• RDBPC trial stratified for DMARD use, N=489, 1:1:1 randomization
• Major adverse events diarrhea and nausea, resolve over time
Kavanaugh A et al, ACR 2012, Washington, #L13
Apremilast 30mg BID Apremilast 20mg BID
ACR20 ACR70
45
% a
ch
ievin
g r
esp
on
se
Placebo
*P<0.05; §P<0.0001
§
ACR50
Week 16 Week 24
ACR20
§
0.0
0.2
0.4
0.6
0.8
1.0
1.2
1.4
Week 0 Week 24 Week 52
Placebo (n=310) Placebo→45 mg at Wk 24 (n=269)*
UST 45 mg (n=308) UST 90 mg (n=309)
PSUMMIT I and II
Change From Baseline in Modified Total vdhS Score Over Time (ITT)
*Patients who did not receive UST are excluded
Mean
Ch
an
ge f
rom
Baselin
e
USTEKINUMAB
FUTURE 1: Radiographic progression in PsA patients stratified by MTX use
Gottlieb AB, et al. EADV 2015, P0348 Sponsored by Novartis Pharma AG
0.08 0.04 0.13
0.57 0.57 0.58
0
0.2
0.4
0.6
0.8
1
Overallpopulation
MTX: Yes MTX: No
Me
an
ch
an
ge
in
vd
H-m
TS
S
Pooled SKB doses PBO
Baseline to Week 24 (full analysis set)
366 179 216
114 150 65 344 206
98
138 54
*P<0.05 vs PBO
Change in mTSS >0.5 considered progression of radiographic disease
* *
SECUKINUMAB
Ixekizumab, an interleukin-17A specific
monoclonal antibody, for the treatment of
biologic-naive patients with active psoriatic
arthritis: results from the 24-week randomised,
double-blind, placebo-controlled and active
(adalimumab)-controlled period of the phase
III trial SPIRIT-P1.
Mease PJ, et al
Ann Rheum Dis. 2017;76:79-87.
IXEKIZUMAB
Ixekizumab, an interleukin-17A specific monoclonal antibody, for the treatment of biologic-naive patients with active psoriatic arthritis: results from the 24-week randomised, double-blind, placebo-controlled and active
(adalimumab)-controlled period of the phase III trial SPIRIT-P1. Mease PJ, et al; Ann Rheum Dis. 2017;76:79-87.
IXEKIZUMAB
Brodalumab Phase 2 PsA study: Clinical response and improvement in psoriasis in subjects with PsA
Mease P, et al. AAD 2014, P7605
ACR20 response rate at Week 24
PBO (n=55) BRO 140 mg q2w (n=57) BRO 280 mg q2w (n=56)
Indicates time point at which all subjects began receiving BRO 280 mg q2w
80
20
0
Pa
tie
nts
(%
± S
E)
2 Week
60
40
4 8 12 16 24
43.5
51.1
64.4
*
*
*
* †
†
ACR20 Open label
2 Week
4 8 12 16 24
19.6
32.7
33.3
*
*
ACR50 Open label
Pa
tie
nts
(%
± S
E)
Number of subjects
PBO 54 52 51 52 49 46
140 mg 56 56 51 53 51 47
280 mg 50 55 53 50 49 45
Number of subjects
PBO 54 53 51 52 50 46
140 mg 56 56 53 53 51 49
280 mg 50 55 53 51 50 45
80
20
0
60
40
BRODALUMAB
Research Gaps
• Can we predict who’ll develop PsA and can we prevent it?
• Can we predict who’ll develop other comorbidities (cardiac, renal, etc) and can we prevent them?
• Can we predict who’ll respond to which therapy?
Risk of myocardial infarction in patients with
psoriasis.
Gelfand JM, Neimann AL, Shin DB, Wang X,
Margolis DJ, Troxel AB.
JAMA 2006;296:1735-41
Does treatment of psoriasis reduce the risk of cardiovascular disease?
Churton S, Brown L, Shin TM, Korman NJ.
Drugs. 2014;74:169-82
Reduction in the incidence of
myocardial infarction in patients with
rheumatoid arthritis who respond to anti-
tumor necrosis factor alpha therapy:
results from the British Society for
Rheumatology Biologics Register.
