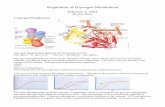
Regulation of glycogen metabolism
-
Upload
namrata-chhabra-md -
Category
Education
-
view
4.352 -
download
4
Transcript of Regulation of glycogen metabolism

05/01/2023 NAMRATA CHHABRA, M.D. 1
Regulation of Glycogen metabolism
NAMRATA CHHABRA, M.D., BIOCHEMISTRY
Glycogen metabolism-Part-3

05/01/2023 NAMRATA CHHABRA, M.D. 2
Learning objectivesTo understand :Purpose of regulation of glycogen metabolismProcesses involved and the reciprocal regulation of glycogenesis and glycogenolysis

05/01/2023 NAMRATA CHHABRA, M.D. 3
General mechanisms involved in the regulation of enzyme activities
Regulation of enzyme activity
Covalent modification
Allosteric modification
Substrate/product concentration
Induction/Repression

05/01/2023 NAMRATA CHHABRA, M.D. 4
Key enzymes involved in the regulation of glycogen metabolism
Glycogen synthase- For Glycogenesis
Glycogen Phosphorylase
Both these enzymes are reciprocally regulated.

05/01/2023 NAMRATA CHHABRA, M.D. 5
Substrate concentration and allosteric modification
Substrate- Glucose-6-P Glycogen Synthase is allosterically activated by glucose-6-P. High blood glucose concentration leads to elevated intracellular glucose-6-P. When glycolytic pathway is saturated, excess glucose-6-P activates Glycogen synthase and thus is stored as glycogen.
High Blood Glucose
High Glucose-6-P
Saturated Glycolytic pathway
GlycogenSynthase
v
Glycogenesis

05/01/2023 NAMRATA CHHABRA, M.D. 6
Covalent modification- General concepts Reversible phosphorylation and dephosphorylation Hormone mediated C-AMP mediated cascadePhosphorylation is mediated by Protein kinase ADephosphorylation is carried out by PhosphataseInsulin causes dephosphorylation by stimulating Phosphatase and Phosphodiesterase (enzyme that breaks down cAMP)Glucagon causes phosphorylation by stimulating Protein kinase A

05/01/2023 NAMRATA CHHABRA, M.D. 7
Regulation of glycogen synthase by covalent modification
Glycogen synthase exists in both phosphorylated or dephosphorylated statesActive glycogen synthase a is dephosphorylated and inactive glycogen synthase b is phosphorylated

05/01/2023 NAMRATA CHHABRA, M.D. 8
Covalent modification of glycogen synthase
Glycogen synthase
a
Glycogen synthase
b
Phosphatase Protein kinase A
ATPPi
H2O ADP
Active
Inactive
p

05/01/2023 NAMRATA CHHABRA, M.D. 9
Mechanism of Phosphorylation of glycogen synthase
The cAMP cascade results in phosphorylation of a serine hydroxyl of Glycogen synthase, which promotes transition to the inactive state.
C AMP cascade is active during fasting or starvation and is activated by glucagon or epinephrine.

05/01/2023 NAMRATA CHHABRA, M.D. 10
Implications of PhosphorylationPhosphorylation of Glycogen Synthase promotes the "b" (less active) conformation. The cAMP cascade thus inhibits glycogen synthesis. Instead of being converted to glycogen, glucose-1-P in liver may be converted to glucose-6-P, and dephosphorylated for release to the blood.

05/01/2023 NAMRATA CHHABRA, M.D. 11
Role of Insulin in Glycogenesis
Insulin promotes Glycogenesis.Insulin, produced in response to high blood glucose causes activation of Phosphoprotein Phosphatase resulting in removal of regulatory phosphate residues from Glycogen Synthase enzyme converting it to dephosphorylated/active form.
In liver insulin increases the activity of phosphodiesterase, promoting hydrolysis of c AMP terminating hormone action.
Insulin thus antagonizes effects of the cAMP cascade induced by glucagon & epinephrine.

