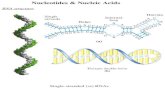
Chapter 26 Nucleosides, Nucleotides, and Nucleic Acids Copyright © The McGraw-Hill Companies, Inc....
-
Upload
maria-polly-logan -
Category
Documents
-
view
223 -
download
6
Transcript of Chapter 26 Nucleosides, Nucleotides, and Nucleic Acids Copyright © The McGraw-Hill Companies, Inc....

Chapter 26Chapter 26Nucleosides, Nucleotides,Nucleosides, Nucleotides,
and Nucleic Acidsand Nucleic Acids
Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display.

26.126.1Pyrimidines and PurinesPyrimidines and Purines

Pyrimidines and Purines
In order to understand the structure and properties of DNA and RNA, one needs to look at the structural components.
One set of important components are certain heterocyclic aromatic compounds called pyrimidines and purines.

Pyrimidines and Purines
Pyrimidine and purine are the names of the parent compounds of two types of nitrogen-containing heterocyclic aromatic compounds.
Pyrimidine Purine
N
N NH
N
NN 1
2
34
57
8
9
1
2
3
4
5
66

Pyrimidines and Purines
Amino-substituted derivatives of pyrimidine and purine have the structures expected from their names, existing as -NH2.
4-Aminopyrimidine 6-Aminopurine
H
N N
NN
HH
NH2
N
N
H
H
HH2N

Pyrimidines and Purines
But hydroxy-substituted pyrimidines exist in the keto, rather than the enol, form. However, the rings are still aromatic.
enol
N
N
H
H
HHO
N
N
H
H
HO
Hketo

Pyrimidines and Purines
The same is true for hydroxy-substituted purines which exist in keto, rather than enol, form.
H
N N
NN
HH
OH
enol keto
H
N N
N
H
H
O
HN

Important Pyrimidines
The pyrimidines that occur in DNA are cytosine and thymine. The pyrimidines that occur in RNA are cytosine and uracil.
HN
NH
O
O
Uracil
HN
NH
O
O
CH3
Thymine
HN
NH
NH2
O
Cytosine

Important Purines
Adenine and guanine are the principal purines of both DNA and RNA.
Adenine
N
N
NH2
N
NH
Guanine
O
HN
NH
N
N
H2N

Caffeine and Theobromine
Caffeine (coffee) and theobromine (coffee and tea) are other naturally occurring purines.
Caffeine
N
N
O
N
N
H3C
O
CH3
CH3
Theobromine
O
HN
NN
N
CH3
CH3
O

26.226.2NucleosidesNucleosides

Nucleosides
The classical structural definition of a nucleoside is a pyrimidine or purine N-glycoside of D-ribofuranose or 2-deoxy-D-ribofuranose.
Base + Ribose = nucleoside
Informal use has extended this definition to apply to purine or pyrimidine N-glycosides of almost any carbohydrate.
The purine or pyrimidine part of a nucleoside is referred to as a purine or pyrimidine base.

NH2
HO OH
OHOCH2
O
N
N
Table 26.2
Pyrimidine nucleosides
Cytidine occurs in RNA; its 2'-deoxy analog occurs in DNA.
Cytidine Note the - glycosidic bond.

O
N
NH
O
HO
O
H3C
HOCH2
Table 26.2
Pyrimidine nucleosides
Thymidine occurs in DNA.
Thymidine

Table 26.2
Pyrimidine nucleosides
HOCH2
O
N
NH
O
OHHO
O
Uridine occurs in RNA.
Uridine

Table 26.2
Purine nucleosides
Adenosine
Adenosine occurs in RNA; its 2'-deoxy analog occurs in DNA.
HOCH2 O
OHHO
N
N
N
NH2
N

Table 26.2
Purine nucleosides
Guanosine
Guanosine occurs in RNA; its 2'-deoxy analog occurs in DNA.
HOCH2
N
NH
O
O
HO
N NH2
N
OH

26.326.3NucleotidesNucleotides
Nucleotides are phosphoric acid Nucleotides are phosphoric acid esters of nucleosides.esters of nucleosides.

