7 glycogen metabolism-_gluconeogenesis
-
Upload
mansoura-university -
Category
Education
-
view
2.007 -
download
1
description
Transcript of 7 glycogen metabolism-_gluconeogenesis

1
23.7 Glycogen Metabolism
23.8 Gluconeogenesis: Glucose Synthesis
Metabolic Pathways for Carbohydrates

2
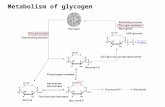
Glycogenesis
Glycogenesis:
Stores glucose by converting glucose to
glycogen.
Operates when high levels of glucose-6-
phosphate are formed in the first reaction of
glycolysis.
Does not operate when energy stores
(glycogen) are full, which means that
additional glucose is converted to body fat.

3
Diagram of Glycogenesis

4
Formation of Glucose-6-Phosphate
Glucose is converted to glucose-6-phosphate
using ATP.
Glucose-6-phosphate
O
OH
OH
OH
OH
CH2OP

5
Formation of Glucose-1-Phosphate
Glucose-6-phosphate is converted
to glucose-1-phosphate.
Glucose-6-phosphate Glucose-1-phosphate
O
O
OH
OH
OH
CH2OH
P
O
OH
OH
OH
OH
CH2OP

6
UDP-Glucose
UTP activates glucose-1-phosphate to form UDP-glucose and pyrophosphate (PPi).
UDP-glucose
O
O
OH
OH
OH
CH2OH
P
O
O-
O P
O
O-
O CH2O
OHOH
N
N
O
H
O

7
Glycogenesis: Glycogen
The glucose in UDP-glucose adds to glycogen.
UDP-Glucose + glycogen glycogen-glucose + UDP
The UDP reacts with ATP to regenerate UTP.
UDP + ATP UTP + ADP

8
Glycogenolysis
Glycogenolysis
is the break
down of
glycogen to
glucose.

9
Glycogenolysis
Glycogenolysis:
Is activated by glucagon (low blood glucose).
Bonds glucose to phosphate to form glucose-1-
phosphate. Glycogen-glucose + Pi Glycogen + glucose-1-phosphate

10
Isomerization of Glucose-1-
phosphate
The glucose-1-phosphate isomerizes to glucose-
6-phosphate, which enters glycolysis for energy
production.

11
Glucose-6-phosphate
Glucose-6-phosphate:
Is not utilized by brain and skeletal muscle
because they lack glucose-6-phosphatase.
Hydrolyzes to glucose in the liver and kidney,
where glucose-6-phosphatase is available providing
free glucose for the brain and skeletal muscle.

12
Utilization of Glucose
Glucose:
Is the primary
energy source for the
brain, skeletal
muscle, and red
blood cells.
Deficiency can
impair the brain and
nervous system.

13
Gluconeogenesis: Glucose
Synthesis
Gluconeogenesis is:
The synthesis of
glucose from
carbon atoms of
noncarbohydrate
compounds.
Required when
glycogen stores are
depleted.

14
Gluconeogenesis: Glucose
Synthesis
Carbon atoms for gluconeogenesis from lactate,
some amino acids, and glycerol are converted to
pyruvate or other intermediates.
Seven reactions are the reverse of glycolysis and
use the same enzymes.
Three reactions are not reversible.
Reaction 1 Hexokinase
Reaction 3 Phosphofructokinase
Reaction 10 Pyruvate kinase

15
Gluconeogenesis: Pyruvate to
Phosphoenolpyruvate
Pyruvate adds a carbon to form oxaloacetate by
two reactions that replace the reverse of reaction
10 of glycolysis.
Then a carbon is removed and a phosphate added
to form phosphoenolpyruvate.

16
Phosphoenolpyruvate to Fructose-
1,6-bisphosphate
Phosphoenolpyruvate is converted to fructose-
1,6-bisphosphate using the same enzymes in
glycolysis.

17
Glucose Formation
A loss of a phosphate from fructose-1,6-
bisphosphate forms fructose-6-phosphate and Pi.
A reversible reaction converts fructose-6-
phosphate to glucose-6-phosphate.
The removal of phosphate from glucose-6-
phosphate forms glucose.

18
Cori Cycle
When anaerobic conditions occur in active
muscle, glycolysis produces lactate.
The lactate moves through the blood stream to the
liver, where it is oxidized back to pyruvate.
Gluconeogenesis converts pyruvate to glucose,
which is carried back to the muscles.
The Cori cycle is the flow of lactate and glucose
between the muscles and the liver.

19
Pathways for Glucose

20
Regulation of Glycolysis and
Gluconeogenesis
High glucose levels and insulin promote glycolysis.
Low glucose levels and glucagon promote
gluconeogenesis.

21
Ethanol
Ethanol is not a carbohydrate, nor is it a precursor for the biosynthesis of carbohydrates.
However, ethanol can replace sizable amounts of carbohydrates as an energy source when large amounts are ingested.
It is present in the blood of most humans, being produced by intestinal flora.
People ingest ethanol in variable amounts in beverages and fermented fruits.
Ethanol is metabolized in the liver to acetate and adds to the caloric content of the diet.
Ethanol has an energy equivalent of 7 kcal/g.
100 mL of table wine has ethanol corresponding to about 72 kcal.
A “jigger” of whiskey furnishes approximately 120 kcal.

22
Ethanol continue: When ethanol is metabolized in the liver, alcohol dehydrogenase oxidizes it first to
acetaldehyde.
CH3CH2OH + NAD+ → CH3CHO + NADH + H+
The acetaldehyde is oxidized further to acetate.
CH3CHO + NAD+ + H2O → CH3COO- + NADH + H+
A small fraction of the alcohol may be oxidized by other systems: Cytochrome P450 oxidase (also involved in detoxification of many drugs);
Catalase
The acetate produced from ethanol largely escapes from the liver and is converted to acetyl CoA and then to carbon dioxide by the way of the Krebs cycle.
The acetyl that stays in the liver may act as a precursor for lipid biosynthesis.
A significant consequence of metabolism of ethanol in the liver is the twofold to threefold increase in the NADH/NAD+ ratio.
With higher concentrations of blood alcohol, the concentration of NADH remains high, and the availability of NAD+ drops and limits both the further oxidation of ethanol and the normal functioning of other metabolic pathways, such as gluconeogenesis.

23
“Fatty liver”
Chronic consumption of significant amounts of alcohol may lead to a “fatty liver”, in which the excess of triacylglyceride is deposited.
This is caused by several contributing factors: Reduced triacylglyceride secretion from the liver
Reduced rates of fatty acid oxidation
Increased rates of lipid biosynthesis
These processes are associated with the increased acetyl CoA and NADH/NAD+ ratio in the liver that results from ethanol oxidation.




![9[1]. Glycogen Metabolism](https://static.fdocuments.in/doc/165x107/577d346f1a28ab3a6b8e0213/91-glycogen-metabolism.jpg)