Chapter 8 Covalent Bonds
-
Upload
currituck-county-high-school -
Category
Technology
-
view
28.461 -
download
1
Transcript of Chapter 8 Covalent Bonds


•The atoms held together by sharing electrons are joined by a Covalent Bond.

2. Covalent bonds- Two atoms share one or more pairs of outer-shell electrons.
Oxygen AtomOxygen Atom Oxygen AtomOxygen Atom
Oxygen Molecule (O2)

Molecules and Molecular Compounds
• Molecule is a neutral group of atoms joined together by covalent bonds.
• Diatomic molecule is a molecule consisting of two atoms.
• A compound composed of molecules is called a molecular compound.

Properties
• Molecular compounds tend to have relatively lower melting and boiling points than ionic compounds.– Many are gases or liquids at room
temperature.– Most are formed from atoms of two or more
nonmetals.

Molecular Formulas
• A molecular formula is the chemical formula of a molecular compound.
• It shows how many atoms of each element a molecule contains.
• CO2 Carbon Dioxide
• 1 Carbon Atom
• 2 Oxygen Atoms

• Ionic Vs. Covalent Bond


Chapter 2 Chemical Principles
Bonding
Covalent bonding

Sowhatarecovalentbonds?

In covalent bonding,atoms still want to achievea noble gas configuration(the octet rule).

In covalent bonding,atoms still want to achievea noble gas configuration(the octet rule).
But rather than losing or gainingelectrons,atoms now share an electron pair.

In covalent bonding,atoms still want to achievea noble gas configuration(the octet rule).
But rather than losing or gainingelectrons,atoms now share an electron pair.
The shared electron pairis called a bonding pair

Cl2
Chlorineforms
acovalent
bondwithitself

ClClHowwilltwochlorineatomsreact?

ClClEach chlorine atom wants to gain one electron to achieve an octet

ClClNeither atom will give up an electron –chlorine is highly electronegative.
What’s the solution – what can theydo to achieve an octet?

ClClNeither atom will give up an electron –chlorine is highly electronegative.
What’s the solution – what can theydo to achieve an octet?

Cl Cloctet

Cl Cl
circle the electrons foreach atom that completestheir octets
octet

Cl Cl
circle the electrons foreach atom that completestheir octets
The octet is achieved byeach atom sharing theelectron pair in the middle

Cl Cl
circle the electrons foreach atom that completestheir octets
The octet is achieved byeach atom sharing theelectron pair in the middle

Cl Cl
circle the electrons foreach atom that completestheir octets
This is the bonding pair

Cl Cl
circle the electrons foreach atom that completestheir octets
It is a single bonding pair

Cl Cl
circle the electrons foreach atom that completestheir octets
It is called a SINGLE BOND

Cl Cl
circle the electrons foreach atom that completestheir octets
Single bonds are abbreviatedwith a dash

Cl Cl
circle the electrons foreach atom that completestheir octets
This is the chlorine molecule,
Cl2

O2
Oxygen is also one of the diatomic molecules

How will two oxygen atoms bond?
OO

OOEach atom has two unpaired electrons

OO

OO

Oxygen atoms are highly electronegative.
So both atoms want to gain two electrons.
OO

OOBoth electron pairs are shared.

6 valence electronsplus 2 shared electrons
= full octet
OO

6 valence electronsplus 2 shared electrons
= full octet
OO

two bonding pairs,
OOmaking a double bond

OO=For convenience, the double bond
can be shown as two dashes.
OO

OO=This is the oxygen molecule,
O2
this is so
cool!!

O
OMultiple Covalent bonds
OSharing One Pair of electrons One Covalent Bond
Only 7 electrons does Not meet Octet Rule!Need to share
Another pair of electrons
O O OSharing Two Pairs of electrons Two Covalent BondsA Double Bond
A Double Bond can be represented by a double line

N
N
Multiple Covalent bonds
N
N
Sharing Three Pairs of electrons Three Covalent BondsA Triple Bond
A Triple Bond can be represented by a Triple line
Nitrogen

Coordinate Covalent Bond
• both electrons contributed by one atom of pair
NH3 + H+ -----> NH4+
H2O + H+ -----> H3O+

Coordinate Covalent Bond
ammonium ion

Drawing Lewis Dot Structures1. Predict the location of the atoms
a. Hydrogen is a terminal atomb. The central atom has the smallest electronegativity.
2. Count the valence electrons.3. Draw a single covalent bond between the
central atom and the surrounding atoms.4. Subtract the number of electrons in the single
covalent bonds from the total number of electrons in 2.
5. Use the remaining electrons to complete the octets of each atom.
6. If the central atom does not have a complete octet then try double or triple bonds.

Drawing Lewis Dot Structures
Draw Lewis Dot Structures for:
PH3
H2S
HCl
CCl4
SiH4
CH2Cl2

Bond Dissociation Energies
•The energy required to break the bond between two covalently bonded atoms.