Adenosine 5'-Monophosphate (AMP)
Adenosine 5'-monophosphate (AMP) is also called 5'-adenylic acid. Note that the primed numbers are on the sugar ring.
OCH2PHO
O
HO
O
OHHO
N
N
N
NH2
N
5'
1'
2'3'
4'

Adenosine Diphosphate (ADP)
OP
O
HO
HO
OCH2P
O
HOO
OHHO
N
N
N
NH2
N
Adenosine 5'-diphosphate (ADP).

Adenosine Triphosphate (ATP)
OP
O
HO
O
OCH2P
O
HO
O
OHHO
N
N
N
NH2
N
P
O
HO
HO
ATP is an important molecule in several biochemical processes including: energy storage (Sections 26.4-26.5) and phosphorylation.

ATP and Phosphorylation
ATP +
hexokinaseThis is the first step in the metabolism of glucose.
ADP +
O
OHHO
HOHO(HO)2POCH2
O
HOCH2 O
OHHO
HOHO

3',5'-cAMP and 3',5'-cGMP
Cyclic adenosine monophosphate (cAMP)
CH2 O
OH
N
N
N
NH2
N
O
P
HO O
O
Cyclic AMP and cyclic GMP are "second messengers" in many biological processes. Hormones (the "first messengers") stimulate the formation of cAMP and cGMP.

O
P
HO O
O
cAMP and cGMP
Cyclic guanosine monophosphate (cGMP)
CH2
N
NH
O
O
N NH2
N
OH

26.426.4BioenergeticsBioenergetics

Bioenergetics
Bioenergetics is the thermodynamics of biological processes.
Emphasis is on free energy changes (G).
When G is negative, reaction is spontaneous in the direction written.
When G is positive, reaction is not spontaneous in direction written.
When G is 0, reaction is at equilibrium.

Standard Free Energy (G°)
Sign and magnitude of G depends on what the reactants and products are and their concentrations.
In order to focus on reactants and products, define a standard state.
The standard concentration is 1 M (for a reaction in homogeneous solution).
G in the standard state is called the standard free-energy change and given the symbol G°.
mA(aq) nB(aq)

Standard Free Energy (G°)
Exergonic: An exergonic reaction is one for which the sign of G° is negative.
Endergonic: An exergonic reaction is one for which the sign of G° is positive.
mA(aq) nB(aq)

Standard Free Energy (G°)
It is useful to define a special standard state for biological reactions.
This special standard state is one for which the pH = 7.
The free-energy change for a process under these conditions is symbolized as G°'.
mA(aq) nB(aq)

26.526.5ATP and BioenergeticsATP and Bioenergetics

Hydrolysis of ATP
G°' for hydrolysis of ATP to ADP is –31 kJ/mol.
Relative to ADP + HPO42–, ATP is a "high-
energy" compound.
When coupled to some other process, the conversion of ATP to ADP can provide the free energy to transform an endergonic process to an exergonic one.
ATP + H2O ADP + HPO42–

Glutamic Acid to Glutamine
+ NH4+–OCCH2CH2CHCO–
+NH3
O O
+ H2OH2NCCH2CH2CHCO–
+NH3
O O
G°' = +14 kJ Reaction is endergonic.

Glutamic Acid to Glutamine
+ NH4+–OCCH2CH2CHCO–
+NH3
O O
Reaction becomes exergonicwhen coupled to the hydrolysisof ATP.
+ ATP
+ HPO42–H2NCCH2CH2CHCO–
+NH3
O O
G°' = –17 kJ
+ ADP

Glutamic Acid to Glutamine
–OCCH2CH2CHCO–
+NH3
O O
Mechanism: step 1 involvesphosphorylation of glutamic acid.
+ ATP
OCCH2CH2CHCO–
+NH3
O O
+ ADPP
O
–O
–O
A mixed anhydride.

