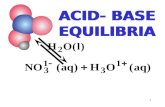Addition of Acid or Base to Solution CE 541. When adding Base or Acid to Solution: Interaction...
-
Upload
brendan-everson -
Category
Documents
-
view
213 -
download
0
Transcript of Addition of Acid or Base to Solution CE 541. When adding Base or Acid to Solution: Interaction...

Addition of Acid Addition of Acid or Base to or Base to SolutionSolution
CE 541CE 541

When adding Base or Acid to Solution:When adding Base or Acid to Solution:Interaction between different chemical species occurInteraction between different chemical species occurChemical equilibrium establishedChemical equilibrium establishedNature of changes depends on Nature of changes depends on
Nature of speciesNature of species Concentration of speciesConcentration of species
Changes can be predicted by solving equilibrium Changes can be predicted by solving equilibrium problemproblemIt is cumbersomeIt is cumbersomeAcid-base titration can be used to help understand Acid-base titration can be used to help understand the processthe process
What is Titration?What is Titration?It is a procedure by which a measured amount It is a procedure by which a measured amount of a chemical is added to solution in order to of a chemical is added to solution in order to bring about the desired change.bring about the desired change.

Titration of Strong Acids Titration of Strong Acids and Basesand Bases
Strong Base Titrated with Strong AcidStrong Base Titrated with Strong Acid High initial pH (12-13)High initial pH (12-13) Slowly declines to pH ≈ 10Slowly declines to pH ≈ 10 Rapid decline to pH ≈ 4Rapid decline to pH ≈ 4 Slow declineSlow decline Between pH 10 – 4 curve is verticalBetween pH 10 – 4 curve is vertical Equivalence point between pH 4 and 10Equivalence point between pH 4 and 10
Equivalence point is reached when Equivalence point is reached when equivalents of base (or acid) added equivalents of base (or acid) added equal to equivalents of base (or acid) equal to equivalents of base (or acid) initially presentinitially present


Strong Acid Titrated with Strong Acid Titrated with Strong BaseStrong Base

Titration ProblemsTitration ProblemsCan be solved using charge balance. If Can be solved using charge balance. If strong HCl is titrated with strong NaOHstrong HCl is titrated with strong NaOH
NaOH + HCl → HNaOH + HCl → H22O + NaO + Na++ + Cl + Cl--
Charge balanceCharge balance
[Na[Na++] + [H] + [H++] = [OH] = [OH--] + [Cl] + [Cl--]]
to get the exact pH, activities must be used. to get the exact pH, activities must be used. However, concentration can produce close However, concentration can produce close solution.solution.

IfIf
CC00 = molar concentration of HCl = molar concentration of HCl
VV00 = volume of HCl to be titrated = volume of HCl to be titrated
C = molar concentration of NaOHC = molar concentration of NaOH
V = volume of NaOH addedV = volume of NaOH added
Then:Then:
VV
CVOHH
VV
VC
0
00
0
][][

Re-arrangeRe-arrange
if C >> Cif C >> C00, then V can be ignored., then V can be ignored.
Study Example 4.18Study Example 4.18
1 f point, eequivalencat function; eequivalenc thecalled is
][][
1
][][1
00
0
00
000
00
VC
CVf
HH
Kf
VV
CV
HOHVC
CV
VV
CV
w

Titration of Weak Acids and Titration of Weak Acids and BasesBases
Weak Acid Titrated with Strong BaseWeak Acid Titrated with Strong Base Curve character depends on acidCurve character depends on acid
Monobasic → yields one HMonobasic → yields one H++ ion ion Polybasic → yields more than one HPolybasic → yields more than one H++
ionion Initial pH depends on degree of Initial pH depends on degree of
ionizationionization Most of natural waters contain weak Most of natural waters contain weak
acids and basesacids and bases


