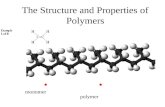
What is the difference between a monomer and polymer? Give an example of each. #1 - Question.
-
Upload
janel-golden -
Category
Documents
-
view
232 -
download
0
Transcript of What is the difference between a monomer and polymer? Give an example of each. #1 - Question.

What is the difference between a monomer and polymer?Give an example of each.
#1 - Question

A monomer is a small molecule that may bind chemically to other monomers to form a polymer.
A polymer is a large molecule (macromolecule) composed of repeating structural units typically
connected by covalent chemical bonds.
The most common monomer is glucose, which is linked by glycosidic bonds into polymers such as cellulose, starch, and glycogen. Amino acids are
monomers linked by peptide bonds to form polymers called polypeptides.
#1 - Answer

What are the subunits of a triglyceride molecule?
What is the bond called between the subunits?
#2 - Question

Three fatty acids (can be saturated or unsaturated) and
a glycerol molecule.
The fatty acids are bonded to the glycerol backbone by
ester bonds.
#2 - Answer
Glycerol Fatty acids
unsaturated
saturated
saturated

What are the subunits of a nucleic acid?
#3 - Question

Chains of Nucleotides:
- Phosphate- Sugar
- Nitrogenous base
#3 - Answer

What is the difference between monosaccharides, disaccharides
and polysaccharides?Give an example of each.
#4 - Question

Monosaccharides, like glucose, fructose, and galactose are the
building blocks of disaccharides (a “two-sugar” containing
molecule) such as maltose, sucrose, lactose and
polysaccharides, such as cellulose, starch and glycogen
(“many-sugar” containing molecule).
#4 - Answer

What is primary source of energy used by cells?
#5 - Question

Carbohydrates
i.e., glucose
#5 - Answer

Describe what happens during a condensation synthesis reaction between two glucose molecules.
What are the products of this reaction?
#6 - Question

One glucose molecule is joined to another glucose molecule through an interaction of the hydroxyl (-OH) functional groups.
Water (H2O) is removed and maltose (a disaccharide) is formed.
#6 - Answer

Describe what happens during a hydrolysis reaction for the disaccharide sucrose.
What are the products of this reaction?
What enzyme catalyzes this reaction?
#7 - Question

The disaccharide sucrose is broken down into glucose and
fructose molecules by the addition of a water (H2O)
molecule. The hydroxyl (-OH) groups are reformed. This
reaction is catalyzed by the enzyme sucrase.
#7 - Answer

Which macromolecule(s) is/are formed by a condensation
synthesis reaction?
#8 - Question

Carbohydrates
Lipids
Proteins
#8 - Answer

Which macromolecule(s) is/are broken down by a hydrolysis
reaction?
#9 - Question

Carbohydrates
Lipids
Proteins
#9 - Answer

What is the ratio of carbon, hydrogen, and oxygen atoms in a carbohydrate molecule?
Explain using the molecular formula of glucose as an example.
#10 - Question

C : H : O 1 : 2 : 1
Glucose C6H12O6
#10 - Answer

Which chemical formula represents a unsaturated acid? How do you know?
a) C17H35COOH b)C17H33COOH
What state will this lipid be at room temperature. Give a food example.
#11 - Question

b) C17H33COOH; there is a double bond between one of the carbon molecules which reduces the number of bonded hydrogen atoms. This lipid will be liquid at
room temperature; for example – olive oil or vegetable oil
#11 - Answer
C17H33COOH

What are the subunits of a phospholipid?
Where are phospholipids found in the cell?
#12 - Question

Two fatty acids, a glycerol backbone, a phosphate group and a nitrogen containing group.
Phospholipids are an important part of the cell membrane.
#12 - Answer
Nitrogen-containing group

Identify which parts of the phospholipid
are hydrophilic and hydrophobic.
#13 - Question

#13 - Answer
Polar Head hydrophilic (water-loving)Non-Polar Tails (fatty acids)
hydrophobic (water-hating)

Identify (circle and label) all of the functional groups present in each
molecule
#14 - Question

#14 - AnswerHydroxyl group
Carbonyl - ketoneCarboxylAmine
Carbonyl - aldehyde

Name these subunits and the macromolecules they belong to.
#15 - Question
(A) (B) (C) (D)

#15 – Answer
(A)
(B)
(C)
(D)
Amino acid
Protein
Glycerol
Lipids (triglyceride or phospholipid
Fatty acid
Lipids (triglyceride or phospholipid)
Glucose
Carbohydrate (starch, cellulose, glycogen)

+
H2O
#16 - Question
What type of reaction is
shown here?
What are the products?