Glutamic Acid to Glutamine
Step 2: reaction of the phosphorylated glutamic acid with ammonia.
H2NCCH2CH2CHCO–
+NH3
O O
+ HPO42–
OCCH2CH2CHCO–
+NH3
O O
+ NH3P
O
–O
–O

26.626.6Phosphodiesters, Phosphodiesters,
Oligonucleotides andOligonucleotides andPolynucleotidesPolynucleotides

Phosphodiesters
A phosphodiester linkage between two nucleotides is analogous to a peptide bond between two amino acids.
Two nucleotides joined by a phosphodiester linkage gives a dinucleotide.
Three nucleotides joined by two phosphodiester linkages gives a trinucleotide, etc. (See next slide)
A polynucleotide of about 50 or fewer nucleotides is called an oligonucleotide.

3'
5'HOCH2
O
O
N
N
N
N
P OCH2O
HO
NH2
OCH2O
NH
N
N
N
HO
O
NH2HO
O P
H3C
O
O
OO
NH
N
Fig. 26.1The
trinucleotide ATG
phosphodiester linkages between 3' of one nucleotide and 5' of the next.
A
T
G
free 5' end
free 3' end

26.726.7Nucleic AcidsNucleic Acids
Nucleic acids are polynucleotides.Nucleic acids are polynucleotides.

Nucleic Acids
Nucleic acids were first isolated in 1869 (Johann Miescher).
Oswald Avery discovered (1945) that a substance which caused a change in the genetically transmitted characteristics of a bacterium was DNA.
Scientists revised their opinion of the function of DNA and began to suspect it was the major functional component of genes.

Composition of DNA
Erwin Chargaff (Columbia Univ.) studied DNAs from various sources and analyzed the distribution of purines and pyrimidines in them.
The distribution of the bases adenine (A), guanine (G), thymine (T), and cytosine (C) varied among species.
But the total purines (A and G) and the total pyrimidines (T and C) were always equal.
Moreover: %A = %T, and %G = %C

Composition of Human DNA
Adenine (A) 30.3% Thymine (T) 30.3%
Guanine (G) 19.5% Cytosine (C) 19.9%
Total purines: 49.8% Total pyrimidines: 50.1%
For example:
Purine Pyrimidine

Structure of DNA
James D. Watson and Francis H. C. Crick proposed a structure for DNA in 1953.
Watson and Crick's structure was based on:•Chargaff's observations•X-ray crystallographic data of Maurice Wilkins and Rosalind Franklin•Model building

26.826.8Secondary Structure of DNA:Secondary Structure of DNA:
The Double HelixThe Double Helix

Base Pairing
Watson and Crick proposed that A and T were present in equal amounts in DNA because of complementary hydrogen bonding.
2-deoxyribose
N
NN
N N
H
H
N
N
O CH3
O
H
2-deoxyriboseA T

Base Pairing
Ball and stick models showing the Watson and Crick base pairing in A=T.

Base Pairing
Likewise, the amounts of G and C in DNA were equal because of complementary hydrogen bonding.
N
NN
N O
NH
H
H
N
N
N
O
HH
2-deoxyribose
2-deoxyribose
G C

Base Pairing
Ball and stick models showing the Watson and Crick base pairing in G≡C.

The DNA Duplex
Watson and Crick proposed a double-stranded structure for DNA in which a purine or pyrimidine base in one chain is hydrogen bonded to its complement in the other.
•Gives proper Chargaff ratios (A=T and G=C)
•Because each pair contains one purine and one pyrimidine, the A---T and G---C distances between strands are approximately equal.
•Complementarity between strands suggests a mechanism for copying genetic information.

O
O
ĞO
ĞO
ĞO
O
O
O
O
OP
O
O
P O
OO
P O
OO
O
O
OĞ
OĞ
OĞ
O
O
O
O
O P
O
O
PO
OO
PO
OO
C G
AT
AT
CG
3'
5'
5'
5'
5' 5'
5'
5'
5'
3'
3'3'
3'3'
3'
3'
Fig. 26.4
Two antiparallel strands of DNA are paired by hydrogen bonds between purine and pyrimidine bases.