Dixon WG, Watson KD, Lunt M, Hyrich KL; British Society for Rheumatology Biologics Register Control Centre Consortium, Silman AJ, Symmons DP; British Society for Rheumatology Biologics Register. Arthritis Rheum. 2007;56(9):2905-12.
Association between tumor necrosis
factor inhibitor therapy and
myocardial infarction risk in patients
with psoriasis.
Wu JJ, Poon KY, Channual JC, Shen AY
Arch Dermatol. 2012;148:1244-50
Risk of moderate to advanced kidney disease in patients with psoriasis: population based cohort study.
Wan J, Wang S, Haynes K, Denburg MR, Shin DB, Gelfand JM.
BMJ. 2013;347:f5961
Research Gaps
• Can we predict who’ll develop PsA and can we prevent it?
• Can we predict who’ll develop other comorbidities (cardiac, renal, etc) and can we prevent them?
• Can we predict who’ll respond to which therapy?
Cost of Biologics: Year 1
• Etanercept: ~$79,623
• Adalimumab: ~ $69,670
• Ustekinumab: ~$52,525-105,015
• Infliximab: ~$22,995-80,482 (50-100 kg q4-
8w. at 5mg/kg)
• Secukinumab: ~$75,007
• Ixekizumab:~$82,891
• Brodalumab:~$31,395
• Guselkumab: ~$71,237
www.goodrx.com 1/21/18
0
10
20
30
40
50
60
70
80
90
100
0 4 8 12 16 20 24 28 32 36 40 44 48 52
ERASURE study results: Secukinumab rapidly improved plaque Ps, and sustained high responses through 52 weeks
Elewski B, et al. EADV 2013: P1501. Sponsored by Novartis Pharma AG
Patients
(%
)
PASI 75 Response
Weeks
* *
*p<0.0001 vs placebo at Wk 12. Grey arrows indicate peak response
aIGA score of 0 (clear) or 1 (almost clear) and an improvement of at least 2 points on the IGA scale compared with baseline; bOne subject did not sign informed consent before starting study procedures and was excluded from analyses
SEK 150 mg (n=245) SEK 300 mg (n=245) Placebo (n=247)b
ERASURE
87.8% SECUKINUMAB
ERASURE study results: Secukinumab rapidly improved plaque Ps, and sustained high responses through 52 weeks
Elewski B, et al. EADV 2013: P1501. Sponsored by Novartis Pharma AG
0
10
20
30
40
50
60
70
80
90
100
0 4 8 12 16 20 24 28 32 36 40 44 48 52
0
10
20
30
40
50
60
70
80
90
100
0 4 8 12 16 20 24 28 32 36 40 44 48 52
PASI 100 Response
*
*
*
*
SEK 150 mg (n=245) SEK 300 mg (n=245) Placebo (n=247)b
*p<0.0001 vs placebo at Wk 12
aIGA score of 0 (clear) or 1 (almost clear) and an improvement of at least 2 points on the IGA scale compared with baseline; bOne subject did not sign informed consent before starting study procedures and was excluded from analyses
PASI 90 Response
Weeks
Patients
(%
)
Weeks
ERASURE
PASI 75 Response Rate (NRI) by Week in the Induction Phase
34
Data on file, Amgen.
140 mg
Q2W Ustekinumab
Placeb
o Weight-Based 210 mg Q2W
0%
80
%
60
%
40
%
20
%
12 1 2 4 6 8 10
Visit Week
8.09%
86.3%
77.0%
66.6% 70.0%
Per
cen
t o
f R
esp
on
der
s
100% AMAGINE-2
BRODALUMAB
PASI 90 Response Rate (NRI) by Week in the Induction Phase
35
Data on file, Amgen.
140 mg
Q2W Ustekinumab
Placeb
o Weight-Based 210 mg Q2W
0%
60
%
40
%
20
%
12 1 2 4 6 8 10
Visit Week
Per
cen
t o
f R
esp
on
der
s
3.24%
70.3%
58.7%
49.3% 47.0%
80% AMAGINE-2
BRODALUMAB
50% AMAGINE-2
PASI 100 Response Rate (NRI) by Week in the Induction Phase
36
Data on file, Amgen.