Relate the strength of covalent bonds to bond length
• The more bonds located between 2 atoms, the shorter the bonds are
• The shorter a bond is, the stronger it is
H – H single bond, not too strong
O=O double bonds, stronger
NΞN triple bonds, strongest

Endothermic/ExothermicIn chemical reactions, bonds are broken,
then new bonds are formedEndothermic
– More energy is required to break the old bonds than is released by the formation of new bonds
• Energy is taken in (colder)
Exothermic– More energy is released when forming new
bonds than is used to break the old bonds• Energy is given off (hotter)

Exceptions to Octet Rule
NO2 nitrogen dioxide
resonance

Exceptions to Octet Rule
PF5
expanded octet

Exceptions to Octet Rule
SF6
Expanded octet

INTRODUCTION
A) Lewis structures do not indicate the three dimensional shape of a molecule. They do not show the arrangement space of the atoms, what we call the molecular geometry or molecular structure.
B) Molecules have definite shapes and the shape of a molecule controls some of its chemical and physical properties.

II. Valence Shell Electron Pair Repulsion Theory - VSEPR - predicts the shapes of a number of molecules and polyatomic ions.
A) Assumptions of VSEPR Theory
1) Electron pairs in the valence shell of an atom tend to orient themselves so that the total energy is minimized. This means that: the electrons will approach the nucleus as close as possible yet take positions as far away from each other as possible to minimize _______________ .

2) Because lone pairs of electrons are spread out more broadly than bond pairs, repulsions are greatest between two lone pairs, intermediate between a lone pair and a bond pair, and weakest between two bonding pairs of electrons.3) Repulsive forces decrease rapidly with increasing interpair angle - greatest at 90o, much weaker at 120o, and very weak at 180o.
B) What are the ideal arrangements of electron pairs to minimize repulsions?

Bond Bond FormationFormation
Bond Bond FormationFormation
A bond can result from anA bond can result from an overlapoverlap of of atomic orbitals on neighboring atoms.atomic orbitals on neighboring atoms.
ClH H Cl••
••
••
••
••
••
+
Overlap of H (1s) and Cl (2p)
Note that each atom has a single, Note that each atom has a single, unpaired electron.unpaired electron.

Double and Double and even triple even triple bonds are bonds are commonly commonly observed for C, observed for C, N, P, O, and SN, P, O, and S
••O OC
•• ••
••
••O OC
•• ••
••
HH22COCO
SOSO33
CC22FF44

Some Common Some Common GeometriesGeometries
LinearLinear
Trigonal PlanarTrigonal Planar TetrahedrTetrahedralal



Structure Determination Structure Determination by VSEPRby VSEPR
Structure Determination Structure Determination by VSEPRby VSEPR
Water, HWater, H22OOThe electron pair The electron pair geometry is geometry is TETRAHEDRALTETRAHEDRAL
The electron pair The electron pair geometry is geometry is TETRAHEDRALTETRAHEDRAL
The molecular The molecular geometry is geometry is BENTBENT..
The molecular The molecular geometry is geometry is BENTBENT..
H O H••
••
H O H••
••
2 bond 2 bond pairspairs
2 lone 2 lone pairspairs

Structure Determination Structure Determination by VSEPRby VSEPR
Structure Determination Structure Determination by VSEPRby VSEPR
Ammonia, NHAmmonia, NH33
The electron pair geometry is tetrahedral.The electron pair geometry is tetrahedral.
H
H
H
lone pair of electronsin tetrahedral position
N
The The MOLECULAR GEOMETRYMOLECULAR GEOMETRY — the — the positions of the atoms — is positions of the atoms — is TRIGONAL TRIGONAL PYRAMIDPYRAMID..
The The MOLECULAR GEOMETRYMOLECULAR GEOMETRY — the — the positions of the atoms — is positions of the atoms — is TRIGONAL TRIGONAL PYRAMIDPYRAMID..

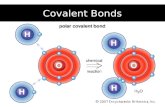
Bond PolarityBond PolarityBond PolarityBond PolarityHCl is HCl is POLARPOLAR because it has a because it has a
positive end and a negative positive end and a negative end. (difference in end. (difference in electronegativity)electronegativity)
Cl has a greater share Cl has a greater share in bonding electrons in bonding electrons than does H.than does H.
Cl has a greater share Cl has a greater share in bonding electrons in bonding electrons than does H.than does H.
Cl has slight negative charge Cl has slight negative charge (-(-)) and H and H has slight positive charge has slight positive charge (+ (+ ))
H Cl••
••
+ -••H Cl
••
••
+ -••

• This is why oil and water will not mix! This is why oil and water will not mix! Oil is nonpolar, and water is polar.Oil is nonpolar, and water is polar.
• The two will repel each other, and so The two will repel each other, and so you can not dissolve one in the otheryou can not dissolve one in the other
Bond PolarityBond PolarityBond PolarityBond Polarity

Bond PolarityBond PolarityBond PolarityBond Polarity
• ““Like Dissolves Like”Like Dissolves Like”
–Polar dissolves Polar dissolves PolarPolar
–Nonpolar Nonpolar dissolves dissolves NonpolarNonpolar

Electronegativity Difference
• If the difference in electronegativities is between:– 1.7 to 4.0: Ionic– 0.3 to 1.7: Polar Covalent– 0.0 to 0.3: Non-Polar Covalent
Example: NaClNa = 0.8, Cl = 3.0Difference is 2.2, sothis is an ionic bond!