Fig. 26.5
Helical structure of DNA. The purine and pyrimidine bases are on the inside, sugars and phosphates on the outside.

26.926.9Tertiary Structure of DNA:Tertiary Structure of DNA:
SupercoilsSupercoils

DNA is coiled
A strand of DNA is too long (about 3 cm in length) to fit inside a cell unless it is coiled.
Random coiling would reduce accessibility to critical regions.
Efficient coiling of DNA is accomplished with the aid of proteins called histones.

Histones
Histones are proteins rich in basic amino acids such as lysine and arginine.
Histones are positively charged at biological pH.DNA is negatively charged due to the phosphate linkages.
DNA winds around histone proteins to form nucleosomes.
Nucleosome = Histone proteins + Supercoiled DNA

Histones
Each nucleosome contains one and three-quarters turns of coil = 146 base pairs.Linker contains about 50 base pairs.

26.1026.10Replication of DNA:Replication of DNA:
Chain ElongationChain Elongation

Elongation of the Growing DNA Chain
The free 3'-OH group of the growing DNA chain reacts with the 5'-triphosphate of the appropriate nucleotide.
The chain elongates from 5' to 3'.

Fig. 26.9: Chain Elongation
OH CH2
OO P
O
O–
O P
O
O–
O P
O
O–
O–Adenine,Guanine,
Cytosine, orThymine
••OH
CH2OPOO
Adenine,Guanine,
Cytosine, orThymine
O
O–
••
Poly-nucleotide
chain
Poly-nucleotide
chain
5' end3' end

Fig. 26.9: Chain Elongation
OH CH2
OO P
O–
Adenine,Guanine,
Cytosine, orThymine
O CH2OPO
OAdenine,Guanine,
Cytosine, orThymine
O
O–
•• ••
Poly-nucleotide
chain
Poly-nucleotide
chain
O
O P
O
O–
O P
O
O–
O––
5' end
3' end

26.1126.11Ribonucleic AcidsRibonucleic Acids

DNA and Protein Biosynthesis
According to Crick, the "central dogma" of molecular biology is:
"DNA makes RNA makes protein."
Three kinds of RNA are involved.Messenger RNA (mRNA)Transfer RNA (tRNA)Ribosomal RNA (rRNA)
There are two main stages.TranscriptionTranslation

Transcription
In transcription, a strand of DNA acts as a template upon which a complementary RNA is biosynthesized.
This complementary RNA is messenger RNA (mRNA).
Mechanism of transcription resembles mechanism of DNA replication.
Transcription begins at the 5' end of DNA and is catalyzed by the enzyme RNA polymerase.

The Genetic Code
The nucleotide sequence of mRNA codes for the different amino acids found in proteins.
There are three nucleotides per codon.
There are 64 possible combinations of A, U, G, and C.
The genetic code is redundant. Some proteins are coded for by more than one codon.

UUU Phe UCU Ser UAU Tyr UGU Cys U
UUC Phe UCC Ser UAC Tyr UGC Cys C
UUA Leu UCA Ser UAA Stop UGA Stop A
UUG Leu UCG Ser UAG Stop UCG Trp G
U
C
A
G
U
C
A
G
U
C
A
G
U C A G
U
C
A
G
First letter
Second letter
Third letter
Table 26.4

UUU Phe UCU Ser UAU Tyr UGU Cys U
UUC Phe UCC Ser UAC Tyr UGC Cys C
UUA Leu UCA Ser UAA Stop UGA Stop A
UUG Leu UCG Ser UAG Stop UCG Trp G
CUU Leu CCU Pro CAU His CGU Arg U
CUC Leu CCC Pro CAC His CGC Arg C
CUA Leu CCA Pro CAA Gln CGA Arg A
CUG Leu CCG Pro CAG Gln CCG Arg G
AUU Ile ACU Thr AAU Asn AGU Ser U
AUC Ile ACC Thr AAC Asn AGC Ser C
AUA Ile ACA Thr AAA Lys AGA Arg A
AUG Met ACG Thr AAG Lys ACG Arg G
GUU Val GCU Ala GAU Asp GGU Gly U
GUC Val GCC Ala GAC Asp GGC Gly C
GUA Val GCA Ala GAA Glu GGA Gly A
GUG Val GCG Ala GAG Glu GCG Gly G
U C A G
U
C
A
G