Titration of Monobasic Titration of Monobasic Weak Acid with Strong Weak Acid with Strong
BaseBaseAcid ionizes as follows:Acid ionizes as follows:
HA ↔ HHA ↔ H+ + + A+ A--
The equilibrium relationships are:The equilibrium relationships are:
)2(]][[
)1(][
]][[
w
A
KOHH
KHA
AH

when weak acid is titrated with when weak acid is titrated with strong basestrong base
HA + B + OHHA + B + OH-- → B → B++ + A + A-- + H + H22OO
BB++ is the cation associated with the is the cation associated with the strong base.strong base.
The charge balance isThe charge balance is
[H[H++] + [B] + [B++] = [A] = [A--] + [OH] + [OH--] (3)] (3)
[H[H++] can be determined by solving ] can be determined by solving equations (1), (2), and (3)equations (1), (2), and (3)

IfIf
CC00 = molar concentration of weak = molar concentration of weak acidacid
VV00 = volume of weak acid to be = volume of weak acid to be titratedtitrated
C = molar concentration of strong C = molar concentration of strong basebase
V = volume of strong base addedV = volume of strong base added
Then:Then:VV
VCAHA
0
00][][

mass balance of strong base cationmass balance of strong base cation
[B+] = (CV) / (V[B+] = (CV) / (V00 + V) + V)
combining mass balance and combining mass balance and equilibrium relationship of weak equilibrium relationship of weak acidacid
1
01
1
00
][1
][
][1
][
A
A
K
H
C
A
H
K
C
HA

The charge balance becomesThe charge balance becomes
RearrangeRearrange
The equation can be used to The equation can be used to generate titration curvegenerate titration curve
Study Example 19Study Example 19
VV
VCOHH
VV
CV
0
001
0
][][
00
01
00
][][ VC
VVH
H
Kf
VC
CV w

Beginning of TitrationBeginning of Titration
In the initial solution containing In the initial solution containing the weak acidthe weak acid
[B[B++] = 0] = 0 pH is low pH is low [OH [OH--] << [H] << [H++]]
So, charge balance becomesSo, charge balance becomes
[H[H++] ≈ [A] ≈ [A--]]
OriginallyOriginally
[H[H++] + [B] + [B++] = [A] = [A--] +[OH] +[OH--]]

Substitute in Substitute in
and solve for [Hand solve for [H++]]
At the beginning of titration (most At the beginning of titration (most of cases)of cases)
[A] << C[A] << C00
AKHA
AH
][
]][[
][][][ 0 ACKHAKH AA

so,so,
then,then,
Study Examples 20-21Study Examples 20-21
0][ CKH A
00 log2
1log
2
1log
2
1]log[ CpKCKHpH AA

Midpoint of TitrationMidpoint of Titration
As titration proceedsAs titration proceeds
HA + BHA + B++ + OH + OH-- → B → B++ + A + A-- + H + H22OO [A[A--] increases] increases [HA] decreases[HA] decreases
when neutralization reaches 50%when neutralization reaches 50% [B[B++] ≈ (1/2) C] ≈ (1/2) C00
[A[A--] increases significantly] increases significantly

so,so,
[H[H++] << [B] << [B++]]
[OH[OH--] << [A] << [A--]]
From charge balance From charge balance
([H([H++] + [B] + [B++] = [A] = [A--] + [OH] + [OH--])])
[B[B++] ≈ [A] ≈ [A--] ≈ (1/2) C] ≈ (1/2) C00
and since and since
[HA] + [A[HA] + [A--] = C] = C00
then then
[HA] ≈ (1/2) C[HA] ≈ (1/2) C00

Substitute inSubstitute inAKHA
AH
][
]][[
A
AA
pKpH
or
KHKC
CH
][
2
12
1][
0
0

Equivalence Point of Equivalence Point of TitrationTitration
At that point, equivalents of base At that point, equivalents of base added are equal to equivalents of added are equal to equivalents of acid in the original solution (f = 1)acid in the original solution (f = 1)So,So,
[B[B++] = C] = C00
The pH value is high enough to The pH value is high enough to assume assume [H[H++] << [OH] << [OH--]]
CC00 ≈ [A ≈ [A--] + [OH] + [OH--]]
CC00 – [A – [A--] ≈ [OH] ≈ [OH--]]

