The Stuff of Everything… …even water and life. What is matter? Three Properties of Matter: 1....
-
Upload
herbert-horn -
Category
Documents
-
view
214 -
download
0
Transcript of The Stuff of Everything… …even water and life. What is matter? Three Properties of Matter: 1....

The Stuff of Everything……even water and life

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Three Properties of Matter:
1. has mass2. takes up space3. shows inertia
(force needed to change motion)

What is matter?Four states of matter:
1.Solid2.Liquid3.Gas4.Plasma

What is matter?Four states of matter:
1.Solid2.Liquid3.Gas4.Plasma

What is matter?Four states of matter:
1.Solid2.Liquid3.Gas4.Plasma

What is matter?Four states of matter:
1.Solid2.Liquid3.Gas4.Plasma

What is matter?Four states of matter:
1.Solid2.Liquid3.Gas4.Plasma

What is matter?Four states of matter:
1.Solid2.Liquid3.Gas4.Plasma

Can matter change?Matter can change in two ways:
1. Physical – a change in the appearance or state of matter
2. Chemical – a change in the chemical make-up of matter

Can matter change?Matter can change in two ways:
1. Physical – a change in the appearance or state of matter
2. Chemical – a change in the chemical make-up of matter

Can matter change?Matter can change in two ways:
1. Physical – a change in the appearance or state of matter
2. Chemical – a change in the chemical make-up of matter

Can matter change?Matter can change in two ways:
1. Physical – a change in the appearance or state of matter
2. Chemical – a change in the chemical make-up of matter

Can matter change?Matter can change in two ways:
1. Physical – a change in the appearance or state of matter
2. Chemical – a change in the chemical make-up of matter

Can matter change?Matter can change in two ways:
1. Physical – a change in the appearance or state of matter
2. Chemical – a change in the chemical make-up of matter

Can matter change?Matter can change in two ways:
1. Physical – a change in the appearance or state of matter
2. Chemical – a change in the chemical make-up of matter

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

Can matter change?
P Melting Ice P Water Freezing P
Water Evaporatin
g
C Burning Wood C Rotting
Wood PMaking
Glass from Sand
C Rusting Iron C Souring
Milk P Water Boiling
C Animal Decaying C
Enzymes Digesting
FoodP
Salt Dissolving in Water

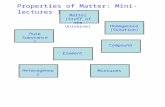
What is matter made of?Composition of matter:
1. Elements2. Compounds3. Mixtures

What is matter made of?Composition of matter:
1. Elements2. Compounds3. Mixtures

What is matter made of?Composition of matter:
1. Elements2. Compounds3. Mixtures

What is matter made of?Composition of matter:
1. Elements2. Compounds3. Mixtures

What is matter made of?Composition of matter:
1. Elements2. Compounds3. Mixtures

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?An element is a basic substance of matter and cannot be broken down by chemical means.
There are about 90 naturally occurring elements. 117 have been discovered.

What is an element?Elements are represented by chemical symbols (letters) shown in the Periodic Table of the Elements.

What is an element?Elements are represented by chemical symbols (letters) shown in the Periodic Table of the Elements.

What is an element?Elements are represented by chemical symbols (letters) shown in the Periodic Table of the Elements.

What is an element?Elements are represented by chemical symbols (letters) shown in the Periodic Table of the Elements.

Common Elements of LifeName Symbol Name Symbol
Hydrogen H Phosphorus P
Oxygen O Potassium K
Carbon C Sulfur S
Sodium Na Calcium Ca
Chlorine Cl Iron Fe
Nitrogen N Magnesium Mg

What is an element?The smallest unit of an element is an atom.
Atoms are the building blocks of matter.

What is an element?The smallest unit of an element is an atom.
Atoms are the building blocks of matter.

What is an element?The smallest unit of an element is an atom.
Atoms are the building blocks of matter.

What is an element?The smallest unit of an element is an atom.
Atoms are the building blocks of matter.

What is an element?The smallest unit of an element is an atom.
Atoms are the building blocks of matter.

What is an atom?
ElectronProton
Neutron
Nucleus
Atom

What is an atom?
ElectronProton
Neutron
Nucleus
Atom

What is an atom?
ElectronProton
Neutron
Nucleus
Atom

What is an atom?
ElectronProton
Neutron
Nucleus
Atom

What is an atom?
ElectronProton
Neutron
Nucleus
Atom

What is an atom?
ElectronProton
Neutron
Nucleus
Atom

What is an atom?Atoms have 3 subatomic particles:Particle Charge Mass Function Symbol Location
Proton + 1 Add mass
p+ Nucleus
Neutron 0 1 Add mass
N Nucleus
Electron - 0 Bonding e- Electron Cloud

What is an atom?Electrons orbit atoms in levels called shells.
Atoms like to have their shells filled.