U
C
UAA Stop UGA Stop A
UAG Stop G
U
C
A
G
AUU Ile ACU Thr AAU Asn AGU Ser U
AUC Ile ACC Thr AAC Asn AGC Ser C
AUA Ile ACA Thr AAA Lys AGA Arg A
AUG Met ACG Thr AAG Lys ACG Arg G
U
C
A
G
U C A G
U
C
A
G
AUG is the "start" codon. Biosynthesis of allproteins begins with methionine as the first aminoacid. This methionine is eventually removed afterprotein synthesis is complete.
UAA, UGA, and UAG are "stop" codons thatsignal the end of the polypeptide chain.

Transfer tRNA
There are 20 different tRNAs, one for each amino acid.
Each tRNA is single stranded with a CCA triplet at its 3' end.
A particular amino acid is attached to the tRNA by an ester linkage involving the carboxyl group of the amino acid and the 3' oxygen of the tRNA.

Transfer RNA
Example—Phenylalanine transfer RNA
One of the mRNA codons for phenylalanine is:
UUC5' 3'
AAG3' 5'
The complementary sequence in tRNA is calledthe anticodon.

Fig. 26.11: Phenylalanine tRNA

Ribosomal RNA
Most of the RNA in a cell is ribosomal RNA.
Ribosomes are the site of protein synthesis. They are where translation of the mRNA sequence to an amino acid sequence occurs.
Ribosomes are about two-thirds RNA and one-third protein.
It is believed that the ribosomal RNA acts as a catalyst—a ribozyme.

26.1226.12Protein BiosynthesisProtein Biosynthesis

Protein Biosynthesis
During translation the protein is synthesized beginning at its N-terminus.
mRNA is read in its 5'-3' direction.Begins at the start codon AUGEnds at stop codon (UAA, UAG, or UGA)

26.1426.14DNA SequencingDNA Sequencing
The Sanger MethodThe Sanger Method

DNA Sequencing
Restriction enzymes cleave the polynucleotide to smaller fragments.
These smaller fragments (100-200 base pairs) are sequenced.
First the two strands are separated.

DNA Sequencing
Single stranded DNA divided in four portions.
Each protion contains adenosine, thymidine, guanosine, and cytidine plus the triphosphates of their 2'-deoxy analogs. POCH2
OH
OO
O
OH
P
O
OH
P
O
HO base
HHO
O

DNA Sequencing
To the first portion, 2,'3'-dideoxyadenosine triphosphate (ddATP) is added; to the second portion, the 2,'3'-dideoxythymidine triphosphate (ddTTP) is added; to the third, ddGTP; and to the fourth, ddCTP. POCH2
OH
OO
O
OH
P
O
OH
P
O
HO base
HH
O
2,'3'-dideoxynucleoside triphosphate (ddNTP)

DNA Sequencing
Each tube also contains a "primer", a short section of the complementary DNA strand, labeled with radioactive phosphorus (32P).
DNA synthesis takes place, producing a complementary strand of the DNA strand used as a template.
DNA synthesis stops when a dideoxynucleotide is incorporated into the growing chain.

DNA Sequencing
The contents of each tube are separated by electrophoresis and analyzed by autoradiography.
There are four lanes on the electrophoresis gel.
Each DNA fragment will be one nucleotide longer than the previous one.

Figure 26.13

End of Chapter 26End of Chapter 26Nucleosides, Nucleotides,Nucleosides, Nucleotides,
and Nucleic Acidsand Nucleic Acids