Since Since
CC00 – [A – [A--] = [HA]] = [HA]
Thus, Thus,
Substitute inSubstitute in
GivesGives
ww KOHHH
KOHHA
]][[....
][][][
AKHA
AH
][
]][[
21
][
][
1
AwKK
A
H

At the end of titration At the end of titration
[A[A--] ≈ C] ≈ C00
then,then,
wA pKpKCpH 0log2
1

When titrating weak base with When titrating weak base with strong acid, pH at various points strong acid, pH at various points can be determined using the same can be determined using the same approach.approach.
Beginning pointBeginning point
pH ≈ pKpH ≈ pKww – (1/2) pK – (1/2) pKBB + (1/2) log C + (1/2) log C00
MidpointMidpoint
pH ≈ pKpH ≈ pKww – pK – pKBB
Equivalence pointEquivalence point
pH ≈ (1/2) (pKpH ≈ (1/2) (pKww – pK – pKBB – logC – logC00))

And when titrating weak acid with And when titrating weak acid with strong basestrong base
Beginning of titrationBeginning of titration
pH ≈ (1/2)(pKpH ≈ (1/2)(pKAA – log C – log C00))MidpointMidpoint
pH ≈ pKpH ≈ pKAA
Equivalence pointEquivalence point
pH ≈ (1/2) (log CpH ≈ (1/2) (log C00 + pK + pKAA + pK + pKww))
Study Examples 22 and 23Study Examples 22 and 23


Calculate the pH of the equivalence point in Calculate the pH of the equivalence point in the titration of solutions containing the the titration of solutions containing the following concentrations of sodium following concentrations of sodium bicarbonate with sulfuric acid: (a) 10 mg/l; (b) bicarbonate with sulfuric acid: (a) 10 mg/l; (b) 100 mg/l; © 1000 mg/l100 mg/l; © 1000 mg/l

A 500ml solution contains 100 mg of NaOH. A 500ml solution contains 100 mg of NaOH. Calculate the followingCalculate the following1. initial pH of the solution1. initial pH of the solution2. pH after addition of 2 ml of 1 N H2. pH after addition of 2 ml of 1 N H22SOSO44
3. pH after addition of 4 ml of 1 N H3. pH after addition of 4 ml of 1 N H22SOSO44
4. ml of 1 N H4. ml of 1 N H22SOSO44 required to reach the required to reach the equivalence pointequivalence point

BuffersBuffers
are substances in solution that offer are substances in solution that offer resistance to changes in pH as acids resistance to changes in pH as acids or bases are added or formed within or bases are added or formed within the solutionthe solution
buffer solutions usually contain buffer solutions usually contain mixtures of weak acids and their mixtures of weak acids and their salts or weak bases and their acids salts or weak bases and their acids (conjugates)(conjugates)
ifif ][
]][[
HA
AHK A

RearrangeRearrange
So, pH of a buffer solution So, pH of a buffer solution depends on the ratio of salt depends on the ratio of salt concentration to acid concentration to acid concentrationconcentration
Study Example 24Study Example 24
][
][log
][
][1
][
1
acid
saltpKpH
thus
HA
A
KH
A
A

Calculate the pH of a buffer solution prepared Calculate the pH of a buffer solution prepared by mixing 2.4 g of acetic acid (CHby mixing 2.4 g of acetic acid (CH33COOH) and COOH) and 0.73 g of sodium acetate (CH0.73 g of sodium acetate (CH33COONa) in 1 COONa) in 1 liter of water.liter of water.

Buffering Index or Buffering Buffering Index or Buffering Capacity can be determined by:Capacity can be determined by:
where β is the buffering capacity where β is the buffering capacity or index of weak acid plus its or index of weak acid plus its conjugate salt.conjugate salt.If we have weak salt (base) plus its If we have weak salt (base) plus its conjugate acid, thenconjugate acid, then
KKAA is replaced by (K is replaced by (Kww / K / KBB))
Study Example 25Study Example 25
][
][][
][303.2
HK
HKCH
H
K
A
ATw