O-LINKED N-ACETYLGLUCOSAMINE PROTEIN MODIFICATION IN...
Transcript of O-LINKED N-ACETYLGLUCOSAMINE PROTEIN MODIFICATION IN...

O-LINKED N-ACETYLGLUCOSAMINE PROTEIN MODIFICATION IN MOUSE MODELS OF
NEURODEGENERATIVE DISEASES
by
Xiao Yang Shan Bachelor of Medicine, Guangxi Medical University, 1989 Master of Medicine, Guangxi Medical University, 1997
M.Sc., Simon Fraser University, 2005
THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF
DOCTOR OF PHILOSOPHY
In the
Department of Molecular Biology and Biochemistry Faculty of Science
© Xiao Yang Shan 2011
SIMON FRASER UNIVERSITY
Summer 2011
All rights reserved. However, in accordance with the Copyright Act of Canada, this work
may be reproduced, without authorization, under the conditions for Fair Dealing. Therefore, limited reproduction of this work for the purposes of private study, research,
criticism, review and news reporting is likely to be in accordance with the law, particularly if cited appropriately.

ii
Approval
Name: Xiao Yang Shan
Degree: Doctor of Philosophy
Title of Thesis: O-linked N-acetylglucosamine protein modification in mouse models of neurodegenerative diseases
Examining Committee:
Chair: Dr. Fiona Brinkman Professor, Department of Molecular Biology and Biochemistry
_____________________________________________________
Dr. David Vocadlo Senior Supervisor Associate Professor, Department of Chemistry
_____________________________________________________
Dr. Charles Krieger Co-Supervisor Professor, Department of Biomedical Physiology and Kinesiology
_____________________________________________________
Dr. Esther Verheyen Supervisor Professor, Department of Molecular Biology and Biochemistry
_____________________________________________________
Dr. Nicholas Harden Supervisor Professor, Department of Molecular Biology and Biochemistry
_____________________________________________________
Dr. Glen Tibbits Internal Examiner Professor, Department of Biomedical Physiology and Kinesiology
_____________________________________________________
Dr. Neil Cashman External Examiner Professor, Department of Medicine University of British Columbia
Date Defended/Approved: August 15, 2011 1

Last revision: Spring 09
Declaration of Partial Copyright Licence The author, whose copyright is declared on the title page of this work, has granted to Simon Fraser University the right to lend this thesis, project or extended essay to users of the Simon Fraser University Library, and to make partial or single copies only for such users or in response to a request from the library of any other university, or other educational institution, on its own behalf or for one of its users.
The author has further granted permission to Simon Fraser University to keep or make a digital copy for use in its circulating collection (currently available to the public at the “Institutional Repository” link of the SFU Library website <www.lib.sfu.ca> at: <http://ir.lib.sfu.ca/handle/1892/112>) and, without changing the content, to translate the thesis/project or extended essays, if technically possible, to any medium or format for the purpose of preservation of the digital work.
The author has further agreed that permission for multiple copying of this work for scholarly purposes may be granted by either the author or the Dean of Graduate Studies.
It is understood that copying or publication of this work for financial gain shall not be allowed without the author’s written permission.
Permission for public performance, or limited permission for private scholarly use, of any multimedia materials forming part of this work, may have been granted by the author. This information may be found on the separately catalogued multimedia material and in the signed Partial Copyright Licence.
While licensing SFU to permit the above uses, the author retains copyright in the thesis, project or extended essays, including the right to change the work for subsequent purposes, including editing and publishing the work in whole or in part, and licensing other parties, as the author may desire.
The original Partial Copyright Licence attesting to these terms, and signed by this author, may be found in the original bound copy of this work, retained in the Simon Fraser University Archive.
Simon Fraser University Library Burnaby, BC, Canada

STATEMENT OF ETHICS APPROVAL
The author, whose name appears on the title page of this work, has obtained, for the research described in this work, either:
(a) Human research ethics approval from the Simon Fraser University Office of Research Ethics,
or
(b) Advance approval of the animal care protocol from the University Animal Care Committee of Simon Fraser University;
or has conducted the research
(c) as a co-investigator, collaborator or research assistant in a research project approved in advance,
or
(d) as a member of a course approved in advance for minimal risk human research, by the Office of Research Ethics.
A copy of the approval letter has been filed at the Theses Office of the University Library at the time of submission of this thesis or project.
The original application for approval and letter of approval are filed with the relevant offices. Inquiries may be directed to those authorities.
Simon Fraser University Library
Simon Fraser University Burnaby, BC, Canada
Last update: Spring 2010

iii
Abstract
The O-linked addition of β-N-acetylglucosamine to proteins (O-GlcNAc) is a form of intracellular glycosylation that has gained increasing attention for its potential involvement in neurodegenerative diseases including amyotrophic lateral sclerosis (ALS) and Alzheimer’s disease (AD). ALS has several causes including abnormal phosphorylation of neuronal proteins as well as several gene mutations including those in the sod1 gene, which encodes superoxide dismutase 1, as well as the TARDBP gene, which encosed TDP-43. Abnormally elevated phosphorylation of proteins including TDP-43, neurofilaments, and tau are all implicated in neurodegeneration. Tau, for example, forms intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein. O-GlcNAc transferase (OGT) catalyzes the installation of GlcNAc onto specific serine and threonine residues of target proteins, while O-GlcNAcase (OGA) removes the modification. It is known that O-GlcNAc modification of tau and other proteins is reciprocal to phosphorylation. The objective of this thesis was to improve our understanding of the role that O-GlcNAc has on proteins implicated in neurodegeneration in animal models of ALS and AD. The main findings are (1) O-GlcNAc levels were reduced in spinal cord tissue from the mSOD mouse model of ALS specifically in motor neurons; (2) mislocalization of TDP-43 occurs in aged mSOD mice; (3) mouse brain TDP-43 was found to be O-GlcNAc modified and four O-GlcNAc modification sites were mapped on recombinant full-length human TDP-43; (4) OGA inhibitor Thiamet-G treatment to JNPL3 mouse model of AD increased tau O-GlcNAc modification, hindered tau aggregation, and protected mice against neuronal cell loss. These results suggest that the neurodegeneration found in mSOD mice might be associated with a reduction of O-GlcNAc levels in motor neurons, and O-GlcNAc modification might influence the abnormal phosphorylation of TDP-43 in ALS. These studies also provide new insight into the potential association of SOD1 and TDP-43, and offer support for OGA as a viable therapeutic targets that might provide an opportunity to alter disease progression in AD and offer benefits in other diseases characterized by the aggregation of proteins that can be O-GlcNAc modified. Keywords: O-GlcNAc; TDP-43; JNPL3; NButGT; Thiamet-G; ALS

iv
Dedication
This thesis is dedicated to my beloved parents.

v
Acknowledgements
It has taken five years to complete this Ph.D. thesis. I would like to express my great appreciation to the people who have led me or helped me reach this milestone.
I am very grateful to my senior supervisor, Dr. David Vocadlo, for his guidance, advice, and support of this exciting research and my life in the past, present, and future. I would also like to thank my co-supervisor, Dr. Charles Krieger, for his supervision, encouragement and constant support. Their knowledge and support will have a lifelong impact on me.
I would also like to extend my sincere appreciation to Dr. Esther Verheyen and Dr. Nicholas Harden (supervisors), for their insight and expert advice in molecular biology and protein biochemistry and Dr. Glen Tibbits and Dr. Neil Cashman (examiners), for their critical review and constructive opinions of this thesis.
The following colleagues are very much appreciated for their advice, assistance and collaboration: Dr. Renee Mosi, Dr. Matthew Macauley, Scott Yuzwa, Tom Clark, Dr. Tracey Gloster, Garrett Whitworth, David Shen, Dr. Anuj Yadav, Dr. Wesley Zandberg, Dr. Ian Greig, Zarina Madden, Dr. Lehua Deng, Shirley Ko, Julia Heinonen, Dr. Ernest McEachern and Dr. Yanshen Deng (Vocadlo Lab), as well as Dr. Coral Lewis, Dr. Jennifer Solomon, Amy Tsai, Jing Yang, John Manning, and Sapana Thakore (Krieger Lab). Dr. Michael Silverman is thanked for providing primary hippocampal neurons.
For the work discussed in Chapter 2, 3, 4 and 5, I would like to thank Audrey Wang, Mary Dearden and rest of staff at the SFU Animal Care Facility for their assistance. I also thank the Canadian Institute for Health Research (CIHR), the ALS Society of Canada, the Scottish Rite Charitable Foundation (SRCF), the Michael Smith Foundation for Health Research (MSFHR), and the National Institute for Health (NIH) for providing funding. Dr. Peter Davies is thanked for the kind gift of the CP27 and PHF-1 antibodies used in the JNPL3 study described in Chapter 5.
Finally, I would like to thank my family for their constant support.

vi
Table of Contents
Approval .......................................................................................................................................... ii Abstract .......................................................................................................................................... iii Dedication ....................................................................................................................................... iv Acknowledgements .......................................................................................................................... v Table of Contents ............................................................................................................................ vi List of Figures ................................................................................................................................. ix List of Tables ................................................................................................................................... xi List of Abbreviations ..................................................................................................................... xii
1: Introduction ................................................................................................................................ 1
1.1 O-GlcNAc post-translational modification ............................................................................. 1 1.1.1 A simple, abundant and dynamic modification .......................................................... 1 1.1.2 Tissue and cellular localization of O-GlcNAc ........................................................... 2 1.1.3 Regulation of O-GlcNAc ........................................................................................... 3 1.1.4 Biological roles of O-GlcNAc modification .............................................................. 7
1.2 Amyotrophic lateral sclerosis (ALS) ..................................................................................... 12 1.2.1 Overview of ALS and its pathogenesis .................................................................... 12 1.2.2 Cu/Zn superoxide dismutase (SOD1) and ALS ....................................................... 14 1.2.3 TAR DNA binding protein-43 (TDP-43) and ALS .................................................. 19 1.2.4 O-GlcNAc in ALS .................................................................................................... 27
1.3 Alzheimer’s disease (AD) ..................................................................................................... 29 1.3.1 Overview of AD, neuropathological features and major hypotheses ....................... 29 1.3.2 Tau and AD .............................................................................................................. 32 1.3.3 Transgenic animal models of AD ............................................................................. 39
1.4 Reference List ....................................................................................................................... 41
2: Reduction of O-GlcNAc in the motor neurons of G93A mutant SOD1 transgenic mouse model of ALS ..................................................................................................................... 60
2.1 Abstract ................................................................................................................................. 60 2.2 Introduction ........................................................................................................................... 61 2.3 Methods ................................................................................................................................. 64
2.3.1 Antibodies ................................................................................................................ 64 2.3.2 Animals and treatment with NButGT ...................................................................... 64 2.3.3 Tissue homogenization and Immunoblotting ........................................................... 65 2.3.4 Perfusion and immunohistochemistry ...................................................................... 66 2.3.5 A densitometry analysis of O-GlcNAc immunohistochemistry ............................... 67 2.3.6 Motor neuron counts ................................................................................................ 68 2.3.7 Statistical analysis .................................................................................................... 69
2.4 Results ................................................................................................................................... 69 2.4.1 Distribution of O-GlcNAc modified protein in CNS ............................................... 69

vii
2.4.2 Level of O-GlcNAc modified protein in spinal cords and motor neurons ............... 70 2.4.3 NButGT significantly enhanced O-GlcNAc level in spinal cords ........................... 72
2.5 Discussion ............................................................................................................................. 73 2.6 Figures ................................................................................................................................... 78 2.7 Reference List ....................................................................................................................... 83
3: Mislocalization of TDP-43 in the G93A mutant SOD1 transgenic mouse model of ALS ................................................................................................................................................ 86
3.1 Abstract ................................................................................................................................. 86 3.2 Introduction ........................................................................................................................... 87 3.3 Methods ................................................................................................................................. 89
3.3.1 Animals .................................................................................................................... 89 3.3.2 Immunohistochemistry ............................................................................................. 89 3.3.3 Immunoblotting ........................................................................................................ 90 3.3.4 Sequential extraction ................................................................................................ 91
3.4 Results ................................................................................................................................... 91 3.5 Discussion ............................................................................................................................. 93 3.6 Figures ................................................................................................................................... 96 3.7 Reference List ....................................................................................................................... 99
4: Brain TDP-43 is modified with O-GlcNAc and the O-linked glycosylation sites map to the C-terminal region of recombinant human TDP-43 .............................................. 102
4.1 Abstract ............................................................................................................................... 102 4.2 Introduction ......................................................................................................................... 103 4.3 Methods ............................................................................................................................... 106
4.3.1 Administration of NButGT and animal tissue preparation ..................................... 106 4.3.2 Immunoprecipitation of TDP-43 from mouse brain ............................................... 107 4.3.3 Immunoblotting ...................................................................................................... 107 4.3.4 Antibodies .............................................................................................................. 108 4.3.5 Molecular cloning .................................................................................................. 109 4.3.6 Production of recombinant O-GlcNAc modified TDP-43 ..................................... 109 4.3.7 Sample preparation for mass spectrometry analysis .............................................. 110 4.3.8 On-line dual liquid chromatography ...................................................................... 112 4.3.9 Information dependant acquisition (IDA) mass spectrometry (MS) ...................... 113
4.4 Results ................................................................................................................................. 114 4.4.1 O-GlcNAc modification of TDP-43 in mouse brains ............................................ 114 4.4.2 O-GlcNAc modification of recombinant human TDP-43 ...................................... 115 4.4.3 O-GlcNAc modification sites map to the C-terminal region of TDP-43 ............... 117
4.5 Discussion ........................................................................................................................... 118 4.6 Figures and Table ................................................................................................................ 121 4.7 Reference List ..................................................................................................................... 124
5: O-GlcNAc stabilizes tau againsts aggregation and slows neurodegeneration in an AD mouse model ......................................................................................................................... 128
5.1 Abstract ............................................................................................................................... 128 5.2 Introduction ......................................................................................................................... 129 5.3 Methods ............................................................................................................................... 132
5.3.1 Animal dosing, handling, and motor testing .......................................................... 132

viii
5.3.2 Rotarod methods .................................................................................................... 133 5.3.3 Tissue collection and blinding ................................................................................ 134 5.3.4 Tissue homogenization ........................................................................................... 134 5.3.5 Fluorescent immunohistochemistry (IHC) ............................................................. 135 5.3.6 Fluorescent microscopy and densitometry analysis ............................................... 136 5.3.7 Motor neuron counts .............................................................................................. 137 5.3.8 AT8 IHC before and after btOGA digestion in tissue ............................................ 137 5.3.9 AT8 IHC by using ABC method ............................................................................ 138 5.3.10 Gallyas silver staining ............................................................................................ 139 5.3.11 Immunoblotting ...................................................................................................... 139 5.3.12 Statistical analyses.................................................................................................. 140 5.3.13 Antibodies used in this study ................................................................................. 141 5.3.14 Molecular cloning .................................................................................................. 142 5.3.15 Production of recombinant O-GlcNAc modified tau ............................................. 142 5.3.16 Purification of control and modified Tau244-441 or S400A Tau244-441 by
reversed-phase HPLC ............................................................................................. 143 5.3.17 In vitro aggregation of control and modified Tau244-441 or S400A
Tau244-441 ............................................................................................................ 144 5.3.18 Filtertrap assay and quantitiation of the sarkosyl insoluble pellet tau by Slot
blot .......................................................................................................................... 145 5.3.19 Production of O-GlcNAc modified sTAB1 and in vitro aggregation .................... 146
5.4 Results and discussion ......................................................................................................... 146 5.5 Conclusion ........................................................................................................................... 154 5.6 Figures ................................................................................................................................. 156 5.7 Reference List ..................................................................................................................... 183
6: Summary, conclusions, and future studies........................................................................... 187
6.1 Summary ............................................................................................................................. 187 6.2 Conclusions ......................................................................................................................... 189 6.3 Future studies ...................................................................................................................... 192 6.4 Reference List ..................................................................................................................... 194
Appendices .................................................................................................................................. 197
Appendix A .................................................................................................................................. 198 Appendix B ................................................................................................................................... 199 Appendix C ................................................................................................................................... 203 Appendix D .................................................................................................................................. 204

ix
List of Figures
Figure 1- 1 Schematic diagram of cycling of O-GlcNAc and O-phosphate on the same residue of a protein. ........................................................................................................ 8
Figure 1- 2 Schematic diagram of TDP-43 with characteristic functional domains of TDP-43 and sites of the identified mutations in familial (fALS) and sporadic (sALS) Amyotrophic Lateral Sclerosis (ALS). ............................................................ 25
Figure 1- 3 Alternative splicing and domain structure of tau. ........................................................ 33
Figure 2- 1 Distribution of O-GlcNAc modified proteins in spinal cord. ...................................... 78
Figure 2- 2 Levels of O-GlcNAc modified proteins in spinal cord and motor neurons. ................ 80
Figure 2- 3 NButGT significantly enhances O-GlcNAc level in spinal cords. .............................. 81
Figure 2- 4 NButGT induces increased O-GlcNAc modification of NFM in spinal cord. ............. 82
Figure 3- 1 TDP-43 redistribution in end stage mSOD lumbar spinal cord ventral horn cells. ............................................................................................................................. 97
Figure 3- 2 TDP-43 protein levels in lumbar spinal cord do not change in end stagemSOD mice. ......................................................................................................... 98
Figure 4- 1 Mouse brain TDP-43 is O-GlcNAc modified. ........................................................... 121
Figure 4- 2 Production of recombinant O-GlcNAc modified TDP-43. ........................................ 122
Figure 5 - 1 Treatment of JNPL3 animals slows motor impairment and reduces neurodegeneration. ..................................................................................................... 156
Figure 5 - 2 JNPL3 body weights and Rotarod Performance. ...................................................... 157
Figure 5 - 3 Motor neuron counting method. ............................................................................... 158
Figure 5 - 4 O-GlcNAc levels are increased in motor neurons of the ventral horn. ..................... 159
Figure 5 - 5 Immunohistological analysis of TG treated JNPL3 animals reveal significant reductions in AT8 immunoreactivity in the brainstem. .............................................. 160
Figure 5 - 6 AT8 immunoreacitivity densitometry analysis method for region of interest (ROI) selection ........................................................................................................... 161
Figure 5 - 7 AT8 tau phosphorylation is decreased and global O-GlcNAc levels are increased in the cervical spinal cord. ......................................................................... 162

x
Figure 5 - 8 AT8 tau phosphorylation is decreased and global O-GlcNAc levels are increased in the hypothalamus. .................................................................................. 163
Figure 5 - 9 AT8 tau phosphorylation is decreased and global O-GlcNAc levels are increased in the cortex. ............................................................................................... 164
Figure 5 - 10 Immunohistological analysis of TG treated JNPL3 animals reveals significant reductions in NFTs. .................................................................................. 165
Figure 5 - 11 MS/MS and MS/MS/MS spectra from an in vitro O-GlcNAc modified Tau tryptic digest identifies a site of glycosylation at Ser208. .......................................... 166
Figure 5 - 12 Increased O-GlcNAc modification does not block binding of the AT8 antibody binding in the JNPL3 mouse brain. ............................................................. 168
Figure 5 - 13 Immunohistological analysis of TG treated JNPL3 animals reveal significant reductions in pSer422 immunoreactivity. ................................................. 169
Figure 5 - 14 PHF-1 tau immunoreactivity is decreased and global O-GlcNAc levels are increased in the brainstem of treated mice. ................................................................ 170
Figure 5 - 15 Immunohistological analysis of TG treated JNPL3 animals reveals antibodies recognize NFTs, which have classical morphologies. .............................. 171
Figure 5 - 16 Gallyas-Braak silver staining reveals typical NFT morphology. ............................ 172
Figure 5 - 17 Immunohistological analysis of TG treated JNPL3 animals reveals NFT immunoreactivity measured using phosphor-depedent antibody overlaps with phosphor-independent antibody known to detect tau pathology in the brainstem. ................................................................................................................... 173
Figure 5 - 18 Nitrotyrosine on Y29 of tau (nY29) is reduced in the hypothalamus. .................... 174
Figure 5 - 19 Nitrotyrosine on Y29 of tau (nY29) is reduced in the cortex. ................................ 175
Figure 5 - 20 Nitrotyrosine on Y29 of tau (nY29) is reduced in the cervical spinal cord. ........... 176
Figure 5 - 21 Biochemical analysis of TG treated JNPL3 animals. ............................................. 177
Figure 5 - 22 Total and phosphorylated tau levels on the 64-kDa human tau species. ................ 178
Figure 5 - 23 Tau Isoforms and Constructs. ................................................................................. 179
Figure 5 - 24 O-GlcNAc on Tau244-441 slows aggregation in vitro. ............................................... 180
Figure 5 - 25 In vitro Tau244-441 aggregation using arachidonic acid as the inducer. .................... 181
Figure 5 - 26 Production of O-GlcNAc modified sTAB1 inhibits its aggregation in vitro. ......... 182

xi
List of Tables
Table 4- 1 O-GlcNAc modification sites mapped from digestion of recombinant human TDP-43. ...................................................................................................................... 123

xii
List of Abbreviations
AD Alzheimer’s Disease APP amyloid presusor protein ALS Amyotrophic Lateral Scelosis ME -mercaptoethanol Abs absorbance ApoA-II apolipoprotein A-II ApoE4 apolipoprotein E4 ATP adenosine triphosphate BtGH84 Bacteroides thetaiotaomicron -N-acetylglucosaminidase BSA bovine serum albumin cDNA complementary DNA cdk5 cyclin dependent kinase 5 CFTR cystic fibrosis transmembrane regulator CNS central nerious system CpGH84 Clostridium perfringens -N-acetylglucosaminidase CTD110.6 O-GlcNAc-specific antibody raised a glycosylated peptide from
the C-terminal domain of RNA polymerase II Cy3 water-soluble fluorescent dye of the cyanine dye family, Cy3 dye
is fluorescent in the red region Da Dalton DAPI 4',6-diamidinO-2-phenylindole DN dystrophic neurites DNA deoxyribonucleic acid DTT dithiothreitol EDTA ethylenediaminetetraacetic acid EAAT2 excitatory amino acid transporter 2 ER endoplasmic reticulum fALS familial Amyotrophic Lateral Scelosis FBS fetal bovine serum FITC fluorescein isothiocyanate, FITC dye is fluorescent in the green
region FL full length FTLD frontotemporal lobar degeneration FTLP-17 frontotemporal dementia with parkinsonism-17 FTLD-U frontotemporal lobar degeneration with ubiquitin inclusions FUS/TLS fused in sarcoma/translocation in liposarcoma HBSP hexosamine biosynthetic pathway GCI glial cytoplasmic inclusions

xiii
GFAT glutamine-fructose-6-phosphate-transaminase GFP green fluorescent protein GH glycoside hydrolase Glc glucose GlcN glucosamine GlcNAc N-acetylglucosamine GLUT glucose transporters GSK glycogen synthase kinase GT glycosyl transferase GWAS genome-wide association HAT histone acetyl transferase HexA hexosaminidase A HexB hexosaminidase B His6 hexahistadine peptide HnRNP heterogeneous ribonucleoprotein HPLC high pressure liquid chromatography hr hour HSP heat shock protein I inhibitor Ig immunoglobulin IPTG isopropyl -D-1-thiogalactopyranoside JNPL3 transgenic mice expressing mutant P301L human tau kb kilobase kDa killodalton Ki inhibition constant LB Luria broth MAP microtubule associate protein MARK1 microtubulr associate protein MAPT gene encoding tau MBP maltose binding protein min minute mg milligram MGEA5 meningioma expressed antigen 5: gene encoding O-GlcNAcase mmol millimoles mRNA messenger ribonucleic acid mSOD mutant SOD1 NButGT 1,2-dideoxy-2'-propyl--D-glucopyranosO-[2,1-d]-2'-thiazoline NCI neuronal cytoplasmic inclusions NES nuclear export signal NF neurofilaments NFH neurofilament heavy chain NFL neurofilament light chain NFM neurofilament medium chain NFT neurofibrillary tangle NII neuronal intranuclear inclusions NLS nuclear localization signal

xiv
NP-40 nonyl phenoxylpolyethoxylethanol NV nuclear variant OD optical density OGA O-GlcNAcase O-GlcNAc O-linked β-N-acetylglucosamine OGT UDP-GlcNAc:polypeptidyl transferase O-GlcNAc transferase PAGE polyacrylamide gel electrophoresis PBS phosphate buffered saline PBST phosphate buffered saline containing 0.3% Triton X-100 PBS-T phosphate buffered saline containing 0.1% Tween20 PCR polymerase chain reaction pET plasmid for protein expression containing the T7 promoter PHF paired helical filaments PKA cyclic AMP depedent protein kinase A (protein kinase A) PFA paraformaldehyde PMSF phenylmethylsulfonyl fluoride ppm parts per million PS1 presenilin 1 PS2 presenilin 2 PTM post-translational modification PUGNAc O-(2-acetamidO-2-deoxy-D-glucopyranosylidene)aminO-N-
phenylcarbamate rcf relative centrifugal force RRM RNA recognition motif ROS reactive oxygen species RL2 O-GlcNAc-specific antibody raised against nucleoporins rpm revolutions per minute sALS sporadic Amyotrophic Lateral Scelosis SDS sodium dodecyl sulphate SMN survival of motor neuron SOD1 superoxide dismutase 1 STZ streptozotocin TAB1 TAK1-binding protein TAPP double transgenic mice expressing both huan mutant tau (P301L)
and Swedish APP mutations (K670N and M671L) TARDBP gene encoding TDP-43 TDP-43 trans activating response region (TAR) DNA-binding protein with
a molecular mass of 43 KDa TG Thiamet-G Thiamet-G 1,2-dideoxy-2'-aminoethyl--D-glucopyranosO-[2,1-d]-2'-
thiazoline Tg transgenic TPR tetratratricopeptide repeat Tris tris(hydroxymethyl)aminomethane UBIs ubiquitin positive inclusions UDP uridine diphosphate

xv
UDP-GlcNAc UDP-N-acetyl--D-glucosamine UV ultraviololet WT wild-type

1
1: Introduction
1.1 O-GlcNAc post-translational modification
Protein post-translational modifications (PTM) involve covalent additions of functional
chemical groups to amino acid side chains and play important roles in influencing the
biological functions of proteins. One example of a PTM is the O-GlcNAc modification,
the addition of O-linked β-N-acetylglucosamine (O-GlcNAc) to the hydroxyl side chains
of serine or threonine residues of various nuclear and cytoplasmic proteins [215]. Since
its discovery less than three decades ago, evidence has suggested the O-GlcNAc
modification of cellular proteins plays key roles in many physiological processes
including glucose homeostasis [51], cell survival [51, 174], cell progression [202], and
synaptic transmission [209]. O-GlcNAc modified proteins may also play critical roles in
some pathological processes in neurodegenerative diseases such as amyotrophic lateral
sclerosis (ALS) [145] and Alzheimer’s disease (AD) [49, 138].
1.1.1 A simple, abundant and dynamic modification
Since its discovery in 1984, the O-GlcNAc modification has been confirmed to be a
ubiquitous post-translational modification present within all multicellular eukaryotes
[87]. More than 1,000 nucleocytoplasmic proteins bear this monosaccharide including
transcription factors, translational regulators, cytoskeletal proteins, and signal
transduction molecules [87, 212]. Unlike other types of glycosylation such as aspargine-
linked or mucin-type O-glycosylation which occurs on membrane-bound or secreted

2
proteins, O-GlcNAc modified proteins localize exclusively in nucleocytoplasmic
compartments [87, 215].
Similar to phosphorylation, O-GlcNAc is a dynamic and inducible post-translational
modification. For example, similar to the way kinases and phosphatases regulate
phosphorylation, two nucleocytoplasmic enzymes regulate O-GlcNAc modification of
proteins. Uridine diphospho (UDP)-N-acetylglucosamine:polypeptide -N-
acetylglucosamineyltransferase, known as O-GlcNAc transferase (OGT), catalyzes the
transfer of N-acetylglucosamine from the donor sugar UDP-GlcNAc to attach the sugar
via a -glycosidic linkage to the target hydroxyl group of serine and threonine residues of
proteins[120]. An N-acetylglucosaminidase, known as O-GlcNAcase (OGA), catalyzes
the hydrolysis of the -glycosidic linkage to remove O-GlcNAc from modified proteins
[59]. The basis by which OGT and OGA localization is controlled remains unknown
though it is known OGT contains a nuclear localization sequence [142] . O-GlcNAc can
turn over on proteins more quickly than the protein itself [38, 192]. O-GlcNAc residues
can consequently respond dynamically to changes in the cellular environment such as
stress, growth factors, or nutrients [84, 100].
1.1.2 Tissue and cellular localization of O-GlcNAc
Earlier studies using specific antibodies to detect the cellular localization of O-GlcNAc
immunoreactivity in multiple cell lines, have determined that O-GlcNAc modified
proteins are predominantly found within the nucleus, and the modification appears
particularly abundant in the nuclear envelope [94, 95, 203]. In rat brain, O-GlcNAc

3
directed antibodies stain most neurons, especially Purkinje cells, within which the
nucleus, perikaryon, and dendrites are most intensely stained. Using cell fractionation
techniques, numerous synaptosomal proteins have been shown to be O-GlcNAc modified
and are abundant in nerve terminals [2, 42]. The localization of O-GlcNAc within spinal
cords and in non-neuronal cells in central nervous system (CNS) has not been reported.
1.1.3 Regulation of O-GlcNAc
In contrast to the regulation of phosphorylation which involves hundreds of individual
kinases and phosphatases, there is only a single O-GlcNAc transferase, OGT, which
catalyzes the addition of O-GlcNAc, and similarly, there is only a single O-GlcNAcase,
OGA, which catalyzes the removal of O-GlcNAc. Notably, however, there are splice
variants of both of these enzymes. OGT has three different splice variants [85, 120, 142]
whereas OGA is known to have two splice variants [214].
1.1.3.1 O-GlcNAc transferase and hexosamine biosynthetic pathway
In a similar way to a kinase, which transfers a phosphate moiety from a unit of ATP,
OGT transfers GlcNAc to serine or threonine residues of proteins from a unit of UDP-
GlcNAc. OGT has been cloned and characterized [120, 142]. The amino acid sequence of
OGT is highly conserved (at the amino acid level sequence identity is greater than 99%
between human and rat, and 61% between rat and Caenorhabditis elegans) between all
eukaryotes examined. OGT is ubiquitously expressed in all tissues examined, however, it
has been shown to be more abundant in the brain and pancreas [142]. OGT comprises
two distinct domains: an N-terminal region consisting of a series of 13.5 tetratricopeptide
repeats (TPR) [120, 142], and a catalytic domain located at C-terminal region [158]. The

4
TPR motif is composed of 34 amino acids that form an -helical structure that is found in
many proteins. These TPR motifs have been found to mediate protein-protein
interactions as well as substrate recognition [109, 143]. The C-terminal region of OGT
has a catalytic domain composed of the traditional Rossman fold found for
glycosyltransferases and is categorized within the GT-B superfamily of
glycosyltransferases [191], and is modified by O-GlcNAc and tyrosine phosphorylation,
suggesting that tyrosine kinase signal transduction cascades might play a role in
modulating OGT activity [121]. The OGT gene is localized on the X chromosome, and
OGT deletion in mice leads to lethality at the single cell level [199], suggesting the OGT
and the O-GlcNAc modification is essential for embryonic stem cell viability. Until
recently, the enzymatic mechanism of OGT has been poorly understood. Two crystal
structures of homologues of human OGT have been reported [110, 158], as well as a
separate crystal structure comprising 11.5 TPR units of human OGT [110]. More recently
the actual structure of human OGT has been solved, including two crystal structures of a
human OGT containing 4.5 TPR units and the C-terminal catalytic domains, and one of
them is a complex with UDP [125]. Using structure analysis of an OGT-UDP-peptide
complex, information was obtained on the enzymatic mechanism of OGT in which UDP-
GlcNAc binds before the polypeptide substrate binds [125].
Currently, very little is known about the processes that serve to regulate OGT. OGT has
been proposed to be sensitive to levels of UDP-GlcNAc over a broad concentration range
[121]. UDP-GlcNAc is the major end product of the hexosamine biosynthetic pathway
(HBSP) [22] and GlcNAc salvage pathway [63]. The HBSP is a branch of the glucose

5
metabolic pathway, which takes in about 2–5% of the total cellular glucose [30]. Thus,
the level of O-GlcNAc on proteins is often considered to be sensitive to changes in
nutrient influx through the HBSP, which can either increase or decrease UDP-GlcNAc
levels [96, 226]. Glutamine–fructose-6-phosphate amidotransferase (GFAT) is the rate-
limiting enzyme of the HBP pathway [249]. This enzyme converts fructose-6-phosphate
to glucosamine-6-phosphate. Depending upon the cell type and organism, GFAT is
feedback inhibited by UDP-GlcNAc [165]. Disruption of UDP-GlcNAc synthesis by
inhibiting GFAT causes notable cellular defects in proliferation and adhesiveness[17].
Therefore, altering UDP-GlcNAc levels by inhibition of the HBP enzymes or by addition
of glucosamine was one of the early approaches used in studies of O-GlcNAc
modification. This approach, however, leads to significant effects on other forms of
glycosylation and is known to cause endoplasmic reticulum stress [160]. With increasing
knowledge of O-GlcNAc regulatory enzymes, additional biological tools have been
applied to study O-GlcNAc modifications, including over-expression, and knockdown of
these enzymes. More recently, inhibitors of both OGT and OGA have been developed
that are able to act in cells [63, 153].
1.1.3.2 O-GlcNAcase and its inhibitors
In an analogous way to protein phosphatases, which remove O-phosphate from proteins,
OGA removes O-GlcNAc. Like OGT, OGA has also been purified, cloned and
characterized [54, 59]. OGA is a neutral glucosaminidase that specifically catalyzes the
removal of β-O-linked GlcNAc [33, 54]. The OGA gene was identified as meninginoma
expressed antigen 5 (MGEA5) [91], and is localized at chromosome 10q24.1–q24.3,

6
which is a region associated with increased risk of late-onset Alzheimer's disease [15].
Recently, a single nucleotide polymorphism in the MGEA5 gene has been identified to be
associated with type II diabetes in a Mexican population [129] although other studies,
including several genome-wide association studies, have failed to find a link suggesting
the earlier observation may stem from linkage disequilibrium.
The OGA sequence is highly conserved in mammals (great than 97% identity at the
amino acid level between humans and mouse) and is expressed in all types of tissues
[59]. Just like OGT, OGA is also particularly abundant in the brain and pancreas [218].
The N-terminal region (1-350 amino acids) contains the catalytic glycoside hydrolase
domain of OGA [82], whereas the C-terminal domain has some homology to
acetyltransferases. OGA was originally shown to have both histone acetyltransferase
(HAT) and OGA activities in vitro [214], though more recently others have failed to
reproduce results showing HAT activity. During apoptosis, caspase-3 cleaves OGA into
the N-terminal glycosidase domain and the C-terminal domain, and cleaved OGA
remains catalytically active in vitro [233]. Unlike functionally related hexosaminidases A
and B (HexA and HexB, collectively known as the -hexosaminidases) that are located in
the lysosome [156], the long 130-kDa isoform (OGA-FL, 916 amino acids) localizes
predominantly in the cytoplasm [59], while the short 75-kDa isoform of OGA (OGA-NV,
677 amino acids) lacking the C-terminal domain of OGA localizes in the nucleus [59,
233]. The two isoforms differ in enzymatic activity that the catalytic efficiency of OGA-
NV is 400-fold lower than that found for OGA-FL [151, 233].