05/01/2023 NAMRATA CHHABRA, M.D. 12
ATP
Glycogen synthase a
Glycogen synthase b
C AMP 5’AMP
Adenylate cyclase
Protein Phosphatase
Phosphodiesterase
ATP ADPProtein
Kinase A
H2OPi
IG
G
IIG InsulinGlucagon
Glucagon favors phosphorylation thus inhibits glycogenesis
Insulin favors dephosphorylation thus stimulates Glycogenesis
cAMP Cascade and the role of hormones

05/01/2023 NAMRATA CHHABRA, M.D. 13
Regulation of GlycogenolysisGlycogen Synthase and Glycogen Phosphorylase are reciprocally regulated, by allosteric effectors and by phosphorylation.The control of phosphorylase differs between liver & muscleIn the liver the role of glycogen is to provide free glucose for export to maintain the blood concentration of glucose; In muscle the role of glycogen is to provide a source of glucose 6-phosphate for glycolysis in response to the need for ATP for muscle contraction.

05/01/2023 NAMRATA CHHABRA, M.D. 14
Regulation of Muscle Phosphorylase by allosteric modification
Phosphorylase
Negative effectors
ATP Glucose-6-P
Positive effectors
AMP
Liver phosphorylase is less sensitive to these allosteric modifier.
Phosphorylase is inhibited by excess Glucose-P, the product of the reaction sequence. On the contrary, Glycogen synthase is stimulated by excess Glucose-6-P, the substrate of this pathway.
Glycogen breakdown is inhibited when ATP and glucose-6-phosphate are plentiful.

05/01/2023 NAMRATA CHHABRA, M.D. 15
Regulation of Glycogenolysis by Covalent Modification (phosphorylation):
The cAMP cascade results in phosphorylation of a serine hydroxyl of Glycogen Phosphorylase, which promotes transition to the active state. The phosphorylated enzyme is less sensitive to allosteric inhibitors. Thus, even if cellular ATP and glucose-6-phosphate are high, Phosphorylase will be active.

05/01/2023 NAMRATA CHHABRA, M.D. 16
Phosphorylation of PhosphorylaseThe enzyme phosphorylase is activated by phosphorylation catalyzed by phosphorylase kinase (to yield phosphorylase a) andInactivated by dephosphorylation catalyzed by phosphoprotein phosphatase (to yield phosphorylase b), in response to hormonal and other signals.
C AMPPhosphorylase
Kinase(Active)
Protein Kinase A
Phosphorylase(Active)

05/01/2023 NAMRATA CHHABRA, M.D. 17
Role of cAMP In Glycogen degradationIncreasing the concentration of cAMP activates cAMP-dependent protein kinase, which catalyzes the phosphorylation by ATP of inactive phosphorylase kinase b to active phosphorylase kinase a, which in turn, phosphorylates phosphorylase b to phosphorylase a.In the liver, cAMP is formed in response to glucagon, which is secreted in response to falling blood glucose; muscle is insensitive to glucagon. In muscle, the signal for increased cAMP formation is the action of norepinephrine, which is secreted in response to fear or fright, when there is a need for increased glycogenolysis to permit rapid muscle activity.

05/01/2023 NAMRATA CHHABRA, M.D. 18
ATP
Phosphorylase kinase
(Inactive)
Phosphorylase(Inactive)
C AMP 5’AMP
Adenylate cyclase
Protein Phosphatase
Phosphodiesterase
ATP ADPProtein
Kinase A
H2OPi
IG
G
IIGInsulin
Glucagon
Glucagon favors phosphorylation thus promotes Glycogenolysis
Insulin favors dephosphorylation
thus inhibits Glycogenolysis
cAMP Cascade and the role of hormones
Phosphorylase(Active)
Phosphorylase kinase
(Active)
ATP
ADP
Phosphatase
H2O
Pi

05/01/2023 NAMRATA CHHABRA, M.D. 19
Role of Ca++ in glycogen degradation
Ca++ also regulates glycogen breakdown in muscle. During activation of contraction in skeletal muscle, Ca++ is released from the sarcoplasmic reticulum to promote actin/myosin interactions. The released Ca++ also activates Phosphorylase Kinase, which in muscle includes calmodulin as its δ subunit.Phosphorylase Kinase is partly activated by binding of Ca++ to this subunit.

