Day 14 Covalent bonds Sci 10Chemistry. Covalent Bonds Non-metal + non-metal.
-
Upload
bonnie-briggs -
Category
Documents
-
view
228 -
download
0
description
Transcript of Day 14 Covalent bonds Sci 10Chemistry. Covalent Bonds Non-metal + non-metal.

Day 14 – Covalent bonds
Sci 10 Chemistry

Covalent Bonds
Non-metal + non-metal

A compound without any metals?!???!A compound without any metals?!???!What happens when 2 nonmetals react?
Remember: Metals want to give away their electrons and nonmetals want to accept them.
•A compound made of only nonmetals (Hydrogen counts as a non-metal) is called a covalent or molecular compound.
Atoms in molecular compounds don’t want to lose their electrons SO …
• electrons are shared amoung multiple atoms to have full valence shells
Animation:http://bcs.whfreeman.com/thelifewire/content/chp02/02020.html

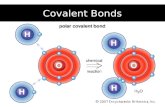
H and ClH and ClHydrogen and chlorine are both gases and nonmetals (even though hydrogen is found in Group 1 (the alkali metals)).
Neither H nor Cl want to give up their electrons to form an ionic sompound so they share them instead, forming a covalent bond (which gives us a covalent compound)
H now has 2e- in its valence shell and Cl has 8, so they are both full.
The line between H and Cl indicates there are 2e- shared between the 2 atoms.
This form is called a Lewis Dot Diagram.

HOBrFINCl’sHOBrFINCl’sThe following elements exist in a diatomic form (2 atoms are always bonded together). These diatomic molecules only exist in pairs of atoms.
Draw Lewis Diagrams for H2, O2, Br2, F2, I2, N2 and Cl2
Triple bond Double bond

Molecular compounds
Give the structural formula (Lewis diagram with lines representing shared electrons) for the molecular compound that will form from the following pairs of elements:
1. bromine + iodine2. nitrogen + chlorine3. carbon + fluorine4. sulfur + bromine

AnswersStructural formula
Br I
Cl N ClClF
F C FF
Br S Br

PracticePractice
Draw Lewis structures for the following compounds:
H2O, CO2, NH3, SCl2, CH4, C2H6, P4, CBr4, CH2Cl2

PP44 et S et S88: Elemental shapes (Chem : Elemental shapes (Chem 11)11)
• No need to memorize this but just to show you that certain atoms bond to others forming specific angles. The compounds that are formed with have specific shapes.

Practice . . . againPractice . . . again
Show the bond formation between each of the following pairs of elements by drawing Lewis diagrams. Indicate if the compound formed is ionic or covalent. Try to write the formula for the compound.
a) K and Se f) C and Cl
b) Br and Cl g) C and O
c) Sr and Br h) N and I
d) B and H i) Ca and N
e) Al and S j) Al and Br

Naming Covalent Compounds
1. Is it a covalent compound (non-metal + non-metal)? If yes,
2. Name the element that is furthest left on the periodic table first. Use a prefix if there is more than one atom of this element.
3. Name the second element. Use a prefix to indicate the # of atoms (even if there is only 1) and add the ending –ide.

Prefixes# of atoms Prefix1 mono-
(only use when naming the 2nd element i.e. names of covalent compounds never start with mono-…)
2 di-
3 tri-
4 tetra-
5 penta-
6 hexa-
7 hepta-
8 octa-
9 nona-
10 deca-

Examples and practice
• NO = nitrogen monoxide• N2O = dinitrogen monoxide
• Name NO2
– nitrogen dioxide • Name CO
– carbon monoxide

Try It!
Name the following molecular compounds:1. PI3
phosphorus triiodide2. SO2
sulfur dioxide3. SO3
sulfur trioxide4. S2F10
disulfur decafluoride5. CCl4
carbon tetrachloride6. N2O5
dinitrogen pentoxide

Name to formula
• Look at the prefixes to write the formula • Ex: phosphorus pentachloride
• PCl5

Find the formula
• nitrogen tribromide – NBr3
• nitrogen dioxide – NO2
• sulfur pentoxide– SO5

Naming diatomic molecules
• For the HOFBrINCl elements, they always exist in pairs of atoms as gases
• To name them, just use the name of the element– H2 (g) = hydrogen
– O2 (g) = oxygen
– N2 (g) = nitrogen

To do
• Day 14 Covalent Practice