COVALENT BONDING Hold on to your electrons!. What is a covalent bond? Atoms ___________ one or more...
-
Upload
lynn-black -
Category
Documents
-
view
214 -
download
0
Transcript of COVALENT BONDING Hold on to your electrons!. What is a covalent bond? Atoms ___________ one or more...

COVALENT BONDING
Hold on to your electrons!

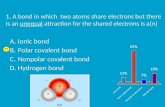
What is a covalent bond?Atoms ___________ one or more electrons with each other to form the bond.
Each atom is left with a ________________ outer shell or ___________________ .
A covalent bond forms between two _________________ or between a ______________ and a ________________.
The compound is held together by a mutual attraction for the shared pair of electrons.

There are two types of covalent bonds:• nonpolar covalent bonds• polar covalent bonds
The type of covalent bond depends on the electronegativity of the atoms involved. Consider it like a tug-o-war for electrons.

Nonpolar Covalent Bonding
Nonpolar covalent bonding occurs between atoms of the same element or between two atoms with very similar electronegativity.
The shared pair of electrons will spend equal time revolving around each nucleus resulting in equal sharing of the electron pair forming a covalent bond.

Two atoms of the same element bonded together are said to be diatomic.

There are seven elements which exist as diatomic molecules. These include:
H2 O2
F2 N2
Cl2 I2
Br2

Polar Covalent Bonding
Polar covalent bonding is the unequal sharing of electrons between two atoms with different electronegativity.

Polar Covalent BondingThe shared pair of electrons spends more time around the atom with the greater electron attracting ability.
This makes the atom with the higher electronegativity become slightly negatively charged (δ-) and the atom with a lower electronegativity, a slightly positively charged (δ +).

Some compounds containing polar-covalent bonds:
HCl CH4
H2O

Now it's your turn!
NH3
BCl3
H2S
CCl4