7
To date the structure of mammalian OGA has not been solved. However, the structure of
enzymes similar to human OGA from Bacteroides thetaiotaomicron (BtGH4) has been
crystallized [50] as has one from Clostridium perfringens (CpGH84) [36, 37]. BtGH84,
CpGH84, and mammalian OGA share a conserved catalytic mechanism and conserved
key catalytic residues [36, 37, 50]. OGA has been shown to use a substrate assisted
catalytic mechanism, which involves the N-acetyl group of GlcNAc acting as a
nucleophile during the course of the reaction [153]. Mutations at the catalytic residues
Asp174 and Asp175 greatly decrease the catalytic efficiency of these enzymes [33]. With
knowledge of OGA mechanism and structure, several cell permeable potent and highly
selective OGA inhibitors have been generated to study the role of OGA both in vitro and
in vivo [147, 148, 150, 244]. These inhibitors include NButGT and Thiamet-G. In
contrast to previously commonly used inhibitors, such as O-(2-acetamidO-2-deoxy-D-
glucopyranosylidene) aminO-N-phenylcarbamate (PUGNAc) [54] or Streptozotocin
(STZ) [213], the newer inhibitors have selectivity toward OGA over functionally related
HexA and HexB, and are not toxic to cells. Therefore, these new inhibitors have promise
as powerful tools that can be used to study O-GlcNAc function. They also hold some
potential as leads to potential therapeutics that could be used to treat certain
neurodegenerative diseases.
1.1.4 Biological roles of O-GlcNAc modification
Significant efforts are being directed toward studies regarding the cellular role of O-
GlcNAc. Nutrient sensing, cellular stress response, and regulation of gene transcription
and signal transduction are believed to be the major functions of O-GlcNAc [88, 178].

8
Accumulating lines of evidence have suggested that the O-GlcNAc modification shows
interplay with phosphorylation, and this may be a mechanism leading to regulation of
protein function in both transcription and signal transduction [88].
1.1.4.1 O-GlcNAc interplay with phosphorylation
O-GlcNAc modified proteins are sometimes also phosphoproteins. Interestingly, on
several proteins, such as c-myc, it has been shown that some phosphorylation sites are
also O-GlcNAc modification sites [40]. Consistent with this observation, it has been
found that increases in phosphorylation levels result in decreased O-GlcNAc levels, and
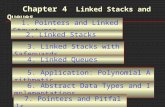
increased O-GlcNAc levels correlate with decreased phosphorylation levels (Figure 1-1)
[71].
Figure 1- 1 Schematic diagram of cycling of O-GlcNAc and O-phosphate on the same residue of a protein. Abbreviations: OGT, O-GlcNAc transferase; OGA, O-GlcNAcase.
This finding suggests that O-GlcNAc modification may have the potential to regulate
important cellular processes in which protein phosphorylation is involved, including
signal transduction and gene transcription. The levels of phosphorylation and O-GlcNAc
on neurofilaments (NF) and tau neuronal proteins vary reciprocally in culture, in
metabolically active rat brain slices, and in rats in vivo [49, 128, 138, 244]. These
findings are interesting as they suggest that O-GlcNAc modification might also be

9
involved in several disease conditions that are associated with tau and neurofilament
proteins. This reciprocal relationship between O-GlcNAc and phosphorylation has been
termed the “Ying-Yang” hypothesis [113].
The relationship between these two PTMs is not, however, simply reciprocal. PTM site-
mapping studies suggest that there are several different types of dynamic interplay
between O-GlcNAc and phosphorylation [87]. They include competitive occupancy at
the same site, as shown above for the transcription factor c-Myc[40], or competitive
alternative occupancy at adjacent sites, as has been observed for synapsin I [43]. One
quantitative mass spectrometry study demonstrated that acutely elevated O-GlcNAc
levels perturbed phosphorylation on ~60% of the 711 phosphoepitopes that were
examined [230]. Specifically, elevated O-GlcNAc modifications resulted in lower
phosphorylation levels at 280 sites and resulted in increased phosphorylation levels at
148 sites. The interplay between these two PTMs might result from several possible
mechanisms including; (1) direct competition for modification site occupancy at the same
or adjacent sites, (2) indirect competition via protein secondary structure at proximal
sites, and (3) regulation of protein kinases and phosphatases via O-GlcNAc.
1.1.4.2 Potential beneficial role of elevated O-GlcNAc modification
One factor that is worth considering is that, unlike phosphorylation where there are
hundreds of protein kinases and phosphatases which regulate phosphorylation levels, O-
GlcNAc cycling is regulated only by two key enzymes, OGT and OGT. This implies that

10
O-GlcNAc might play a more general cellular role, like regulating cellular stress
responses or/and nutrient sensing as other groups have proposed previously.
Within cells O-GlcNAc levels have been found to increase in response to various cellular
stresses in several cell lines [245]. Stress induced elevation of O-GlcNAc modifications
is dynamic and normal basal levels are obtained after 24 to 48 h. Experimentally
increasing O-GlcNAc levels using transfection of OGT or OGA inhibitors results in
improved cell survival, whereas decreasing the O-GlcNAc level results in cells which are
less stress tolerant. Moreover, the Chatham group have intensively investigated a
potential cardioprotective role by elevating O-GlcNAc levels under ischemic conditions
[35]. They demonstrated that acute elevation of O-GlcNAc levels by inhibition of OGA
using PUGNAc, NButGT, or Thiamet-G leads to remarkable ischemic cardioprotection.
Studies using neonatal cardiomyocytes provided further support for OGT and OGA
playing critical roles in regulating the response to acute ischemic and oxidative stress
[34]. However, the specific mechanisms underlying the protection associated with
increased O-GlcNAc levels remains elusive. The protection may be due at least in part to
increased levels of heat shock proteins [77, 78]. Other explanations include the possibility
that O-GlcNAc may help to stabilize protein structures, or prevent protein aggregation
through conferring different biophysical properties to modified proteins.
As mentioned earlier, the O-GlcNAc modification has been proposed to be a nutrient
sensor. Numerous studies support the idea that a global increase in O-GlcNAc levels
causes insulin resistance and impaired glucohomeostasis [88]. For example, in various in

11
vivo studies using different models, increased cellular O-GlcNAc levels have been
correlated with insulin resistance [51, 90, 96, 164, 242]. These studies have involved
overexpression of OGT in specific tissues. The most compelling of these studies have
produced insulin resistance through overexpression of OGT in the liver, using adenovirus
[51, 242], or the muscle and fat, through knock-in of an extra copy of OGT [164].
Supporting these findings are studies having shown that increasing O-GlcNAc levels in
cultured cells using PUGNAc leads to insulin resistance [81]. PUGNAc competitively
inhibits OGA, resulting in elevated O-GlcNAc modification levels in treated cells. Such
studies making use of PUGNAc have shown that it decreases insulin stimulated glucose
uptake in cultured adipocytes [224, 242] and muscle tissue [12] as well as altered
expression of gluconeogenic genes in cultured hepatocytes [51]. Collectively, studies
overexpressing OGT and using PUGNAc have provided compelling evidence that
elevated O-GlcNAc levels cause insulin resistance.
However, recently, the interpretation of these previous findings have been brought into
question by a number of new results. Firstly, it has been demonstrated that a reduction in
the levels of O-GlcNAc-modified proteins in cultured adipocytes did not relieve the
insulin resistance caused by culturing these cells in media containing high glucose [190].
Second, PUGNAc has been shown to be a promiscuous inhibitor with the ability to
inhibit a variety of different glycoside hydrolases [152]. PUGNAc inhibits the
functionally related HexA and HexB enzymes that reside in the lysosome and are
responsible for removing GlcNAc and GalNAc off of a variety of glycoconjugates [150,
207]. Studies using the more selective OGA inhibitor, NButGT, have shown that the off-

12
target effects of PUGNAc appear to be of serious concern since NButGT does not
recapitulate the insulin desensitizing effects of PUGNAc in cultured 3T3-L1 adipocytes
[147], and does not replicate the ability of PUGNAc to impair insulin-stimulated
activation of Akt in either cultured astrocytes or in 3T3-L1 adipocytes [161]. More
recently, results from animal studies have clearly demonstrated that treatment of rats and
mice with NButGT for various time regimens and doses dramatically increases O-
GlcNAc levels throughout all tissues, yet does not perturb insulin sensitivity or alter
glucohomeostasis [149]. These results suggest that pharmacological increases in global
O-GlcNAc levels do not cause insulin resistance nor do they appear to disrupt
glucohomeostasis. Consequently, these findings are intriguing in the context of the
proposed beneficial effects of elevated O-GlcNAc levels on the survival of cells in
response to various stresses [175, 245, 250]. Furthermore, these studies might open the
possibility that elevated O-GlcNAc levels, induced by inhibitors of OGA, could be
harnessed for therapeutic benefit without disruption of glucohomeostasis.
1.2 Amyotrophic lateral sclerosis (ALS)
1.2.1 Overview of ALS and its pathogenesis
Amyotrophic lateral sclerosis (ALS) is an adult-onset motor neuron disease first
described by the French neurologist Jean-Martin Charcot in 1869. It is characterized by
selective degeneration and death of motor neurons in the spinal cord and brainstem
causing progressive muscle weakness, atrophy, and eventual muscle paralysis and death
due to respiratory failure, most often within 2-5 years of diagnosis [32, 57].

13
Most cases of ALS are sporadic, having no known genetic component, and are referred to
as sporadic ALS (sALS). The remaining 10% of patients inherit the disease in an
autosomal dominant pattern, referred to as familial ALS (fALS). Approximately 20% of
familial cases have mutations in the Cu/Zn superoxide dismutase (SOD1) gene mapped to
chromosome 21q22 [193]. Mutations in other genes have also been found in various
subsets of familial ALS and rare cases of sporadic ALS, including the recently discovered
RNA processing proteins TAR DNA binding protein-43 (TDP-43) [112, 205].
Despite substantial efforts, the basis for the selective death of motor neurons in ALS
remains largely unknown, and no effective treatments have been developed to halt the
progression of the disease except for the glutamate release inhibitor, Riluzole, which has
limited benefit [184]. In the past decade, the pathogenesis of familial ALS has been
intensely investigated because of the development of transgenic mouse models with
human SOD1 mutations [18]. Since sporadic and familial ALS are clinically and
neuropathologically similar, progress in elucidating the mechanisms underlying familial
ALS may provide insight into both forms of the disease [27]. Based on the studies from
ALS patients and transgenic animal models overexpressing human SOD1 mutations,
multiple factors in multiple cell types might underlie the pathogenesis of ALS [18, 19,
130, 170, 241]. Many mechanisms have been proposed to be involved in the pathology of
ALS such as aberrant protein phosphorylation [122], defective axonal transport, oxidative
stress, mitochondrial damage and excitotoxicity [27]. More recently, aberrant RNA
metabolism has been proposed as yet another possible pathogenic mechanism [44, 124].

14
In 2006, TDP-43 was discovered as a major disease associated protein in ubiquitin
positive inclusions of ALS and other neurodegenerative diseases [10, 173]. However,
whether TDP-43 is a causative factor or merely a by-product of the disease process is
unclear. Since mutations of TDP-43 have been uncovered in several familial ALS and
sporadic ALS cases [62, 74, 112, 205, 222], it is possible that TDP-43 may be a causative
factor for neuronal death in some patients. Furthermore, the pathological aggregation of
TDP-43 is seen in almost all sporadic ALS and most familial ALS cases examined,
making TDP-43 an important disease protein in the understanding of sporadic and
familial ALS.
1.2.2 Cu/Zn superoxide dismutase (SOD1) and ALS
1.2.2.1 Mutant SOD1 (mSOD) and gain of neurotoxicity
Currently, more than 140 different mutations have been identified in the SOD1 gene in
about 20% of fALS patients distributed throughout the 153-amino acid SOD1
polypeptide [18]. SOD1 is a homodimer of a ubiquitously expressed cytoplasmic
enzyme, each monomer of the mature form contains a copper ion, a zinc ion, and a
disulfide bond [220]. As an antioxidant, its normal function is to convert toxic superoxide
anions to hydrogen peroxide which is then converted into water by glutathione
peroxidase. The exact mechanisms underlying selective motor neuron death induced by
mutant SOD1 are still unclear. It was initially thought that SOD1 mutations would reduce
overall SOD1 activity, and thereby promote the accumulation of toxic superoxide radicals
[193]. The loss of normal dismutase function as a major causative factor in familiar ALS
was further supported by the observations of a 25-50% reduction of total cellular SOD1

15
activity in familial ALS patients with a SOD1 mutation [23, 24]. However, transgenic
mice expressing mutant human SOD1 G93A (glycine to alanine point mutant at amino
acid residue 93), SOD1 G37R , or SOD1 G85R developed an ALS-like phenotype, even
in the presence of the endogenous murine SOD1 [25, 79, 238], whereas SOD1 knockout
mice do not develop ALS [26]. Currently, it is believed that mutant SOD1-mediated
toxicity in ALS is not due to loss of function but instead to a gain of toxic functions that
are independent of the levels of SOD1 activity [18]. However, the exact molecular
mechanisms of the toxicity and pathways leading to motor neuron degeneration are still
largely debated [18, 181].
Pathological intracellular aggregates are characteristic neuropathological hallmarks of
ALS and consist of SOD1 and/or intermediate filament proteins such as neurofilaments
[39, 93, 101, 206]. Insoluble SOD1-containing intracellular aggregates typical of familial
ALS have been observed in motor neurons and surrounding glial cells in mutant SOD1
mice [26]. Moreover, aggregates have been shown to appear either before, or coincident
with, the onset of disease symptoms and accumulate along with disease progression. This
observation suggests that SOD1 aggregation may be an early event in disease
pathogenesis [25]. The mutations in SOD1 are known to destabilize the protein, and may
induce conformational changes leading to SOD1 misfolding [26, 229]. SOD1 misfolding
may subsequently trigger a downstream cascade that includes protein accumulation
and/or aggregation, possibly followed by impaired axonal transportation as well as
mitochondrial and/or proteasome dysfunction. These events may indirectly lead to
accumulation of reactive oxygen species and activation of the caspase cascade [18, 181].

16
Overall, SOD1 mutations have been detected in about 2% of ALS patients. However,
how SOD1 contributes to the disease in the majority ALS patients without mutant SOD1
is large unknown. There is some evidence that suggests that wild-type and mutant SOD1
may share a common SOD1 dependent neurotoxic mechanism. For example, one study
identified an aberrant SOD1-containing protein in spinal cord tissue from patients with
sporadic ALS and from patients with familial ALS, suggesting a possible role for SOD1
in both forms of the disease [74]. Furthermore, misfolded SOD1 with conformational
abnormalities has been identified in all forms of ALS. The misfolded, wild-type SOD1
from sporadic ALS inhibits fast axonal transport though a mechanism involving specific
activation of p38 kinase in the same manner as mutant SOD1 from familial ALS [20],
suggesting that sporadic and familial ALS patients may share a common disease
mechanism involving misfolded SOD1.
1.2.2.2 Transgenic murine models of mutant SOD1
The first transgenic mouse model of human G93A SOD1 mutation was generated in 1994
[79]. Since then, several lines of mice overexpressing human mutant SOD1 (mSOD) have
been characterized as models of ALS [25, 238]. It has been suggested that in mSOD
transgenic mice, the age of onset of the disease, the severity of pathological abnormalities
and clinical features are crucially dependent on the copy numbers of the mutant
transgene, or on the specific mSOD mutation [47, 183, 238]. Comparative studies reveal
a striking similarity in pathology between the mSOD mouse and human ALS, which
include the loss of motoneurons, muscle atrophy, and the presence of large numbers of

17
vacuoles and protein aggregates in the axons and perikaryal regions of motoneurons [79,
87, 238].
The G93A mSOD mouse is one of the most commonly used mouse models of ALS, and
these mice overexpress G93A mutant human SOD by 10-20 fold compared to
endogenous murine SOD. Clinical symptoms typically develop at about 100 days of age
when they manifest as hind limb weakness that results in a slow, unstable gait. With
disease progression, mice develop muscle atrophy mainly in the hindlimbs with body
weight loss. However, mice are still able to ambulate, feed, and right themselves. At 120-
125 days of age (end-stage of disease), eventual muscle paralysis occurs, and many mice
are unable to right themselves and have difficulty feeding [79].
The G93A mSOD mouse is a good model of ALS as the specific mSOD mutation in
these mice correlates with disease onset and the rate of progression [79]. Furthermore, as
in ALS, selective neuron vulnerability is observed in these G93A mice. In ALS, there is
variable involvement of motoneurons, and descending motor tracts such as the
corticospinal, bulbospinal, and rubrospinal tracts [57]. Remarkably, mice overexpressing
mSOD also demonstrate similar motor system defects in both motoneurons and
descending motor tracts [246]. It has also been found that the spinal nucleus of the
bulbocavernosus muscle (SNB), a homologous structure to the nucleus of Onuf in
humans, is spared in mSOD mice, paralleling observations in patients with ALS [83].
Sparing of motoneurons innervating extraocular muscles has been reported in these mice,
again paralleling observations in ALS tissue [80]. Generally, a reduction of motoneuron

18
numbers by 40-60% are seen in mSOD mice at the disease “end-stage” [83, 168]. These
reductions in motoneuron number are also parallel the extent of motoneuron loss in ALS
patients observed at autopsy [216].
1.2.2.3 Elevated phosphoprotein level in mSOD mice and ALS
The increase in protein levels of many protein kinases and phosphoproteins have been
found in mSOD mice compared to wild type littermates and in nervous system tissue
from ALS patients as compared to controls. For instance, elevated protein level and/or
activation of many protein kinases have been observed in G93A mSOD mice and ALS
spinal cord tissue [98, 99]. These protein kinases include PKC and GSK3, which may
augment neural death in ALS. Furthermore, p38, a member of the stress-activated
protein kinase family, has been found to be associated with the abnormally
phosphorylated side arms of neurofilament heavy and neurofilament middle proteins in
ALS tissue, as well as in mSOD mice [1]. The exact mechanisms underlying the
abnormality of protein phosphorylation in ALS are unclear. Protein phosphatases are
responsible for the de-phosphorylation of phosphoproteins including several kinases.
Therefore, it has been previously hypothesized that the identification of
hyperphosphorylation of a number of phosphoproteins in ALS tissue could also be the
result of impaired activation of one or more protein phosphatases [225]. Although the
previously reported findings were consistent with the above hypothesis [225], recent
observations have not demonstrated impairments in the activation and protein content of
the protein phosphatase known as calcineurin in mSOD mice [134]. It also could be
possible that abnormal phosphorylation of proteins might be due to the influence of other

19
types of post-translational modifications on these phosphoproteins, including O-GlcNAc
modification [71, 87].
1.2.3 TAR DNA binding protein-43 (TDP-43) and ALS
1.2.3.1 TDP-43 and its pathology in ALS
Recently, there has been extremely exciting progress in linking sporadic ALS and
familial ALS [10, 62, 112, 173, 205, 222]. Studies of ALS and frontotemporal lobar
degeneration (FTLD) with ubiquitin inclusions (UBI, FTLD-U) demonstrated related
neuropathology as well as involvement of the protein TDP-43 [10, 173]. Insoluble
cytoplasmic inclusions and nuclear to cytoplasmic mislocalization of this poorly
understood protein were observed. Since the initial reports, characteristic TDP-43
neuropathological features have been observed in most of the sporadic and familial ALS
tissue examined. Moreover, mutations in the gene TARDBP encoding TDP-43 have been
identified in both sporadic and familial ALS cases [62, 112, 205, 222]. Thus, TDP-43
mutations and resulting alterations in protein localization and function may become the
most important agent of pathogenesis associated with ALS to date.
TDP-43 is a highly conserved and ubiquitously expressed nuclear protein among
metazoans such as Caenorhabditis elegans, Drosophila melanogaster, mouse, and human
(> 55% amino acid conservation) [227]. In human, TARDBP is located at the
chromosomal locus 1p36.22 and is comprised of six exons, five of which encode a
predominantly nuclear protein, TDP-43, which is structurally related to the heterogeneous

20
ribonucleoprotein (hnRNP) family, includes two RNA recognition motifs (RRM)
domains flanked by the N-terminal and C-terminal regions [227]. The RRM motif
domains of TDP-43 are highly homologous among species; however, the glycine-rich
sequence varies significantly among all species, reflecting likely species-specific
functions in different organisms. TDP-43 has also been shown to be essential for
embryogenesis [198, 239].
The biological function of TDP-43 was relatively unexplored before it was identified as a
major disease protein in 2006. Its only well characterized roles includes suppression of
HIV gene expression mediated by blocking assembly of transcription complexes [177],
and alternative RNA splicing associated with exon skipping in both cystic fibrosis
transmembrane regulator (CFTR) [29] and apolipoprotein A-II (apoA-II) transcripts
[166]. Recently, TDP-43 has been shown to be capable of promoting exon inclusion
during splicing of the survival of motor neuron (SMN) pre-mRNA [21], and to play roles
in mRNA stability and transport [197], as well as microRNA biogenesis [137]. In terms
of mRNA stability, a precise molecular mechanism remains unclear. However, like many
members of the hnRNP family, the C-terminal region appears able to interact with other
cellular factors and permit the hnRNP complex to regulate splicing [157]. There is
evidence that TDP-43 binds to the C-terminal region (at 321-366 aa) of hnRNP A2
protein , where hnRNP complex formation is known to be critical for mRNA stability and
transport [46]. Its specific neuronal function is also largely unknown, however, two large
scale screening studies have identified either 6304 or 192 RNA binding targets for TDP-
43. Many of these targets are known to be involved in neuronal activities [182, 240].

21
Accordingly, this growing body of evidence indicates that TDP-43 functions in RNA
metabolism in neurons.
Consistent with its role as a dual DNA/RNA binding protein, under normal conditions,
most (more than 90%) of the TDP-43 proteins are located in the nucleus, while minor
fractions of TDP-43 are located in the cytoplasm of normal cells [228]. TDP-43 is
normally observed as yielding punctate nuclear staining with minimal cytosolic staining.
However, in brains and spinal cords from ALS cases, there is an abundance of cytosolic
TDP-43, some of which forms aggregates and adopts a compact round structure or fibril-
like structures, termed skeins, in surviving neurons, glia cells, as well as less dystrophic
neurites (DN). Currently in the field, these characteristic compact round or skein-like
structures are defined as neuronal cytoplasmic inclusions (NCI) and neuronal intranuclear
inclusions (NII) when they are localized in the neurons, and as glial cytoplasmic
inclusions (GCI) when they are localized in glial cells. Some of these aggregates and
inclusions are ubiquitinated [10, 173]. Analysis by gel electrophoresis of TDP-43
obtained from insoluble protein extracts isolated from affected brain regions of ALS
patients reveals it forms a high molecular weight smear, a hyperphosphorylated ~ 45 kDa
species, abnormal C-terminal fragments of ~ 25 kDa, all in addition to the expected 43
kDa band [10, 173].
After the initial report that the characteristic lesions obtained in sporadic ALS were
composed of TDP-43, the role of TDP-43 in familial ALS was explored further. Despite
the fact that the majority of studies on TDP-43 in human ALS cases are generally

22
consistent [10, 61, 155, 173], some aspects of the TDP-43 pathology remain
controversial. For instance, while TDP-43 pathology is a consistent feature in all sporadic
ALS and most familial ALS cases, there is neither neuropathological nor biochemical
evidence for TDP-43 pathology in most familial ALS cases caused by SOD1 mutations
[154, 210]. In contrast, others investigating familial ALS cases carrying various SOD1
mutations show mislocalization of TDP-43 to the cytoplasm as well as association with
UBIs [189, 208]. Studies of some lines of mSOD mouse models have claimed that there
is no TDP-43 redistribution in these mice [189, 217], while others reported that many
neurons from the lumbar spinal cords of mSOD mice do show cytoplasmic TDP-43
inclusions similar to those seen in CNS tissue from patients with ALS [146, 208]. Thus, it
remains unclear whether familial ALS with a SOD1 mutation and other familial and
sporadic ALS cases are caused by a shared mechanism that involves TDP-43 and leads to
motor neuron degeneration.
The mechanistic aspects leading to accumulation of pathologic TDP-43 in cytoplasmic
and nuclear inclusions and the functional consequences of TDP-43 accumulation are not
well understood. The subcellular redistribution of TDP-43 from the nucleus to the
cytoplasm in affected cells in ALS suggests, however, that loss of its normal nuclear
function in transcription and mRNA processing might play a pathogenic role. The
formation of pathologic TDP-43 in cytoplasmic and nuclear inclusions might have arisen
because TDP-43 trafficking between the nucleus and the cytoplasm is disturbed and/or
there is an impairment of TDP-43 protein degradation. Supporting this view, studies
using immunoelectron microscopy have shown that immunogold-labeled TDP-43 is

23
deposited primarily in the nucleus, and frequently in the rough endoplasmic reticulum
(rER), and to a lesser extent, in the mitochondria and the synaptic vesicles of the
presynaptic terminals in spinal cord neurons from both controls and ALS cases. However,
in ALS, a reduction in TDP-43 immunogold-labeled deposits was observed in the nuclei,
while elevated TDP-43 immunogold-labeled deposits were observed at the rER within
ALS cases [194]. These findings suggest that TDP-43 is synthesized at the rER surface
within the cytoplasm and translocated to the nucleus, and in ALS, TDP-43 trafficking
between the nucleus and the cytoplasm might be disturbed. Other recent cell biology
studies have shown that TDP-43 continuously shuttles between the nucleus and
cytoplasm, a process partially regulated by nuclear localization signal (NLS) and nuclear
export signal (NES) motifs [14, 236]. Restricting TDP-43 from entering the nucleus by
mutation of the NLS motif in cell culture systems is reported to lead to cytoplasmic TDP-
43 aggregates, involving sequestration of endogenous TDP-43, and leads to a depletion of
nuclear TDP-43 [236]. Thus, perturbing the normal shuttling of TDP-43 between the
nucleus and cytoplasm may lead to both the formation of cytoplasmic inclusions and loss
of nuclear TDP-43.
Alternatively, generation of abnormal TDP-43 species such as hyperphosphorylated
TDP-43 enriched in inclusions might induce a toxic gain of function in a similar manner
as pathological hyperphosphorylated tau does in AD [107]. Recent studies have
demonstrated that pathological hyperphosphorylated TDP-43 is detected in the CNS of
ALS cases but not in normal control CNS [10, 89, 173]. Furthermore, TDP-43 serine
residues 409 and 410 (S409/410) have been identified as the major pathological

24
phosphorylation sites through detection with rabbit polyclonal antibodies or rat or mouse
monoclonal antibodies These residues are consistently phosphorylated in the TDP-43
inclusions of ALS patients but not on nonpathological TDP-43 [89, 102, 172].
Phosphorylation specific antibodies to pS409/410 label all types of pathological
inclusions, such as dystrophic neurites, neuronal cytoplasmic and nuclear inclusions, as
well as glial cytoplasmic inclusions in ALS as well as FTLD-U [89, 172]. In some cases,
a pS409/410 specific antibody has been shown to label granular, cytoplasmic, ‘pre-
inclusions’ that are not detectable by anti-ubiquitin, suggesting that phosphorylation at
S409/410 may precede ubiquitination [172]. Furthermore, it has also been shown in a
Caenorhabditis elegans model of TDP-43 that phosphorylation at S409/410 promotes
mutant TDP-43 toxicity [135]. Taken together, the pathological significance of
phosphorylation on TDP-43 for ALS disease development is still unclear, however, these
data have led to the view that hyperphosphorylation of TDP-43 might contribute to
neurotoxicity. With respect to regulation of TDP-43 phosphorylation, it has been shown
that casein kinase-1 may be involved in hyperphosphorylation of TDP-43 in ALS brains
[89, 102]. Consistent with this finding, 29 Ser/Thr residues on recombinant TDP-43 have
been identified as casein kinase-1 phosphorylation sites. Of note, 18 of them were
localized in the C-terminal glycine-rich domain including S409/410 [114]. It remains
unknown as to whether and why abnormal phosphorylation of TDP-43 leads to its
accumulation to form inclusions. Given that TDP-43 mutations have only been found in ~
3-5% of ALS patients, yet TDP-43 hyperphosphorylation and pathology has been
observed in almost all ALS cases examined, it is reasonable to speculate that abnormal
phosphorylation might be responsible for TDP-43 aggregation.

