pH scale
description
Transcript of pH scale


pH scalepH scale
• Logarithmic scale –expressing H+1 concentration, [H+1]
• If pH changes by factor of 1,
[H+1] changes by factor of 10• pH = -log[HpH = -log[H+1+1]]

Molarity to pH
To determine pH:
• express [H+1] in scientific notation
• remember, [ ] means concentration of whatever is inside brackets
• log is the power of 10log is the power of 10

Molarity to pH for Pure water
there’s a tiny bit of ionization:
H2O H+1 + OH-1
• [H+1] = [OH-1] = 1 X 10-7-7 M
• pH = -log[Hlog[H+1+1]] = -(-7-7) = 7

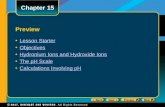
pH
33221100pH
.001 M
or
1 X 10-3-3
M
0.01 M
or
1 X 10-2-2
M
0.1 M
or
1 X 10-1-1
M
1 M
or
1X1000 M
[H+1]

pH to Molarity
pH = -log[H+1], solve for [H+1]
• -pH = log[H+1]
• antilog(-pH) = [H+1]
• say pH = 5, then –pH = -5
• antilog(-5) = 10-5
• the –pH becomes the power of 10!the –pH becomes the power of 10!

pOH
• by analogy: pOH is defined as –log[OH-1]
• express [OH-1] in scientific notation
• if [OH-1] = 1.0 X 10-3 M
• then pOH = -log(10-3) = -(-3) = 3

pH + pOH = 14pH + pOH = 14
• pH = 3, pOH =
• pH = 7, pOH =
• pH = 10, pOH =
• pH = 9, pOH =
• pH = 1, pOH =
1111
77
44
55
1313

ACID or BASE?
• acids: [H+1] [OH-1]
• bases: [OH-1] [H+1]


pH range
• 0 to 14 –with 7 being neutral
• pH = 0, strongly acidic
• pH = 14, strongly basic

•if pH = 4if pH = 4
• [H+1] = ?
• pOH = ?
• [OH-1] = ?
1010
antilog(-pH) = [Hantilog(-pH) = [H+1+1]]antilog(-4) = 1 x 10antilog(-4) = 1 x 10-4 -4 MM
antilog(-10) = [OHantilog(-10) = [OH-1-1]]1x101x10-10-10 M = [OH M = [OH-1-1]]

• if the [OH-1] = 1 X 10-3
• pOH = ?
• pH = ?
• [H+1] = ?
pOH = -log[OHpOH = -log[OH-1-1]]= -log(10= -log(10-3-3) = -(-3) = 3) = -(-3) = 3
14 – 3 = 1114 – 3 = 11
antilog(-11) = 1x10antilog(-11) = 1x10-11-11 M M

• if the [H+1] = 1 x 10-5 M
• pH = ?
• pOH = ?
• [OH-] = ?
55
14 – 5 = 914 – 5 = 9
antilog(-9) = 1x10antilog(-9) = 1x10-9-9MM

6
1x10-2
1x10-5
3
Acidic or
Basic
pOH[OH-]
(M)(M)
pH[H+]
(M)(M)
11111x101x10-3-3 1x101x10-11-11AA
55991x101x10-9-9
BB
22 12121x101x10-12-12 AA
881x101x10-8-8 1x101x10-6-6 BB

How to safely test pH
• instruments – use a pH meter
• indicators – use a series of indicators
• see if substance reacts with a metal other than Cu, Ag, or Au
• NEVER “taste”


Indicator
• substance that changes color over narrow pH range
• use several indicators to narrow down pH range of substance










![The pH Scale and Indicators. The pH Scale pH- “potency hydrogen” [H + ], a measure of how acidic or basic a solution is, the concentration of H + ions.](https://static.fdocuments.in/doc/165x107/56649dbe5503460f94ab0c87/the-ph-scale-and-indicators-the-ph-scale-ph-potency-hydrogen-h-.jpg)
![pH Scale and Calculations Chapter 14 pH Scale We use this scale to measure the strength of an acid or base. pH is defined as the –log[H+] pH can use.](https://static.fdocuments.in/doc/165x107/551adb23550346856e8b6439/ph-scale-and-calculations-chapter-14-ph-scale-we-use-this-scale-to-measure-the-strength-of-an-acid-or-base-ph-is-defined-as-the-logh-ph-can-use.jpg)