#16 - Answer
+
H2O
Hydrolysis Reaction
Dipeptide and water (H2O)

#17 - Question
What are the nitrogen bases called in a DNA molecule and how do
they pair up?

#17 - Answer
AdenineGuanineThymineCytosine
Adenine must pair with Thymine
Guanine must pair with Cytosine

#18 - Question
Match the nucleotides with the correct label
a)
b)
c)
-Sugar-Phosphate-Nitrogen base

#18 - Answer
a)
b)
c)
c) Sugara)Phosphateb)Nitrogen base

#41 - Question
Is the nitrogen base shown here a purine or a pyrimidine?
Phosphate
Sugar

#41 - AnswerPurine – double ring
Pyrimidine – single ring
Phosphate
Sugar
Phosphate
Sugar

#19 - Question
If the blood is too acidic, this reaction moves to the left or to the right?
CO2
H2O
H+H+
H2CO3 (aq)
H+
H+
carbonic acid
H+
surplus hydroge
n ion
HCO3-
(aq)
H+
bicarbonate ion

CO2
H2O
H+H+
H2CO3 (aq)
H+
H+
carbonic acid
H+
surplus hydroge
n ion
HCO3-
(aq)
H+
bicarbonate ion
If blood too acidic ( H+), rxn moves left
#19 - Question

#20 - Question
If the blood is too basic, this reaction moves to the left or to the right?
CO2
H2O
H+H+
H2CO3 (aq)
H+
H+
carbonic acid
H+
surplus hydroge
n ion
HCO3-
(aq)
H+
bicarbonate ion

CO2
H2O
H+H+
H2CO3 (aq)
H+
H+
carbonic acid
H+
surplus hydroge
n ion
HCO3-
(aq)
H+
bicarbonate ion
#20 - Question
If blood too basic ( H+), rxn moves right

#21 - Question
Nutrient Test
Based on the nutrient test conducted, is sample E most likely to be steak, bread, or butter?

#21 - Answer
Steak – sample E tested positive for protein and fat.

#22 - Question
Nutrient Test
Based on the nutrient test conducted, is sample G most likely to be table sugar, pasta, or lard?

#22 - Answer
Table sugar – sample G tested negative for monosaccharides but may contain disaccharides like sucrose.

#23 - Question
Match the enzymes with their specific substrates.
a)Proteaseb)Phosphatasec)Lipased)Amylasee)Lactasef) Sucrase
1. Chicken2. Table sugar (sucrose)3. Triglyceride4. Starch5. Phosphate6. Milk

#23 - Answer
a)Proteaseb)Phosphatasec)Lipased)Amylasee)Lactasef) Sucrase
1. Chicken2. Table sugar (sucrose)3. Triglyceride4. Starch5. Phosphate6. Milk

#24 - Question
Which of the foll0wing correctly represents the mechanism of enzyme function?a)S + P E-P E +Pb)E + P E-P E-S -> E + Sc)E + P E-S E-P E + Pd)E + S E-S E-P E + Pe)E + S E-P E-S E + S
Where:E = enzymeS = substrateP = product

#24 - Answer
a)S + P E-P E +Pb)E + P E-P E-S -> E + Sc)E + P E-S E-P E + Pd)E + S E-S E-P E + Pe)E + S E-P E-S E + S
Enzyme + substrate forms an enzyme-substrate complex the substrate is converted to a product by the enzyme the product is released from the enzyme

#25 - Question
Which factor(s) denature enzymes?
a)Cookingb)NaOHc)H+ ionsd)All of the above

#25 - Answer
a)Cooking (high heat)b)NaOH (basic solution)c)H+ ions (acidic solution)d)All of the above

#26 - Question
You are testing a new inhibitor of catalase – sodium fluoride (liquid). This inhibitor is structurally different from hydrogen peroxide. Using the assay technique from the enzyme lab, you found that no reaction occurred when you incubated the enzyme with the substrate in the presence of the inhibitor.
Is the inhibitor acting via competitive or non-competitive inhibition? How do you know?

#26 - AnswerIf the inhibitor is structurally different from hydrogen peroxide then it will not be competing with the substrate for a position on the enzyme’s active site.
Therefore, the inhibitor is acting via non-competitive inhibition – specifically via allosteric inhibition. The inhibitor bound to the allosteric site which change the shape of the active site causing the enzyme to become deactivated and stop the reaction altogether, or cause a reduced affinity for the substrate (decreased attraction) and slow down the reaction.

#27 - Question
What is an example of an organic co-factor?