25
1.2.3.2 TDP-43 mutations – loss of function or gain of toxic function?
The presence of TDP-43 pathology in ALS made TARDBP, the gene encoding TDP-43, a
promising candidate for genetic screening. Analysis of large ALS cohorts have now led
to the identification of more than 30 different mutations in unrelated ALS patients
(Figure 1-2), which were absent in healthy controls [62, 112, 179, 205, 222, 243]. Less
than 50% of the mutations were found in familial ALS following an autosomal dominant
pattern of inheritance, while others were reported only in sporadic ALS cases. TDP-43
neuropathology has been examined in some ALS cases with TDP-43 mutations including
G294A [179], G298S [222], and Q343R [243] showing the distribution of the
characteristic TDP-43 positive neuronal and glial inclusions in addition to motor neuron
loss. Notably, TDP-43 mutations have only been identified in ALS cases, and have not
been found in other neurodegenerative diseases that also have TDP-43 pathology,
providing strong evidence for a direct link between TDP-43 dysfunction and ALS.
Figure 1- 2 Schematic diagram of TDP-43 with characteristic functional domains of TDP-43 and sites of the identified mutations in familial (fALS) and sporadic (sALS) Amyotrophic Lateral Sclerosis (ALS). Abbreviations: RRM (RNA Recognition Motif); NLS (nuclear localization sequence); NES (nuclear export sequence). Currently, neither the functional consequences of TDP-43 mutations nor the mechanisms
of mutant TDP-43 leads to neuronal degeneration are fully understood. Interestingly,

26
except for the A90V mutation localized in the NLS region [237], the Y374X truncation
mutation [48], and the missense D169G mutation localized in the RRM1 [112], all other
missense TDP-43 mutations are clustered in exon 6. Mutations in the exon 6 region affect
highly conserved amino residues located in the glycine-rich C-terminal region of TDP-
43, indicating abnormal RNA metabolism may be associated with mutated TDP-43 [46,
136, 236]. These new findings highlight a previous observation that showed aberrant
exon and intron splicing are the cause of abnormal RNA metabolism in sporadic ALS
[136]. Further support for this view is the discovery of another functionally related
DNA/RNA-binding protein, fused in sarcoma/translocation in liposarcoma (FUS/TLS),
which also causes some cases of familial ALS [123].
Recent studies have suggested that certain missense mutations may cause a toxic gain of
function through the intracellular accumulation and aggregation of proteolytically
cleaved TDP-43 fragments. Mutations may also cause a loss of function through the
aberrant phosphorylation and cellular distribution of TDP-43. For example, compared
with human wild-type TDP-43, human Q331K and M337V TDP-43 mutations promote
dendritic branching to a much lesser extent in Drosophila neurons [141]. Some mutations
(Q331K, M337V, Q343R, N345K, R361S and N390D) accelerate the aggregation of
TDP-43 in vitro and enhance aggregate formation and toxicity in yeast [111], while other
mutations appear to increase the propensity of TDP-43 to become fragmented. For
instance, immunoblot analysis of TDP-43 from frozen lymphocyte lysates revealed that
the expression level of an aberrant 30–32 kDa band is elevated in samples from familial
ALS cases with A382T or S393L mutations, compared with samples from patients

27
without mutations or from controls [45]. In contrast, some other mutations (D169G,
G294A, Q331K, M337V, Q343R, N390D and N390S) do not facilitate inclusion
formation of full-length TDP-43 in SH-SY5Y cells, but they do enhance the aggregation
of the C-terminal fragment of TDP-43, suggesting that ALS associated mutations, in
combination with the C-terminal fragment, promote abnormal TDP-43 accumulation in
mammalian cells [176]. Finally, many mutations result in substitutions to threonine and
serine residues [112] and may thus increase TDP-43 phosphorylation, which could
adversely impact various TDP-43 functions and alter its aggregation potential.
Interestingly, based on phosphorylation site prediction scores from 0.0 (minimal
probability) to 1.0 (maximum probability), the disease related N390D and N390S
mutations have been predicted to increase the probability of phosphorylation at the S387
(0.959) and S393 (0.583) sites, respectively [112]. This is of interest, as both N390D and
N390S mutations both close to the major pathological phosphorylation sites identified at
serine residues 409 and 410 (S409/410) [89, 102, 172].
1.2.4 O-GlcNAc in ALS
It has been shown previously that the components of characteristic pathological
intracellular inclusions of ALS are all abnormally phosphorylated including
neurofilaments and TDP-43 [39, 89, 93, 101, 206]. Very recently, it has even been shown
that human SOD1 from human erythrocytes is phosphorylated [235]. In addition to
phosphorylation, reports have suggested that human neurofilaments [55, 56], rat SOD1
[204, 211] and TDP-43 from HeLa cells [171] are all post-translationally modified by O-

28
GlcNAc. However, whether or not SOD1 and TDP-43 are modified by O-GlcNAc within
CNS has not been investigated.
The process of O-GlcNAc modification of neurofilaments has been studied.
Phosphorylation of neurofilaments medium chain (NFM) is regulated by O-GlcNAc
modification both in vitro and in vivo [145]. Additionally, it has been demonstrated that
O-GlcNAc and phosphorylation levels on neurofilaments medium chain are reciprocal,
and O-GlcNAc modified neurofilaments medium chain levels are markedly decreased in
the spinal cords of rats having an ALS like disorder, while phosphorylation of
neurofilament medium chain is increased [145]. Altered O-GlcNAc levels have been
shown to be associated with hyperphosphorylation of neurofilaments, which may be
involved in the pathogenesis of ALS [31]. However, the role of O-GlcNAc modification
of disease proteins implicated in ALS remains unknown. Accordingly, mechanisms that
can regulate the phosphorylation and /or stability of disease proteins are of interest since
it may be possible to exploit them to intervene in the progression of disease.
Putative pathogenic mechanisms mediated by mutant SOD1 may be relevant in both
familial and sporadic cases of ALS given their clinical similarity, but the disparity
between the success in treating SOD1-based animal models and the failure to find human
therapeutics is troubling [18]. New findings that wild-type and mutant SOD1 can share a
common aberrant conformation in ALS [20] has focused researchers on investigating
ways to stabilize protein structure [28]. Increased O-GlcNAc modification of these
disease proteins might contribute to increased stability and/or limit their

29
hyperphosphorylation, which in turn could offer sorely needed new approaches to
altering disease progression [28].
1.3 Alzheimer’s disease (AD)
1.3.1 Overview of AD, neuropathological features and major hypotheses
Alzheimer’s disease (AD), the most common cause of dementia in adults, is a
neurodegenerative disease of the brain that is clinically characterized by a progressive
decline of cognitive function and memory. Pathologically, the two hallmarks of disease
are extracellular amyloid plaques composed of amyloid β-peptide (Aβ) derived from
amyloid precursor protein (APP) [86], intracellular neurofibrillary tangles (NFT)
composed of hyperphosphorylated tau protein [72, 73], and extensive loss of both
synapses and neurons. In the last two decades, many advances have been made
concerning the mechanisms of the disease, especially with regard to APP processing, Aβ
deposition, and tau abnormalities. However, the mechanisms leading to the impairment of
cognition and memory in AD patients remain poorly understood and controversial.
Despite scientific advances, there is still no treatment available for delaying the onset or
preventing cognitive symptoms of AD [104].
There are two main forms of the disease; ~ 5 % of patients have familial AD, the
inherited form which mainly affects people younger than 60 (early-onset AD, EOAD),
while the remaining ~ 95 % of all AD patients occurs most often in adults aged 60 and
older (later-onset AD, LOAD) and is not inherited. This later and larger group are
denoted as suffering from sporadic AD [185]. Familiar AD cases are caused by mutations

30
in the following three different transmembrane proteins: amyloid precursor protein,
presenilin 1 (PS1), and presenilin 2 (PS2). In contrast, the majority of sporadic AD cases
are not linked to any known gene mutations, but are suggested to be linked to
neuroinflammation, diabetes, and environmental factors, as well as the presence of one or
two alleles of apolipoprotein 4 (ApoE4) [185].
The neuropathological features of AD have been described in detail [64, 234]. At the
macroscopic level, there is the atrophy of the brain, and this is largely due to extensive
neuronal loss. At the microscopic level, the neuronal loss is primarily in the entorhinal
cortex, hippocampus and amydala, frontal, parietal and temporal cortices of the brain.
Neurons in layer II of the entorhinal cortex and the pyramidal layer of the hippocampal
CA1 region are particularly vulnerable. The reason for this selective vulnerability
remains unclear. In addition to neuronal loss, synaptic loss in the hippocampus and
neocortex has been implicated as an early event [92, 159], and is the major structural
alteration which is correlated to cognitive dysfunction [16]. However, the exact
mechanism for synapse loss, especially in affected regions remains unclear, although it
has been speculated that it might be due to disrupted axonal transport or impaired
mitochondrial function.
Extracellular amyloid plaques (AP), are found in specific brain regions within the limbic
system and neocortex, and are composed of Aβ40 and Aβ42 peptides, 40 and 42 amino
acid peptides derived from a much larger protein APP [86]. Within amyloid plaques, Aβ
is present in aggregated forms including insoluble fibrils and soluble oligomers [116].
The amyloid plaques are surrounded by degenerating neurites, which are composed of

31
axons and/or dendrites. In areas surrounding amyloid plaques, there is also gliosis with
cells having a hypertrophic morphology, as well as the proliferation of astrocytes and
microglia. It is likely that this inflammatory response contributes to brain injury, although
there is evidence that glial cells can also play a protective role [144]. Interestingly,
amyloid plaques are also found in the neocortex of normal elderly people, however, these
amyloid plaques lack dystrophic neurites with neurofibrillary changes surrounding the
amyloid plaque cores [52]. The other characteristic hallmark of AD, intracellular
neurofibrillary tangles, are observed primarily in the neocortex and hippocampal regions,
and are made up of insoluble paired helical filaments (PHF) in the cell soma and
dystrophic neurites sometimes surrounding the amyloid plaque core, as well as in distal
dendrites, also known as neuropil threads, scattered throughout grey matter.
The relationship between these two core lesions and their roles in the etiology and
pathogenesis of AD has long been debated. People who favor the amyloid cascade
hypothesis (colloquially known as “Baptists”) believe that increased Aβ42 production
and accumulation leads to AD primarily through Aβ42 oligomerization and deposition as
AP which induces NFT. The lesions are then associated with widespread
neuronal/neuritic dysfunction and cell death in specific brain regions resulting in
cognitive and memory impairment [86]. People who favor the tau hypothesis
(colloquially known as “Tauists”) believe that hyperphosphorylated tau aggregates into
NFT in neuronal perikarya and NFT or soluble tau aggregates alone leads to AD [107].

32
It has emerged that Aβ toxicity is tau dependent [223], although mechanistically this link
remains unknown. One recent study made a link between tau and APP by showing that
tau mediated Aβ-induced excitotoxicity at the synapse through binding of tau with the
Src kinase known as Fyn to the NMDA receptor. It was also shown that depletion of tau
in a mouse model of AD rescues memory and longevity without changing Aβ levels and
amyloid plaque burden, therefore suggesting that removal of tau may exert
neuroprotection against Aβ excitotoxic insults in this mouse model [108]. A further
discussion of tau protein and its association with the pathogenesis of AD will be
discussed in detail below.
1.3.2 Tau and AD
1.3.2.1 Tau structure and its biological function
Tau is a microtubule-associated protein (MAP) in the CNS encoded by a single gene,
MTBP, on chromosome 17. In human brain, six isoforms of tau are expressed as a result
of alternatively splicing of the tau gene, and the resulting proteins range from 352 to 441
amino acids in length [67]. The presence or absence of the second microtubule binding
domain/repeat of 31 or 32 amino acids near the C-terminal region, which results from
alternative splicing of tau exon 10, divides tau isoforms into two groups: 3R-tau
(containing three microtubule binding repeats) and 4R-tau (containing four three
microtubule binding repeats), and each group includes three isoforms respectively,
depending on whether these isoforms contain zero, one, or two inserts of 29 amino acids
at the N-terminal region of tau [9, 68] (Figure 1-3). Normal adult human brain expresses
approximately equal levels of 3R-tau and 4R-tau [119]. Equal levels of 3R-tau and 4R-
tau have been suggested to be important for maintaining optimal neuronal physiology

33
[66]. Tau has little secondary structure, and it is a mostly random coil with -structure in
the central microtubule binding repeats.
Figure 1- 3 Alternative splicing and domain structure of tau. Six isoforms of tau expressed in adult human brain are derived from a single gene, MTBP, localized on chromosome 17 via alternative splicing. Microtubule binding repeats (R) are located in the C-terminal of tau. Two N-terminal inserts (N) are characteristic of the larger isoforms. 4R2N is the longest isoform of tau and contains 441 amino acids (aa). Tau is known to promote the stability and assembly of microtubules via interactions with
tubulin in neurons [232], thereby facilitating the distribution of proteins and nutrients
within neurons [115]. Consistent with this role, tau is present within neurons
predominantly in the axons where it is known to interact with dynein and kinesin, two
motor proteins which move along tau decorated microtubules. These observations
suggest that tau could modulate axonal transport in neurons [53]. Tau is a
phosphoprotein, and its biological function is regulated by its phosphorylation status.

34
Fetal tau is more highly phosphorylated than adult tau, and the degree of phosphorylation
of the six isoforms decreases with age, perhaps because of the activation of phosphatases
[162]. Hyperphosphorylation of tau depresses its normal ability to be involved in
microtubule assembly and stabilization [6, 7]. In addition to abnormal
hyperphosphorylation, tau also undergoes several other abnormal post-translational
modifications such as nitration, ubiquitination, and acetylation [41, 69, 167].
1.3.2.2 Mechanisms for the formation of NFT
Tau from normal human brain contains ~ 3 mol of phosphate per mol of tau [117].
However, tau from AD brain is abnormally hyperphosphorylated with ~ 7 to 10 mol of
phosphate per mol of tau [117]. In the adult human brain, 4R2N/441aa tau, the longest
isoform of adult human brain tau, has 80 serine (Ser) and threonine (Thr) residues [68].
Of these, at least 30 different residues have been found to be phosphorylated on tau from
AD brain [69]. Many of these sites are also phosphorylated in normal brain without NFT
but to a lesser extent. These phosphorylation sites mainly localize in the regions that
flank the microtubule binding repeats, specifically, in the proline-rich region (residues
172-251) and the C-terminal region (residues 368-441) [139]. Studies suggest that the
phosphorylation of tau at different sites or regions affects tau function in different ways.
For example, by using affinity chromatography to isolate tau with various levels of
phosphorylation, it was found that neither under-phosphorylated nor highly
phosphorylated tau isoforms are associated with microtubules, suggesting that
phosphorylation at certain sites is needed for tau binding to microtubules while
phosphorylation at some other sites may prevent the association [60].

35
Numerous studies have demonstrated that hyperphosphorylated tau is the major
component of PHF, which forms NFT, neuropil threads, and plaque dystrophic neurites
in AD [72, 73, 105, 106, 126]. In patients with AD, the proper assembly of tau
microtubule complexes is disrupted by hyperphosphorylation of tau which is generally
believed to result in its self-aggregation into PHF, which leads to the formation of NFT.
The correlation between the levels of NFT in the brains of AD patients, but not the AP,
and the severity of dementia strongly supports a role for tau dysfunction in AD [3, 4, 6,
72, 73].
Although the precise molecular causes of tau hyperphosphorylation have remained
elusive, several protein kinases and protein phosphatases are implicated [5, 103].
Dynamic regulation of phosphorylation by protein kinases and phosphatases determines
the phosphorylation level on tau, and abnormal hyperphosphorylation of tau could arise
from the dysregulation of this dynamic process. Much effort was made in the last decade
to identify the kinases and phosphatases that regulate tau phosphorylation in the brain.
Accumulated studies have demonstrated that the major tau kinases include GSK-3β,
cyclin-dependent protein kinase 5 (cdk5), cAMP-dependent protein kinase (PKA), and
microtubule affinity-regulating kinase 1 (MARK1) [103, 163]. Each kinase favors its
own set of phosphorylation sites or regions on tau. For example, PKA mainly
phosphorylates tau in multiple regions including Ser214, Ser262, Ser356 and Ser409,
while GSK-3β prefers to phosphorylate at Ser-Pro or Thr-Pro motifs located in proline-
rich regions. The most favorable phosphorylation sites of GSK-3β are Ser396 and Ser404

36
in the C-terminal region, and when certain sites such as Ser214 and Ser262 are
phosphorylated, it inhibits tau from binding to microtubules [103, 196, 201]. Abnormally
hyperphosphorylated tau not only loses its biological activity and disassociates from
microtubules, but it also promotes its self-polymerization. Instead of stimulating
microtubule assembly, the cytosolic abnormally hyperphosphorylated and oligomeric tau
acts to sequester normal tau and other MAPs and causes disruption of microtubules [133].
Furthermore, other observations have shown that a conformational change of tau protein
precedes its aggregation into PHF and is one of the earliest events in AD [231]. The
conformational change in tau could also arise from the hyperphosphorylation of tau [8].
Besides abnormal hyperphosphorylation, other tau abnormalities also appear to contribute
to tau pathology. For instance, truncation of tau has been demonstrated in AD brains [58].
Studies using a mouse monoclonal antibody, Tau C3, which only reacts with C-terminal
truncated tau at D421, have shown that the truncated tau appears to reside in a highly
stable detergent-insoluble fraction in AD brain and immunohistochemically stains NFT-
like morphology [58, 75, 76, 169]. Moreover, pathological tau is also
hyperphosphorylated, and can undergo conformational changes which are recognized by
pathological conformation specific antibodies such as MC1 and Alz50 [188]. Truncation
of tau has also been shown in NFT in a transgenic mouse model of AD with a P301L tau
mutation [247, 248]. These data suggest that truncation of tau may be involved in tau
pathology. In addition, tau in AD brain has been shown to be nitrated [187]. There are
five tyrosine (Tyr) residues (Tyr18, Tyr29, Tyr197, Tyr310, and Tyr394) that span the
length of the longest tau isoform [68]. Notably, one study has shown that site-specific tau
nitration promotes a pathological conformation of tau as demonstrated by using the Alz50

37
antibody [186]. In addition, an antibody (tau-nY29) that site-specifically detects tau
nitration at Y29, robustly stains the typical NFT lesions in AD brain [187]. This
observation suggests the nitration of tau can be another disease specific marker for AD.
1.3.2.3 O-GlcNAc modification regulates tau phosphorylation
It is known previously that bovine tau is O-GlcNAc modified [13], and in neuroblastoma
cells, O-GlcNAc residues are mainly found on less phosphorylated tau, whereas highly
phosphorylated tau has less O-GlcNAc [127], raising the possibility that O-GlcNAc
modification might regulate tau phosphorylation. This observation has been supported by
the fact that human tau is O-GlcNAc modified [138]. Besides, it is also known that
impaired glucose metabolism is an early hallmark of Alzheimer disease [195]. Also, tau
O-GlcNAc levels in human AD brains are markedly lower than in healthy brains and
hyperphosphorylated tau bears little O-GlcNAc, further supporting a reciprocal
relationship between O-GlcNAc and phosphorylation on tau [138]. Together, these data
suggest that impaired brain glucose uptake/metabolism might cause dysfunction of O-
GlcNAc processing, lead to tau hyperphosphorylation, and contribute to the pathological
aggregation of tau in AD.
Consistent with this view, genome-wide association (GWAS) and biochemical studies
have linked decreased levels of glucose transporters to AD pathology [140, 200].
Specifically, the major glucose transporters (GLUT) responsible for glucose uptake into
neurons from the blood stream, GLUT1 and GLUT3, were found to be decreased in AD
brain [140]. This decrease in GLUT1 and 3 correlated with a decrease in protein O-

38
GlcNAc modification, hyperphosphorylation of tau, and to the density of NFT in human
brains [140]. Based on these findings, it has been proposed that a decrease in tau O-
GlcNAc results in abnormal hyperphosphorylation of tau and leads to Alzheimer
neurofibrillary degeneration [244].
Although cellular stresses contribute to the pathological hyperphosphorylation of tau [65,
118, 180], they are also known to increase O-GlcNAc levels [245]. The molecular
rationale for elevated O-GlcNAc levels in response to cellular stress remains unknown
but it seems possible that O-GlcNAc is an adaptive protective response that could limit
pathological protein phosphorylation under conditions of cellular stress. Elevated O-
GlcNAc levels may therefore serve a protective function in healthy mammalian brain by
limiting tau hyperphosphorylation. The lower levels of O-GlcNAc observed in the AD
brain, and within NFT, could consequently reflect a failure of this protective mechanism.
Tau O-GlcNAc levels are dynamically regulated by OGT and OGA. This regulation
offers the possibility of altering tau phosphorylation by targeting these two enzymes.
Small potent inhibitory compounds that selectively inhibit OGA, the enzyme that
removes O-GlcNAc from proteins, have recently been developed [153]. A new OGA
inhibitor, with more selectivity and potency, thiamet-G, has been shown to decrease tau
phosphorylation at pathologically relevant sites both in cultured PC12 cells and in rat
brains [244]. These inhibitors have the potential to be used to inhibit tau
hyperphosphorylation by increasing tau O-GlcNAc levels.

39
1.3.3 Transgenic animal models of AD
The discoveries that the mutations of the genes (APP and MAPT, respectively) that
encode the proteins that are deposited in the AP and NFT in familial AD and
Frontotemporal Dementia with Parkinsonism-17 (FTDP-17) suggest a causal role for
these proteins in neurodegenerative disease, and have led to the generation of transgenic
animal models of AD. These mice have become a widely used tool to study disease
pathogenic mechanisms and to test for various potential therapeutic strategies [70].
1.3.3.1 JNPL3 transgenic mouse model
JNPL3 transgenic mice, which express the human FTDP-17 P301L mutation in the
shortest four repeat tau isoform under the mouse prion promoter, reproduce tau
aggregation and NFT formation in the spinal cord, brainstem, midbrain, and
hypothalamus. These mice also develop motor deficits and neuronal loss in the spinal
cord [132]. These JNPL3 mice are an aggressive transgenic model showing impaired
motor function as early as ~ 6.5 months of age that is associated with the formation of
NFTs primarily in the spinal cord and hindbrain. JNPL3 mice also show body mass loss
during disease progression due to neurogenic atrophy of skeletal muscle [132, 219].
Biochemically, JNPL3 mice show hyperphosphorylated sarkosyl insoluble tau in spinal
cord and brain. A recent study also showed a correlation between the number of NFT and
decline in performance on the Morris water maze in JNPL3 mice, indicating a detrimental
effect of NFT or NFT precursors on memory [11]. Taken together, it is clear that the
JNPL3 mice recapitulate neurofibrillary pathology and tau-related neuronal dysfunction
and loss in vivo. Therefore, the JNPL3 mouse model is a popular animal model used to
study tau-mediated pathogenesis and to test treatments which target tau [221].

40
1.3.3.2 TAPP transgenic mouse model
To study amyloid pathology, another transgenic mouse model of AD, Tg2576, which
overexpresses human APP with the double Swedish mutation (K670N and M671L) under
the control of the hamster prion protein promoter, has also been developed. Tg2576 mice
produce APP at 5.5 fold over endogenous levels and develop diffuse and neuritic plaques
in the hippocampus, cortex, subiculum, and cerebellum at around 9 to 11 months of age
similar to those seen in AD [97].
Thus, JNPL3 and Tg2576 mice independently reproduce the typical neuropathological
features of NFT and AP respectively, however, TAPP mice, a double hemizygous
transgenic animal obtained by crossing JNPL3 mice and Tg2576 mice, exhibit both NFT
and AP [131]. Whereas the presence of tau does not affect amyloid pathology, the
presence of amyloid enhanced NFT formation in TAPP mice in the subiculum and
hippocampus - areas that do not show NFT in JNPL3 mice. At ~ 9 to 11 months of age
overall NFT levels are higher in TAPP mice than in JNPL3 mice of comparable age.
Amyloid deposits, composed of both Aβ40 and Aβ42, appear at ~ 6 months and
accumulate gradually, only increasing after ~ 8 months of age. The development of these
two pathologies result in a graduate onset of marked cognitive and motor deficiencies
manifesting as early as 6.5 months, and developing into severe deficiencies with about
90% penetrance by ~ 10 months of age. TAPP mice, with a complete AD-like pathology,
provide an excellent tool for studying the relationship between tau and APP as well as for
evaluating potential therapeutic strategies [221].

41
1.4 Reference List
[1] S. Ackerley, A.J. Grierson, S. Banner, M.S. Perkinton, J. Brownlees, H.L. Byers, M. Ward, P. Thornhill, K. Hussain, J.S. Waby, B.H. Anderton, J.D. Cooper, C. Dingwall, P.N. Leigh, C.E. Shaw, C.C. Miller, p38alpha stress-activated protein kinase phosphorylates neurofilaments and is associated with neurofilament pathology in amyotrophic lateral sclerosis, Molecular and cellular neurosciences 26 (2004) 354-364.
[2] Y. Akimoto, F.I. Comer, R.N. Cole, A. Kudo, H. Kawakami, H. Hirano, G.W. Hart, Localization of the O-GlcNAc transferase and O-GlcNAc-modified proteins in rat cerebellar cortex, Brain Res 966 (2003) 194-205.
[3] I. Alafuzoff, K. Iqbal, H. Friden, R. Adolfsson, B. Winblad, Histopathological criteria for progressive dementia disorders: clinical-pathological correlation and classification by multivariate data analysis, Acta neuropathologica 74 (1987) 209-225.
[4] A. Alonso, T. Zaidi, M. Novak, I. Grundke-Iqbal, K. Iqbal, Hyperphosphorylation induces self-assembly of tau into tangles of paired helical filaments/straight filaments, Proc Natl Acad Sci U S A 98 (2001) 6923-6928.
[5] A.C. Alonso, B. Li, I. Grundke-Iqbal, K. Iqbal, Mechanism of tau-induced neurodegeneration in Alzheimer disease and related tauopathies, Curr Alzheimer Res 5 (2008) 375-384.
[6] A.C. Alonso, T. Zaidi, I. Grundke-Iqbal, K. Iqbal, Role of abnormally phosphorylated tau in the breakdown of microtubules in Alzheimer disease, Proc Natl Acad Sci U S A 91 (1994) 5562-5566.
[7] A.D. Alonso, I. Grundke-Iqbal, H.S. Barra, K. Iqbal, Abnormal phosphorylation of tau and the mechanism of Alzheimer neurofibrillary degeneration: sequestration of microtubule-associated proteins 1 and 2 and the disassembly of microtubules by the abnormal tau, Proc Natl Acad Sci U S A 94 (1997) 298-303.
[8] C. Alonso Adel, A. Mederlyova, M. Novak, I. Grundke-Iqbal, K. Iqbal, Promotion of hyperphosphorylation by frontotemporal dementia tau mutations, J Biol Chem 279 (2004) 34873-34881.
[9] A. Andreadis, W.M. Brown, K.S. Kosik, Structure and novel exons of the human tau gene, Biochemistry 31 (1992) 10626-10633.
[10] T. Arai, M. Hasegawa, H. Akiyama, K. Ikeda, T. Nonaka, H. Mori, D. Mann, K. Tsuchiya, M. Yoshida, Y. Hashizume, T. Oda, TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Biochem Biophys Res Commun 351 (2006) 602-611.
[11] G.W. Arendash, J. Lewis, R.E. Leighty, E. McGowan, J.R. Cracchiolo, M. Hutton, M.F. Garcia, Multi-metric behavioral comparison of APPsw and P301L models for Alzheimer's disease: linkage of poorer cognitive performance to tau pathology in forebrain, Brain Res 1012 (2004) 29-41.
[12] E.B. Arias, J. Kim, G.D. Cartee, Prolonged incubation in PUGNAc results in increased protein O-Linked glycosylation and insulin resistance in rat skeletal muscle, Diabetes 53 (2004) 921-930.

42
[13] C.S. Arnold, G.V. Johnson, R.N. Cole, D.L. Dong, M. Lee, G.W. Hart, The microtubule-associated protein tau is extensively modified with O-linked N-acetylglucosamine, J Biol Chem 271 (1996) 28741-28744.
[14] Y.M. Ayala, P. Zago, A. D'Ambrogio, Y.F. Xu, L. Petrucelli, E. Buratti, F.E. Baralle, Structural determinants of the cellular localization and shuttling of TDP-43, J Cell Sci 121 (2008) 3778-3785.
[15] L. Bertram, D. Blacker, K. Mullin, D. Keeney, J. Jones, S. Basu, S. Yhu, M.G. McInnis, R.C. Go, K. Vekrellis, D.J. Selkoe, A.J. Saunders, R.E. Tanzi, Evidence for genetic linkage of Alzheimer's disease to chromosome 10q, Science 290 (2000) 2302-2303.
[16] K. Blennow, N. Bogdanovic, I. Alafuzoff, R. Ekman, P. Davidsson, Synaptic pathology in Alzheimer's disease: relation to severity of dementia, but not to senile plaques, neurofibrillary tangles, or the ApoE4 allele, J Neural Transm 103 (1996) 603-618.
[17] G. Boehmelt, A. Wakeham, A. Elia, T. Sasaki, S. Plyte, J. Potter, Y. Yang, E. Tsang, J. Ruland, N.N. Iscove, J.W. Dennis, T.W. Mak, Decreased UDP-GlcNAc levels abrogate proliferation control in EMeg32-deficient cells, EMBO J 19 (2000) 5092-5104.
[18] S. Boillee, C. Vande Velde, D.W. Cleveland, ALS: a disease of motor neurons and their nonneuronal neighbors, Neuron 52 (2006) 39-59.
[19] S. Boillee, K. Yamanaka, C.S. Lobsiger, N.G. Copeland, N.A. Jenkins, G. Kassiotis, G. Kollias, D.W. Cleveland, Onset and progression in inherited ALS determined by motor neurons and microglia, Science (New York, N.Y) 312 (2006) 1389-1392.
[20] D.A. Bosco, G. Morfini, N.M. Karabacak, Y. Song, F. Gros-Louis, P. Pasinelli, H. Goolsby, B.A. Fontaine, N. Lemay, D. McKenna-Yasek, M.P. Frosch, J.N. Agar, J.P. Julien, S.T. Brady, R.H. Brown, Jr., Wild-type and mutant SOD1 share an aberrant conformation and a common pathogenic pathway in ALS, Nat neurosci 13 (2010) 1396-1403.
[21] J.K. Bose, I.F. Wang, L. Hung, W.Y. Tarn, C.K. Shen, TDP-43 overexpression enhances exon 7 inclusion during the survival of motor neuron pre-mRNA splicing, J Biol Chem 283 (2008) 28852-28859.
[22] C. Bouche, S. Serdy, C.R. Kahn, A.B. Goldfine, The cellular fate of glucose and its relevance in type 2 diabetes, Endocr Rev 25 (2004) 807-830.
[23] A.C. Bowling, E.E. Barkowski, D. McKenna-Yasek, P. Sapp, H.R. Horvitz, M.F. Beal, R.H. Brown, Jr., Superoxide dismutase concentration and activity in familial amyotrophic lateral sclerosis, J Neurochem 64 (1995) 2366-2369.
[24] A.C. Bowling, J.B. Schulz, R.H. Brown, Jr., M.F. Beal, Superoxide dismutase activity, oxidative damage, and mitochondrial energy metabolism in familial and sporadic amyotrophic lateral sclerosis, J Neurochem 61 (1993) 2322-2325.
[25] L.I. Bruijn, M.W. Becher, M.K. Lee, K.L. Anderson, N.A. Jenkins, N.G. Copeland, S.S. Sisodia, J.D. Rothstein, D.R. Borchelt, D.L. Price, D.W. Cleveland, ALS-linked SOD1 mutant G85R mediates damage to astrocytes and promotes rapidly progressive disease with SOD1-containing inclusions, Neuron 18 (1997) 327-338.