140 mg
Q2W Ustekinumab
Placeb
o Weight-Based 210 mg Q2W
0%
30
%
20
%
10
%
12 1 2 4 6 8 10
Visit Week
Per
cen
t o
f R
esp
on
der
s
0.65%
44.4%
33.6%
25.7%
21.7%
40
%
BRODALUMAB
PASI 50 at Wk 4 optimal for predicting PASI 75 at Wk 12
• Good overall sensitivity (83%), specificity (87%), positive predictive value (90%),
and negative predictive value (77%)
• Combining results for 75 and 150 mg groups showed similar results
• PASI 50 responders at Wk 4 were called early responders
Predictability for PASI 75 at Wk 12 at different percentages of
PASI improvement at Wk 4 (combined groups)
Zhu B, et al. EADV 2012: P952
Sensitiv
ity / S
pecific
ity
PASI improvement at Week 4
10 20 30 40 50 60 70 90 100 0.0
0.4
0.6
0.8
0.2
1.0
80 -10 -20 0
*Optimal threshold at PASI48;
PASI 50 selected for practicality Optimal
threshold*
Sensitivity
Specificity
IXEKIZUMAB
Early responders had significantly higher improvement in PASI 75 and 100 than non-responders
*p<0.05; **p<0.01; ***p<0.001 vs
non-responder group
Zhu B, et al. EADV 2012: P952
Weeks
% P
atients
Weeks
*** *** ***
*** **
*
PASI 75 PASI 10S0
IXEKIZUMAB
Week 16 Partial Responders (50 ≤ PASI < 75) and Nonresponders (25 ≤ PASI < 50) That Achieved PASI 50, 75, or 90 at Week 28
UST Combined*
50 ≤ PASI < 75 at Week 16 25 ≤ PASI < 50 at Week 16
Patients at Week 16, n 98 48
Achieve PASI 50 at Week 28 98% 71%
Achieve PASI 75 at Week 28 52% 13%
Achieve PASI 90 at Week 28 13% 2%
*Combined data for PHOENIX 1 and 2 patients ≤100kg
receiving 45 mg UST and >100kg receiving 90 mg UST. Sobell J, et al. Poster Presentation at FCD 2010.
USTEKINUMAB
Successful treatment of recalcitrant
palmoplantar psoriasis with
etanercept. Weinberg JM.
Cutis. 2003;72:396-8.
Successful treatment of hand and foot psoriasis
with infliximab.
Di Lernia V, Guareschi E.
Dermatol Online J. 2010 ;16:8.
Severe psoriasis pustulosa palmaris et plantaris
(Barber-Königsbeck) treated successfully with
soluble tumour necrosis factor receptor fusion
protein (etanercept).
Kasche A, et al
J Eur Acad Dermatol Venereol.2007;21:255-7.
Safety and efficacy of Adalimumab in
the treatment of moderate to severe
palmo-plantar psoriasis: an open label
study.
Clin Ter. 2012;163(2):e61-6.
Richetta AG, et al
4/11→clear
5/11→50% improvement
Increased expression of IL-17A and limited
involvement of IL-23 in patients with palmo-plantar
(PP) pustular psoriasis of PP pustulosis; results from a
randomized controlled trial. Bissonnette R et al.
JEADV 2013 DOI: 10.1111/jdv.12272.
• UST 45mg doesn’t work
Investigator-initiated, open-label trial of
ustekinumab for the treatment of moderate-to-
severe palmoplantar psoriasis.
J Dermatolog Treat. 2012 May 8. [Epub ahead of print]
Au SC, Goldminz AM, Kim N, Dumont N, Michelon M, Volf E,
Hession M, Lizzul PF, Andrews ID, Kerensky T, Wang A, Yaniv S,
Gottlieb AB.
7/20 → clear (90 mg:6/9; 45mg:1/11)
12/20 → >2point PGA approval
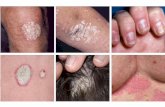
An investigator-initiated, open-label study evaluating the efficacy and safety of UST in patients with moderate-to- severe palmar/plantar psoriasis
• 24 subjects with palmar/plantar psoriasis with PGA ≥3 treated with FDA-approved dose of UST using weight-based dosing
• Report of 20/24 subjects, 11 in 45-mg dose, 9 in 90-mg dose. Mean weight of subjects not reported
Shimrat Y, et al. AAD 2012: P4733; Study sponsored by Centocor
Apremilast, an oral phosphodiesterase-4 inhibitor, in
the treatment of palmoplantar psoriasis: Results of a
pooled analysis from phase II PSOR-005 and phase
III Efficacy and Safety Trial Evaluating the Effects
of Apremilast in Psoriasis (ESTEEM) clinical trials
in patients with moderate to severe psoriasis.
