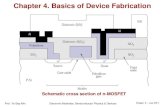
Chapter 12: Structures & Properties of...
Transcript of Chapter 12: Structures & Properties of...
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 1
Chapter 12: Structures & Properties of
Ceramics
ISSUES TO ADDRESS...
• How do the crystal structures of ceramic materials
differ from those for metals?
• How do point defects in ceramics differ from those
defects found in metals?
• How are impurities accommodated in the ceramic lattice?
• How are the mechanical properties of ceramics
measured, and how do they differ from those for metals?
• In what ways are ceramic phase diagrams different from
phase diagrams for metals?
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 2
• Bonding: -- Can be ionic and/or covalent in character.
-- % ionic character increases with difference in
electronegativity of atoms.
• Degree of ionic character may be large or small:
Atomic Bonding in Ceramics
SiC: small
CaF2: large
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 3
Ceramic Crystal Structures
Oxide structures – oxygen anions larger than metal cations
– close packed oxygen in a lattice (usually FCC)
– cations fit into interstitial sites among oxygen ions
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 4
Factors that Determine Crystal Structure 1. Relative sizes of ions – Formation of stable structures: --maximize the # of oppositely charged ion neighbors.
- -
- - +
unstable
- -
- - +
stable
- -
- - +
stable
2. Maintenance of
Charge Neutrality : --Net charge in ceramic
should be zero.
--Reflected in chemical
formula:
CaF 2 : Ca 2+
cation
F -
F -
anions +
A m X p
m, p values to achieve charge neutrality
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 5
• Coordination # increases with
Coordination # and Ionic Radii
2
r cation r anion
Coord
#
< 0.155
0.155 - 0.225
0.225 - 0.414
0.414 - 0.732
0.732 - 1.0
3
4
6
8
linear
triangular
tetrahedral
octahedral
cubic
ZnS
NaCl
CsCl
r cation r anion
To form a stable structure, how many anions can surround
around a cation? Coordination number
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 6
Computation of Minimum Cation-Anion
Radius Ratio • Determine minimum rcation/ranion for an octahedral site
(C.N. = 6)
a = 2ranion
2ranion 2rcation= 2 2ranion
ranion rcation= 2ranion
rcation= ( 2 1)ranion
arr 222 cationanion =
414.012anion
cation ==r
r
//upload.wikimedia.org/wikipedia/commons/1/14/Octahedron.gif
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 7
Bond Hybridization
Bond Hybridization is possible when there is significant
covalent bonding
– hybrid electron orbitals form
– For example for SiC
• XSi = 1.8 and XC = 2.5
% ionic character = 100 {1- exp[-0.25(XSi XC)2]} =11.5%
• ~ 89% covalent bonding
• Both Si and C prefer sp3 hybridization
• Therefore, for SiC, Si atoms occupy tetrahedral sites
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 8
• On the basis of ionic radii, what crystal structure
would you predict for FeO?
• Answer:
5500
1400
0770
anion
cation
.
.
.
r
r
=
=
based on this ratio,
-- coord # = 6 because
0.414 < 0.550 < 0.732
-- crystal structure is NaCl
Example Problem: Predicting the Crystal Structure of FeO
Ionic radius (nm)
0.053
0.077
0.069
0.100
0.140
0.181
0.133
Cation
Anion
Al 3+
Fe 2 +
Fe 3+
Ca 2+
O 2-
Cl -
F -
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 9
AX-Type Crystal Structures: Zinc Blende Structure
• ZnS (zinc blende or sphalerite) structure
rZn = 0.075 nm
rZn/rS = 0.408
cations (Zn2+) prefer tetrahedral sites
rS = 0.184 nm
So each Zn2+ has 4 neighbor S2-
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 10
AX-Type Crystal Structures: Rock Salt Structure
• NaCl (rock salt) structure
rNa = 0.102 nm
rNa/rCl = 0.564
cations (Na+) prefer octahedral sites
rCl = 0.181 nm
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 11
MgO and FeO
O2- rO = 0.140 nm
Mg2+ rMg = 0.072 nm
rMg/rO = 0.514
cations prefer octahedral sites
So each Mg2+ (or Fe2+) has 6 neighbor oxygen atoms
MgO and FeO also have the NaCl structure
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 12
939.0181.0
170.0
Cl
Cs ==
r
r
Cesium Chloride structure:
Since 0.732 < 0.939 < 1.0,
cubic sites preferred
So each Cs+ has 8 neighbor Cl-
AX-Type Crystal Structures: CsCl Structure
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 13
AmXp Crystal Structures: AX2
• Calcium Fluorite (CaF2)
• Cations in cubic sites
• UO2, ThO2, ZrO2, CeO2
• Antifluorite structure –
positions of cations and
anions reversed
Fluorite structure
75201330
1000.
.
.==
F
Ca
r
r
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 14
AmBnXp Crystal Structures: ABX3
• Perovskite structure
Ex: complex oxide
BaTiO3
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 15
AmBnXp Crystal Structures: AB2X4
• Spinel structure:
Ex: MgAl2O4
Mg2+ in tetrahedral sites
Al3+ in octahedral sites
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 16
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 17
Density Computations for Ceramics
A
AC )(
NV
AAn
C
=
Number of formula units/unit cell
Volume of unit cell
Avogadro’s number
= sum of atomic weights of all anions in formula unit
AA
AC = sum of atomic weights of all cations in formula unit
-
Prof. Yo-Sep Min Fusion Technology of Chemical & Materials Engineering Lecture 21, Chapter 12 - 18
ANNOUNCEMENTS
Reading: pp. 464 ~ 491





![Konkuk International Winter Program Jan 4 · Field Trip KRW 400,000 Upon Registration Tuition KRW 0 Waived [Students whose parents are Konkuk University Alumni] Payments Amount Deadline](https://static.fdocuments.in/doc/165x107/5f9a65472ddeb973d844ab1f/konkuk-international-winter-program-jan-4-field-trip-krw-400000-upon-registration.jpg)