43
[26] L.I. Bruijn, M.K. Houseweart, S. Kato, K.L. Anderson, S.D. Anderson, E. Ohama, A.G. Reaume, R.W. Scott, D.W. Cleveland, Aggregation and motor neuron toxicity of an ALS-linked SOD1 mutant independent from wild-type SOD1, Science (New York, N.Y 281) (1998) 1851-1854.
[27] L.I. Bruijn, T.M. Miller, D.W. Cleveland, Unraveling the mechanisms involved in motor neuron degeneration in ALS, Annu Rev Neurosci 27 (2004) 723-749.
[28] K.R. Brunden, J.Q. Trojanowski, V.M. Lee, Advances in tau-focused drug discovery for Alzheimer's disease and related tauopathies, Nat Rev Drug Discov 8 (2009) 783-793.
[29] E. Buratti, F.E. Baralle, Characterization and functional implications of the RNA binding properties of nuclear factor TDP-43, a novel splicing regulator of CFTR exon 9, J Biol Chem 276 (2001) 36337-36343.
[30] M.G. Buse, Hexosamines, insulin resistance, and the complications of diabetes: current status, Am J Physiol Endocrinol Metab 290 (2006) E1-E8.
[31] C. Butkinaree, K. Park, G.W. Hart, O-linked beta-N-acetylglucosamine (O-GlcNAc): Extensive crosstalk with phosphorylation to regulate signaling and transcription in response to nutrients and stress, Biochimica et biophysica acta 1800 (2009) 96-106.
[32] E.A.a.K. C., Amyotrophic lateral sclerosis: A synthesis of research and clinical practice. , Cambridge, UK., 1998.
[33] N. Cetinbas, M.S. Macauley, K.A. Stubbs, R. Drapala, D.J. Vocadlo, Identification of Asp174 and Asp175 as the key catalytic residues of human O-GlcNAcase by functional analysis of site-directed mutants, Biochemistry 45 (2006) 3835-3844.
[34] V. Champattanachai, R.B. Marchase, J.C. Chatham, Glucosamine protects neonatal cardiomyocytes from ischemia-reperfusion injury via increased protein O-GlcNAc and increased mitochondrial Bcl-2, American journal of physiology 294 (2008) C1509-1520.
[35] J.C. Chatham, R.B. Marchase, The role of protein O-linked beta-N-acetylglucosamine in mediating cardiac stress responses, Biochim Biophys Acta 1800 (2010) 57-66.
[36] S. Chitayat, J.J. Adams, H.S. Furness, E.A. Bayer, S.P. Smith, The solution structure of the C-terminal modular pair from Clostridium perfringens mu-toxin reveals a noncellulosomal dockerin module, J Mol Biol 381 (2008) 1202-1212.
[37] S. Chitayat, K. Gregg, J.J. Adams, E. Ficko-Blean, E.A. Bayer, A.B. Boraston, S.P. Smith, Three-dimensional structure of a putative non-cellulosomal cohesin module from a Clostridium perfringens family 84 glycoside hydrolase, J Mol Biol 375 (2008) 20-28.
[38] C.F. Chou, A.J. Smith, M.B. Omary, Characterization and dynamics of O-linked glycosylation of human cytokeratin 8 and 18, J Biol Chem 267 (1992) 3901-3906.
[39] S.M. Chou, H.S. Wang, K. Komai, Colocalization of NOS and SOD1 in neurofilament accumulation within motor neurons of amyotrophic lateral sclerosis: an immunohistochemical study, Journal of chemical neuroanatomy 10 (1996) 249-258.

44
[40] T.Y. Chou, G.W. Hart, C.V. Dang, c-Myc is glycosylated at threonine 58, a known phosphorylation site and a mutational hot spot in lymphomas, J Biol Chem 270 (1995) 18961-18965.
[41] T.J. Cohen, J.L. Guo, D.E. Hurtado, L.K. Kwong, I.P. Mills, J.Q. Trojanowski, V.M. Lee, The acetylation of tau inhibits its function and promotes pathological tau aggregation, Nature communications 2 (2011) 252.
[42] R.N. Cole, G.W. Hart, Cytosolic O-glycosylation is abundant in nerve terminals, J Neurochem 79 (2001) 1080-1089.
[43] R.N. Cole, G.W. Hart, Glycosylation sites flank phosphorylation sites on synapsin I: O-linked N-acetylglucosamine residues are localized within domains mediating synapsin I interactions, J Neurochem 73 (1999) 418-428.
[44] C. Colombrita, E. Onesto, C. Tiloca, N. Ticozzi, V. Silani, A. Ratti, RNA-binding proteins and RNA metabolism: a new scenario in the pathogenesis of amyotrophic lateral sclerosis, Arch Ital Biol 149 (2011) 83-99.
[45] L. Corrado, A. Ratti, C. Gellera, E. Buratti, B. Castellotti, Y. Carlomagno, N. Ticozzi, L. Mazzini, L. Testa, F. Taroni, F.E. Baralle, V. Silani, S. D'Alfonso, High frequency of TARDBP gene mutations in Italian patients with amyotrophic lateral sclerosis, Human mutation 30 (2009) 688-694.
[46] A. D'Ambrogio, E. Buratti, C. Stuani, C. Guarnaccia, M. Romano, Y.M. Ayala, F.E. Baralle, Functional mapping of the interaction between TDP-43 and hnRNP A2 in vivo, Nucleic acids research 37 (2009) 4116-4126.
[47] M.C. Dal Canto, M.E. Gurney, A low expressor line of transgenic mice carrying a mutant human Cu,Zn superoxide dismutase (SOD1) gene develops pathological changes that most closely resemble those in human amyotrophic lateral sclerosis, Acta neuropathologica 93 (1997) 537-550.
[48] H. Daoud, P.N. Valdmanis, E. Kabashi, P. Dion, N. Dupre, W. Camu, V. Meininger, G.A. Rouleau, Contribution of TARDBP mutations to sporadic amyotrophic lateral sclerosis, Journal of medical genetics 46 (2009) 112-114.
[49] Y. Deng, B. Li, F. Liu, K. Iqbal, I. Grundke-Iqbal, R. Brandt, C.X. Gong, Regulation between O-GlcNAcylation and phosphorylation of neurofilament-M and their dysregulation in Alzheimer disease, FASEB J 22 (2008) 138-145.
[50] R.J. Dennis, E.J. Taylor, M.S. Macauley, K.A. Stubbs, J.P. Turkenburg, S.J. Hart, G.N. Black, D.J. Vocadlo, G.J. Davies, Structure and mechanism of a bacterial beta-glucosaminidase having O-GlcNAcase activity, Nat Struct Mol Biol 13 (2006) 365-371.
[51] R. Dentin, S. Hedrick, J. Xie, J. Yates, 3rd, M. Montminy, Hepatic glucose sensing via the CREB coactivator CRTC2, Science (New York, N.Y) 319 (2008) 1402-1405.
[52] D.W. Dickson, H.A. Crystal, L.A. Mattiace, D.M. Masur, A.D. Blau, P. Davies, S.H. Yen, M.K. Aronson, Identification of normal and pathological aging in prospectively studied nondemented elderly humans, Neurobiology of aging 13 (1992) 179-189.
[53] R. Dixit, J.L. Ross, Y.E. Goldman, E.L. Holzbaur, Differential regulation of dynein and kinesin motor proteins by tau, Science (New York, N.Y) 319 (2008) 1086-1089.

45
[54] D.L. Dong, G.W. Hart, Purification and characterization of an O-GlcNAc selective N-acetyl-beta-D-glucosaminidase from rat spleen cytosol, J Biol Chem 269 (1994) 19321-19330.
[55] D.L. Dong, Z.S. Xu, M.R. Chevrier, R.J. Cotter, D.W. Cleveland, G.W. Hart, Glycosylation of mammalian neurofilaments. Localization of multiple O-linked N-acetylglucosamine moieties on neurofilament polypeptides L and M, J Biol Chem 268 (1993) 16679-16687.
[56] D.L. Dong, Z.S. Xu, G.W. Hart, D.W. Cleveland, Cytoplasmic O-GlcNAc modification of the head domain and the KSP repeat motif of the neurofilament protein neurofilament-H, J Biol Chem 271 (1996) 20845-20852.
[57] A. Eisen, and, C. Krieger, Amyotrophic lateral sclerosis: A synthesis of research and clinical practice. , Cambridge, UK., 1998.
[58] T.C. Gamblin, F. Chen, A. Zambrano, A. Abraha, S. Lagalwar, A.L. Guillozet, M. Lu, Y. Fu, F. Garcia-Sierra, N. LaPointe, R. Miller, R.W. Berry, L.I. Binder, V.L. Cryns, Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer's disease, Proc Natl Acad Sci U S A 100 (2003) 10032-10037.
[59] Y. Gao, L. Wells, F.I. Comer, G.J. Parker, G.W. Hart, Dynamic O-glycosylation of nuclear and cytosolic proteins: cloning and characterization of a neutral, cytosolic beta-N-acetylglucosaminidase from human brain, J Biol Chem 276 (2001) 9838-9845.
[60] J. Garcia de Ancos, I. Correas, J. Avila, Differences in microtubule binding and self-association abilities of bovine brain tau isoforms, J Biol Chem 268 (1993) 7976-7982.
[61] F. Geser, M. Martinez-Lage, L.K. Kwong, V.M. Lee, J.Q. Trojanowski, Amyotrophic lateral sclerosis, frontotemporal dementia and beyond: the TDP-43 diseases, J Neurol (2009).
[62] M.A. Gitcho, R.H. Baloh, S. Chakraverty, K. Mayo, J.B. Norton, D. Levitch, K.J. Hatanpaa, C.L. White, 3rd, E.H. Bigio, R. Caselli, M. Baker, M.T. Al-Lozi, J.C. Morris, A. Pestronk, R. Rademakers, A.M. Goate, N.J. Cairns, TDP-43 A315T mutation in familial motor neuron disease, Ann Neurol 63 (2008) 535-538.
[63] T.M. Gloster, W.F. Zandberg, J.E. Heinonen, D.L. Shen, L. Deng, D.J. Vocadlo, Hijacking a biosynthetic pathway yields a glycosyltransferase inhibitor within cells, Nat Chem Biol 7 (2011) 174-181.
[64] M. Goedert, Oskar Fischer and the study of dementia, Brain 132 (2009) 1102-1111.
[65] M. Goedert, M. Hasegawa, R. Jakes, S. Lawler, A. Cuenda, P. Cohen, Phosphorylation of microtubule-associated protein tau by stress-activated protein kinases, FEBS Lett 409 (1997) 57-62.
[66] M. Goedert, R. Jakes, Mutations causing neurodegenerative tauopathies, Biochim Biophys Acta 1739 (2005) 240-250.
[67] M. Goedert, M.G. Spillantini, R. Jakes, D. Rutherford, R.A. Crowther, Multiple isoforms of human microtubule-associated protein tau: sequences and localization in neurofibrillary tangles of Alzheimer's disease, Neuron 3 (1989) 519-526.
[68] M. Goedert, M.G. Spillantini, M.C. Potier, J. Ulrich, R.A. Crowther, Cloning and sequencing of the cDNA encoding an isoform of microtubule-associated protein

46
tau containing four tandem repeats: differential expression of tau protein mRNAs in human brain, Embo J 8 (1989) 393-399.
[69] C.X. Gong, F. Liu, I. Grundke-Iqbal, K. Iqbal, Post-translational modifications of tau protein in Alzheimer's disease, J Neural Transm 112 (2005) 813-838.
[70] J. Gotz, N. Deters, A. Doldissen, L. Bokhari, Y. Ke, A. Wiesner, N. Schonrock, L.M. Ittner, A decade of tau transgenic animal models and beyond, Brain pathology (Zurich, Switzerland) 17 (2007) 91-103.
[71] L.S. Griffith, B. Schmitz, O-linked N-acetylglucosamine levels in cerebellar neurons respond reciprocally to pertubations of phosphorylation, Eur J Biochem 262 (1999) 824-831.
[72] I. Grundke-Iqbal, K. Iqbal, M. Quinlan, Y.C. Tung, M.S. Zaidi, H.M. Wisniewski, Microtubule-associated protein tau. A component of Alzheimer paired helical filaments, J Biol Chem 261 (1986) 6084-6089.
[73] I. Grundke-Iqbal, K. Iqbal, Y.C. Tung, M. Quinlan, H.M. Wisniewski, L.I. Binder, Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology, Proc Natl Acad Sci U S A 83 (1986) 4913-4917.
[74] A. Gruzman, W.L. Wood, E. Alpert, M.D. Prasad, R.G. Miller, J.D. Rothstein, R. Bowser, R. Hamilton, T.D. Wood, D.W. Cleveland, V.R. Lingappa, J. Liu, Common molecular signature in SOD1 for both sporadic and familial amyotrophic lateral sclerosis, Proc Natl Acad Sci U S A 104 (2007) 12524-12529.
[75] A.L. Guillozet-Bongaarts, M.E. Cahill, V.L. Cryns, M.R. Reynolds, R.W. Berry, L.I. Binder, Pseudophosphorylation of tau at serine 422 inhibits caspase cleavage: in vitro evidence and implications for tangle formation in vivo, J Neurochem 97 (2006) 1005-1014.
[76] A.L. Guillozet-Bongaarts, F. Garcia-Sierra, M.R. Reynolds, P.M. Horowitz, Y. Fu, T. Wang, M.E. Cahill, E.H. Bigio, R.W. Berry, L.I. Binder, Tau truncation during neurofibrillary tangle evolution in Alzheimer's disease, Neurobiology of aging 26 (2005) 1015-1022.
[77] C. Guinez, A.M. Mir, Y. Leroy, R. Cacan, J.C. Michalski, T. Lefebvre, Hsp70-GlcNAc-binding activity is released by stress, proteasome inhibition, and protein misfolding, Biochem Biophys Res Commun 361 (2007) 414-420.
[78] C. Guinez, A.M. Mir, N. Martin, D. Leprince, J.C. Michalski, G. Vergoten, T. Lefebvre, Arginine 469 is a pivotal residue for the Hsc70-GlcNAc-binding property, Biochem Biophys Res Commun 400 (2010) 537-542.
[79] M.E. Gurney, H. Pu, A.Y. Chiu, M.C. Dal Canto, C.Y. Polchow, D.D. Alexander, J. Caliendo, A. Hentati, Y.W. Kwon, H.X. Deng, et al., Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation, Science (New York, N.Y 264 (1994) 1772-1775.
[80] C. Haenggeli, A.C. Kato, Differential vulnerability of cranial motoneurons in mouse models with motor neuron degeneration, Neurosci Lett 335 (2002) 39-43.
[81] R.S. Haltiwanger, K. Grove, G.A. Philipsberg, Modulation of O-linked N-acetylglucosamine levels on nuclear and cytoplasmic proteins in vivo using the peptide O-GlcNAc-beta-N-acetylglucosaminidase inhibitor O-(2-acetamido-2-deoxy-D-glucopyranosylidene)amino-N-phenylcarbamate, J Biol Chem 273 (1998) 3611-3617.

47
[82] R.S. Haltiwanger, G.D. Holt, G.W. Hart, Enzymatic addition of O-GlcNAc to nuclear and cytoplasmic proteins. Identification of a uridine diphospho-N-acetylglucosamine:peptide beta-N-acetylglucosaminyltransferase, J Biol Chem 265 (1990) 2563-2568.
[83] D.K. Hamson, J.H. Hu, C. Krieger, N.V. Watson, Lumbar motoneuron fate in a mouse model of amyotrophic lateral sclerosis, Neuroreport 13 (2002) 2291-2294.
[84] J.A. Hanover, M.W. Krause, D.C. Love, The hexosamine signaling pathway: O-GlcNAc cycling in feast or famine, Biochim Biophys Acta 1800 (2009) 80-95.
[85] J.A. Hanover, S. Yu, W.B. Lubas, S.H. Shin, M. Ragano-Caracciola, J. Kochran, D.C. Love, Mitochondrial and nucleocytoplasmic isoforms of O-linked GlcNAc transferase encoded by a single mammalian gene, Arch Biochem Biophys 409 (2003) 287-297.
[86] J. Hardy, D.J. Selkoe, The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics, Science (New York, N.Y) 297 (2002) 353-356.
[87] G.W. Hart, M.P. Housley, C. Slawson, Cycling of O-linked beta-N-acetylglucosamine on nucleocytoplasmic proteins, Nature 446 (2007) 1017-1022.
[88] G.W. Hart, C. Slawson, G. Ramirez-Correa, O. Lagerlof, Cross Talk Between O-GlcNAcylation and Phosphorylation: Roles in Signaling, Transcription, and Chronic Disease, Annu Rev Biochem (2011).
[89] M. Hasegawa, T. Arai, T. Nonaka, F. Kametani, M. Yoshida, Y. Hashizume, T.G. Beach, E. Buratti, F. Baralle, M. Morita, I. Nakano, T. Oda, K. Tsuchiya, H. Akiyama, Phosphorylated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Ann Neurol 64 (2008) 60-70.
[90] M. Hazel, R.C. Cooksey, D. Jones, G. Parker, J.L. Neidigh, B. Witherbee, E.A. Gulve, D.A. McClain, Activation of the hexosamine signaling pathway in adipose tissue results in decreased serum adiponectin and skeletal muscle insulin resistance, Endocrinology 145 (2004) 2118-2128.
[91] D. Heckel, N. Comtesse, N. Brass, N. Blin, K.D. Zang, E. Meese, Novel immunogenic antigen homologous to hyaluronidase in meningioma, Hum Mol Genet 7 (1998) 1859-1872.
[92] O. Heinonen, H. Soininen, H. Sorvari, O. Kosunen, L. Paljarvi, E. Koivisto, P.J. Riekkinen, Sr., Loss of synaptophysin-like immunoreactivity in the hippocampal formation is an early phenomenon in Alzheimer's disease, Neuroscience 64 (1995) 375-384.
[93] A. Hirano, H. Donnenfeld, S. Sasaki, I. Nakano, Fine structural observations of neurofilamentous changes in amyotrophic lateral sclerosis, J Neuropathol Exp Neurol 43 (1984) 461-470.
[94] G.D. Holt, G.W. Hart, The subcellular distribution of terminal N-acetylglucosamine moieties. Localization of a novel protein-saccharide linkage, O-linked GlcNAc, J Biol Chem 261 (1986) 8049-8057.
[95] G.D. Holt, C.M. Snow, A. Senior, R.S. Haltiwanger, L. Gerace, G.W. Hart, Nuclear pore complex glycoproteins contain cytoplasmically disposed O-linked N-acetylglucosamine, J Cell Biol 104 (1987) 1157-1164.

48
[96] M.P. Housley, J.T. Rodgers, N.D. Udeshi, T.J. Kelly, J. Shabanowitz, D.F. Hunt, P. Puigserver, G.W. Hart, O-GlcNAc regulates FoxO activation in response to glucose, J Biol Chem 283 (2008) 16283-16292.
[97] K. Hsiao, P. Chapman, S. Nilsen, C. Eckman, Y. Harigaya, S. Younkin, F. Yang, G. Cole, Correlative memory deficits, Abeta elevation, and amyloid plaques in transgenic mice, Science (New York, N.Y) 274 (1996) 99-102.
[98] J.H. Hu, K. Chernoff, S. Pelech, C. Krieger, Protein kinase and protein phosphatase expression in the central nervous system of G93A mSOD over-expressing mice, J Neurochem 85 (2003) 422-431.
[99] J.H. Hu, H. Zhang, R. Wagey, C. Krieger, S.L. Pelech, Protein kinase and protein phosphatase expression in amyotrophic lateral sclerosis spinal cord, J Neurochem 85 (2003) 432-442.
[100] P. Hu, S. Shimoji, G.W. Hart, Site-specific interplay between O-GlcNAcylation and phosphorylation in cellular regulation, FEBS Lett 584 2526-2538.
[101] P.G. Ince, J. Tomkins, J.Y. Slade, N.M. Thatcher, P.J. Shaw, Amyotrophic lateral sclerosis associated with genetic abnormalities in the gene encoding Cu/Zn superoxide dismutase: molecular pathology of five new cases, and comparison with previous reports and 73 sporadic cases of ALS, J Neuropathol Exp Neurol 57 (1998) 895-904.
[102] Y. Inukai, T. Nonaka, T. Arai, M. Yoshida, Y. Hashizume, T.G. Beach, E. Buratti, F.E. Baralle, H. Akiyama, S. Hisanaga, M. Hasegawa, Abnormal phosphorylation of Ser409/410 of TDP-43 in FTLD-U and ALS, FEBS letters 582 (2008) 2899-2904.
[103] K. Iqbal, C. Alonso Adel, S. Chen, M.O. Chohan, E. El-Akkad, C.X. Gong, S. Khatoon, B. Li, F. Liu, A. Rahman, H. Tanimukai, I. Grundke-Iqbal, Tau pathology in Alzheimer disease and other tauopathies, Biochim Biophys Acta 1739 (2005) 198-210.
[104] K. Iqbal, I. Grundke-Iqbal, Alzheimer's disease, a multifactorial disorder seeking multitherapies, Alzheimers Dement 6 (2010) 420-424.
[105] K. Iqbal, I. Grundke-Iqbal, A.J. Smith, L. George, Y.C. Tung, T. Zaidi, Identification and localization of a tau peptide to paired helical filaments of Alzheimer disease, Proc Natl Acad Sci U S A 86 (1989) 5646-5650.
[106] K. Iqbal, I. Grundke-Iqbal, T. Zaidi, P.A. Merz, G.Y. Wen, S.S. Shaikh, H.M. Wisniewski, I. Alafuzoff, B. Winblad, Defective brain microtubule assembly in Alzheimer's disease, Lancet 2 (1986) 421-426.
[107] K. Iqbal, F. Liu, C.X. Gong, C. Alonso Adel, I. Grundke-Iqbal, Mechanisms of tau-induced neurodegeneration, Acta neuropathologica 118 (2009) 53-69.
[108] L.M. Ittner, Y.D. Ke, F. Delerue, M. Bi, A. Gladbach, J. van Eersel, H. Wolfing, B.C. Chieng, M.J. Christie, I.A. Napier, A. Eckert, M. Staufenbiel, E. Hardeman, J. Gotz, Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer's disease mouse models, Cell 142 (2010) 387-397.
[109] S.P. Iyer, Y. Akimoto, G.W. Hart, Identification and cloning of a novel family of coiled-coil domain proteins that interact with O-GlcNAc transferase, J Biol Chem 278 (2003) 5399-5409.
[110] M. Jinek, J. Rehwinkel, B.D. Lazarus, E. Izaurralde, J.A. Hanover, E. Conti, The superhelical TPR-repeat domain of O-linked GlcNAc transferase exhibits

49
structural similarities to importin alpha, Nat Struct Mol Biol 11 (2004) 1001-1007.
[111] B.S. Johnson, D. Snead, J.J. Lee, J.M. McCaffery, J. Shorter, A.D. Gitler, TDP-43 is intrinsically aggregation-prone, and amyotrophic lateral sclerosis-linked mutations accelerate aggregation and increase toxicity, J Biol Chem 284 (2009) 20329-20339.
[112] E. Kabashi, P.N. Valdmanis, P. Dion, D. Spiegelman, B.J. McConkey, C. Vande Velde, J.P. Bouchard, L. Lacomblez, K. Pochigaeva, F. Salachas, P.F. Pradat, W. Camu, V. Meininger, N. Dupre, G.A. Rouleau, TARDBP mutations in individuals with sporadic and familial amyotrophic lateral sclerosis, Nat Genet 40 (2008) 572-574.
[113] K. Kamemura, G.W. Hart, Dynamic interplay between O-glycosylation and O-phosphorylation of nucleocytoplasmic proteins: a new paradigm for metabolic control of signal transduction and transcription, Prog Nucleic Acid Res Mol Biol 73 (2003) 107-136.
[114] F. Kametani, T. Nonaka, T. Suzuki, T. Arai, N. Dohmae, H. Akiyama, M. Hasegawa, Identification of casein kinase-1 phosphorylation sites on TDP-43, Biochem Biophys Res Commun 382 (2009) 405-409.
[115] S. Kar, J. Fan, M.J. Smith, M. Goedert, L.A. Amos, Repeat motifs of tau bind to the insides of microtubules in the absence of taxol, Embo J 22 (2003) 70-77.
[116] R.M. Koffie, M. Meyer-Luehmann, T. Hashimoto, K.W. Adams, M.L. Mielke, M. Garcia-Alloza, K.D. Micheva, S.J. Smith, M.L. Kim, V.M. Lee, B.T. Hyman, T.L. Spires-Jones, Oligomeric amyloid beta associates with postsynaptic densities and correlates with excitatory synapse loss near senile plaques, Proc Natl Acad Sci U S A 106 (2009) 4012-4017.
[117] E. Kopke, Y.C. Tung, S. Shaikh, A.C. Alonso, K. Iqbal, I. Grundke-Iqbal, Microtubule-associated protein tau. Abnormal phosphorylation of a non-paired helical filament pool in Alzheimer disease, J Biol Chem 268 (1993) 24374-24384.
[118] A. Korneyev, L. Binder, J. Bernardis, Rapid reversible phosphorylation of rat brain tau proteins in response to cold water stress, Neurosci Lett 191 (1995) 19-22.
[119] K.S. Kosik, L.D. Orecchio, S. Bakalis, R.L. Neve, Developmentally regulated expression of specific tau sequences, Neuron 2 (1989) 1389-1397.
[120] L.K. Kreppel, M.A. Blomberg, G.W. Hart, Dynamic glycosylation of nuclear and cytosolic proteins. Cloning and characterization of a unique O-GlcNAc transferase with multiple tetratricopeptide repeats, J Biol Chem 272 (1997) 9308-9315.
[121] L.K. Kreppel, G.W. Hart, Regulation of a cytosolic and nuclear O-GlcNAc transferase. Role of the tetratricopeptide repeats, J Biol Chem 274 (1999) 32015-32022.
[122] C. Krieger, J.H. Hu, S. Pelech, Aberrant protein kinases and phosphoproteins in amyotrophic lateral sclerosis, Trends Pharmacol Sci 24 (2003) 535-541.
[123] T.J. Kwiatkowski, Jr., D.A. Bosco, A.L. Leclerc, E. Tamrazian, C.R. Vanderburg, C. Russ, A. Davis, J. Gilchrist, E.J. Kasarskis, T. Munsat, P. Valdmanis, G.A. Rouleau, B.A. Hosler, P. Cortelli, P.J. de Jong, Y. Yoshinaga, J.L. Haines, M.A. Pericak-Vance, J. Yan, N. Ticozzi, T. Siddique, D. McKenna-Yasek, P.C. Sapp,

50
H.R. Horvitz, J.E. Landers, R.H. Brown, Jr., Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis, Science (New York, N.Y) 323 (2009) 1205-1208.
[124] C. Lagier-Tourenne, M. Polymenidou, D.W. Cleveland, TDP-43 and FUS/TLS: emerging roles in RNA processing and neurodegeneration, Hum Mol Genet 19 (2010) R46-64.
[125] M.B. Lazarus, Y. Nam, J. Jiang, P. Sliz, S. Walker, Structure of human O-GlcNAc transferase and its complex with a peptide substrate, Nature 469 (2011) 564-567.
[126] V.M. Lee, B.J. Balin, L. Otvos, Jr., J.Q. Trojanowski, A68: a major subunit of paired helical filaments and derivatized forms of normal Tau, Science (New York, N.Y) 251 (1991) 675-678.
[127] T. Lefebvre, M.L. Caillet-Boudin, L. Buee, A. Delacourte, J.C. Michalski, O-GlcNAc glycosylation and neurological disorders, Adv Exp Med Biol 535 (2003) 189-202.
[128] T. Lefebvre, S. Ferreira, L. Dupont-Wallois, T. Bussiere, M.J. Dupire, A. Delacourte, J.C. Michalski, M.L. Caillet-Boudin, Evidence of a balance between phosphorylation and O-GlcNAc glycosylation of Tau proteins--a role in nuclear localization, Biochim Biophys Acta 1619 (2003) 167-176.
[129] D.M. Lehman, D.J. Fu, A.B. Freeman, K.J. Hunt, R.J. Leach, T. Johnson-Pais, J. Hamlington, T.D. Dyer, R. Arya, H. Abboud, H.H. Goring, R. Duggirala, J. Blangero, R.J. Konrad, M.P. Stern, A single nucleotide polymorphism in MGEA5 encoding O-GlcNAc-selective N-acetyl-beta-D glucosaminidase is associated with type 2 diabetes in Mexican Americans, Diabetes 54 (2005) 1214-1221.
[130] J.B. Levine, J. Kong, M. Nadler, Z. Xu, Astrocytes interact intimately with degenerating motor neurons in mouse amyotrophic lateral sclerosis (ALS), Glia 28 (1999) 215-224.
[131] J. Lewis, D.W. Dickson, W.L. Lin, L. Chisholm, A. Corral, G. Jones, S.H. Yen, N. Sahara, L. Skipper, D. Yager, C. Eckman, J. Hardy, M. Hutton, E. McGowan, Enhanced neurofibrillary degeneration in transgenic mice expressing mutant tau and APP, Science (New York, N.Y) 293 (2001) 1487-1491.
[132] J. Lewis, E. McGowan, J. Rockwood, H. Melrose, P. Nacharaju, M. Van Slegtenhorst, K. Gwinn-Hardy, M. Paul Murphy, M. Baker, X. Yu, K. Duff, J. Hardy, A. Corral, W.L. Lin, S.H. Yen, D.W. Dickson, P. Davies, M. Hutton, Neurofibrillary tangles, amyotrophy and progressive motor disturbance in mice expressing mutant (P301L) tau protein, Nat Genet 25 (2000) 402-405.
[133] B. Li, M.O. Chohan, I. Grundke-Iqbal, K. Iqbal, Disruption of microtubule network by Alzheimer abnormally hyperphosphorylated tau, Acta neuropathologica 113 (2007) 501-511.
[134] S. Li, X. Wang, C.B. Klee, C. Krieger, Overexpressed mutant G93A superoxide dismutase protects calcineurin from inactivation, Brain research 125 (2004) 156-161.
[135] N.F. Liachko, C.R. Guthrie, B.C. Kraemer, Phosphorylation promotes neurotoxicity in a Caenorhabditis elegans model of TDP-43 proteinopathy, J Neurosci 30 16208-16219.

51
[136] C.L. Lin, L.A. Bristol, L. Jin, M. Dykes-Hoberg, T. Crawford, L. Clawson, J.D. Rothstein, Aberrant RNA processing in a neurodegenerative disease: the cause for absent EAAT2, a glutamate transporter, in amyotrophic lateral sclerosis, Neuron 20 (1998) 589-602.
[137] S.C. Ling, C.P. Albuquerque, J.S. Han, C. Lagier-Tourenne, S. Tokunaga, H. Zhou, D.W. Cleveland, ALS-associated mutations in TDP-43 increase its stability and promote TDP-43 complexes with FUS/TLS, Proc Natl Acad Sci U S A 107 (2010) 13318-13323.
[138] F. Liu, K. Iqbal, I. Grundke-Iqbal, G.W. Hart, C.X. Gong, O-GlcNAcylation regulates phosphorylation of tau: a mechanism involved in Alzheimer's disease, Proc Natl Acad Sci U S A 101 (2004) 10804-10809.
[139] F. Liu, Z. Liang, C.X. Gong, Hyperphosphorylation of tau and protein phosphatases in Alzheimer disease, Panminerva medica 48 (2006) 97-108.
[140] Y. Liu, F. Liu, I. Grundke-Iqbal, K. Iqbal, C.X. Gong, Brain glucose transporters, O-GlcNAcylation and phosphorylation of tau in diabetes and Alzheimer's disease, J Neurochem 111 (2009) 242-249.
[141] Y. Lu, J. Ferris, F.B. Gao, Frontotemporal dementia and amyotrophic lateral sclerosis-associated disease protein TDP-43 promotes dendritic branching, Molecular brain 2 (2009) 30.
[142] W.A. Lubas, D.W. Frank, M. Krause, J.A. Hanover, O-Linked GlcNAc transferase is a conserved nucleocytoplasmic protein containing tetratricopeptide repeats, J Biol Chem 272 (1997) 9316-9324.
[143] W.A. Lubas, J.A. Hanover, Functional expression of O-linked GlcNAc transferase. Domain structure and substrate specificity, J Biol Chem 275 (2000) 10983-10988.
[144] K.M. Lucin, T. Wyss-Coray, Immune activation in brain aging and neurodegeneration: too much or too little?, Neuron 64 (2009) 110-122.
[145] N. Ludemann, A. Clement, V.H. Hans, J. Leschik, C. Behl, R. Brandt, O-glycosylation of the tail domain of neurofilament protein M in human neurons and in spinal cord tissue of a rat model of amyotrophic lateral sclerosis (ALS), The Journal of biological chemistry 280 (2005) 31648-31658.
[146] B.T. M. Kiaei, A. Neymotin, N. Y. Calingassan, E. G. Wille, F. Yin, A. Ding, M. F. Beal, Altered TDP-43 expression patterns in the spinal cord of SOD1 transgenic mice and PGRN knockout mice, . Soc. for Neurosci. Abstr., (2008)
[147] M.S. Macauley, A.K. Bubb, C. Martinez-Fleites, G.J. Davies, D.J. Vocadlo, Elevation of global O-GlcNAc levels in 3T3-L1 adipocytes by selective inhibition of O-GlcNAcase does not induce insulin resistance, J Biol Chem 283 (2008) 34687-34695.
[148] M.S. Macauley, Y. He, T.M. Gloster, K.A. Stubbs, G.J. Davies, D.J. Vocadlo, Inhibition of O-GlcNAcase using a potent and cell-permeable inhibitor does not induce insulin resistance in 3T3-L1 adipocytes, Chem Biol 17 (2010) 937-948.
[149] M.S. Macauley, X. Shan, S.A. Yuzwa, T.M. Gloster, D.J. Vocadlo, Elevation of Global O-GlcNAc in rodents using a selective O-GlcNAcase inhibitor does not cause insulin resistance or perturb glucohomeostasis, Chem Biol 17 (2010) 949-958.