05/01/2023 NAMRATA CHHABRA, M.D. 20
Role of calcium in muscle degradation
Phosphorylase Kinase is partly activated by binding of Ca++ to this subunit.
Further activation is brought by phosphorylation.
Phosphorylase KinaseDephosphorylated (inactive)
Phosphorylase kinase- Ca++Partly active
Phosphorylase kinase- Ca++Phosphorylated- active
Ca++
ATP

05/01/2023 NAMRATA CHHABRA, M.D. 21
Role of calcium in the activation of phosphorylation kinase
Muscle phosphorylase kinase, which activates glycogen phosphorylase, is a tetramer of four different subunits-α, β ,Υ and δ .
The α and β subunits contain serine residues that are phosphorylated by cAMP-dependent protein kinase. The δ subunit is identical to the Ca2+-binding protein calmodulin.
The binding of Ca2+ activates the catalytic site of the subunit even while the enzyme is in the dephosphorylated b state; the phosphorylated a form is only fully activated in the presence of Ca2+.
Phosphorylase Kinase
α γδβ
Phosphorylase Kinase
α β δ γ
Protein kinase A Ca++
Phosphatase
Ca ++P P
Active EnzymeInactive Enzyme

05/01/2023 NAMRATA CHHABRA, M.D. 22
Role of Insulin in Glycogen degradation
Both phosphorylase and phosphorylase kinase are dephosphorylated and inactivated by protein phosphatase. Protein phosphatase is stimulated by Insulin, Therefore Insulin by inhibiting the activation of these enzymes inhibits the overall process of glycogenolysis.

05/01/2023 NAMRATA CHHABRA, M.D. 23
Reciprocal regulation of Glycogenesis and glycogenolysisGlycogen Synthase & Phosphorylase activity are reciprocally regulatedAt the same time as phosphorylase is activated by a rise in concentration of cAMP (via phosphorylase kinase), glycogen synthase is converted to the inactive form.Thus, inhibition of glycogenolysis enhances net glycogenesis, and inhibition of glycogenesis enhances net glycogenolysisBoth processes do not occur at the same time.

05/01/2023 NAMRATA CHHABRA, M.D. 24
Biological significance When the blood glucose is low as in fasting or starvation, the predominant hormones such as Glucagon and epinephrine trigger the C- AMP mediated phosphorylation cascade.In the phosphorylated state glycogen synthase becomes inactive whereas Phosphorylase becomes active, Glycogenesis is switched “off” and Glycogenolysis is switched “on”. Liver glycogen breakdown restores the lowered blood glucose concentration back to normal

05/01/2023 NAMRATA CHHABRA, M.D. 25
Biological significanceWhen the blood glucose concentration is high- Insulin, the main hormone, promotes the dephosphorylated forms of the enzymes by disrupting the c AMP mediated phosphorylation cascade and by stimulating the phosphatase activities.Phosphorylase in the dephosphorylated form becomes inactive whereas the Glycogen synthase in that state becomes active.Hence extra glucose is used for glycogen synthesis and blood glucose concentration is restored back to normal.

05/01/2023 NAMRATA CHHABRA, M.D. 26
ConclusionGlycogenesis and glycogenolysis are reciprocally regulated.Insulin promotes glycogenesis.Glucagon and epinephrine promote glycogenolysis.Glycogenesis is the process of well-fed state.Glycogenolysis is the process of Fasting or starvation.Both these processes are meant for maintaining the blood glucose concentration within the normal range.

05/01/2023 NAMRATA CHHABRA, M.D. 27
Thank you