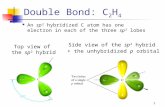
all C are sp 3 hybridized
description
Transcript of all C are sp 3 hybridized

all C are sp3 hybridized bondsC
3 prop ane C3H8
4 but ane C4H10
5 pent ane C5H12
6 hex ane C6H14
7 hept ane C7H16
8 oct ane C8H18
9 non ane C9H20
10 dec ane C10H22
n root suffix formula
1 meth ane C
nH
2n+2
H4
2 C2H6eth ane
Alkanes

Alkanes
CH4 16 -164C2H6 30 -88.6C3H8 44 -42.1C4H10 58 -0.5C5H12 72 +36.0
compound m.w. b.p.(Co)
n = 1-4 gasesn = 5-15 liquidsn > 15 solids

Alkanes
IMF = q1q2
____
rn = 1-4 gasesn = 5-15 liquidsn > 15 solids
q # electronsinstantaneous dipole moments
LDF

Alkanes
m.w. b.p.
IMF = q1q2
r
____
compound
72 36.1
72 27.9
72 9.5
structural isomers
r same formula different shape
branching


Reactions of Alkanes
combustion
__ C3H8 __ CO2 + 45
exothermic reactioncombustion with insufficient O2
__ C3H8 + __ O2 __ CO + __ H2O3 47/2
improperly vented spaces and cigarettes
+ __ O2 __ H2O3

Reactions of AlkanesSubstitution
step 1 Cl2 2Cl.h
initiation
step 2Cl.+ CH4 CH3
. + HClpropagation
step 3CH3
.+ Cl2 CH3Cl + Cl.propagation
step 4Cl.+ Cl. Cl2
termination
get a mixture of chlorinated methanes
of halogens

Halogenation of Alkanes
CH3Cl chloro methyl chloride
CH2Cl2 chloromethane methylene chloride
CHCl3 chloromethane chloroform
CCl4 chloromethane carbon tetrachloride
all liquids at room temperature methane is gase- rich Cl increases LDF
methane
di
tri
tetra

Nomenclature
1. Pick longest C chain as parent2. Number C beginning at end with first branch3. Identify branching substituents Assign each a number4. Use commas to separate numbers
hyphens to separate numbers from names5. Alphabetize substituents

NomenclatureStructural isomers of C5H12
I.
I. pentane
II.1 2 3 4
II. butanemethyl2-
III.
III.
1 2 3
propanemethyldi2,2-
1
2 3 41234

NomenclatureR = “alkyl” group
primary carbon1o
secondary carbon
2o
tertiary carbon
3o
quaternary carbon
4o

Nomenclature“common” names to learn:
propyl alphabetized as i
butyl alphabetized as i
butyl alphabetized as b
butyl alphabetized as b
iso
iso
sec-
tert-
H