52
[150] M.S. Macauley, K.A. Stubbs, D.J. Vocadlo, O-GlcNAcase catalyzes cleavage of thioglycosides without general acid catalysis, J Am Chem Soc 127 (2005) 17202-17203.
[151] M.S. Macauley, D.J. Vocadlo, Enzymatic characterization and inhibition of the nuclear variant of human O-GlcNAcase, Carbohydrate research 344 (2009) 1079-1084.
[152] M.S. Macauley, D.J. Vocadlo, Increasing O-GlcNAc levels: An overview of small-molecule inhibitors of O-GlcNAcase, Biochim Biophys Acta 1800 (2010) 107-121.
[153] M.S. Macauley, G.E. Whitworth, A.W. Debowski, D. Chin, D.J. Vocadlo, O-GlcNAcase uses substrate-assisted catalysis: kinetic analysis and development of highly selective mechanism-inspired inhibitors, J Biol Chem 280 (2005) 25313-25322.
[154] I.R. Mackenzie, E.H. Bigio, P.G. Ince, F. Geser, M. Neumann, N.J. Cairns, L.K. Kwong, M.S. Forman, J. Ravits, H. Stewart, A. Eisen, L. McClusky, H.A. Kretzschmar, C.M. Monoranu, J.R. Highley, J. Kirby, T. Siddique, P.J. Shaw, V.M. Lee, J.Q. Trojanowski, Pathological TDP-43 distinguishes sporadic amyotrophic lateral sclerosis from amyotrophic lateral sclerosis with SOD1 mutations, Ann Neurol 61 (2007) 427-434.
[155] I.R. Mackenzie, R. Rademakers, The role of transactive response DNA-binding protein-43 in amyotrophic lateral sclerosis and frontotemporal dementia, Current opinion in neurology 21 (2008) 693-700.
[156] D.J. Mahuran, R.A. Gravel, The beta-hexosaminidase story in Toronto: from enzyme structure to gene mutation, Adv Genet 44 (2001) 145-163.
[157] R. Martinez-Contreras, P. Cloutier, L. Shkreta, J.F. Fisette, T. Revil, B. Chabot, hnRNP proteins and splicing control, Adv Exp Med Biol 623 (2007) 123-147.
[158] C. Martinez-Fleites, M.S. Macauley, Y. He, D.L. Shen, D.J. Vocadlo, G.J. Davies, Structure of an O-GlcNAc transferase homolog provides insight into intracellular glycosylation, Nat Struct Mol Biol 15 (2008) 764-765.
[159] E. Masliah, M. Mallory, N. Ge, M. Alford, I. Veinbergs, A.D. Roses, Neurodegeneration in the central nervous system of apoE-deficient mice, Experimental neurology 136 (1995) 107-122.
[160] J.A. Matthews, J.L. Belof, M. Acevedo-Duncan, R.L. Potter, Glucosamine-induced increase in Akt phosphorylation corresponds to increased endoplasmic reticulum stress in astroglial cells, Molecular and cellular biochemistry 298 (2007) 109-123.
[161] J.A. Matthews, J.L. Belof, M. Acevedo-Duncan, R.L. Potter, Glucosamine-induced increase in Akt phosphorylation corresponds to increased endoplasmic reticulum stress in astroglial cells, Mol. Cell. Biochem. 298 (2007) 109-123.
[162] M. Mawal-Dewan, J. Henley, A. Van de Voorde, J.Q. Trojanowski, V.M. Lee, The phosphorylation state of tau in the developing rat brain is regulated by phosphoprotein phosphatases, J Biol Chem 269 (1994) 30981-30987.
[163] M.P. Mazanetz, P.M. Fischer, Untangling tau hyperphosphorylation in drug design for neurodegenerative diseases, Nat Rev Drug Discov 6 (2007) 464-479.

53
[164] D.A. McClain, W.A. Lubas, R.C. Cooksey, M. Hazel, G.J. Parker, D.C. Love, J.A. Hanover, Altered glycan-dependent signaling induces insulin resistance and hyperleptinemia, Proc Natl Acad Sci U S A 99 (2002) 10695-10699.
[165] G.L. McKnight, S.L. Mudri, S.L. Mathewes, R.R. Traxinger, S. Marshall, P.O. Sheppard, P.J. O'Hara, Molecular cloning, cDNA sequence, and bacterial expression of human glutamine:fructose-6-phosphate amidotransferase, J Biol Chem 267 (1992) 25208-25212.
[166] P.A. Mercado, Y.M. Ayala, M. Romano, E. Buratti, F.E. Baralle, Depletion of TDP 43 overrides the need for exonic and intronic splicing enhancers in the human apoA-II gene, Nucleic acids research 33 (2005) 6000-6010.
[167] S.W. Min, S.H. Cho, Y. Zhou, S. Schroeder, V. Haroutunian, W.W. Seeley, E.J. Huang, Y. Shen, E. Masliah, C. Mukherjee, D. Meyers, P.A. Cole, M. Ott, L. Gan, Acetylation of tau inhibits its degradation and contributes to tauopathy, Neuron 67 (2010) 953-966.
[168] M.H. Mohajeri, D.A. Figlewicz, M.C. Bohn, Selective loss of alpha motoneurons innervating the medial gastrocnemius muscle in a mouse model of amyotrophic lateral sclerosis, Exp Neurol 150 (1998) 329-336.
[169] S. Mondragon-Rodriguez, G. Basurto-Islas, I. Santa-Maria, R. Mena, L.I. Binder, J. Avila, M.A. Smith, G. Perry, F. Garcia-Sierra, Cleavage and conformational changes of tau protein follow phosphorylation during Alzheimer's disease, International journal of experimental pathology 89 (2008) 81-90.
[170] P.N. Monk, P.J. Shaw, ALS: life and death in a bad neighborhood, Nature medicine 12 (2006) 885-887.
[171] A. Nandi, R. Sprung, D.K. Barma, Y. Zhao, S.C. Kim, J.R. Falck, Y. Zhao, Global identification of O-GlcNAc-modified proteins, Analytical chemistry 78 (2006) 452-458.
[172] M. Neumann, L.K. Kwong, E.B. Lee, E. Kremmer, A. Flatley, Y. Xu, M.S. Forman, D. Troost, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Phosphorylation of S409/410 of TDP-43 is a consistent feature in all sporadic and familial forms of TDP-43 proteinopathies, Acta neuropathologica 117 (2009) 137-149.
[173] M. Neumann, D.M. Sampathu, L.K. Kwong, A.C. Truax, M.C. Micsenyi, T.T. Chou, J. Bruce, T. Schuck, M. Grossman, C.M. Clark, L.F. McCluskey, B.L. Miller, E. Masliah, I.R. Mackenzie, H. Feldman, W. Feiden, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Science 314 (2006) 130-133.
[174] G.A. Ngoh, H.T. Facundo, T. Hamid, W. Dillmann, N.E. Zachara, S.P. Jones, Unique hexosaminidase reduces metabolic survival signal and sensitizes cardiac myocytes to hypoxia/reoxygenation injury, Circ Res 104 (2009) 41-49.
[175] G.A. Ngoh, L.J. Watson, H.T. Facundo, W. Dillmann, S.P. Jones, Non-canonical glycosyltransferase modulates post-hypoxic cardiac myocyte death and mitochondrial permeability transition, Journal of molecular and cellular cardiology 45 (2008) 313-325.
[176] T. Nonaka, F. Kametani, T. Arai, H. Akiyama, M. Hasegawa, Truncation and pathogenic mutations facilitate the formation of intracellular aggregates of TDP-43, Hum Mol Genet 18 (2009) 3353-3364.

54
[177] S.H. Ou, F. Wu, D. Harrich, L.F. Garcia-Martinez, R.B. Gaynor, Cloning and characterization of a novel cellular protein, TDP-43, that binds to human immunodeficiency virus type 1 TAR DNA sequence motifs, Journal of virology 69 (1995) 3584-3596.
[178] S. Ozcan, S.S. Andrali, J.E. Cantrell, Modulation of transcription factor function by O-GlcNAc modification, Biochim Biophys Acta 1799 (2010) 353-364.
[179] R. Pamphlett, N. Luquin, C. McLean, S.K. Jew, L. Adams, TDP-43 neuropathology is similar in sporadic amyotrophic lateral sclerosis with or without TDP-43 mutations, Neuropathology and applied neurobiology 35 (2009) 222-225.
[180] S.C. Papasozomenos, Y. Su, Altered phosphorylation of tau protein in heat-shocked rats and patients with Alzheimer disease, Proc Natl Acad Sci U S A 88 (1991) 4543-4547.
[181] P. Pasinelli, R.H. Brown, Molecular biology of amyotrophic lateral sclerosis: insights from genetics, Nat Rev Neurosci 7 (2006) 710-723.
[182] M. Polymenidou, C. Lagier-Tourenne, K.R. Hutt, S.C. Huelga, J. Moran, T.Y. Liang, S.C. Ling, E. Sun, E. Wancewicz, C. Mazur, H. Kordasiewicz, Y. Sedaghat, J.P. Donohue, L. Shiue, C.F. Bennett, G.W. Yeo, D.W. Cleveland, Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43, Nature neuroscience 14 (2011) 459-468.
[183] A. Pramatarova, J. Laganiere, J. Roussel, K. Brisebois, G.A. Rouleau, Neuron-specific expression of mutant superoxide dismutase 1 in transgenic mice does not lead to motor impairment, J Neurosci 21 (2001) 3369-3374.
[184] A. Radunovic, H. Mitsumoto, P.N. Leigh, Clinical care of patients with amyotrophic lateral sclerosis, Lancet Neurol 6 (2007) 913-925.
[185] C. Reitz, C. Brayne, R. Mayeux, Epidemiology of Alzheimer disease, Nat reviews 7 (2011) 137-152.
[186] M.R. Reynolds, T.J. Lukas, R.W. Berry, L.I. Binder, Peroxynitrite-mediated tau modifications stabilize preformed filaments and destabilize microtubules through distinct mechanisms, Biochemistry 45 (2006) 4314-4326.
[187] M.R. Reynolds, J.F. Reyes, Y. Fu, E.H. Bigio, A.L. Guillozet-Bongaarts, R.W. Berry, L.I. Binder, Tau nitration occurs at tyrosine 29 in the fibrillar lesions of Alzheimer's disease and other tauopathies, J Neurosci 26 (2006) 10636-10645.
[188] R.A. Rissman, W.W. Poon, M. Blurton-Jones, S. Oddo, R. Torp, M.P. Vitek, F.M. LaFerla, T.T. Rohn, C.W. Cotman, Caspase-cleavage of tau is an early event in Alzheimer disease tangle pathology, J of clinical investigation 114 (2004) 121-130.
[189] J. Robertson, T. Sanelli, S. Xiao, W. Yang, P. Horne, R. Hammond, E.P. Pioro, M.J. Strong, Lack of TDP-43 abnormalities in mutant SOD1 transgenic mice shows disparity with ALS, Neurosci Lett 420 (2007) 128-132.
[190] K.A. Robinson, L.E. Ball, M.G. Buse, Reduction of O-GlcNAc protein modification does not prevent insulin resistance in 3T3-L1 adipocytes, Am. J. Physiol. Endocrinol. Metab. 292 (2007) E884-890.
[191] M.D. Roos, J.A. Hanover, Structure of O-linked GlcNAc transferase: mediator of glycan-dependent signaling, Biochem Biophys Res Commun 271 (2000) 275-280.

55
[192] E.P. Roquemore, M.R. Chevrier, R.J. Cotter, G.W. Hart, Dynamic O-GlcNAcylation of the small heat shock protein alpha B-crystallin, Biochemistry 35 (1996) 3578-3586.
[193] D.R. Rosen, T. Siddique, D. Patterson, D.A. Figlewicz, P. Sapp, A. Hentati, D. Donaldson, J. Goto, J.P. O'Regan, H.X. Deng, et al., Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis, Nature 362 (1993) 59-62.
[194] S. Sasaki, T. Takeda, N. Shibata, M. Kobayashi, Alterations in subcellular localization of TDP-43 immunoreactivity in the anterior horns in sporadic amyotrophic lateral sclerosis, Neurosci Lett 478 (2010) 72-76.
[195] D. Schubert, Glucose metabolism and Alzheimer's disease, Ageing research reviews 4 (2005) 240-257.
[196] A. Sengupta, M. Novak, I. Grundke-Iqbal, K. Iqbal, Regulation of phosphorylation of tau by cyclin-dependent kinase 5 and glycogen synthase kinase-3 at substrate level, FEBS Lett 580 (2006) 5925-5933.
[197] C.F. Sephton, C. Cenik, A. Kucukural, E.B. Dammer, B. Cenik, Y. Han, C.M. Dewey, F.P. Roth, J. Herz, J. Peng, M.J. Moore, G. Yu, Identification of neuronal RNA targets of TDP-43-containing ribonucleoprotein complexes, J Biol Chem 286 (2011) 1204-1215.
[198] C.F. Sephton, S.K. Good, S. Atkin, C.M. Dewey, P. Mayer, 3rd, J. Herz, G. Yu, TDP-43 is a developmentally regulated protein essential for early embryonic development, J Biol Chem 285 (2010) 6826-6834.
[199] R. Shafi, S.P. Iyer, L.G. Ellies, N. O'Donnell, K.W. Marek, D. Chui, G.W. Hart, J.D. Marth, The O-GlcNAc transferase gene resides on the X chromosome and is essential for embryonic stem cell viability and mouse ontogeny, Proc Natl Acad Sci U S A 97 (2000) 5735-5739.
[200] J.M. Shulman, P. Chipendo, L.B. Chibnik, C. Aubin, D. Tran, B.T. Keenan, P.L. Kramer, J.A. Schneider, D.A. Bennett, M.B. Feany, P.L. De Jager, Functional Screening of Alzheimer Pathology Genome-wide Association Signals in Drosophila, The American J of Human Genetics 88 (2011) 232-238.
[201] T.J. Singh, J.Z. Wang, M. Novak, E. Kontzekova, I. Grundke-Iqbal, K. Iqbal, Calcium/calmodulin-dependent protein kinase II phosphorylates tau at Ser-262 but only partially inhibits its binding to microtubules, FEBS Lett 387 (1996) 145-148.
[202] C. Slawson, N.E. Zachara, K. Vosseller, W.D. Cheung, M.D. Lane, G.W. Hart, Perturbations in O-linked beta-N-acetylglucosamine protein modification cause severe defects in mitotic progression and cytokinesis, J Biol Chem 280 (2005) 32944-32956.
[203] C.M. Snow, A. Senior, L. Gerace, Monoclonal antibodies identify a group of nuclear pore complex glycoproteins, J cell biol 104 (1987) 1143-1156.
[204] R. Sprung, A. Nandi, Y. Chen, S.C. Kim, D. Barma, J.R. Falck, Y. Zhao, Tagging-via-substrate strategy for probing O-GlcNAc modified proteins, J Proteome Res 4 (2005) 950-957.
[205] J. Sreedharan, I.P. Blair, V.B. Tripathi, X. Hu, C. Vance, B. Rogelj, S. Ackerley, J.C. Durnall, K.L. Williams, E. Buratti, F. Baralle, J. de Belleroche, J.D. Mitchell, P.N. Leigh, A. Al-Chalabi, C.C. Miller, G. Nicholson, C.E. Shaw, TDP-43

56
mutations in familial and sporadic amyotrophic lateral sclerosis, Science (New York, N.Y) 319 (2008) 1668-1672.
[206] M.J. Strong, S. Kesavapany, H.C. Pant, The pathobiology of amyotrophic lateral sclerosis: a proteinopathy?, J Neuropathol Exp Neurol 64 (2005) 649-664.
[207] K.A. Stubbs, M.S. Macauley, D.J. Vocadlo, A selective inhibitor Gal-PUGNAc of human lysosomal beta-hexosaminidases modulates levels of the ganglioside GM2 in neuroblastoma cells, Angewandte Chemie (International ed 48 (2009) 1300-1303.
[208] H. Sumi, S. Kato, Y. Mochimaru, H. Fujimura, M. Etoh, S. Sakoda, Nuclear TAR DNA binding protein 43 expression in spinal cord neurons correlates with the clinical course in amyotrophic lateral sclerosis, J Neuropathol Exp Neurol 68 (2009) 37-47.
[209] M.K. Tallent, N. Varghis, Y. Skorobogatko, L. Hernandez-Cuebas, K. Whelan, D.J. Vocadlo, K. Vosseller, In vivo modulation of O-GlcNAc levels regulates hippocampal synaptic plasticity through interplay with phosphorylation, J Biol Chem 284 (2009) 174-181.
[210] C.F. Tan, H. Eguchi, A. Tagawa, O. Onodera, T. Iwasaki, A. Tsujino, M. Nishizawa, A. Kakita, H. Takahashi, TDP-43 immunoreactivity in neuronal inclusions in familial amyotrophic lateral sclerosis with or without SOD1 gene mutation, Acta neuropathologica 113 (2007) 535-542.
[211] C.F. Teo, S. Ingale, M.A. Wolfert, G.A. Elsayed, L.G. Not, J.C. Chatham, L. Wells, G.J. Boons, Glycopeptide-specific monoclonal antibodies suggest new roles for O-GlcNAc, Nat Chem Biol 6 (2010) 338-343.
[212] C.F. Teo, S. Ingale, M.A. Wolfert, G.A. Elsayed, L.G. Not, J.C. Chatham, L. Wells, G.J. Boons, Glycopeptide-specific monoclonal antibodies suggest new roles for O-GlcNAc, Nat Chem Biol 6 (2010) 338-343.
[213] C. Toleman, A.J. Paterson, R. Shin, J.E. Kudlow, Streptozotocin inhibits O-GlcNAcase via the production of a transition state analog, Biochem Biophys Res Commun 340 (2006) 526-534.
[214] C. Toleman, A.J. Paterson, T.R. Whisenhunt, J.E. Kudlow, Characterization of the histone acetyltransferase (HAT) domain of a bifunctional protein with activable O-GlcNAcase and HAT activities, J Biol Chem 279 (2004) 53665-53673.
[215] C.R. Torres, G.W. Hart, Topography and polypeptide distribution of terminal N-acetylglucosamine residues on the surfaces of intact lymphocytes. Evidence for O-linked GlcNAc, J Biol Chem 259 (1984) 3308-3317.
[216] H. Tsukagoshi, N. Yanagisawa, K. Oguchi, K. Nagashima, T. Murakami, Morphometric quantification of the cervical limb motor cells in controls and in amyotrophic lateral sclerosis, J Neurol Sci 41 (1979) 287-297.
[217] B.J. Turner, D. Baumer, N.J. Parkinson, J. Scaber, O. Ansorge, K. Talbot, TDP-43 expression in mouse models of amyotrophic lateral sclerosis and spinal muscular atrophy, BMC Neurosci 9 (2008) 104.
[218] J.R. Turner, A.M. Tartakoff, N.S. Greenspan, Cytologic assessment of nuclear and cytoplasmic O-linked N-acetylglucosamine distribution by using anti-streptococcal monoclonal antibodies, Proc Natl Acad Sci U S A 87 (1990) 5608-5612.

57
[219] Y. Uno, H. Iwashita, T. Tsukamoto, N. Uchiyama, T. Kawamoto, M. Kori, A. Nakanishi, Efficacy of a novel, orally active GSK-3 inhibitor 6-Methyl-N-[3-[[3-(1-methylethoxy)propyl]carbamoyl]-1H-pyrazol-4-yl]pyridi ne-3-carboxamide in tau transgenic mice, Brain Res 1296 (2009) 148-163.
[220] J.S. Valentine, P.A. Doucette, S. Zittin Potter, Copper-zinc superoxide dismutase and amyotrophic lateral sclerosis, Annu Rev Biochem 74 (2005) 563-593.
[221] D. Van Dam, P.P. De Deyn, Drug discovery in dementia: the role of rodent models, Nat Rev Drug Discov 5 (2006) 956-970.
[222] V.M. Van Deerlin, J.B. Leverenz, L.M. Bekris, T.D. Bird, W. Yuan, L.B. Elman, D. Clay, E.M. Wood, A.S. Chen-Plotkin, M. Martinez-Lage, E. Steinbart, L. McCluskey, M. Grossman, M. Neumann, I.L. Wu, W.S. Yang, R. Kalb, D.R. Galasko, T.J. Montine, J.Q. Trojanowski, V.M. Lee, G.D. Schellenberg, C.E. Yu, TARDBP mutations in amyotrophic lateral sclerosis with TDP-43 neuropathology: a genetic and histopathological analysis, Lancet Neurol 7 (2008) 409-416.
[223] K.A. Vossel, K. Zhang, J. Brodbeck, A.C. Daub, P. Sharma, S. Finkbeiner, B. Cui, L. Mucke, Tau reduction prevents Abeta-induced defects in axonal transport, Science (New York, N.Y 330 (2010) 198.
[224] K. Vosseller, L. Wells, M.D. Lane, G.W. Hart, Elevated nucleocytoplasmic glycosylation by O-GlcNAc results in insulin resistance associated with defects in Akt activation in 3T3-L1 adipocytes, Proceedings of the National Academy of Sciences of the United States of America 99 (2002) 5313-5318.
[225] R. Wagey, C. Krieger, C.A. Shaw, Abnormal dephosphorylation effect on NMDA receptor regulation in ALS spinal cord, Neurobiology of disease 4 (1997) 350-355.
[226] J.L. Walgren, T.S. Vincent, K.L. Schey, M.G. Buse, High glucose and insulin promote O-GlcNAc modification of proteins, including alpha-tubulin, Am J Physiol Endocrinol Metab 284 (2003) E424-434.
[227] H.Y. Wang, I.F. Wang, J. Bose, C.K. Shen, Structural diversity and functional implications of the eukaryotic TDP gene family, Genomics 83 (2004) 130-139.
[228] I.F. Wang, L.S. Wu, H.Y. Chang, C.K. Shen, TDP-43, the signature protein of FTLD-U, is a neuronal activity-responsive factor, J Neurochem 105 (2008) 797-806.
[229] Q. Wang, J.L. Johnson, N.Y. Agar, J.N. Agar, Protein aggregation and protein instability govern familial amyotrophic lateral sclerosis patient survival, PLoS Biol 6 (2008) e170.
[230] Z. Wang, M. Gucek, G.W. Hart, Cross-talk between GlcNAcylation and phosphorylation: site-specific phosphorylation dynamics in response to globally elevated O-GlcNAc, Proc Natl Acad Sci U S A 105 (2008) 13793-13798.
[231] C.L. Weaver, M. Espinoza, Y. Kress, P. Davies, Conformational change as one of the earliest alterations of tau in Alzheimer's disease, Neurobiology of aging 21 (2000) 719-727.
[232] M.D. Weingarten, A.H. Lockwood, S.Y. Hwo, M.W. Kirschner, A protein factor essential for microtubule assembly, Proc Natl Acad Sci U S A 72 (1975) 1858-1862.

58
[233] L. Wells, Y. Gao, J.A. Mahoney, K. Vosseller, C. Chen, A. Rosen, G.W. Hart, Dynamic O-glycosylation of nuclear and cytosolic proteins: further characterization of the nucleocytoplasmic beta-N-acetylglucosaminidase, O-GlcNAcase, J Biol Chem 277 (2002) 1755-1761.
[234] M.J. West, P.D. Coleman, D.G. Flood, J.C. Troncoso, Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer's disease, Lancet 344 (1994) 769-772.
[235] K.C. Wilcox, L. Zhou, J.K. Jordon, Y. Huang, Y. Yu, R.L. Redler, X. Chen, M. Caplow, N.V. Dokholyan, Modifications of superoxide dismutase (SOD1) in human erythrocytes: a possible role in amyotrophic lateral sclerosis, J Biol Chem 284 (2009) 13940-13947.
[236] M.J. Winton, L.M. Igaz, M.M. Wong, L.K. Kwong, J.Q. Trojanowski, V.M. Lee, Disturbance of nuclear and cytoplasmic TAR DNA-binding protein (TDP-43) induces disease-like redistribution, sequestration, and aggregate formation, J Biol Chem 283 (2008) 13302-13309.
[237] M.J. Winton, V.M. Van Deerlin, L.K. Kwong, W. Yuan, E.M. Wood, C.E. Yu, G.D. Schellenberg, R. Rademakers, R. Caselli, A. Karydas, J.Q. Trojanowski, B.L. Miller, V.M. Lee, A90V TDP-43 variant results in the aberrant localization of TDP-43 in vitro, FEBS Lett 582 (2008) 2252-2256.
[238] P.C. Wong, C.A. Pardo, D.R. Borchelt, M.K. Lee, N.G. Copeland, N.A. Jenkins, S.S. Sisodia, D.W. Cleveland, D.L. Price, An adverse property of a familial ALS-linked SOD1 mutation causes motor neuron disease characterized by vacuolar degeneration of mitochondria, Neuron 14 (1995) 1105-1116.
[239] L.S. Wu, W.C. Cheng, S.C. Hou, Y.T. Yan, S.T. Jiang, C.K. Shen, TDP-43, a neuro-pathosignature factor, is essential for early mouse embryogenesis, Genesis 48 (2009) 56-62.
[240] S. Xiao, T. Sanelli, S. Dib, D. Sheps, J. Findlater, J. Bilbao, J. Keith, L. Zinman, E. Rogaeva, J. Robertson, RNA targets of TDP-43 identified by UV-CLIP are deregulated in ALS, Molecular and cellular neurosciences (2011).
[241] K. Yamanaka, S.J. Chun, S. Boillee, N. Fujimori-Tonou, H. Yamashita, D.H. Gutmann, R. Takahashi, H. Misawa, D.W. Cleveland, Astrocytes as determinants of disease progression in inherited amyotrophic lateral sclerosis, Nature neuroscience 11 (2008) 251-253.
[242] X. Yang, P.P. Ongusaha, P.D. Miles, J.C. Havstad, F. Zhang, W.V. So, J.E. Kudlow, R.H. Michell, J.M. Olefsky, S.J. Field, R.M. Evans, Phosphoinositide signalling links O-GlcNAc transferase to insulin resistance, Nature 451 (2008) 964-969.
[243] A. Yokoseki, A. Shiga, C.F. Tan, A. Tagawa, H. Kaneko, A. Koyama, H. Eguchi, A. Tsujino, T. Ikeuchi, A. Kakita, K. Okamoto, M. Nishizawa, H. Takahashi, O. Onodera, TDP-43 mutation in familial amyotrophic lateral sclerosis, Ann Neurol 63 (2008) 538-542.
[244] S.A. Yuzwa, M.S. Macauley, J.E. Heinonen, X. Shan, R.J. Dennis, Y. He, G.E. Whitworth, K.A. Stubbs, E.J. McEachern, G.J. Davies, D.J. Vocadlo, A potent mechanism-inspired O-GlcNAcase inhibitor that blocks phosphorylation of tau in vivo, Nat Chem Biol 4 (2008) 483-490.

59
[245] N.E. Zachara, N. O'Donnell, W.D. Cheung, J.J. Mercer, J.D. Marth, G.W. Hart, Dynamic O-GlcNAc modification of nucleocytoplasmic proteins in response to stress. A survival response of mammalian cells, J Biol Chem 279 (2004) 30133-30142.
[246] D.W. Zang, S.S. Cheema, Degeneration of corticospinal and bulbospinal systems in the superoxide dismutase 1(G93A G1H) transgenic mouse model of familial amyotrophic lateral sclerosis, Neurosci Lett 332 (2002) 99-102.
[247] Q. Zhang, X. Zhang, J. Chen, Y. Miao, A. Sun, Role of caspase-3 in tau truncation at D421 is restricted in transgenic mouse models for tauopathies, J Neurochem 109 (2009) 476-484.
[248] Q. Zhang, X. Zhang, A. Sun, Truncated tau at D421 is associated with neurodegeneration and tangle formation in the brain of Alzheimer transgenic models, Acta neuropathologica 117 (2009) 687-697.
[249] J. Zhou, Q.K. Huynh, R.T. Hoffman, E.D. Crook, M.C. Daniels, E.A. Gulve, D.A. McClain, Regulation of glutamine:fructose-6-phosphate amidotransferase by cAMP-dependent protein kinase, Diabetes 47 (1998) 1836-1840.
[250] L. Zou, S. Yang, S. Hu, I.H. Chaudry, R.B. Marchase, J.C. Chatham, The protective effects of PUGNAc on cardiac function after trauma-hemorrhage are mediated via increased protein O-GlcNAc levels, Shock 27 (2007) 402-408.

60
2: Reduction of O-GlcNAc in the motor neurons of G93A mutant SOD1 transgenic mouse model of ALS
Manuscript in revision for re-submission to the Neuroscience
Authors: Xiaoyang Shan, David Vocadlo and Charles Krieger.
Author contribution: X.S., D.V. and C.K. designed the experiments. X.S. performed all experiments. X.S., D.V. and C.K. analysed data and wrote the manuscript.
2.1 Abstract
Many intracellular proteins are O-glycosylated on serine and threonine residues with β-
N-acetylglucosamine residues (O-GlcNAc). It has been found in some cases that O-
GlcNAc modifies proteins competitively with protein phosphorylation, so increased O-
GlcNAc can in this way reduce phosphorylation at specific sites. In the
neurodegenerative disease amyotrophic lateral sclerosis (ALS), a number of proteins
have been found to be hyperphosphorylated, including neurofilament proteins (NFs).
Here we evaluated a transgenic mouse model of ALS that overexpress mutant superoxide
dismutase (G93A mSOD) and found that O-GlcNAc levels are decreased in spinal cord
tissue from mSOD mice, compared to controls. This reduction in O-GlcNAc levels is
prominent in the motor neurons localized in ventral horn region of grey matter of spinal
cord. We find that inhibition of O-GlcNAcase (OGA), the enzyme catalyzing removal of
O-GlcNAc, using the inhibitor NButGT for 3 days resulted in increased O-GlcNAc levels

61
in spinal cord, both in mSOD and control mice. Furthermore, NButGT increased levels of
O-GlcNAc modified NF-medium in spinal cords of control mice but, not in mSOD mice.
These observations suggest that the neurodegeneration found in mSOD mice might be
associated with a reduction of O-GlcNAc levels in motor neurons.
2.2 Introduction
Neurofilaments (NFs) are a major component of the cytoskeleton of neurons, particularly
those with large calibers, such as motorneurons. In mature neurons, NFs are composed of
three subunits: neurofilament light, medium, and heavy chains (NFL, NFM, and NFH).
NFM and NFH possess extended carboxy-terminal domains forming side-arms that
project from the filament which can be heavily phosphorylated on serine residues within
repeating lys-ser-pro (KSP) motifs [18]. Interestingly, the phosphorylation states of NFM
and NFH side-arms determine the axonal diameter, which regulates conduction velocity,
and affects axonal transport [18, 27].
It is likely that the hyperphosphorylation of NFs is deleterious and accumulation of
hyperphosphorylated NFs in cell bodies and proximal axons of motor neurons contributes
to the impairment found in both the sporadic and familial forms of amyotrophic lateral
sclerosis (ALS) [14, 32]. ALS is an adult onset neurodegenerative disease characterized
by neuron and motorneuron death in the spinal cord and brain. The phosphorylation state
of NFs impacts on slow axonal transport [1] and it is thought that disorganized and
hyperphosphorylated NFs impair axonal transport and thereby contribute to NF-induced
pathology in ALS [26, 35].

