Thrombotic Microangiopathies and AntiPhospholipid Syndrome
-
Upload
richard-mccrory -
Category
Health & Medicine
-
view
1.385 -
download
1
description
Transcript of Thrombotic Microangiopathies and AntiPhospholipid Syndrome

Thrombotic Microangiopathies
Richard McCrory

Objectives• Thrombotic Microangiopathy: Pathology – 5 mins
• The Alternative Complement Pathway – 5 minutes
• STEC related HUS – 5 mins• Atypical HUS – 5 mins• TTP and ADAMTS13 – 5 minutes
• Treatment and Transplantation in TMA – 10 minutes• Antiphospholipid Syndrome – 10 minutes

Thrombotic Microangiopathy
Describes a process comprising:
1) Consumptive Thrombocytopenia
2) Microangiopathic Haemolytic Anaemia
3) Microvascular Thrombosis

Clinical Spectrum of TMA
• Haemolytic uremic syndrome• Thrombotic thrombocytopenic purpura• Systemic sclerosis• Malignant hypertension• Preeclampsia – eclampsia• Systemic lupus erythematosus• Antiphospholipid antibody syndrome• Renal transplant associated• Drug induced• Radiation therapy associated

Pathophysiology
Endothelial Cells key players in pathogenesis of TMA
All TMA biopsies reflect endothelial injury and thrombus formation

General Lab Findings in TMA• Full blood count
– Severe thrombocytopenia and Anaemia• Blood film
– Red cell fragmentation (‘schistocytosis’ >1%)– Reticulocytosis– Absent or Giant platelets
• Coombs test– Negative
• Haemolysis screen– Elevated LDH– Serum haptoglobins low
• Liver enzymes and coagulation screen: Normal• Creatinine: Elevated in renal involvement

Pathological Findings in TMA
TMA
Glomerular Vascular

Glomerular Lesions
Acute
Glomerular intracapillary thrombosis,– Accumulation of fragmented erythrocytes– Focally ischaemic or congested glomerular
tufts.
Chronic• Membranoproliferative pattern with mesangial
interposition, double contours of the GBM, and prominent mesangiolysis


Glomerular Tuft

Immunofluorescence

Bloodless glomeruli in TMA

Double Contours on Silver Stain

Vascular Changes
• Acute
• Thrombotic Occlusion of medium vessels

Vascular Changes
• Chronic– Vessel-wall sclerosis, intraluminal organizing
thrombi– Elastic lamina destruction with resultant
aneurysmal dilatation of the vessel wall


The Alternative Complement Pathway


The Alternative Complement Pathway

Regulation of C3 ‘Tickover’
MCP – Membrane Cofactor Protein; found on all endothelial tisues

Haemolytic Uraemic Syndrome

HUS: Epidemiology
Prevalence: 2 per 100,000 persons per year– Peaks in children <5 yrs old
Shiga Toxin producing E. Coli (STEC) contributes 90% of cases worldwide– Can be sporadic or epidemic

Shiga Toxin

Pathogenesis of Shiga Toxin (1)
Localises to endothelium via globotriaosylceramide (Gb3)– Alpha subunit = N-glycosidase = Protein
Shutdown
Endothelial cell damage– In the gut →Bloody Diarrhoea– In the kidney → Intravascular thrombogenesis

Pathogenesis of Shiga Toxin (2)
Shiga toxin can de-regulate complement activation
Binds to Factor HOrth et al. Journal of Immunology (2009)
Encourages expression of P-selectin on endothelium, attracting C3a
Factor B deficient mice demonstrate less severe disease with STEC organisms
Morigi et al. Journal of Immunology (2011)

E.Coli O157:H7 Outbreaks
Nov – Dec 1996 - Lanarkshire21 elderly people die and another 500 were
made ill eating contaminated meat
2006 CaliforniaLinked to Spinach consumption from fields
contaminated with sewage water.205 total cases, with 31 HUS cases, for an HUS
rate of about 15%.

Shiga-Toxin–Producing Escherichia coli O104:H4 Outbreak in Germany
3816 lab confirmed cases
857 cases involved HUS
54 deaths
Frank et al. (NEJM 2011)


Question
Which of these causes of TMA can be associated with a Coombs positive test?
Shiga Toxin AssociatedInvasive Pneumococcal InfectionCalcineurin InhibitorTiclopidineQuinine

Pneumococcus associated HUS
• Streptococcal Neuramidase exposes a novel antigen [Thomsen-Freidenreich (T) Antigen] on RBCs/Platelets/ Glomeruli
• Antibodies lead to cell aggregation and deposition in small vessels

‘Atypical’ HUS
Non-STEC, Non-Pneumococcal
• Genetic and acquired factors leading to dysregulation of the alternative complement pathway.
• Much rarer (~1000 cases in registry data from Europe/US)

Mutations in aHUS Registries
Factor HMCPFactor IC3Factor BHybrid GenesCombinedFactor H antibody
The presence of a mutation in an individual with aHUS does not prove causation

Testing for Complement Dysregulation
C3 /C4Factor H, factor I, factor BMCP (CD46) expression on PBMCsFactor H autoantibodies
Mutations• Direct exon sequencing of CFH, MCP,
CFI, CFB, C3• Copy number variation across CFH-CFHR
locus