Bissonnette R, et al.
J Am Acad Dermatol. 2016 Mar 24. pii: S0190-
9622(16)01400-6. doi: 10.1016/j.jaad.2016.02.1164.
[Epub ahead of print]
PPPGA 0 or 1 Achievement at Week 16 in Patients
With Baseline PPPGA ≥3
20.0
30.8 31.3
66.7
38.6
65.4
0
10
20
30
40
50
60
70
80*
ESTEEM 2
Pa
tien
ts A
ch
ievin
g P
PP
GA
0 o
r 1
(%
)
ESTEEM 1 *P<0.05 vs. placebo.
Includes patients with Palmoplantar Psoriasis Physician Global Assessment (PPPGA) ≥3, indicating moderate or severe
palmoplantar psoriasis, at baseline; all data are last observation carried forward. n/m=number of patients with response/number
of patients with sufficient data for evaluation.
*
PSOR-005
6/
9
n/m = 8/26 22/57 5/16 17/2
6
2/10
Placebo
Apremilast 30 mg BID
Clinical and patient-reported improvements of hand and/or foot psoriasis with ADA 40 mg qow: Subanalysis of REACH
• Subanalysis of REACH study looking at elements of erythema, scaling, induration and fissuring (ESIF) score along with DLQI
0102030405060708090
100
Week 16 Week 28% P
ati
en
ts A
ch
iev
ing
Ch
an
ge
fro
m
Ba
se
lin
e in
ES
IF S
co
re o
f >
4.9
8
Patients achieving a change from baseline in ESIF score of >4.98
PBO
ADA
Non-responder
imputation.
ESIF: Erythema,
scaling, induration,
fissuring.
Menter A, et al. AAD 2012: P5061; Study sponsored by Abbott Laboratories
Secukinumab shows significant efficacy in palmoplantar
psoriasis: results from GESTURE, a randomized
controlled trial.
Gottlieb A, Sullivan J, van Doorn M, Kubanov A, You
R, Parneix A, Hugot S, Milutinovic M.
J Am Acad Dermatol. 2016 Oct (epub ahead of print)
More than Half of All Subjects on Secukinumab
300 mg Achieved Clear/Almost Clear Palms and
Soles at 1.5 Years
Palmoplantar disease improved by approximately 70% at 1.5 years
in subjects receiving secukinumab 300 mg
ppIGA 0/1†Response Out to 1.5 Years ppPASI Change Out to 1.5 Years
39.4%**
23.1%*
1.5%
–54.6%***
–35.3%***
34.9%
57.2%
–69.5%
–52.4%***
UNCOVER-3: Ixekizumab in patients with palmoplantar involvement: ppPASI 75 response rates
Menter A, et. al. EADV 2016, FC03.08; Sponsored by Eli Lilly and Company
• These patients have plaque psoriasis of the hands and feet, this does not address efficacy in
pustular disease nor patients with predominantly palmoplantar disease
0 2 4 8 12 16 20 24 28 32
Weeks
36 40 44 48 52 56 60
100 Induction
dosing period
Open label, long term extension period (IXE q4w)
78.1*
68.4*†
44.0
20.0
80.0
80.0
71.1
68.8
0
Pa
tie
nts
(%
)
ETN/IXE q4w (n=25) Placebo washout
IXE q4w/IXE q4w (n=32) IXE q2w/IXE q4w (n=38)
Placebo/IXE q4w (n=20)
*†
*
40
20
80
60
































































![Drug safety of systemic treatments for psoriasis: results from The German Psoriasis … · 2017-04-10 · systemic treatment of plaque type psoriasis and psoriatic arthritis [4].](https://static.fdocuments.in/doc/165x107/5f317903e0e5ab2579444a88/drug-safety-of-systemic-treatments-for-psoriasis-results-from-the-german-psoriasis.jpg)







