Work How Cells Work Chapter 5. Learning Objectives 1.Physics tells us that in any energy...
-
date post
21-Dec-2015 -
Category
Documents
-
view
217 -
download
0
Transcript of Work How Cells Work Chapter 5. Learning Objectives 1.Physics tells us that in any energy...
Learning Objectives
1. Physics tells us that in any energy transformation: a) energy is neither created nor destroyed, and b) there is always some energy lost in an unusable form such as heat. Explain what this means to living systems.
2. What is ATP and what is its role in cell metabolism. Describe the ATP cycle.
3. What are enzymes and what is their role in cell metabolism? What factors affect the efficiency of enzyme function? Give some examples of conditions that might interfere with normal metabolism (i.e. enzyme function).
4. Explain what is involved in redox reactions, and why this process is important in living systems.
5. Describe the relationship between photosynthesis and cellular respiration in the biosphere.
What Is Energy?• Total E =
usable E + unusable E (heat)
• Capacity to do work• Forms of energy
– Potential energy– Kinetic energy
How would you describe the energy in a chemical bond?
What type of energy drives osmosis?
Energy Transformations• Energy release is coupled
to energy-requiring
processes
• Cells use energy for:
– Chemical work
– Mechanical work
– Electrochemical work
• Reactants (describe)• Energy • Enzymes• Coenzymes/Cofactors• Products
What molecule is the only one directly used by cells to perform work?
Can some cellular processes be done without this molecule? Explain.
Energy Transformations
• First Law of Thermodynamics– The total amount of energy in the universe
remains constant– Energy can undergo conversions from one form to
another, but it cannot be created or destroyed
• Second Law of Thermodynamics– No energy conversion is ever 100 percent efficient
– The total amount of energy is flowing from high-energy forms to forms lower in energy
p. 74
One-Way Flow of Energy• The sun is life’s primary
energy source• Producers trap energy
from the sun and convert it into chemical bond energy
• All organisms use the energy stored in the bonds of organic compounds to do work
There is a tendency for increasing disorder (entropy) in the universe.
How is it possible that living systems can resist this move toward disorder?
How would you describe the molecules that contain food energy for other organisms (stable/unstable; how many bonds, etc)?
p. 74
Activation Energy
• For a chemical reaction to occur, some energy must be “invested” in the reactant(s)– Some reactions have a net
release of energy– Some reactions have a net
storage energy
• Enzymes make conditions more favorable, so less energy is needed to activate the reaction
activation energywithout enzyme
activation energywith enzyme
energyreleased
by thereaction
products
starting substance
p. 78
Enzyme Function (review)
• Highly specific• Very efficient
– speed up reaction up to 10 billion times faster
– have a maximum rate– organize metabolic pathways– Can be re-used (are
unchanged by the reaction)• Temperature & pH can affect
the natural shape of any enzyme
p. 78
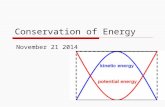
Endergonic ReactionsProduct has more energy than reactants, and
potential energy is stored.
glucose, a high energy product
ENERGY IN
low energy starting substances
6
+ 6O2
666
Name the reactants. What metabolic process does this describe?
What does “potential energy of molecules” mean?
Anabolic (molecule building) reactions, e.g. ADP + P
p. 75
Exergonic Reactions• Products have less energy than starting substance, and energy is released to fuel cell processes.
• Catabolic (breakdown) reactions (e.g. digestion, ATP use)
glucose, a high energy starting substance
+ 6O2
6 6
low energy productsENERGY OUT
Name the products. What metabolic process does this describe?
p. 75
Red-Ox Reactions• Oxidation is the loss of electrons from a molecule
– decreases its potential energy– acceptor of the electron (often associated with H) is
often oxygen (thus, described as “oxidation”)• Reduction is the gain of electrons by a molecule
– increases its potential energy• In the body, oxidation-reduction reactions are coupled &
occur simultaneously; tightly controlled by enzymes
LEO
the Lion says
GER
Loss of Electrons is Oxidation
Gain of Electrons is Reduction
p. 76
Main Metabolic Pathways in Ecosystems
glucose is oxidized, to
produce ATP
(what is the other form of energy released?)
CO2 is reduced, to
produce glucose
(where does the energy
come from to fuel these
reactions?)
p. 77
The Role of ATP
Cells “earn” ATP in exergonic reactions
Cells “spend” ATP in endergonic reactionsName some examples of ‘cellular work”. Are these energy transformations completely efficient?
p. 75