Thrombotic Thrombocytopenic Purpura

TTP• Prevalence 1 in 50,000 hospitalisations• Mortality previously >90%, now around 20-30% with current therapy
The ‘classical’ pentad• Neurological findings – 75% of patients• Neurological Findings / Haemolytic Anaemia / Thrombocytopaenia –
74%• Fever / Renal Impairment – 40%• Elevated creatinine is a poor prognostic indicator

Pathophysiology
Reduced activity of the ADAMTS13 cleaving protease with consequent disruption of von Willebrand factor multimer processing– Can be congenital or ‘functional’

TTP and Complement Activation
• Increased C3a activity detected in TTP flares
• C3a activity declines with plasma exchange treatment
Réti et al J Thromb Haemost.(2012)

Treatment and Transplantation

Treatment
Supportive Management• Avoid antibiotics in STEC infection -
increases HUS 17-fold• Fluid balance, fluid resusciation,
electrolytes, early RRT if indicated• Avoid platelet transfusion

Treatment
• ‘Urgent Empirical Plasma Exchange’– Commonly when STEC/Pneumococcal
Negative• May have a role in severe adult cases of STEC-
HUS but controversial– Recommended in all other HUS/TTP settings

Transplantation
All forms of renal transplant available for:– STEC-HUS– MCP-aHUS
Other aHUS phenotypes– Living-related donation ‘contraindicated’– Unknown risk factors within family probands– High risk recurrence in grafts with transplant
failure common.

Liver/Kidney Transplantation
May be considered as a solution to deficiencies in complement proteins
Performed with limited success in paediatric cases.

Eculizumab – The Great White Hope?
• Monoclonal Antibody to C5• Prevents formation of membrane attack
complexes
• Multiple Case series in aHUS– Also used in STEC-HUS during German
epidemic outbreak– Use in transplantation to prevent graft
recurrence

Antiphospholipid Syndrome

Common autoimmune diseases associated with APL antibodies
SLE -25-50%
Sjogren’s – 42%
Rheumatoid Disease - 33%
Autoimmune Thrombocytopenic Purpura - 30%
PA - 28%
Systemic Sclerosis - 25%
Mixed Connective Tissue Disease - 22%
Bechet - 20%
Polymyalgia Rheumatica - 20%

Pathogenesis of APL
Homeostatic regulation of blood coagulation is altered.
Exposure of membrane phospholipids to the binding of various plasma proteins (b2gp1)
Production of antibodies against prothrombin, Protein C/ Protein S and Annexins.
Activation of:– platelets to enhance endothelial adherence.– vascular endothelium—platelet and monocyte binding.
Antibodies against oxidized LDL—atherosclerosis.
Complement activation has been increasingly recognized as a possible significant role in the pathogenesis of APS.
Blood. Jan 15 2007;109(2):422-30.
Nat Med. Nov 2004;10(11):1222-6.

The role of Complement in Miscarriage


The renal manifestations of APS
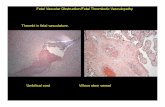
Systemic hypertension in association with livedo reticularis
Renal Arterial Stenosis
Renal infarction
APS-associated nephropathy
Acutely – Thrombotic Microangiopathy
Chronic - Arteriosclerosis, Fibrous intimal hyperplasia, focal cortical atrophy
Renal vein thrombosis and increased allograft vascular thrombosis

Antiphospholipid syndrome (APS) - Diagnosis
• At least one clinical and one laboratory criteria• Clinical – vascular thrombosis or pregnancy
morbidity– ≥ 1 venous, arterial or small vessel thrombosis with
unequivocal imaging or histology– Unexplained fetal death ≥ 10wks gestation– One or more premature births before 34wks gestation
because of eclampsia, pre-eclampsia or placental insufficiency
– ≥ 3 pregnancy losses unexplained by chromosomal, maternal or hormonal causes

APS - diagnosis
• Laboratory criteria
• The presence of APL ABs on ≥ 2 occasions at least 12 wks apart
• No more than 5 yrs prior to clinical manifestations– Anticardiolipin Ab in moderate/high titre– Β2-glycoprotein 1 Ab elevation– Lupus anticoagulant (LA) activity ( APTT in vitro,
not corrected with normal plasma)

Catastrophic Antiphospholipid Syndrome(CAPS)
• A syndrome of multisystem involvement (<1% of patients)
• Acute onset– 3 different organ systems within 2 weeks
– Acute microangiopathy is characteristic
TriggersInfection (25-30%)
Surgery / Trauma (10%)
Anticoagulation Withdrawal (10%)


SLE with antiphospholipid Abs
risk of thromboembolism & recurrent pregnancy loss• 20-50% met criteria for APS at 10-20 yrs• The higher the ACL Ab titre the greater risk of
thrombosis• Aspirin if cardiovascular or genetic risk factors for
thromboembolic disease• Address cardiovascular risk factors• Avoid OCP - esp if high oestrogen content• SLE pts should be regularly assessed for APL Abs
(ACL Ab, β2GP1 Ab, LAC)• Aspirin and HCQ prophylaxis for those with persistent
LAC or ACL Ab at medium/high titre

TMA: Take Home Points
Pathological findings similar – causes are multiple
Unifying pathological event appear to be related to dysregulation of the alternative complement pathway
Treatments aiming to stop the complement cascade appear to promising targets for therapy