What is an atom?Electrons orbit atoms in levels called shells.
Atoms like to have their shells filled.

What is an atom?Electrons orbit atoms in levels called shells.
Atoms like to have their shells filled.

What is an atom?Electrons orbit atoms in levels called shells.
Atoms like to have their shells filled.

What is an atom?K or 1st Shell – closest to the nucleus holding up to 2 electrons
L or 2nd Shell – can hold up to a maximum of 8 electrons
M or 3rd shell – can hold up to a maximum of 18 electrons

What is an atom?K or 1st Shell – closest to the nucleus holding up to 2 electrons
L or 2nd Shell – can hold up to a maximum of 8 electrons
M or 3rd shell – can hold up to a maximum of 18 electrons

What is an atom?K or 1st Shell – closest to the nucleus holding up to 2 electrons
L or 2nd Shell – can hold up to a maximum of 8 electrons
M or 3rd shell – can hold up to a maximum of 18 electrons

What is an atom?K or 1st Shell – closest to the nucleus holding up to 2 electrons
L or 2nd Shell – can hold up to a maximum of 8 electrons
M or 3rd shell – can hold up to a maximum of 18 electrons

What is an atom?K or 1st Shell – closest to the nucleus holding up to 2 electrons
L or 2nd Shell – can hold up to a maximum of 8 electrons
M or 3rd shell – can hold up to a maximum of 18 electrons

What is an atom?K or 1st Shell – closest to the nucleus holding up to 2 electrons
L or 2nd Shell – can hold up to a maximum of 8 electrons
M or 3rd shell – can hold up to a maximum of 18 electrons

What is an atom?K or 1st Shell – closest to the nucleus holding up to 2 electrons
L or 2nd Shell – can hold up to a maximum of 8 electrons
M or 3rd shell – can hold up to a maximum of 18 electrons

What is an atom?The number of protons (atomic number) in an atom will determine its elemental properties.
The periodic table of the elements shows known elements by their atomic number.

What is an atom?The number of protons (atomic number) in an atom will determine its elemental properties.
The periodic table of the elements shows known elements by their atomic number.

What is an atom?The number of protons (atomic number) in an atom will determine its elemental properties.
The periodic table of the elements shows known elements by their atomic number.

What is an atom?In atoms, the number of protons equals the number of electrons giving the atom an overall neutral charge.
The number of protons plus neutrons in an atom is the atomic mass number.

What is an atom?In atoms, the number of protons equals the number of electrons giving the atom an overall neutral charge.
The number of protons plus neutrons in an atom is the atomic mass number.

What is an atom?In atoms, the number of protons equals the number of electrons giving the atom an overall neutral charge.
The number of protons plus neutrons in an atom is the atomic mass number.

What is an atom?In atoms, the number of protons equals the number of electrons giving the atom an overall neutral charge.
The number of protons plus neutrons in an atom is the atomic mass number.

What is an atom?In atoms, the number of protons equals the number of electrons giving the atom an overall neutral charge.
The number of protons plus neutrons in an atom is the atomic mass number.

Using The Periodic Table1
HHydrogen
1.0079
Atomic Number
Element Symbol
Element Name
Atomic Mass

Using The Periodic Table1
HHydrogen
1.0079
Atomic Number
Element Symbol
Element Name
Atomic Mass

Using The Periodic Table1
HHydrogen
1.0079
Atomic Number
Element Symbol
Element Name
Atomic Mass

Using The Periodic Table1
HHydrogen
1.0079
Atomic Number
Element Symbol
Element Name
Atomic Mass

Using The Periodic Table1
HHydrogen
1.0079
Atomic Number
Element Symbol
Element Name
Atomic Mass

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic Table
Atomic Number Mass Number
Number of protons Number of electrons Number of Neutrons
= 8= 16= 8= 8= 8
88

Using The Periodic TableElement Atomic
NumberMass Number
# of protons
# of electrons
# of neutrons
Calcium 20 40 20 20 20Silver 47 108 47 47 61Gold 79 197 79 79 118Uranium 92 238 92 92 146