62
Recently, it has been shown that NFs can also be post-translationally modified by a form
of O-glycosylation, O-linked β-N-acetylglucosamine, known as O-GlcNAc [11]. This
modification has been found to occur on amino acid residues that are also known to be
phosphorylated in some proteins [4, 5]. Consistent with this observation, it has been
previously shown that increased phosphorylation levels of specific proteins result in
decreased O-GlcNAc levels, and increased O-GlcNAc levels correlate with decreased
phosphorylation levels [12, 33, 37]. The reciprocal relationship between O-
GlcNAcylation and phosphorylation has been termed the Ying-Yang hypothesis [17], and
has gained biochemical support from the recent discovery that the enzyme installing the
O-GlcNAc residue, termed O-GlcNAc transferase (OGT) [21], appears to form a
functional complex with certain protein phosphatases [34]. Like phosphorylation, O-
GlcNAcylation is a dynamic modification that can be removed and installed several times
during the lifespan of a protein [29]. The enzyme catalyzing the removal of O-GlcNAc
from modified proteins is a β-N-acetylglucosamindase known as OGA [10]. A potent and
highly selective small molecule inhibitor of OGA, termed NButGT, has been described
and shown to increase global levels of O-GlcNAc modified proteins in a wide range of
tissues including those of the central nervous system (CNS) of rodents [23-25].
Phosphorylation of NFM appears to be regulated by O-GlcNAc modification both in vitro
and in vivo [22]. Additionally, it has been proposed that O-GlcNAc and phosphorylation
levels on NFM are reciprocal, and O-GlcNAc modified NFM levels are markedly
decreased in spinal cords of rats having an ALS-like disorder, while phosphorylation of
NFM is increased [22]. However, it is unknown where O-GlcNAc modified proteins are

63
distributed and whether levels of O-GlcNAc modified proteins are altered in motor
neurons in ALS.
Although the pathogenesis of ALS is still unclear, it has been found that about 20% of
patients with the familial form of ALS (fALS) are associated with mutations in the Cu/Zn
superoxide dismutase (SOD1) gene. A transgenic mouse strain that over-expresses a
mutant form of human SOD1 seen in fALS (G93A mSOD) has been commonly used as a
mouse model for study of ALS [13]. Mice that over-express G93A mSOD develop a
disorder resembling ALS in humans. In G93A mSOD mice, aberrant accumulation of
hyperphosphorylated NFs occurs in motor neurons of spinal cords [31]. Remarkably, it
has been demonstrated that the depletion of heavily phosphorylated side-arms of NFM
and NFH, containing the KSP repeats, increases survival of motor neurons in G37R
mSOD mice [20]. Accordingly, mechanisms that can regulate the phosphorylation of NFs
are of interest since it may be possible to exploit them to yield methods for intervening in
the progression of disease symptoms.
In this work, we describe the distribution of O-GlcNAc modified proteins in the spinal
cords of G93A mSOD mice and controls, and explore whether modulation of O-GlcNAc
using small molecule inhibitors of OGA will alter levels of O-GlcNAc modified proteins
and NFs phosphorylation in the CNS of G93A mSOD mice.

64
2.3 Methods
2.3.1 Antibodies
Mouse monoclonal IgG antibody SMI31 against the phosphorylated epitopes of the
repeating KSP motifs of NFH and NFM and mouse monoclonal IgM antibody CTD110.6
was purchased from Covance Research Products (Berkeley, CA). CTD110.6 was raised
against a synthetic peptide containing Ser-O-GlcNAc, but it detects both serine-O-
GlcNAc and threonine-O-GlcNAc. Mouse monoclonal IgG antibody NL6 that recognizes
O-glycosylated NFM at, or close to, its repeating KSP tail domain was purchased from
Sigma (Sigma-Aldrich Canada, Oakville, ON). Mouse monoclonal IgG antibodies anti-
NeuN and anti- 2', 3'-cyclic nucleotide 3'-phosphodi-esterase (CNPase) were purchased
from Chemicon International (Temecula, CA). Rat polyclonal IgG anti-glial fibrillary
acidic protein (GFAP) antibody was from Calbiochem (Temicula, CA). Rabbit polyclonal
IgG anti-ionized calcium binding adapter molecule 1 (Iba1) antibody was from Wako
Pure Chemical Industries (Osaka, Japan). Mouse monoclonal IgG antibody against β-
actin was purchased from Sigma. Mouse monoclonal IgG antibody against total NFM
(NF-09), and HRP conjugated secondary antibodies for immunoblotting were from Santa
Cruz Biotechnology (Santa Cruz, CA). Cy3 or FITC conjugated secondary antibodies for
fluorescent immunohistochemistry were from Jackson ImmunoResearch Laboratories
(West Grove, PA).
2.3.2 Animals and treatment with NButGT
The strain of transgenic mice over expressing mutant human SOD1 (mSOD) [B6SJL-
TgN(SOD1-G93A)1Gur] were purchased from Jackson Laboratories (Bar Harbor, MA)
or bred locally from progenitor animals. Genotyping of mSOD mice was performed using

65
a PCR-based assay described on the Jackson Laboratory website. The mSOD mice
overexpress the G93A mutant form of SOD1, which is found in some familial ALS
patients. A total of 24 mSOD transgenic mice and 24 wild-type (wt) littermate control
mice were used. mSOD mice were studied when animals exhibited severe symptoms (a
stage where animals have demonstrated maximum flexion in the hind limbs, partial
paralysis of one or both of the hind legs, muscle atrophy, and have considerable difficulty
with ambulation), mild symptoms, and no symptoms. Protocols governing the use of
animals were approved by the Animal Care Review Committee of Simon Fraser
University and were in compliance with guidelines published by the Canadian Council on
Animal Care. All efforts were made to minimize animal stress and to reduce the number
of animals used.
A selective OGA inhibitor, 1,2-dideoxy-2′-propyl-α-D-glucopyranosO-[2,1-d]-Δ2′-
thiazoline (NButGT) [24], was used in this study to treat mSOD with severe symptoms
and wt control mice. mSOD and wt littermate mice were fed chow with or without
NButGT (100mg/kg/day) for three days (n= 6 for each group). Following treatment,
animals were sacrificed using CO2 and tissues were then collected.
2.3.3 Tissue homogenization and Immunoblotting
Protein lysates were extracted from freshly frozen spinal cord tissue. Modified RIPA
buffer was used (50 mM Tris-HCl, 150 mM NaCl, 1% NP-40, I mM EDTA) and a
cocktail of protease/phosphatase/OGA inhibitors was added to the buffer before use (1
µg/mL aprotinin; 1 µg/mL leupeptin; 1 µg/mL pepstatin; 1mM PMSF; 2mM Na3VO4; 1
mM NaF; 1 mM NButGT). The buffer was added to the tissue using a total volume of

66
10 x the weight of the tissue. The tissue was sonicated at 4 °C, and then clarified by
centrifugation at 17,900 g (Eppendorf 5410 centrifuge, Hamburg, Germany) for 15 min at
4 oC. Supernatant was kept on ice for Bradford assays, aliquoted, and finally stored at -
80 oC for subsequent use.
Immunoblotting was carried out as previously described [30]. Briefly, protein samples
were diluted with Laemmli sample buffer, boiled for 5 min, and electrophoresed through
7-12.5% SDS-PAGE gels. Proteins were electrophoresed for 1.5 hr at constant voltage,
transferred onto nitrocellulose membrane for 2 hr, and then the membranes were blocked
with 1% BSA or gelatin dissolved in 1 x PBS containing 0.1% Tween-20 (PBS-T) for 2
hr at room temperature on a shaker plate. Membranes were then incubated with the
specific primary antibodies overnight at 4 °C. The membrane was washed three times
with PBST at room temperature and blocked again in 1% BSA or gelatin for 0.5 hr at
room temperature, then incubated with HRP-conjugated secondary antibodies for 1 hr at
room temperature. The membrane was washed four times with PBS-T and visualized
using ECL reagents and film (Amersham Bioscience, Piscataway, NJ). Developed films
were scanned and quantified using Scion Image Beta 4.03 software (Scion Corp.,
Frederick, MD).
2.3.4 Perfusion and immunohistochemistry
mSOD and littermate control mice were sacrificed using CO2, and rapidly perfused
transcardially with 30 mL phosphate buffered saline (0.1 M PBS, pH 7.4) followed by 30
mL 4% paraformaldehyde (PFA; pH 7.4). Spinal cords were removed, post-fixed in 4%
PFA for 24 hr, transferred to 20% sucrose in PBS overnight for cryoprotection, frozen in

67
Tissue-Tek O.C.T compound (Sakura, Zoeterwoude, Netherlands), and finally sectioned
in the transverse plane to provide 50 m thick sections using a Leica cryostat (Leica,
Wetzlar, Germany).
Free floating sections were permeabilized using 1 x PBS (pH7.4) containing 0.3% Triton
X-100 (PBST) for 15 min. After blocking with 10% normal goat serum and 5% BSA for
1 hr at room temperature, sections were incubated with specific primary antibodies
overnight at 4 °C. The sections then were washed three times with 0.3% Triton X-100 in
PBS for 15 min each and incubated with specific secondary antibody conjugated with
Cy3 or FITC for 1.5 hr at room temperature in the dark. Finally, sections were washed
and mounted with Vectashield Mounting Medium containing 4′-6-diamidinO-2-
phenylindole (DAPI) (H-1200, Vector Laboratories). Images were collected and analyzed
using a Leica fluorescent microscope (DM4000B) equipped with the Spot digital camera
(DFC350FX, Diagnostic Instruments, Sterling Heights, MI) and Leica Application Suite
(LAS 2.5.0 R1). In control experiments, sections were performed in parallel but without
primary antibody. When pre-treating spinal cord sections, transverse slices were
incubated with either wt OGA [9] at 105 g/mL or mutant OGA [3] at 86 g/mL for 3 hr
in a 25 °C water bath, rinsed, and immunostained as described above.
2.3.5 A densitometry analysis of O-GlcNAc immunohistochemistry
Densitometry analysis of O-GlcNAc immunoreactivity in spinal cords was performed
using a protocol described previously [30]. Briefly, images were taken using Leica 10 x
objective lens and imported into NIH Image J software (NIH, Bethesda, MD) for analysis,

68
regions of interest (ROI) were defined morphologically in the ventral horns of spinal
cord. Two ROIs of identical size (200 x 300 pixels) were defined in each transverse
section of spinal cord including both ventral horns. To insure the uniformity of analysis,
spinal cord sections were processed in parallel from both mSOD mice and littermate wt
controls. The O-GlcNAc immunoreactivities in the ROIs were expressed as the mean
pixel grey scale values from these ROIs. Measurements taken from the ventral horns in
each section were averaged to get one mean value for ventral horn from each section.
Data from five sections obtained from the lower lumbar region (L3-5) of each animal
were averaged for each data point. For densitometry analysis of O-GlcNAc
immunoreactivity within motor neurons, high magnification images were acquired by
using a Leica 40 x objective lens. All images were obtained using identical camera gain
and other settings using tissue processed in parallel.
2.3.6 Motor neuron counts
Motor neuron counts were performed using modified methods previously used by others
for counting neurons in animal preparations [19]. Briefly, lumbar spinal cord sections
from mSOD mice (n=3) and littermate wt controls (n=3) were stained with NeuN
antibody and counterstained with DAPI. Under 40 x objective lens, motor neurons
meeting the following criteria were counted: 1) size > 25 m; 2) possession of at least
one thick process; 3) location in ventral grey matter regions below a horizontal level
through the center of central canal. Data were collected by averaging five sections per
animal and a total of 30 sections were counted.

69
2.3.7 Statistical analysis
Results were expressed as mean SD. Comparisons between mSOD and control mouse
samples were analyzed by a Student t-test using SPSS 17.0 software. A p-value of less
than 0.05 was considered significant.
2.4 Results
2.4.1 Distribution of O-GlcNAc modified protein in CNS
Immunoreactivity against O-GlcNAc-modified proteins was evaluated in transverse
sections of spinal cord from wt mice using an antibody that preferentially detects O-
GlcNAc modified residues [6]. As shown in Figure 2-1A, O-GlcNAc immunolabelling
was evident in cells of the spinal cord, largely within the grey matter and in both dorsal
and ventral horns. To confirm that the observed pattern of O-GlcNAc immunoreactivity
was specific for O-GlcNAc modified proteins, sections were treated with a bacterial β-N-
acetylglucosamindase having OGA activity (Bacteroides thetaiotamicron GH84,
BtGH84) in order to cleave this moiety off from modified proteins [9]. As shown in
Figure 2-1B, following treatment with wt OGA, immunoreactivity was absent,
confirming that the O-GlcNAc-directed antibody used here detects only O-GlcNAc
modified proteins. When spinal cord sections were treated in an identical manner using
an inactive mutant of BtGH84 (D174A, D175A) that does not possess enzymatic activity
[3], O-GlcNAc immunoreactivity was essentially identical to that seen in control tissues
(Figure 2-1C).

70
Under high magnification, O-GlcNAc immunoreactivity was observed in large motor
neurons, smaller interneurons, and other cells (Figure 2-1D), generally in the perinuclear
regions of cells, but also within the cytoplasm and nucleus. The immunolabelling of
nuclei was evident as shown by colocalization with DAPI (Figure 2-1E, and overlay in
Figure 2-1F). To determine the cell types that exhibit O-GlcNAc immunoreactivity in
spinal cord, we employed cell-type specific markers. We observed that O-GlcNAc
immunoreactivity was displayed by neurons (Figure 2-1G) using the neuron-specific
marker NeuN, both within the nucleus and cytoplasm, but especially in the perinuclear
region; oligodendroglia, as demonstrated by CNPase, showed O-GlcNAc
immunoreactivity largely within the nucleus (Figure 2-1H), and astrocytes (Figure 2-1I;
GFAP+ cells) showed immunoreactivity largely within the nucleus. Immunoreactivity
against O-GlcNAc was not apparent in microglia (Figure 2-1J).
2.4.2 Level of O-GlcNAc modified protein in spinal cords and motor neurons
To evaluate the levels and identities of O-GlcNAc modified proteins in spinal cord tissue
from littermate wt mice (n=9) and mice over-expressing mSOD (n=9) with neurological
deficits at early stage (no symptoms), middle stage (mild symptoms), and end stage
(severe symptoms), Immunoblotting was performed (Figure 2-2A). O-GlcNAc
immunoreactivity was evident for a large number of protein bands in the immunoblot,
both in wt and mSOD tissue. No significant difference in total O-GlcNAc levels was seen
as measured by integrating total band densities for both mSOD and wt tissues (Figure 2-
2B).

71
A previous study has shown that O-GlcNAc modified NFM levels are decreased mainly
at end stage in spinal cords of ALS-like rats [22]. As the levels of total O-GlcNAc
density in immunoblots are a function of O-GlcNAc modification of many proteins from
a mixture of cell types across various tissue regions, we evaluated levels of
immunoreactivity of O-GlcNAc modified proteins as determined by
immunocytochemistry in spinal cord sections from end stage mSOD (n=3) and wt mice
(n=3). As shown in Figure 2-2C, densitometric analysis of O-GlcNAc immunoreactivities
in ventral horn regions of spinal cord demonstrated that immunoreactivity was
significantly lower (p < 0.01) in mSOD than control spinal cord. As neurons have O-
GlcNAc immunoreactivity in spinal cord, the decreased O-GlcNAc immunoreactivity in
end stage mSOD spinal cord could stem from a specific reduction in the levels of O-
GlcNAc protein modification in neurons or from a reduction in the number of motor
neurons.
To examine these possibilities, we conducted motor neurons counts and performed
densitometry analysis of O-GlcNAc immunoreactivity in the ventral horns of both end
stage mSOD mice and wt controls. As shown in Figure 2-2C, O-GlcNAc
immunoreactivity in the ventral horns of spinal cord was significantly lower (p < 0.001)
in end stage mSOD than control spinal cord. As expected, we found the mSOD mice had
significantly lower numbers of motor neurons remaining than the control wt mice (p <
0.001) (Figure 2-2D).

72
2.4.3 NButGT significantly enhanced O-GlcNAc level in spinal cords
Modification of proteins by O-GlcNAc occurs through the action of a specific
glycosyltransferase known as OGT [21] and hydrolytic cleavage of the O-GlcNAc
moiety is catalyzed byOGA [10]. In an effort to modulate levels of O-GlcNAc modified
proteins in spinal cord we treated mSOD (n=6) and control mice (n=6) with a specific
inhibitor of OGA (NButGT) for 3 days. As shown in Figure 2-3C, O-GlcNAc
immunoreactivity in NButGT treated mice was increased in spinal cord sections as
compared to tissue from non-treated mice in both gray and white matter of spinal cord.
Enhanced O-GlcNAc immunoreactivity was found in the nuclei of astrocytes and
oligodendroglia, and in the cytoplasm and nuclei of neurons, including surviving
motorneurons from mSOD mice (Figure 2-3C). Figure 2-3A shows immunoblotting data
indicating that levels of O-GlcNAc modified proteins were significantly increased (P <
0.05) both in wt and mSOD spinal cord tissue in the NButGT treated groups, as
compared to non-treated animals (Figure 3B). Although it appears that levels of the O-
GlcNAc modified proteins were higher in wt as compared to mSOD tissue, there was no
statistically significant difference in total O-GlcNAc band densities between wt and
mSOD (Figure 2-3B). O-GlcNAc immunoreactivity was evaluated by densitometric
analysis in ventral horn regions of spinal cord where wt spinal cord exhibited
significantly higher (p < 0.01) immunoreactivity as compared to mSOD spinal cord.
NButGT treatment significantly increased both wt and mSOD O-GlcNAc
immunoreactivities (p < 0.01 and p < 0.001, respectively) as measured using
immunohistochemistry (Figure 2-3C-D).

73
Spinal cord homogenates were probed with the antibody (NL6), which recongnizes an O-
GlcNAc modified NFM epitope (OG-NFM) [22]. As seen in Figure 2-4A,
immunoblotting revealed that levels of OG-NFM proteins were significant reduced (p <
0.01) in mSOD mice versus wt mice in both NButGT treated and non-treated groups. We
also found that levels of OG-NFM proteins were significantly increased (p < 0.05) in wt
spinal cord tissues in the NButGT treated groups, as compared to tissue from non-treated
animals. There was, however, no significant difference in OG-NFM band densities in
mSOD spinal cord tissue arising from treatment with NButGT (p > 0.05). As we
observed that NButGT could modulate global O-GlcNAc and specific OG-NFM levels,
we further examined phospho-NFM levels in these tissues using SMI31 antibody, which
is known to recognize phosphorylated epitopes within the repeating KSP motifs of NFH
and NFM. In Figure 2-4B, we observed that levels of phospho-NFM in spinal cord tissues
appear lower in wt mice treated with NButGT compared to non-treated wt mice,
however, this difference did not reach statistical significance (p > 0.05) in either wt or
mSOD groups after densitometry was corrected by normalization using total NFM levels.
2.5 Discussion
Previous studies have demonstrated that when compared to appropriate controls, CNS
tissues from ALS patients, and murine models of ALS, have increased levels of protein
kinases and phosphoproteins [15, 16]. Notably, increased immunoreactivity of the highly
phosphorylated isoform of NFH has been reported and it has been postulated that NFH
acts as a sink for phosphorylation by hyperactive cyclin-dependant kinase-5 (cdk5) to
hinder uncontrolled phsophorylation of proteins having critical cellular functions [26].

74
Extensive hyperphosphorylation of the KSP-repeat domain of NFs, however, has also
been implicated in progression of symptoms of ALS [31]. Interestingly, the microtubule-
associated protein, tau, has also been found to be hyperphosphorylated in ALS tissue
[36]. Since there appears to be a reciprocal relationship between O-GlcNAc and
phosphate on some proteins including tau [8, 37] we were intrigued by the possible
reciprocal relationship between O-GlcNAc levels and phosphorylation of NFs. If
phosphorylation and O-GlcNAc are generally reciprocal then we might observe that O-
GlcNAc levels in a mouse model of ALS would be significantly decreased. Furthermore,
if this holds true for NFs in addition to other proteins pharmacological elevation of O-
GlcNAc levels achieved by blocking the action of OGA, the enzyme responsible for
removing this modification, might hinder hyperphosphorylation of NFs in this model.
As a first step to evaluating this question, we first investigated distribution of O-GlcNAc
modified proteins in CNS and whether O-GlcNAc levels were decreased in G93A mSOD
mice. Using a specific antibody against O-GlcNAc modified proteins, we evaluated
which CNS cell types exhibited O-GlcNAc immunoreactivety to determine whether
modulating O-GlcNAc levels might have a possible effect on NF hyperphosphorylation
in neurons in this mouse model of ALS. Immunocytochemical detection of O-GlcNAc
and use of cell-specific markers revealed that O-GlcNAc was abundant in neurons, in
oligodendrocytes, and astrocytes. We did not detect O-GlcNAc immunoreactivity in
microglia. In both oligodendrocytes and astrocytes, O-GlcNAc immunoreactivity was
most evident in the nuclei of these cells, whereas in neurons O-GlcNAc
immunoreactivity was found at similar levels in both the nucleus and cytoplasm, but must

75
notably in the peri-nuclear region, as described previously in human astroglial cells,
mouse cerebellar neurons, and mouse brain tissue [2, 25, 28]. Significantly, we found that
O-GlcNAc levels are significantly decreased in spinal cord tissue from mSOD mice as
compared to controls. Using immunohistochemistry, we find that the reduction in O-
GlcNAc levels found in mSOD spinal cord is prominent in the grey matter of spinal cord,
furthermore, we observed low O-GlcNAc levels in remaining neurons as well as neuronal
cell loss which likely lead to decreased overall O-GlcNAc levels in spinal cord from
mSOD mice with severe symptoms at end stage.
Having established that O-GlcNAc levels were decreased in symptomatic G93A mSOD
mice and that the spinal motor neurons had significantly lower O-GlcNAc modification
of proteins in this mouse model, we investigated whether we could modulate O-GlcNAc
levels through pharmacological methods. To determine if O-GlcNAc levels could be
modulated, we employed a small molecule inhibitor of OGA, NButGT, that is highly
selective for OGA. While PUGNAc has been widely used as an inhibitor of OGA to
modulate O-GlcNAc in cells and tissues it is known that this compound also inhibits
lysosomal β-hexosaminidases, the enzymes that cleave both GlcNAc and GalNAc from
many glycosylated proteins [24]. Furthermore, it has been recently reported that
PUGNAc does not increase O-GlcNAc levels in treated animals [38]. Therefore, we use
NButGT, a potent (KI = 230 nM) and highly selective (1200-fold selectivity) small
molecule inhibitor of OGA, which has been shown to increase global levels of O-GlcNAc
modified proteins in various tissues [24, 25]. We found that a three-day oral treatment
with NButGT significantly increased O-GlcNAc levels, as measured both by immunoblot

76
and immunohistochemistry. Interestingly, the treatment increased O-GlcNAc levels in
spinal cord to similar levels, both in mSOD and control mice, when compared to
untreated mSOD and control animals, respectively. This data suggests that in addition to
neuronal loss, some of the reduction in O-GlcNAc levels in mSOD spinal cord is likely
due to decreased O-GlcNAc modification of surviving neurons and other cells, an
observation consistent with the known activation of protein kinases and increased
phosphorylation of neuronal proteins in this animal model [15]. These observations were
supported by immunohistochemical analysis of O-GlcNAc levels that showed
significantly enhanced O-GlcNAc levels in neurons, astrocytes, and oligodendroglia in
both mSOD and wt spinal cord tissue following NButGT treatment. NButGT is therefore
able to modulate O-GlcNAc effectively in this mouse model of ALS.
To test whether inhibitor treatment was modulating O-GlcNAc levels on NFs, we used an
antibody (NL6) that has been raised against rat O-GlcNAc modified NFM [22]. As
outlined above, there is considerable evidence for hyperphosphorylated NFs playing a
role in the progression of ALS in humans [14]. Recent studies have shown reductions in
O-GlcNAc modified NFM in a rat model of ALS over-expressing mSOD [22]. Here, we
observed significant reductions of NL6 immunoreactivity in mSOD versus wt mice both
in NButGT treated and non-treated groups by using immunoblots. Our immunoblot
results corroborate the findings reported by Ludemann et al. [22]. Interestingly, use of
NButGT significantly increased O-GlcNAc modified NFM in wt but not mSOD mice as
determined using the NL6 antibody. Given these observations, we examined the effects
of short term three days NButGT treatment on phosphorylation of the KSP repeats of

77
NFM using the SMI31 antibody, which recognizes the phosphorylated KSP epitope.
Given that it has been proposed that the KSP repeat region of NFM contains several O-
GlcNAc modification sites, we anticipated that treatment might decrease phosphorylation
of this region. immunoblot analysis revealed a decrease in the mean SMI31
imunoreactivity in the wt mice upon inhibitor treatment, although this effect did not reach
statistical significance. No difference in mean SMI31 immunoreactivity was observed
between treated and untreated mSOD mice. We speculate that this lack of difference in
SMI31 immunoreactivity in mSOD mice may arise from the aggregation of
hyperphosphorylated NFs in these severe symptomatic stage animals [7], which may
prevent dephosphorylation and subsequent O-GlcNAc modification. Overall, however,
these results indicate that O-GlcNAc levels can be modulated in vivo to increase levels of
O-GlcNAc on NFs. To address whether elevated O-GlcNAc levels induced by NButGT
or other OGA inhibitors might alter hyperphosphorylation of NFs, further long term
treatment studies monitoring progression of symptoms in disease models are required to
clarify this possibility.

78
2.6 Figures
Figure 2- 1 Distribution of O-GlcNAc modified proteins in spinal cord. Immunoreactivity of O-GlcNAc modified proteins in transverse sections of lumbar spinal cord from wild type (WT) mice detected using CTD110.6 antibody in the absence of O-GlcNAcase (OGA) (A); in the presence of functional OGA (wtOGA) (B); and in the presence of mutant, non-functional OGA (mOGA) (C). Note that immunofluorescence is observed in cells largely in grey matter and in both ventral and dorsal horns (A, C, D); immunostaining is absent after wtOGA treatment (B). Immunolabelling of O-GlcNAc (green) in ventral horns of lumbar spinal cord sections from WT mice localize with many nuclei (blue, DAPI) (D-F), and cells exhibiting the neuronal marker, NeuN (red) (G); the oligodendrocyte marker CNPase (red) (H); and the astrocyte marker, GFAP (red) (I); but absent with cells exhibiting microglia marker, Iba1 (red) (J). Scale bars: G, 15 µm; H-J, 5 µm.

79

80
Figure 2- 2 Levels of O-GlcNAc modified proteins in spinal cord and motor neurons. (A) Immunoblotting shows many CTD110.6 immunoreactive bands corresponding to O-GlcNAc modified proteins from spinal cord homogenates from both WT and mSOD mice at various disease stages. There is no significant difference in total band density between WT and mSOD tissue (p>0.05) (B). (C) Densitometric analysis of O-GlcNAc immunoreactivity in ventral horn regions of spinal cords shows that immunoreactivity is significantly lower in mSOD than WT ( * P<0.01). (D) Densitometric analysis of O-GlcNAc immunoreactivity in motor neurons of spinal cords shows that immunoreactivity is significantly lower in mSOD than WT ( ** P<0.001) (upper panel). Motor neuron counts show that significantly lower survival neurons labelled with NeuN (red) in mSOD than wt (** P<0.001) (lower panel).

81
Figure 2- 3 NButGT significantly enhances O-GlcNAc level in spinal cords.
mSOD and wild type (WT) mice were treated orally with a specific inhibitor of OGA (NButGT) for 3 days. (A-B) Immunoblotting shows that levels of O-GlcNAc modified proteins were significantly increased both in WT and mSOD spinal cord tissues in the NButGT treated groups, compared with non-treated animals ( * p<0.05). There is no significant difference in total O-GlcNAc band densities between WT and mSOD spinal cord tissue (B). (C-D) This observation is evaluated further by densitometric analysis of O-GlcNAc immunoreactivity in the ventral horn regions of spinal cord (* p<0.01, ** p<0.001, respectively).

82
Figure 2- 4 NButGT induces increased O-GlcNAc modification of NFM in spinal cord.
(A) Immunoblotting shows levels of OG-NFM proteins in spinal cord tissue of mSOD and wt mice from NButGT treated and non-treated groups. Levels of OG-NFM proteins are significantly reduced (* p < 0.01) in mSOD spinal cord tissue in both NButGT treated and non-treated groups, compared to WT mice. There is a significantly increase in OG-NFM protein in WT spinal cord tissue in the NButGT treated group, compared with non-treated (control) mice ( * p < 0.05) (B) Immunoblotting shows levels of phosphorylation of NFM proteins from spinal cord homogenates using SMI31 antibody. Levels were quantified by densitometry and normalized to total NFM level (NF-09 immunoreactivity), are not significant difference (p> 0.05) between WT and mSOD tissue in either NButGT treated or non-treated (control) groups.

83
2.7 Reference List
[1] S. Ackerley, P. Thornhill, A.J. Grierson, J. Brownlees, B.H. Anderton, P.N. Leigh, C.E. Shaw, C.C. Miller, Neurofilament heavy chain side arm phosphorylation regulates axonal transport of neurofilaments, The Journal of cell biology 161 (2003) 489-495.
[2] Y. Akimoto, F.I. Comer, R.N. Cole, A. Kudo, H. Kawakami, H. Hirano, G.W. Hart, Localization of the O-GlcNAc transferase and O-GlcNAc-modified proteins in rat cerebellar cortex, Brain Res 966 (2003) 194-205.
[3] N. Cetinbas, M.S. Macauley, K.A. Stubbs, R. Drapala, D.J. Vocadlo, Identification of Asp174 and Asp175 as the key catalytic residues of human O-GlcNAcase by functional analysis of site-directed mutants, Biochemistry 45 (2006) 3835-3844.
[4] X. Cheng, G.W. Hart, Alternative O-glycosylation/O-phosphorylation of serine-16 in murine estrogen receptor beta: post-translational regulation of turnover and transactivation activity, J Biol Chem 276 (2001) 10570-10575.
[5] T.Y. Chou, G.W. Hart, C.V. Dang, c-Myc is glycosylated at threonine 58, a known phosphorylation site and a mutational hot spot in lymphomas, J Biol Chem 270 (1995) 18961-18965.
[6] F.I. Comer, K. Vosseller, L. Wells, M.A. Accavitti, G.W. Hart, Characterization of a mouse monoclonal antibody specific for O-linked N-acetylglucosamine, Anal Biochem 293 (2001) 169-177.
[7] M.C. Dal Canto, M.E. Gurney, Neuropathological changes in two lines of mice carrying a transgene for mutant human Cu,Zn SOD, and in mice overexpressing wild type human SOD: a model of familial amyotrophic lateral sclerosis (FALS), Brain Res 676 (1995) 25-40.
[8] Y. Deng, B. Li, F. Liu, K. Iqbal, I. Grundke-Iqbal, R. Brandt, C.X. Gong, Regulation between O-GlcNAcylation and phosphorylation of neurofilament-M and their dysregulation in Alzheimer disease, FASEB J 22 (2008) 138-145.
[9] R.J. Dennis, E.J. Taylor, M.S. Macauley, K.A. Stubbs, J.P. Turkenburg, S.J. Hart, G.N. Black, D.J. Vocadlo, G.J. Davies, Structure and mechanism of a bacterial beta-glucosaminidase having O-GlcNAcase activity, Nat Struct Mol Biol 13 (2006) 365-371.
[10] D.L. Dong, G.W. Hart, Purification and characterization of an O-GlcNAc selective N-acetyl-beta-D-glucosaminidase from rat spleen cytosol, J Biol Chem 269 (1994) 19321-19330.
[11] D.L. Dong, Z.S. Xu, G.W. Hart, D.W. Cleveland, Cytoplasmic O-GlcNAc modification of the head domain and the KSP repeat motif of the neurofilament protein neurofilament-H, J Biol Chem 271 (1996) 20845-20852.
[12] L.S. Griffith, B. Schmitz, O-linked N-acetylglucosamine levels in cerebellar neurons respond reciprocally to pertubations of phosphorylation, Eur J Biochem 262 (1999) 824-831.
[13] M.E. Gurney, H. Pu, A.Y. Chiu, M.C. Dal Canto, C.Y. Polchow, D.D. Alexander, J. Caliendo, A. Hentati, Y.W. Kwon, H.X. Deng, et al., Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation, Science 264 (1994) 1772-1775.

