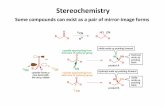
Isomers: Different Compounds With the Same Molecular Formula Constitutional Isomers:
Types of Isomers. Enantiomers Enantiomers are nonsuperimposable mirror images. Molecules are...
-
Upload
opal-roberts -
Category
Documents
-
view
219 -
download
2
Transcript of Types of Isomers. Enantiomers Enantiomers are nonsuperimposable mirror images. Molecules are...

Types of Isomers

Enantiomers
• Enantiomers are nonsuperimposable mirror images.
• Molecules are nonsuperimposable if there is no orientation in which all atoms of both molecules can be superimposed.

Ibuprofen
R formnon-activeside effects
S formactive
anti-inflammatory

Thalidomide is now being used to treat plasma cell cancer, leprosy, and has shown anti-HIV activity

Chirality• A molecule is chiral if it has an enantiomer.
– Does NOT have mirror symmetry
• A molecule is achiral if it does not have an enantiomer.
– DOES have mirror symmetry

Terminology in Stereochemistry
• Stereocenter– An atom about which exchange of 2 groups
produces a stereoisomer
• Chiral Center– Type of Stereocenter– A tetrahedral atom that is bonded to four
different groups


Fischer Projections
• The Fischer projection is a convenient way to depict complex molecules having more than one stereocenter.
• The intersection of a horizontal line and a vertical line indicates a carbon stereocenter.
• The substituents on the horizontal bonds are understood to point toward you (like a bowtie), whereas the substituents on the vertical bonds are understood to point away from you.

Chirality and Conformational Isomers
• Rotation about single bonds can determine whether a molecule is chiral or achiral.
• If a molecule and its mirror image are rapidly interconverting conformational isomers, then the molecule is effectively achiral.
• The mirror image of 1,2-dibromoethane in one of its gauche conformations is its second gauche conformation.

Haworth Projections
• Haworth projections are helpful in determining the chirality of all cyclics.
• Taking the chair conformation of cyclohexane to a flat hexagon allows us to quickly and accurately determine whether it is chiral or achiral.

biphenyls
has been resolvedhalf-life for racemizationis 78 min at 118oC
“Frozen” Conformational Isomers

Physical and Chemical Properties of Isomers
Constitutional Isomers
Due to different connectivities, these isomers must have different physical and chemical properties.
Enantiomers
Have the same connectivities and precisely the same polarities.
Diastereomers
Have the same connectivities but different 3-D arrangement, so they have different properties.

Optical Activity
• Chiral molecules interact with plane-polarized light.
• When all photons from a light source have their electric fields oscillating in the same plane, then the light is plane polarized.

The Polarizer• Most light sources emit light that is unpolarized.• A polarizer generates plane-polarized light by
filtering out light whose electric field oscillates in any other plane.
• If plane-polarized light passes through a sample of a compound, the plane in which the light is polarized can change, depending upon whether the compound is chiral or achiral.

Enantiomers rotate Plane Polarized Light
• One enantiomer rotates polarized light in one direction while the other enantiomer rotates it in the opposite direction.
• Enantiomers have identical physical and chemical properties except the direction at which they rotate polarized light.


Louis Pasteur
• Louis Pasteur was the first to isolate a pair of enantiomers from each other.
• Pasteur noted that the crystals appeared to grow in one of two varieties—left-handed crystals and right-handed crystals—that are mirror images of each other
• Pasteur physically separated the two types of crystals using tweezers.

Option for Separating Enantiomers1.Pasteur Method
2.Conversion to Diastereomers1. Temporarily convert the enantiomers into a pair of diastereomers (will
now have different physical properties).
2. Separate those diastereomers from each other by exploiting their different physical and chemical properties.
3. Regenerate the enantiomers from the separated diastereomers.
3.Enzymes (version of #2)
4.Chiral Chromatography (version of #2)
Separating (Resolving) Enantiomers

enantiomers to be separated
one enantiomer of a chiral amine
diastereomeric salts
one diastereomer
one pure enantiomer