Diatomic ElementsDiatomic Elements
• These elements do not exist as a single atom; they always appear as pairs
• When atoms turn into ions, this NO LONGER HAPPENS!– Hydrogen– Nitrogen– Oxygen– Fluorine– Chlorine– Bromine– Iodine
Remember: Remember: BrINClHOFBrINClHOF

Polar Covalent Bonds: Unevenly matched, but willing to share.

Van der Waals Forces
Small, weak interactions between molecules

Van der Waals Forces
Intermolecular: between molecules (not a bond)
Intramolecular: bonds within molecules (stronger)

3 Types of Van der Waals Forces
1) dipole-dipole
2) dipole-induced dipole
3) dispersion

Dipole-DipoleTwo polar molecules align so that + and - are matched (electrostatic attraction)
Ex: ethane (C2H6) vs. fluromethane (CH3F)
Occurs when polar molecules are attracted to one another.The slightly region of a polar molecule is weakly attracted to the slightly positive region of another polar molecule.
Similar to but much weaker than ionic bonds.

Dispersion Forces
The weakest of all molecular interactions, are caused by the motion of electrons.
Dispersion is the ONLY intermolecular attraction that occurs between non-polar molecules

Review
Dipole –
between two polar molecules
Dispersion-
between two non-polar molecules

Hydrogen Bonding
STRONGEST Intermolecular Force!!
A special type of dipole-dipole attraction
Bonds form due to the polarity of water.
Ice Liquid

Hydrogen Bonding con’t
Hydrogen bonds keep water in the liquid phase over a wider range of temperatures than is found for any other molecule of its size

How many drops can you get on a penny?
Water?
Why is there a difference???Water has strong Hydrogen Bonds and
TTE has weaker intermolecular forces
http://www.msnucleus.org/membership/html/k-6/wc/water/1/images/penny.jpg

How is surface tension affected by soap?
Breaks the surface tension!
http://www.chemistryland.com/CHM107/Water/SoapDisruptsWater.jpg http://www.chemistry.nus.edu.sg/2500/micelle.jpg

Intermolecular Attractions and Molecular Properties
• The physical properties of a compound depend on the type of bonding it displays-in particular, on whether it is ionic or covalent.
• Network Solids are solids in which all of the atoms are covalently bonded together.
• Melting a network solid would require breaking covalent bonds throughout the solid.
• Diamond does not melt; rather it vaporizes to a gas at 3500 degrees Celsius and above.

Review of Chemical Review of Chemical BondsBonds
• There are 3 forms of bonding:There are 3 forms of bonding:• __________________—complete —complete transfer transfer of of
1 or more electrons from one atom 1 or more electrons from one atom to another (one loses, the other to another (one loses, the other gains) forming oppositely charged gains) forming oppositely charged ions that attract one anotherions that attract one another
• __________________——some valence some valence electrons electrons sharedshared between atoms between atoms
• __________________ – holds atoms of a – holds atoms of a metal togethermetal together
Most bonds are Most bonds are somewhere in somewhere in between ionic between ionic and covalent.and covalent.

Review of Valence Review of Valence ElectronsElectrons
Review of Valence Review of Valence ElectronsElectrons
Number of valence electrons of a main Number of valence electrons of a main (A) group atom = Group number(A) group atom = Group number

Review of Valence Review of Valence ElectronsElectrons
Review of Valence Review of Valence ElectronsElectrons
• Remember from the electron Remember from the electron chapter that valence electrons are chapter that valence electrons are the electrons in the OUTERMOST the electrons in the OUTERMOST energy level… that’s why we did energy level… that’s why we did all those electron configurations!all those electron configurations!
• B is 1sB is 1s22 2s 2s22 2p 2p11; so the outer ; so the outer energy level is 2, and there are energy level is 2, and there are 2+1 = 3 electrons in level 2. 2+1 = 3 electrons in level 2. These are the valence electrons!These are the valence electrons!
• Br is Br is [Ar] 4s[Ar] 4s22 3d 3d1010 4p 4p55
How many valence electrons are How many valence electrons are present?present?

Bond and Lone Bond and Lone PairsPairs
Bond and Lone Bond and Lone PairsPairs
• Valence electrons are distributed Valence electrons are distributed as shared oras shared or BOND PAIRSBOND PAIRS and and unshared orunshared or LONE PAIRS.LONE PAIRS.
•
••
•
••
H Cllone pair (LP)
shared orbond pair
This is called a This is called a LEWIS LEWIS structure.structure.