#27 - Answer
NAD or FAD
Active site
Enzyme
Coenzyme(detaches)
Coenzyme
e.g., NAD
Active site
Enzyme
Prosthetic group(perm. attached)
Prosthetic Group
e.g., FAD

#28 - Question
How does an enzyme affect activation energy?

#28 - Answer
Enzymes catalyze biochemical reactions by lowering the amount of energy required to activate the reaction.

#29 - Question
Name two structures that can
be identified
on the HSL enzyme.

#29 - Answer
Beta-pleated sheets
Alpha helices

#30 - Question
What is one difference and one similarity between competitive and non-competitive enzyme
inhibition?

#30 - Answer
Competitive
Non-competitive
Normal enzyme reaction

#31 - Question
What is one similarity and one difference between facilitated diffusion and active transport via the
Na+/K+ pump?

#31 - Answer
Similarities• Both move polar
molecules across the membrane
• Both use highly selective carrier proteins that bind to the molecules
Differences• Facilitated diffusion does not
require ATP to move molecules across the membrane and the Na+/K+ pump does
• Facilitated diffusion and the Na+/K+ pump move molecules from [high] to [low] and [low] to [high], respectively

#32 - Question
Describe the six steps of the Na+/K+ pump

#3
2 -
Answ
er

#33 - Question
How is direct diffusion and protein channel diffusion the
same and different?

#33 - Answer
Similarities• Both move
molecules down their concentration gradient from [high] to [low]
• Both are passive processes
Differences• Direct diffusion is the
movement of non-polar molecules across the membrane, while protein channel diffusion is the movement of polar molecules through a hydrophilic protein channel that spans the membrane.

#34 - Question
How is protein channel and facilitated diffusion the same
and different?

#34 - Answer
Similarities• Both move polar
molecules down their concentration gradient from [high] to [low]
• Both are passive processes
Differences• Protein channel diffusion is not as
selective with respect to the type of molecules that pass through the channels. Facilitated diffusion via protein carriers involves the binding of specific molecules to protein carriers that are highly specific (like enzymes are with their substrates)
• The specific nature of facilitated diffusion is way for the cell to tightly regulate the entry or exit of certain molecules through the principle of saturation. When all protein carriers are occupied, the rate at which molecules can pass through is held constant (much like ticket holders entering a stadium through packed turn style gates)

#35 - Question
How is the Na+/K+ pump involved in helping to facilitate coupled
transport?

#3
5 -
Answ
er

#36 - Question
How is endocytosis and exocytosis the same and
different from other active processes (e.g., Na+/K+
pump and coupled transport ?

#36 - AnswerSimilarities• Like the Na+/K+ pump
and coupled transport, receptor-mediated endocytosis involves the use of a proteins to help move across the mombrane
• Both are active processes that require ATP
Differences• Endocytosis and exocytosis transport
large polar molecules in bulk amounts across the membrane
• Phagocytosis and pinocytosis do not use proteins to move molecules across the membrane, but instead involve the membrane enveloping around molecules or liquids to bring them in
• Endocytosis (all types) only bring in materials, while the Na+/K+ pump and coupled transport bring molecules in and out of the cell

#37 - Question
How is phagocytosis
and pinocytosis the same
and different?

#37 - AnswerSimilarities• Both involve the bulk
movement of molecules
• Both move bulk amounts of molecules into the cell
Differences• Phagocytosis moves
particles into the cell (phagein = “to eat”), while pinosytosis moves liquid into the cells (pinein = “to drink”)

#38 - Question
What is the main
difference between
endocytosis and
exocytosis?

#38 - Answer
Endycytosis moves molecules into the cell, while exocytosis moves molecules out of the cell.

#39 - Question
Explain the process of osmosis.

#3
9 -
Answ
er

#40 - Question
Explain how the movement of water will be affected in an
animal cell under hypotonic, isotonic, and hypertonic
conditions.

#4
0 -
Answ
er

#42 - Question
Explain how electronegativity is related to molecular
polarity, and why this is important with respect to
functional groups and macromolecules.

#42 - AnswerElectronegativity is a measure of the attraction of an atom for electrons in a covalent bond (like a game of tug-of-war). The electronegative difference (ΔEN) between two atoms can be used to predict the bond type formed (e.g., <0.5 = non-polar covalent, 0.5-1.7 = polar-covalent, >1.7 = ionic).
ΔEN = larger minus (-) smaller electronegative numbere.g., oxygen(3.5) – hydrogen(2.1) = 1.4 (polar-covalent; oxygen δ-, hydrogen δ+)
Functional groups are groups of polar molecules that are attached to macromolecules. They are involved in reactions to make or break macromolecules and help make them soluble in the aqueous environments of human body cells and the blood.