84
[14] A. Hirano, Cytopathology of amyotrophic lateral sclerosis, Adv Neurol 56 (1991) 91-101.
[15] J.H. Hu, K. Chernoff, S. Pelech, C. Krieger, Protein kinase and protein phosphatase expression in the central nervous system of G93A mSOD over-expressing mice, J Neurochem 85 (2003) 422-431.
[16] J.H. Hu, H. Zhang, R. Wagey, C. Krieger, S.L. Pelech, Protein kinase and protein phosphatase expression in amyotrophic lateral sclerosis spinal cord, J Neurochem 85 (2003) 432-442.
[17] K. Kamemura, G.W. Hart, Dynamic interplay between O-glycosylation and O-phosphorylation of nucleocytoplasmic proteins: a new paradigm for metabolic control of signal transduction and transcription, Prog Nucleic Acid Res Mol Biol 73 (2003) 107-136.
[18] V.M. Lee, L. Otvos, Jr., M.J. Carden, M. Hollosi, B. Dietzschold, R.A. Lazzarini, Identification of the major multiphosphorylation site in mammalian neurofilaments, Proc Natl Acad Sci U S A 85 (1988) 1998-2002.
[19] J. Lewis, E. McGowan, J. Rockwood, H. Melrose, P. Nacharaju, M. Van Slegtenhorst, K. Gwinn-Hardy, M. Paul Murphy, M. Baker, X. Yu, K. Duff, J. Hardy, A. Corral, W.L. Lin, S.H. Yen, D.W. Dickson, P. Davies, M. Hutton, Neurofibrillary tangles, amyotrophy and progressive motor disturbance in mice expressing mutant (P301L) tau protein, Nat genetics 25 (2000) 402-405.
[20] C.S. Lobsiger, M.L. Garcia, C.M. Ward, D.W. Cleveland, Altered axonal architecture by removal of the heavily phosphorylated neurofilament tail domains strongly slows superoxide dismutase 1 mutant-mediated ALS, Proc Natl Acad Sci U S A 102 (2005) 10351-10356.
[21] W.A. Lubas, D.W. Frank, M. Krause, J.A. Hanover, O-Linked GlcNAc transferase is a conserved nucleocytoplasmic protein containing tetratricopeptide repeats, J Biol Chem 272 (1997) 9316-9324.
[22] N. Ludemann, A. Clement, V.H. Hans, J. Leschik, C. Behl, R. Brandt, O-glycosylation of the tail domain of neurofilament protein M in human neurons and in spinal cord tissue of a rat model of amyotrophic lateral sclerosis (ALS), J Biol Chem 280 (2005) 31648-31658.
[23] M.S. Macauley, X. Shan, S.A. Yuzwa, T.M. Gloster, D.J. Vocadlo, Elevation of Global O-GlcNAc in rodents using a selective O-GlcNAcase inhibitor does not cause insulin resistance or perturb glucohomeostasis, Chem Biol 17 (2010) 949-958.
[24] M.S. Macauley, G.E. Whitworth, A.W. Debowski, D. Chin, D.J. Vocadlo, O-GlcNAcase uses substrate-assisted catalysis: kinetic analysis and development of highly selective mechanism-inspired inhibitors, J Biol Chem 280 (2005) 25313-25322.
[25] J.A. Matthews, J.L. Belof, M. Acevedo-Duncan, R.L. Potter, Glucosamine-induced increase in Akt phosphorylation corresponds to increased endoplasmic reticulum stress in astroglial cells, Mol Cell Biochem 298 (2007) 109-123.
[26] M.D. Nguyen, R.C. Lariviere, J.P. Julien, Deregulation of Cdk5 in a mouse model of ALS: toxicity alleviated by perikaryal neurofilament inclusions, Neuron 30 (2001) 135-147.

85
[27] R.A. Nixon, P.A. Paskevich, R.K. Sihag, C.Y. Thayer, Phosphorylation on carboxyl terminus domains of neurofilament proteins in retinal ganglion cell neurons in vivo: influences on regional neurofilament accumulation, interneurofilament spacing, and axon caliber, J Cell Biol 126 (1994) 1031-1046.
[28] M. Rex-Mathes, S. Werner, D. Strutas, L.S. Griffith, C. Viebahn, K. Thelen, B. Schmitz, O-GlcNAc expression in developing and ageing mouse brain, Biochimie 83 (2001) 583-590.
[29] E.P. Roquemore, M.R. Chevrier, R.J. Cotter, G.W. Hart, Dynamic O-GlcNAcylation of the small heat shock protein alpha B-crystallin, Biochemistry 35 (1996) 3578-3586.
[30] X. Shan, J.H. Hu, F.S. Cayabyab, C. Krieger, Increased phospho-adducin immunoreactivity in a murine model of amyotrophic lateral sclerosis, Neuroscience 134 (2005) 833-846.
[31] P.H. Tu, P. Raju, K.A. Robinson, M.E. Gurney, J.Q. Trojanowski, V.M. Lee, Transgenic mice carrying a human mutant superoxide dismutase transgene develop neuronal cytoskeletal pathology resembling human amyotrophic lateral sclerosis lesions, Proc Natl Acad Sci U S A 93 (1996) 3155-3160.
[32] J.S. Valentine, P.A. Doucette, S. Zittin Potter, Copper-zinc superoxide dismutase and amyotrophic lateral sclerosis, Annu Rev Biochem 74 (2005) 563-593.
[33] Z. Wang, M. Gucek, G.W. Hart, Cross-talk between GlcNAcylation and phosphorylation: Site-specific phosphorylation dynamics in response to globally elevated O-GlcNAc, Proc Natl Acad Sci U S A 105 (2008) 13793-13798.
[34] L. Wells, L.K. Kreppel, F.I. Comer, B.E. Wadzinski, G.W. Hart, O-GlcNAc transferase is in a functional complex with protein phosphatase 1 catalytic subunits, J Biol Chem 279 (2004) 38466-38470.
[35] T.L. Williamson, D.W. Cleveland, Slowing of axonal transport is a very early event in the toxicity of ALS-linked SOD1 mutants to motor neurons, Nat Neurosci 2 (1999) 50-56.
[36] W. Yang, M.M. Sopper, C. Leystra-Lantz, M.J. Strong, Microtubule-associated tau protein positive neuronal and glial inclusions in ALS, Neurology 61 (2003) 1766-1773.
[37] S.A. Yuzwa, M.S. Macauley, J.E. Heinonen, X. Shan, R.J. Dennis, Y. He, G.E. Whitworth, K.A. Stubbs, E.J. McEachern, G.J. Davies, D.J. Vocadlo, A potent mechanism-inspired O-GlcNAcase inhibitor that blocks phosphorylation of tau in vivo, Nat Chem Biol 4 (2008) 483-490.
[38] L. Zou, S. Yang, S. Hu, I.H. Chaudry, R.B. Marchase, J.C. Chatham, The protective effects of PUGNAc on cardiac function after trauma-hemorrhage are mediated via increased protein O-GlcNAc levels, Shock (Augusta, Ga 27 (2007) 402-408.

86
3: Mislocalization of TDP-43 in the G93A mutant SOD1 transgenic mouse model of ALS
Published in the Neuroscience Letters (2009) 458: 70-74
Authors: Xiaoyang Shan, David Vocadlo. Charles Krieger.
Author contribution: X.S., D. V. and C. K., designed the experiments. X.S. performed all experiments. X.S., D. V. and C. K., analysed data. X.S. wrote the first draft, and C.K. and D.V. wrote the final manuscript. D.V. and C. K. were both corresponding authors.
3.1 Abstract
Previous evidence demonstrates that TAR DNA binding protein (TDP-43)
mislocalization is a key pathological feature of amyotrophic lateral sclerosis (ALS).
TDP-43 normally shows nuclear localization, but in CNS tissue from patients who have
died with ALS this protein mislocalizes to the cytoplasm. Disease specific TDP-43
species have also been reported to include hyperphosphorylated TDP-43, as well as a C-
terminal fragment. Whether these abnormal TDP-43 features are present in patients with
SOD1- related familial ALS (fALS), or in mutant SOD1 over-expressing transgenic
mouse models of ALS remains controversial. Here we investigate TDP-43 pathology in
transgenic mice expressing the G93A mutant form of SOD1. In contrast to previous
reports we observe redistribution of TDP-43 to the cytoplasm of motor neurons in mutant
SOD1 transgenic mice, but this is seen only in mice having advanced disease.
Furthermore, we also observe rounded TDP-43 immunoreactive inclusions associated

87
with intense ubiquitin immunoreactivity in lumbar spinal cord at end stage disease in
mSOD mice. These data indicate that TDP mislocalization and ubiquitination are present
in end stage mSOD mice. However, we do not observe C-terminal TDP-43 fragments nor
TDP-43 hyperphosphorylated species in these end stage mSOD mice. Our findings
indicate that G93A mutant SOD1 transgenic mice recapitulate some key pathological, but
not all biochemical hallmarks, of TDP-43 pathology previously observed in human ALS.
These studies suggest motor neuron degeneration in the mutant SOD1 transgenic mice is
associated with TDP-43 histopathology.
3.2 Introduction
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease selectively affecting
motor neurons of the brainstem and spinal cord, as well as cortical regions.
Approximately, 5–10% of ALS cases are inherited (fALS), however, the cause of the
remaining 90–95% of sporadic ALS (sALS) cases remains unknown. Mutations of
superoxide dismutase 1 (SOD1) account for approximately 15–20% of fALS cases (1–
2% of all ALS cases), whereas various other gene mutations account for only a small
subset of the remaining fALS cases [10]. Nervous system tissue from patients who died
with fALS and sALS have been extensively studied to gain insight into the pathological
features of ALS. To study the pathogenesis and investigate the details of the progression
of ALS, transgenic rodent models harboring fALS-associated mutations in human mutant
SOD1 (mSOD) have been developed. These models have proven to be of significant
value [9] and replicate several key features observed in both sALS and fALS including
progressive loss of motor neurons, neurofilament aggregation, and the accumulation of
cytoplasmic ubiquitinated inclusions (UBI) within degenerating motor neurons[6, 21].

88
Recently, two independent groups [15] have reported that TAR DNA binding protein
(TDP-43), a nuclear DNA and RNA binding protein proposed to function as a regulator
of transcription [17] and alternative splicing [2, 14] is present in UBIs in sALS.
Furthermore, a number of TDP-43 mutations have been reported in both sALS and fALS
cases, suggesting that TDP-43 may play a causal role in the pathogenesis of ALS [20, 23,
24]. TDP-43 is normally localized to the nucleus, however, in CNS tissue from patients
who died with ALS, TDP-43 is redistributed from the nucleus to the cytoplasm, where it
appears to be distributed diffusely, or to aggregate as a component of UBIs [1, 5, 15].
Although studies of TDP-43 in human ALS cases are generally consistent [1, 4, 13, 15],
some aspects of the TDP-43 pathology remain controversial. For instance, Mackenzie
and colleagues [12] suggest that abnormal localization of TDP-43 is present in most
sALS and fALS cases but is absent in fALS caused by SOD1 mutations. In contrast,
Robertson and colleagues [18] showed, in two fALS cases carrying SOD1 mutations, that
there is mislocalization of TDP-43 to the cytoplasm as well as association with UBIs.
Studies of some lines of mSOD mouse models have claimed that there is no TDP-43
redistribution in these mice [18, 22], although very recently, Kiaei and colleagues [11]
reported in abstract form that many neurons from lumbar spinal cords of mSOD mice do
show cytoplasmic TDP-43 inclusions similar to those seen in CNS tissue from patients
with ALS. Given the discrepancies between previous reports and because transgenic
mSOD mice have been employed extensively to study ALS, we have evaluated whether
pathological changes in TDP-43 localization are found in G93A mSOD mice.

89
3.3 Methods
3.3.1 Animals
Transgenic mice expressing human G93A mutant SOD1 were purchased from Jackson
Laboratories (B6.Cg-Tg(SOD1-G93A)1Gur/J, stock# 004435) or bred locally with
C57BL6 female mice. Mice were genotyped using PCR [7] and wild-type (WT)
littermates were used as controls in these studies. Protocols governing the use of animals
were approved by the Animal Care Review Committee of Simon Fraser University and
were in compliance with guidelines published by the Canadian Council on Animal Care
(CCAC). A minimum of 3 mice per group were used in these studies. We defined end
stage for mSOD mice as the appearance of a set of behavioral markers including an
inability to forage due to paralysis of the hind limbs and an inability of the mice to right
themselves within 10 seconds of lateral recumbency. Mean survival time of the mSOD
mice reaching end stage was 178 ±14 days (mean ± SEM, n = 9).
3.3.2 Immunohistochemistry
Animals were culled with CO2, perfused transcardially with PBS, and subsequently with
a 4% solution of paraformaldehyde (PFA) in PBS. The spinal cords were dissected out
and postfixed in 4% PFA, left overnight in a solution of 20% sucrose in PBS for
cryoprotection, and subsequently embedded in Tissue-Tek O.C.T compound (Sakura,
Zoeterwoude, Netherlands). Transverse lumbar spinal cord sections of 50 µm were cut
using a Leica cryostat. Sections were treated with PBS containing 0.3% Triton X-100
(PBST) for permeabilization, followed by blocking with 5% BSA and 10% NGS. Anti-
TDP-43 rabbit polyclonal antibody (Proteintech, 10782-2-AP) was diluted at 1:500 in
PBST and incubated with free floating cryo-sections overnight at 4 °C. For double

90
labeling experiments, anti-NeuN mouse monoclonal antibody (Chemicon, MAB378),
diluted at 1:1000, anti-ubiquitin mouse monoclonal antibody (Chemicon, MAB1510)
diluted in 1:500 were used, and immunoreactivities of interest detected using appropriate
Cy3- and FITC-conjugated secondary antibodies (Jackson ImmunoResearch
Laboratories). Immunolabeled spinal cord sections were imaged and analyzed using a
Leica DM 4000 microscope and images captured using a Spot digital camera
(DFC350FX, diagnostic Instruments, Sterling Heights, MI) and Leica Application Suite
(LAS2.5.0 R1). Labeling of mSOD transgenic mouse tissue was compared to tissues
obtained from age-matched WT littermates. Control experiments were carried out in
parallel using sections incubated with only secondary antibody and no primary antibody.
3.3.3 Immunoblotting
Protein lysate was extracted from fresh frozen spinal cord tissue using modified RIPA
buffer (50 mM Tris-HCl, 150 mM NaCl, 1% NP-40, 1 mM EDTA) containing a cocktail
of protease/phosphatase inhibitors (1 µg/mL aprotinin; 1 µg/mL leupeptin; 1 µg/mL
pepstatin; 1 mM PMSF; 2 mM Na3VO4; 1 mM NaF) added to the buffer just before use
to generate a solution containing 0.1 mg tissue/mL. The tissue was homogenized on ice
and then the mixture was centrifuged at 17,900 g for 15 min at 4 °C. The resulting
supernatants were diluted with Laemmli sample buffer, boiled for 5 min, and separated
using 10% SDS-PAGE. After electrophoresis, proteins were transferred onto
nitrocellulose membranes at 100 V for 1.5 hr. Membranes were then blocked for 1 hr
with 1% BSA or gelatin dissolved in PBS containing 0.1% Tween 20 (PBS-T) and then
incubated with the specified primary antibodies overnight at 4 °C. The membrane was
washed with PBS-T and blocked for another 0.5 hr after which the membranes were

91
incubated with the appropriate HRP-conjugated secondary antibodies for 1 hr.
Membranes were then washed with PBS-T and immunoreactive proteins visualized using
ECL reagents and film (Amersham Bioscience, Piscataway, NJ).
3.3.4 Sequential extraction
Frozen lumbar spinal cords were weighted and sequentially extracted as previously
described with slight modifications [15]. In brief, tissues were extracted at 200 mg/mL in
low salt (LS) buffer (10 mM Tris, pH 7.5, 5 mM EDTA, 1 mM DTT, 10% sucrose, and a
cocktail of protease inhibitors) by sonicating twice for 20 sec and then centrifuging the
resulting mixture at 25,000 g for 30 min at 4°C. Pellets were sequentially extracted in
high salt buffer containing Triton (TX) (LS containing 1% Triton X-100 and 0.5 M
NaCl), Sarkosyl-containing buffer (SA) (LS containing 1% N-Lauroyl-sarcosine and 0.5
M NaCl) and urea-containing buffer (UR) (7 M urea, 2 M thiourea, 4% 3-[(3-
Cholamidopropyl)dimethylammonio]-1-propanesulfonate (CHAPS), 30 mM Tris, pH
8.5), followed by immunoblotting analysis as described above.
3.4 Results
We examined lumbar spinal cords from 12 week old mSOD mice for NeuN and TDP-43
histopathology, screening all stained sections visually for abnormal neuronal TDP-43
localization (Fig. 3-1). NeuN immunolabelling of 12 week old mSOD ventral horn grey
matter showed many healthy neurons resembling age-matched WT mice (Fig. 3-1a,d).
TDP-43 immunoreactivity was observed exclusively in the nucleus in tissues from both
WT and mSOD mice of this age (Fig. 3-1b,c,e,f). To more comprehensively evaluate
potential TDP-43 pathology, we examined end-stage G93A mSOD mice. Strikingly,

92
motor neurons in lumbar spinal cords from end-stage mSOD mice (Fig. 3-1 g-i), but not
those of WT animals (Fig. 3-1 j-l) of the same age, showed strong cytoplasmic labelling
with anti-TDP-43 antibodies. Furthermore, some of these motoneurons having
cytoplasmic TDP-43 immunoreactivity showed axonal TDP-43 immunoreactivity that
was sometimes also accompanied by punctate nuclear TDP-43 staining (Fig. 3-1 m).
Within the ventral horn of lumbar spinal cord, some TDP-43 immunoreactivity appeared
in dystrophic neurite-like structures as well as rounded inclusions resembling Lewy
body-like hyaline inclusions (LBHI) (Fig. 3-1 h), both of which have been previously
observed and used to define TDP-43 pathology in human ALS tissues [1, 15]. In addition,
some surviving motor neurons displaying normal nuclear TDP-43 immunoreactivity are
observed adjacent to neurons exhibiting a pathological cytoplasmic distribution of TDP-
43 (Fig. 3-1h). Previous studies of spinal cord tissue from patients who died with ALS
has shown that some TDP-43 immunoreactive inclusions also contain ubiquitin, and that
TDP-43 immunoreactivity is associated with the periphery of the ubiquitin-containing
inclusions[1, 3]. It also has been reported that LBHIs are immunoreactive for TDP-43
and ubiquitin in neurons from a sALS patient[16]. To investigate the similarities between
G93A mSOD mice and these human ALS cases we analysed ubiquitin immunoreactivity
and its relation to TDP-43-positive structures in end stage mice. Immunohistochemistry
using a ubiquitin-specific monoclonal antibody revealed strong ubiquitin
immunoreactivity most frequently associated with rounded TDP-43-positive structures
(Fig. 3-1 n-s), but this was not associated with other TDP-43-positive structures such as
dystrophic neurites. Therefore, the TDP-43 histopathological features of end-stage G93A
mSOD mice recapitulate those observed in human ALS cases.

93
These observations suggest TDP-43 redistribution and aggregation occurs in G93A
mSOD mice. We therefore assessed whether abnormal TDP-43 species were present in
these end-stage mSOD mice using immunoblotting. First we examined TDP-43 protein
expression in spinal cord tissue by immunoblotting. Full-length TDP-43 and a lower
migrating species were detected in lysates obtained from both mSOD and WT littermate
mice (Fig. 3-2a), indicating no difference in expression levels of TDP-43 between mSOD
mice and WT controls. The identity of the rapidly migrating species remains unknown.
Secondly, spinal cords were sequentially extracted in buffers of increasing ionic and
detergent strength, essentially as previously described, and these fractions were then
analyzed by immunoblotting. No differences in the abundance or solubility of TDP-43 in
different fractions from end stage mSOD and age-matched WT samples could be
detected. Furthermore, no pathological ~ 25 kDa species nor ~ 45 kDa
hyperphosphorylated species were seen, as have been previously detected in human ALS
(Fig. 3-2c).
3.5 Discussion
Recent reports have shown that TDP-43 is mislocalized in affected neurons in CNS tissue
from ALS patients, moving from normal localization in the nucleus to the cytoplasm. In
addition to TDP-43 mislocalization, both a ~ 25 kDa C-terminally cleaved fragment and a
~ 45 kDa hyperphosphorylated TDP-43 species have been reported in urea-soluble
protein extracts from ALS tissues [15]. In this work we have investigated whether similar
abnormalities in TDP-43 localization occur in transgenic mice expressing the G93A
mutant form of SOD1. In contrast to a previous report on mSOD mice, we observed a

94
redistribution of TDP-43 to the cytoplasm of motor neurons in mutant SOD1 transgenic
mice. The differing results may stem from the stage of the mSOD mice from which
spinal cord sections are derived. Consistent with this possibility, it has been suggested
that some of the TDP-43 pathological features observed in ALS patients may need time
to develop [19]. Previous studies of mSOD mice at 120 days [21], or at 150 days of age
[17] have shown normal nuclear TDP-43 localization. The line of mSOD mice studied in
this work (B6.Cg-Tg(SOD1-G93A)1Gur/J, stock# 004435) is related to the line studied
previously by two other laboratories [17, 21]. Turner and colleagues [21] reported studies
using a G93A mSOD line bred with C57BL6 females, providing (B6SJL-Tg(SOD1-
G93A)1Gur/J, stock# 002726) transgenic mice, which shows disease onset at ~ 90-100
days and a lifespan of ~ 110-120 days. Robertson and colleagues [17] used the same
G93A mSOD line bred with C57BL6 providing (B6.Cg-Tg(SOD1-G93A)1Gur/J)
transgenic mice, which shows disease onset at ~ 4.5 months and a life expectancy of ~ 5
months. Consistent with these previous studies we also observed only normal TDP-43
localization in mSOD mice of 12 weeks of ages but observed TDP-43 pathology in those
mice having advanced to end stage disease with an extended lifespan of ~ 180 days.
Interestingly, despite pathological TDP-43 relocalization we failed to observe C-terminal
TDP-43 fragments (~25 k Da) and TDP-43 hyperphosphorylated species (~45 kDa) in
these end stage mSOD mice. Furthermore, we also find rounded TDP-43
immunoreactivity (LBHI) in lumbar spinal cord tissues in end stage mSOD mice
associated with ubiquitin inclusions, indicating that at least as far as histopathological
features are concerned, all facets of human ALS are present in end stage mSOD mice.
One possible interpretation for the lack of apparent biochemical changes in TDP-43 in

95
G93A mSOD mice, including the lack of TDP-43 C-terminal fragments in spinal cords of
mSOD mice, could be that brain TDP-43 inclusions are enriched in C-terminal fragments,
whereas spinal cord inclusions comprise mostly full length TDP-43; a suggestion first
made by Igaz and colleagues to account for related observations made using human ALS
tissues [8]. Another possibility is that biochemical changes are less prominent in the mice
than in human tissue and that it is more difficult to detect these changes, given the small
volume of tissue available for study in mice.
In brief, our findings suggest that the mutant SOD1 transgenic mice (G93A) do
recapitulate some pathological features, but not all of the biochemical hallmarks, of TDP-
43 pathology that have been observed in human ALS. These studies suggest that motor
neuron degeneration occurring in mutant SOD1 transgenic mice is associated with TDP-
43 histopathology. Given that TDP-43 pathology in G93A mSOD mice of advanced age
does recapitulate features observed in human ALS cases, this animal model may be a
valuable tool for studying the progression and pathophysiology of ALS and as a useful
model to test potential TDP-43-directed therapeutics.

96
3.6 Figures

97
Figure 3- 1 TDP-43 redistribution in end stage mSOD lumbar spinal cord ventral horn cells. mSOD mice and age-matched wild-type (WT) mice were double labelled with NeuN (green) and TDP-43 (red) antibodies. (a–f) At 12 weeks of age, TDP-43 remains localized in the nucleus (large arrow) of ventral horn cells and shows no distinct aggregates in either mSOD (a–c) or wild-type mice (d–f). (g–s) At end stage, TDP-43 aggregates and redistributes from the nucleus to the cytoplasm (small arrow) in some neurons observed in sections from mSOD mice (g–i). In some ventral horn motor neurons punctate nuclear and axonal staining is observed (m) and dystrophic neurite-like (*) and rounded structures (**) are also labelled (h, o and r). Many TDP-43 positive rounded structures colocalize with ubiquitin (Ubiq, green) (n–s). Mislocalized cytoplasmic TDP-43 is not observed inWT mice of the same age as end stagemSOD mice (j–l). Scale bars: a–l = 25 µm; m–s and inserts = 5 µm.

98
Figure 3- 2 TDP-43 protein levels in lumbar spinal cord do not change in end stagemSOD mice. (a–b) Immunoblots of total soluble protein from lumbar spinal cords using rabbit anti-TDP-43 show immunoreactive ~ 43 kDa bands. (a) No significant differences between end stagemSOD mice and age-matchedWTmicewere observed; (b) protein loading is equivalent based on immunoblots of lysates using an anti-actin monoclonal antibody; (c) Immunoblot of sequential extracts from lumbar spinal cords. No pathological species detected as ~ 45 or ~ 25 kDa bands are observed in tissues from mSOD mice (SA, sarkosyl; UA, urea).

99
3.7 Reference List
[1] T. Arai, M. Hasegawa, H. Akiyama, K. Ikeda, T. Nonaka, H. Mori, D. Mann, K. Tsuchiya, M. Yoshida, Y. Hashizume, T. Oda, TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Biochemical and biophysical research communications 351 (2006) 602-611.
[2] E. Buratti, T. Dork, E. Zuccato, F. Pagani, M. Romano, F.E. Baralle, Nuclear factor TDP-43 and SR proteins promote in vitro and in vivo CFTR exon 9 skipping, The EMBO journal 20 (2001) 1774-1784.
[3] Y. Fujita, Y. Mizuno, M. Takatama, K. Okamoto, Anterior horn cells with abnormal TDP-43 immunoreactivities show fragmentation of the Golgi apparatus in ALS, Journal of the neurological sciences 269 (2008) 30-34.
[4] F. Geser, M. Martinez-Lage, L.K. Kwong, V.M. Lee, J.Q. Trojanowski, Amyotrophic lateral sclerosis, frontotemporal dementia and beyond: the TDP-43 diseases, J Neurol (2009).
[5] M.A. Gitcho, R.H. Baloh, S. Chakraverty, K. Mayo, J.B. Norton, D. Levitch, K.J. Hatanpaa, C.L. White, 3rd, E.H. Bigio, R. Caselli, M. Baker, M.T. Al-Lozi, J.C. Morris, A. Pestronk, R. Rademakers, A.M. Goate, N.J. Cairns, TDP-43 A315T mutation in familial motor neuron disease, Ann Neurol 63 (2008) 535-538.
[6] M.E. Gurney, H. Pu, A.Y. Chiu, M.C. Dal Canto, C.Y. Polchow, D.D. Alexander, J. Caliendo, A. Hentati, Y.W. Kwon, H.X. Deng, et al., Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation, Science (New York, N.Y 264 (1994) 1772-1775.
[7] J.H. Hu, K. Chernoff, S. Pelech, C. Krieger, Protein kinase and protein phosphatase expression in the central nervous system of G93A mSOD over-expressing mice, Journal of neurochemistry 85 (2003) 422-431.
[8] L.M. Igaz, L.K. Kwong, Y. Xu, A.C. Truax, K. Uryu, M. Neumann, C.M. Clark, L.B. Elman, B.L. Miller, M. Grossman, L.F. McCluskey, J.Q. Trojanowski, V.M. Lee, Enrichment of C-terminal fragments in TAR DNA-binding protein-43 cytoplasmic inclusions in brain but not in spinal cord of frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Am J Pathol 173 (2008) 182-194.
[9] J.P. Julien, J. Kriz, Transgenic mouse models of amyotrophic lateral sclerosis, Biochimica et biophysica acta 1762 (2006) 1013-1024.
[10] E. Kabashi, P.N. Valdmanis, P. Dion, G.A. Rouleau, Oxidized/misfolded superoxide dismutase-1: the cause of all amyotrophic lateral sclerosis? Annals of neurology 62 (2007) 553-559.
[11] B.T. M. Kiaei, A. Neymotin, N. Y. Calingassan, E. G. Wille, F. Yin, A. Ding, M. F. Beal, Altered TDP-43 expression patterns in the spinal cord of SOD1 transgenic mice and PGRN knockout mice, . Soc. for Neurosci. Abstr., (2008)
[12] I.R. Mackenzie, E.H. Bigio, P.G. Ince, F. Geser, M. Neumann, N.J. Cairns, L.K. Kwong, M.S. Forman, J. Ravits, H. Stewart, A. Eisen, L. McClusky, H.A. Kretzschmar, C.M. Monoranu, J.R. Highley, J. Kirby, T. Siddique, P.J. Shaw, V.M. Lee, J.Q. Trojanowski, Pathological TDP-43 distinguishes sporadic amyotrophic lateral sclerosis from amyotrophic lateral sclerosis with SOD1 mutations, Ann Neurol 61 (2007) 427-434.

100
[13] I.R. Mackenzie, R. Rademakers, The role of transactive response DNA-binding protein-43 in amyotrophic lateral sclerosis and frontotemporal dementia, Current opinion in neurology 21 (2008) 693-700.
[14] P.A. Mercado, Y.M. Ayala, M. Romano, E. Buratti, F.E. Baralle, Depletion of TDP 43 overrides the need for exonic and intronic splicing enhancers in the human apoA-II gene, Nucleic acids research 33 (2005) 6000-6010.
[15] M. Neumann, D.M. Sampathu, L.K. Kwong, A.C. Truax, M.C. Micsenyi, T.T. Chou, J. Bruce, T. Schuck, M. Grossman, C.M. Clark, L.F. McCluskey, B.L. Miller, E. Masliah, I.R. Mackenzie, H. Feldman, W. Feiden, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Science 314 (2006) 130-133.
[16] Y. Nishihira, C.F. Tan, Y. Toyoshima, Y. Yonemochi, H. Kondo, T. Nakajima, H. Takahashi, Sporadic amyotrophic lateral sclerosis: Widespread multisystem degeneration with TDP-43 pathology in a patient after long-term survival on a respirator, Neuropathology (2009).
[17] S.H. Ou, F. Wu, D. Harrich, L.F. Garcia-Martinez, R.B. Gaynor, Cloning and characterization of a novel cellular protein, TDP-43, that binds to human immunodeficiency virus type 1 TAR DNA sequence motifs, Journal of virology 69 (1995) 3584-3596.
[18] J. Robertson, T. Sanelli, S. Xiao, W. Yang, P. Horne, R. Hammond, E.P. Pioro, M.J. Strong, Lack of TDP-43 abnormalities in mutant SOD1 transgenic mice shows disparity with ALS, Neurosci Lett 420 (2007) 128-132.
[19] J.D. Rothstein, TDP-43 in amyotrophic lateral sclerosis: pathophysiology or patho-babel?, Annals of neurology 61 (2007) 382-384.
[20] J. Sreedharan, I.P. Blair, V.B. Tripathi, X. Hu, C. Vance, B. Rogelj, S. Ackerley, J.C. Durnall, K.L. Williams, E. Buratti, F. Baralle, J. de Belleroche, J.D. Mitchell, P.N. Leigh, A. Al-Chalabi, C.C. Miller, G. Nicholson, C.E. Shaw, TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis, Science (New York, N.Y 319 (2008) 1668-1672.
[21] P.H. Tu, P. Raju, K.A. Robinson, M.E. Gurney, J.Q. Trojanowski, V.M. Lee, Transgenic mice carrying a human mutant superoxide dismutase transgene develop neuronal cytoskeletal pathology resembling human amyotrophic lateral sclerosis lesions, Proc Natl Acad Sci U S A 93 (1996) 3155-3160.
[22] B.J. Turner, D. Baumer, N.J. Parkinson, J. Scaber, O. Ansorge, K. Talbot, TDP-43 expression in mouse models of amyotrophic lateral sclerosis and spinal muscular atrophy, BMC Neurosci 9 (2008) 104.
[23] V.M. Van Deerlin, J.B. Leverenz, L.M. Bekris, T.D. Bird, W. Yuan, L.B. Elman, D. Clay, E.M. Wood, A.S. Chen-Plotkin, M. Martinez-Lage, E. Steinbart, L. McCluskey, M. Grossman, M. Neumann, I.L. Wu, W.S. Yang, R. Kalb, D.R. Galasko, T.J. Montine, J.Q. Trojanowski, V.M. Lee, G.D. Schellenberg, C.E. Yu, TARDBP mutations in amyotrophic lateral sclerosis with TDP-43 neuropathology: a genetic and histopathological analysis, Lancet Neurol 7 (2008) 409-416.
[24] M.J. Winton, V.M. Van Deerlin, L.K. Kwong, W. Yuan, E.M. Wood, C.E. Yu, G.D. Schellenberg, R. Rademakers, R. Caselli, A. Karydas, J.Q. Trojanowski,

101
B.L. Miller, V.M. Lee, A90V TDP-43 variant results in the aberrant localization of TDP-43 in vitro, FEBS Lett 582 (2008) 2252-2256.

102
4: Brain TDP-43 is modified with O-GlcNAc and the O-linked glycosylation sites map to the C-terminal region of recombinant human TDP-43
Manuscript in preparation.
Authors: Xiaoyang Shan, Tom Clark, Coral-Ann Lewis, Charles Krieger, and David Vocadlo.
Author contribution: X.S., C.K., and D.V. designed the experiments. X.S. performed all experiments except mass spectrometry analysis; T.C. performed mass spectrometry analysis including sample digestion. C.L. provided animals and performed tail vein injections. X.S., C.K. and D.V. analysed data. X.S. wrote the first draft with T.C., and C.K. and D.V. wrote the final manuscript.
4.1 Abstract
Cytoplasmic and intranuclear inclusions of TAR DNA-binding protein 43 (TDP-43) are
the defining neuropathological feature of several neurodegenerative diseases such as
amyotrophic lateral sclerosis (ALS) and frontotemporal degeneration with ubiquintinated
inclusions (FTLD-U). These diseases are characterized by the presence of cellular
aggregates composed of hyperphosphorylated TDP-43 in the spinal cord and brain.
However, it is unknown what causes TDP-43 hyperphosphorylation and how their
abnormal phosphorylation contributes to the pathogenesis of diseases. Here, we
demonstrate mouse brain TDP-43 is modified by another post-translational modification

103
known as O-GlcNAc. Moreover, using mass spectrometric analysis, we identify four O-
GlcNAc sites on serine residues (S292, S342, S389, S393) at the C-terminal region of
recombinant human TDP-43. These sites overlapped with previously identified sites that
are known to be phosphorylated by casein kinase-1 (CK1). Our results indicate that O-
GlcNAc modification on TDP-43 could influence its level of phosphorylation particularly
in the C-terminal region we identify as O-GlcNAc modified.
4.2 Introduction
TAR DNA-binding protein 43 (TDP-43) is a highly conserved nuclear protein that
belongs to the family of heterogeneous nuclear ribonucleoproteins (hnRNPs) [44].
Structurally similar to other hnRNP family members, TDP-43 contains two RNA
recognition motifs (RRM1 and RRM2) that bind double-stranded DNA, single-stranded
DNA, and RNA [4, 37], as well as a glycine-rich C-terminal domain that is involved in
protein-protein interactions [6]. TDP-43 has been reported to play multiple roles in gene
transcription, RNA splicing, RNA stability and RNA transport [5]. Recently, TDP-43
was identified as an important constituent protein found in the cytoplasmic ubiquitinated
inclusions in amyotrophic lateral sclerosis (ALS) as well as in other neurodegenerative
diseases such as frontotemporal degeneration with ubiquintinated inclusions (FTLD-U)
[1, 36]. ALS is a fatal disorder characterized by the selective degeneration of motor
neurons and descending motor tracts from brain and brainstem and most patients with
ALS have hyperphosphorylated TDP-43 inclusions in the cytoplasm of neurons and glia
[1, 36]. Recently, mutations in the gene coding for TDP-43, TARDBP, have been
identified in both the familial and sporadic forms of ALS [15, 20, 41] suggesting that
TDP-43 is involved in the pathogenesis of ALS. Interestingly, the majority of TARDBP

104
mutations identified in ALS patients localize to exon 6, which encodes the C-terminal
glycine-rich domain (amino acids 261 to 414) of TDP-43 [38].
The TDP-43 extracted from insoluble fractions of CNS tissue from patients with ALS is
hyperphosphorylated, and is disease-specific as hyperphosphorylated TDP-43 is not
detected in normal control CNS tissue [1, 18, 36]. In particular, serine residues 409 and
410 (S409/410) have been identified as major pathological phosphorylation sites on TDP-
43, and these residues are consistently phosphorylated in the TDP-43 inclusions in ALS
tissue, but not in control material [19, 35]. It has also been shown that phosphorylation
of TDP-43 at S409/410 enhances the toxicity of mutant TDP-43 in a C elegans model
[26]. The view that phosphorylation of TDP-43 is neurotoxic has also gained support
from recent studies showing that phosphorylation regulates the solubility or aggregation
of the C-terminal fragment of TDP-43 in several cell lines [3, 49]. Casein kinase-1 (CK1)
may be involved in the hyperphosphorylation of TDP-43 in ALS tissue [18, 19], as 29
Ser/Thr residues on recombinant human TDP-43 are known phosphorylation sites for
CK1 and 18 of these sites localize to the C-terminal glycine-rich domain, including
S409/410 [21]. Other types of post-translational modification of TDP-43 have not been
investigated extensively, except that a high-throughput proteomic studies using a
metabolic labelling approach reported that TDP-43 was a putative O-GlcNAc modified
proteins from HeLa cells and NIH-3T3 cells [34, 48]. However, other proteomic studies
using more sensitive chemoenzymatic tagging method coupled with mass spectrometry,
did not report O-GlcNAc modification found on TDP-43, although they shown a total of

105
18 O-GlcNAc protein from rat brain [22, 23]. Thus, whether or not TDP-43 is modified
with O-GlcNAc in cells or in vivo remains unknown.
A number of neuronal proteins are modified by O-GlcNAc including cytoskeletal
proteins such as neurofilaments [12, 13], synapsins [30], as well as some
neurodegenerative disease-associated proteins such as the microtubule-associated protein
tau (MAPT) [2]. The O-GlcNAc modification is a dynamic post-translational
modification involving attachment of N-acetyl-D-glucosamine (GlcNAc), via a -
glycosidic linkage, to the hydroxyl side chains of serine and threonine residues of nuclear
and cytoplasmic proteins [43]. O-GlcNAc is similar to serine and threonine
phosphorylation in that it can be added or removed several times during the lifespan of a
particular protein. In contrast to the hundreds of kinases and protein phosphatases that are
involved in regulating phosphorylation, only two enzymes regulate O-GlcNAc
modification levels. O-GlcNAc transferase (OGT) catalyzes the transfer of GlcNAc from
the donor sugar uridine 5'-diphospho-N-acetylglucosamine (UDP-GlcNAc) to target
proteins [24, 29] and a glycoside hydrolase known as O-GlcNAcase (OGA) removes the
sugar from modified proteins [11, 14].
O-GlcNAc has been proposed to influence many cellular processes in the CNS,
including: transcriptional regulation, signaling, proteasomal degradation, and axonal
trafficking [25]. Moreover, it has shown that O-GlcNAc levels are elevated in response to
various cellular stresses in several cell lines [47], thus elevated O-GlcNAc levels may
also play a protective role through currently unknown mechanisms, perhaps due in part to
increased levels of heat shock proteins [16]. O-GlcNAc modification of serine and

106
threonine residues have been found in some cases to be reciprocal with phosphorylation
on certain neuronal proteins, and is proposed to be involved in the pathogenesis of
neurodegenerative diseases [17]. For example, tau is abnormally hyperphosphorylated
and aggregated into neurofibrillary tanges (NFT) in the brains of patients who died
having Alzheimer’s Disease (AD) and the levels of O-GlcNAc-modified tau are lower in
AD brain [27, 28]. These observations raise the possibility that decreased O-GlcNAc-
modified tau may contribute to tau hyperphosphorylation. As TDP-43 is also
hyperphosphorylated in ALS, and as there might be reciprocal effects of phosphorylation
and O-GlcNAc modification of TDP-43 in the CNS, we undertook this work to determine
whether TDP-43 is a target for O-GlcNAc modification in vivo and to map potential O-
GlcNAc modification site(s) on recombinant TDP-43 protein using a method that has
been used to successfully map several O-GlcNAc sites on recombinant tau protein [46].
4.3 Methods
4.3.1 Administration of NButGT and animal tissue preparation
C57BL/6 mice (Jackson Laboratories, Bar Habour, ME) were given a 100 mg/kg tail vein
injection of the selective OGA inhibitor, NButGT [32]; after 7 hr, mice were sacrificed
using CO2. For immunoprecipitation experiments, brain tissue were collected and stored
in -80 C. For immunohistochemistry study, mice were perfused transcardially with 30
mL PBS (pH 7.4) followed by 30 mL 4% paraformaldehyde (PFA) in PBS, then brains
were removed, post-fixed in 4% PFA for 24 hr, and then transferred to 20% sucrose in
PBS overnight for cryoprotection. The brains were subsequently sectioned in the sagittal
plane at 30 m on a Leica cryostat (Leica, Wetzlar, Germany). The animal

107
experimentation protocol was approved by the university review committee at Simon
Fraser University and was in compliance with guidelines from the Canadian Council on
Animal Care.
4.3.2 Immunoprecipitation of TDP-43 from mouse brain
Mouse brains were homogenized on ice in lysis buffer containing 1 x PBS, 500 mM
NaCl, 1 mM NButGT, and Roche protease inhibitors in a 1:1 ratio of buffer to frozen
tissue weight. The tissue was homogenized using an IKA tissue homogenizer
(Wilmington, NC, USA) for 1 min at a power setting of 60%, and subsequently spun for
15 min at 13,000 rpm. The 500 L supernatant was added to 50 L Protein A/G agarose
beads to which 5 L of rabbit polyclonal anti-TDP-43 antibody had been pre-bound and
washed. The combined lysates and beads were rocked at 4 C for 2 hr, the beads were
then washed four times with lysis buffer, after which all the buffer was removed.
Immunoprecipitated protein was then eluted from the beads by boiling in 60 L of 1 x
SDS-PAGE loading buffer for 10 min. Immunoblot analyses of the immunoprecipitates
were then carried out as described below. Protein A/G agarose beads without pre-bound
rabbit polyclonal anti-TDP-43 antibody were used in parallel as experimental controls to
address non-specific binding of proteins to the beads.
4.3.3 Immunoblotting
Samples were electrophoresed through a 10 % SDS-PAGE and transferred to
nitrocellulose (Bio-Rad) membranes. Membranes were then blocked for 1 hr at room
temperature (RT) with 2 % BSA (BioShop, Burlington, ON, Canada) in PBS containing
0.1 % Tween-20 (Sigma) (PBS-T) and then probed overnight at 4 ºC with the appropriate

108
primary antibodies diluted in 2 % BSA in PBS-T. Following washing with PBS-T,
membranes were blocked again for 30 min at RT and then probed for 1 hr at RT with the
appropriate HRP conjugated secondary antibody diluted in 2 % BSA in PBS-T. After
extensive washing with PBS-T, the membranes were developed using ECL detection
(SuperSignal West Pico Chemiluminesence substrate, Pierce) in combination with film
(CL-XPosure Film, Pierce). For membranes pretreated with Bacteroides
thetaiotaomicron OGA (btOGA, a bacterial homologue of human OGA) [10], the
membranes were incubated following the first blocking step with 500 g btOGA in 50
mM Tris-HCl, pH 8.5, 0.1 mM EDTA, 0.5 mM MgCl2 for 3 hr at 37 ºC. The membranes
were then washed in PBS-T and the remaining immunoblot steps were followed as
described above.
4.3.4 Antibodies
Rabbit polyclonal anti-TDP-43, which recognizes the N-terminal 1-260 aa of TDP-43 in
a phosphorylation-independent manner, was purchased from ProteinTech (Chicago, IL,
USA). Mouse monoclonal anti-O-GlcNAc antibodies, CTD110.6 and RL2, were
purchased from Covance and Abcam, respectively. O-GlcNAc specific mouse
monoclonal antibody, 1F5.D6 (14), was a gift from Dr. Geert-Jan Boons (University of
Georgia, Athens, Georgia, USA). Secondary antibodies conjugated to HRP for were
obtained from Santa Cruz.

109
4.3.5 Molecular cloning
The full length human TDP-43 gene (TARDBP) purchased from Invitrogen (Clone ID
5498250, Accession# BC071657) was sub-cloned in pET28a expression vector
(Novagen) using the following primers: 5’-cctcagaagcttccatgtctgaatatattcgggtaacc-3’
(HindIII cut site shown in bold) and 5’-cgtcagctcgagttacattccccagccagaagac-3’ (XhoI cut
site shown in bold) to generate pET28aTDP-43.
4.3.6 Production of recombinant O-GlcNAc modified TDP-43
The gene encoding human TDP-43 in pET28a (pET28aTDP-43), was co-transformed
with the plasmid encoding wild-type OGT (wtOGT) or the H558A mutant of OGT
(mutOGT) [33] (pMal-c2XOGT or pMal-c2XH558AOGT), into E. coli BL21 Tuner cells
(Stratagene), and plated onto LB plates containing 50 g/mL kanamycin and 100 g/mL
ampicillin for selection of co-transformed bacteria. Once colonies were obtained, the co-
transformed bacteria were selected by growth in LB solution containing containing
containing 50 g/mL kanamycin and 100 g/mL ampicillin. To induce expression of
TDP-43, IPTG (0.5 mM) was added to bacteria culture in exponential phase (cultures had
an optical density (OD600) of ~ 0.8), and incubated for 3 hr at 22 C. The bacterial cells
were harvested by centrifugation at 5000 rpm for 10 min in a Sorvall RC-6 plus
centrifuge (Thermo Scientific). The bacterial pellets were then resuspended in 25 mL of
Ni-NTA column binding buffer (20 mM sodium phosphate, 500 mM NaCl, 5 mM
imidazole, pH 7.4). wtOGT/TDP-43 and mutOGT/TDP-43 co-transformed cell pellets
were lysed by the addition of 2 mg/mL lysozyme (BioShop) in the presence of one Roche
protease inhibitor tablet per 25 mL of resuspended bacterial pellet. Sonication was carried
out at ~30 % power on a Fischer Scientific sonic dismembrator (model 500) for six

110
cycles consisting of 20 sec on followed by a 40 sec rest period. Cellular debris was
removed by centrifugation (Sorvall RC-6 plus centrifuge, Thermo Scientific) of the
solution at 13,000 rpm for 1 hr at 4 C. The supernatants were then loaded onto two
separate HisTrap Ni-NTA columns (GE Healthcare) using two separate peristaltic pumps.
The columns were washed with 90 mL of Ni-NTA column wash buffer (20 mM sodium
phosphate, 500 mM NaCl, 60 mM imidazole, pH 7.4) and then protein was eluted using
25 mL of Ni-NTA column elution buffer (20 mM sodium phosphate, 500 mM NaCl, 250
mM imidazole, pH 7.4). The eluates were then dialyzed overnight against 4 L of PBS at 4
°C for immunoblot analysis and for subsequent mass spectrometry analysis.
4.3.7 Sample preparation for mass spectrometry analysis
In order to map O-GlcNAc site(s), the recombinant O-GlcNAc modified TDP-43 was
electrophoresed through 10% SDS-PAGE gels, and then stained with Coomassie Blue.
The same bands were cut from mulitple lanes in order to obtain sufficient material. Gel
pieces were further broken down into approximately 1 mm x 1 mm pieces using a glass
pipette. The gel pieces were evenly distributed in three separate eppendorf tubes. Each
tube underwent the same process in parallel but were exposed to different digestive
enzymes.
Gel pieces were covered with a solution of 50% 50 mM ammonium bicarbonate / 50%
ethanol for 20 min. The fluid was removed by pipette and then replaced with pure ethanol
and allowed to stand for 15 min. Ethanol was removed and reducing agent (10 mM
TCEP, Thermo Scientific) was added, after which the samples were immediately heated

111
to 56 °C in the dark for 1 hr. Alkylation of liberated cysteine residues was performed by
first making an alkylating solution by adding 0.010g of iodoacetamide with 500 L of
100 mM ammonium bicarbonate and 500 L of H2O (JT Baker, HPLC grade). The
reducing agent solution was then replaced with the alkylating agent solution and the
mixture was kept in the dark at room temperature for 30 min. Reduction and alkylation of
cysteines was completed by following two cycles of replacing the liquid with 50 mM
Ammonium Bicarbonate for 15 min each.
After this process, the liquid in the tube was replaced with a solution of 25 mM
ammonium bicarbonate / 5% acetonitrile:H2O for 30 min, then replaced again with 25
mM ammonium bicarbonate / 50% acetonitrile:H2O for 30 min, and finally with
acetonitrile for 10 min. All steps were carried out at RT. The resulting white gel pieces
were transferred equally into three tubes containing 5 g of each protease (previously
aliquotted and stored at -80 °C) immediately covered with a 50 mM ammonium
bicarbonate solution and incubatedat 37 °C for 18 hr. Proteases used were trypsin (Gold
mass spectrometry grade, Promega), endoproteinase Glu-C (sequencing grade, Roche)
and endoproteinase Lys-C (sequencing grade, Promega).
After the digestions, the supernatant was removed and formic acid was added to generate
a 0.1% final formic acid concentration. To the gel pieces, 0.1% trifluoroacetic acid / 50%
acetonitrile was added, sufficient to cover the gel pieces and the mixture was gently
vortexed for 10 min. This supernatant was removed and combined with the first

112
supernatant. Enough of 0.1% trifluoroacetic acid / 80% acetonitrile was finally added to
the gel pieces to cover the gel pieces (approximately 100uL) and the mixture was gently
vortexed for 10 min. This supernatant was removed and combined with the first two
supernatants. The combined supernatants wereconcentrated by vacuum centrifuge to less
to less than 5 L and then reconstituted in 0.1% formic acid to a volume of 30 L.
4.3.8 On-line dual liquid chromatography
A U3000 (Dionex) liquid chromatography system with a 1000:1 splitter used to produce
a 300 nL/min flow rate was combined with two external switching valves to enable dual
chromatography. The sample was injected onto two trap columns, C18 (Dionex, P/N
160454 ) and graphitized carbon (Packed in house with 3m particles removed from a
Hypercarb column (Thermo Scientific) in 200 m ID capillary (Idexs), washing the salts
to the waste at a flow rate of 4 L/min for 20 min. The sample on the C18 trap column
was passed by gradient elution over 45 min through a 14 cm analytical column (75 m
diameter in-house packed with 3 m C18 stationary phase (Dr. Maisch GmbH))
interfaced to the mass spectrometer. The sample on the graphitized carbon trap column
was also passed by gradient elution through an 11 cm graphitized carbon column (75 m
diameter packed in house with 3 m graphitous carbon (from a Hypercarb column,
Thermo Scientific)) to the mass spectrometer for 45min. Mobile phase A and the loading
solvent was 0.05% formic acid. Mobile phase B was 90% acetonitrile / 0.05% formic
acid. Additionally, mobile phase A was introduced at a constant flow rate of 50nL/min
by syringe pump just before the nano spray tip, in order to keep the tip wet while electro
spray voltage was applied.

113
4.3.9 Information dependant acquisition (IDA) mass spectrometry (MS)
Mass spectrometric data collection was performed using an API 4000 QTrap instrument
(AB Sciex) using a nano flow electro-spray ionization source fitted with a custom head
and nano spray tips that terminate with a diameter of 5 m (New Objective). Each sample
was digested using a different protease and analyzed independently. An information
dependant acquisition method was established in which the five largest chromatographic
peaks in excess of 100000 cps were further analyzed using an enhanced MS scan at 4000
amu/s over a range from 400-1400 amu. Ions detected in this way were further analyzed
at 250 amu/s using an enhanced resolution scan to more accurately determine the
precursor ion mass and charge state (only charge states 1 through 4 were analyzed). On
each cycle, a maximum of five corresponding MSMS spectra were collected using an
enhanced product ion scan at 1000 amu/s while using Q0 trapping Pressure near the
collision cell was maintained at 4.8 x 10-5 Torr using nitrogen gas. ‘Q0’ refers to the
collimating RF only rod set positioned near the MS interface. ‘Enhanced” refers to any
method whereby the linear ion trap is employed in place of the standard quadrapole.
Mascot Daemon v2.3 was used solely to convert the software files from Analyst 1.4.2
used to acquire data in *.mgf format, which was then analyzed using PEAKS client 5.2
licensed to the British Columbia Proteomics Network.

114
4.4 Results
4.4.1 O-GlcNAc modification of TDP-43 in mouse brains
As TDP-43 has been suggested as a putative O-GlcNAc protein in cell lines [34, 48], we
first determined whether TDP-43 extracted from mouse CNS is subject to O-GlcNAc
modification. Our previous work has shown that TDP-43 protein from mouse spinal cord
is detectable by immunoblot [39]. In this study, in order to obtain larger amounts of CNS
tissue to immunoprecipitate TDP-43 protein, mouse brain tissues were used. It has been
shown that under physiological conditions, the majority O-GlcNAc-modified neuronal
proteins have very low abundance in CNS tissue, and are difficult to detect by regular
biochemical methods [7]. Therefore, to increase the sensitivity of the detection methods
used, wild-type C57BL/6 mice were administrated NButGT, a selective inhibitor of OGA
that acts to increase CNS O-GlcNAc levels, as has been demonstrated in our previous
work [31]. The most commonly used anti-O-GlcNAc (CDT110.6) antibody is a mouse
IgM antibody, which is not suitable for immunoprecipitation, therefore, we used a rabbit
polyclonal anti-TDP-43 antibody for immunoprecipitation assays, which has been used
extensively to evaluate TDP-43 in various model systems [1, 36]. Immunoblot of control
mouse brain lysates were analyzed in parallel and demonstrated two bands having
immunoreactivity against TDP-43 at ~ 43-45 kDa (Figure 4-1A-B). The lower band
appeared to be dominant and the upper band was observed clearly only under longer
exposure conditions (Figure 4-1B). Following immunoprecipitation of TDP-43 a single
band was detected, corresponding to the dominant lower TDP-43 band observed in the
lysate at ~ 43 kDa, in addition to bands associated with the heavy and light chains of the
antibody (Figure 4-1A). Immunoblot using the anti-O-GlcNAc antibody, CTD110.6

115
revealed a single band corresponding to immunoprecipitated TDP-43, as opposed to the
multiple immunoreactive bands seen in the control lysate (Figure 4-1C). These results
suggest that TDP-43 is O-GlcNAc modified in mouse brain under conditions where
global O-GlcNAc levels are elevated. The specificity of the CTD110.6 antibody for O-
GlcNAc was further validated by pre-incubation of the antibody with 10 mM free
GlcNAc which abolishes the immunoreactivity of this antibody (Figure 4-1D), indicating
that the interaction of the CTD110.6 antibody with O-GlcNAc modified proteins in
mouse brains likely depends on recognition of O-GlcNAc.
4.4.2 O-GlcNAc modification of recombinant human TDP-43
With the observation that TDP-43 is modified by O-GlcNAc in mouse brain using
biochemical methods, we set out to map potential O-GlcNAc modification sites on TDP-
43. Given the difficulties in mapping O-GlcNAc modification sites by mass
spectrometry, as glycosidic bond between GlcNAc and serine or threonine residue is
readily cleaved during collision-induced dissociation[23], we took advantage of a
recently developed method which has been demonstrated to produce large amount O-
GlcNAc modified recombinant proteins, and has been used for successfully mapping
several O-GlcNAc sites [47]. This approach involves the recombinant co-expression of a
fusion protein of wtOGT linked to the maltose binding protein (MBP) and the target
protein bearing a hexahistidine (His6) tag in E.coli BL21 Tuner cells. During induction,
wtOGT can transfer O-GlcNAc onto the target proteins of interest, and the His6 tagged
target protein is then purified away from wtOGT by using a Ni-NTA column which
results in milligram quantities of recombinant O-GlcNAc modified target proteins [46].

116
Using the method described in Figure 4-2A-B, we expected to produce recombinant
TDP-43 that has a high stoichiometry of O-GlcNAc to protein. The full length human
TDP-43 construct was subcloned into a pET28a vector containing His6 tag, where the
His6 tag was linked to the N terminal end of TDP-43 (Figure 4-2A). In parallel, we also
expressed TDP-43 in the presence of a catalytically inactive mutOGT which cannot O-
GlcNAc modify target proteins [33] and thus serves as a negative control for the
recombinant O-GlcNAc modification of TDP-43. As shown by Coommassie Blue
staining (Figure 4-2C), equal amounts of proteins were shown to be expressed in the
presence of either wtOGT or mutOGT, and the major bands observed using Coommassie
Blue staining were indeed immunoreactive with an anti-TDP-43 antibody (Figure 4-2C).
In order to determine that the TDP-43 protein obtained was actually the O-GlcNAc
modified form, we used immunoblot analysis and found, as expected, that only the TDP-
43 expressed in the presence of wtOGT appeared to be modified when assayed using the
anti-O-GlcNAc antibody, CTD110.6 (Figure 4-2D), which recognizes a number of O-
GlcNAc modified proteins [8, 40]. Both digestion of the TDP-43 with BtOGA (Figure 4-
2D) and preincubation of the CTD110.6 antibody with free GlcNAc (Figure 4-2D),
resulted in a loss of immunoreactivity for this antibody, confirming the specificity of the
CTD110.6 antibody for O-GlcNAc. Taken together, these data show recombinant human
TDP-43 protein expressed in the presence of wtOGT is O-GlcNAc modified. We further
validated our finding using another two anti-O-GlcNAc antibodies (Figure 4-2E), RL2,
which is also commonly used for O-GlcNAc detection [40], and 1F5.D6(14) which has

117
been generated recently and has been shown to detect more than 200 mammalian O-
GlcNAc modified proteins [42].
In the immunoblot shown in Figure 4-2C, several TDP-43 bands are observed including
an upper band with relative molecular mass of ~ 50 kDa, a major band with relative
molecular mass of approximately 43 kDa, and few lower bands that most likely are
degradation fragments. Interestingly, only the upper band showed immunoreactivity with
all three anti-O-GlcNAc specific antibodies used (Figure 4-2D-E). This upper band likely
corresponds to full length recombinant TDP-43 bearing the His6 tag since the predicted
mass of this recombinant protein is 49.4 kDa.
4.4.3 O-GlcNAc modification sites map to the C-terminal region of TDP-43
In order to achieve the highest possible protein coverage we aimed to increase the
number of peptides that are amenable for MS analysis. To accomplish this aim we used
three different proteases to prepare samples for mapping. The highest coverage resulted
from Trypsin digestion. Using O-GlcNAc modified TDP-43 expressed in the presence of
wtOGT we were able to map four O-GlcNAc modification sites by Information
Dependant Acquisition (IDA) MS as summarized in Table 4-1. The IDA approach led to
the identification and sequencing of an O-GlcNAc modified peptide by monitoring the
fragment ions resulting from collision induced fragmentation of a doubly charged
precursor ion of mass 652.8.
The first site was mapped and confirmed by analysis of three individual peptides as
shown in the Table 4-1. One peptide was determined to be 280-

118
PGGFGNQGGFGNSgRGGGAGLGNNdQ-303, where g stands for the site of
glycosylation and d stands for a site of deamidation (Table 4-1). This peptide spans Pro-
280 to Gln-303 of TDP-43 in which Ser-292 is the modification site. The second site was
mapped to Ser342 from one peptide, and its sequence was determined to be 334-
WGMMGMMGMLASg-342 (Table 4-1). The third site was mapped to Ser389 from one
peptide, and its sequence was determined to be 385-WGSASgNdAG-392 (Table 4-1).
This site was further confirmed from the digest using Lys-C, and the resulting sequence
was determined to be 385-WGSASgNdAGSGSGFNG-399 (Table 4-1). The last site was
mapped to Ser393, and its sequence was determined to be 385-WGSASNdAGSg-393
(Table 4-1).
4.5 Discussion
We have shown for the first time that TDP-43 is post-translationally modified with O-
GlcNAc in mouse brain tissue and that recombinant human TDP-43 is O-GlcNAc
modified when co-expressed with wtOGT in vitro. Site mapping of TDP-43 indicates
that at least four O-GlcNAc modification are found within the C-terminal region of O-
GlcNAc-modified recombinant human TDP-43 protein. Notably, all four O-GlcNAc sites
we mapped are on the same residues, serines 292, 342, 389, and 393 (S292, S342, S389,
S393) that are sites of phosphorylation by CK1 [21]. This observation suggests that O-
GlcNAc modification might regulate the normal function of TDP-43 or that it could
influence the abnormal phosphorylation of TDP-43 in ALS and other neurodegenerative
diseases [1, 18, 36]. This effect on phosphorylation could either arise via a reciprocal
relation with phosphorylation on the same modification site, or through as yet
unidentified mechanisms.

119
In addition to TDP-43 O-GlcNAc modification sites we report in this study, some of the
proteins known to be associated with neurodegenerative diseases are also modified by O-
GlcNAc, such as neurofilaments [13] and tau [2]. While it is speculated that O-GlcNAc
plays a role in the pathogenesis of neurodegenerative diseases, its exact function in this
role remains unknown. What is clear now is that O-GlcNAc can influence the
phosphorylation status of these neuronal proteins [9, 27]. Furthermore, studies in which
O-GlcNAc levels have been increased using highly selective OGA inhibitors, show a
decrease in endogenous tau phosphorylation at several physiologically relevant sites, and
elevation of O-GlcNAc by an OGA inhibitor might prevent tau hyperphosphorylation and
perhaps the formation of NFT [45].
Pathological TDP-43 is also hyperphosphorylated and aggregates into insoluble
ubiquitined intracellular inclusions [1, 18, 36] Given the similarity between TDP-43
pathology in ALS and tau protein in AD, one would speculate that TDP-43 O-GlcNAc
and phosphorylation may also be reciprocally regulated as discussed above. This view
gains some support though our mass spectrometry findings that four O-GlcNAc sites on
serine residues of TDP-43 overlap with previously reported phosphorylation sites [21].
Thus, our findings suggest a possibility that decreased O-GlcNAc modification on certain
sites of TDP-43 might enable hyperphosphorylation of TDP-43 on the same or adjacent
residues. Therefore, it is a possibility that decreased O-GlcNAc modification on TDP-43
might lead to its aggregation at disease condition.

120
We note that one observation of interest is that only the upper band shows strong O-
GlcNAc immunoreactivity. This band likely corresponds to the recombinant full length
TDP-43 as cloned with the His6 tag (Figure 4-1A). This upper band likely contains the
entire C-terminal region of TDP-43, whereas the lower band that still has the His6 tag
likely lacks the C-terminal region, suggesting potential O-GlcNAc modification sites
might cluster within the glycine-rich C-terminal region. Our mass spectrometry data,
demonstrating that all mapped sites are localized within the C-terminal region, strongly
support this notion. This C-terminal region has been implicated in protein-protein
interactions required for TDP-43 dependent RNA processing [6]. More interestingly, the
majority of phosphorylation sites and mutations found on TDP-43 are also found in this
region [21, 38]. Thus, our observation further suggests that O-GlcNAc could play role in
regulating TDP-43 function and might be involved in the pathogenesis of TDP-43
pathology in ALS and perhaps other neurodegenerative diseases. Further investigations
will directly evaluate the roles of O-GlcNAc in TDP-43 major functions such as RNA
stabilization and transportation, as well as in disease associated conditions involving
hyperphosphorylation and known human TDP-43 disease related mutations.

121
4.6 Figures and Table
Figure 4- 1 Mouse brain TDP-43 is O-GlcNAc modified.
(A) Immunoprecipotated TDP-43 from mouse brains. Immunoblotting of tissue lysate and the immunoprecipitated TDP-43 were probed with a rabbit anti-TDP-43 antibody (short exposure). (B) The same blot in A reveals the double bands of TDP-43 in tissue lysates when using a long exposure time. (C) Immunoblotting using anti-O-GlcNAc CTD110.6 antibody revealed the precipitated TDP-43 is O-GlcNAc modified. (D) O-GlcNAc immunoreactive specificity was supported by pre-incubation of CTD110.0 antibody with 10 mM GlcNAc resulting in complete loss of immunoreactivity.

122
Figure 4- 2 Production of recombinant O-GlcNAc modified TDP-43.
(A) Recombinant human full length TDP-43 sequence showing the His6 tag linked to the N-terminus. (B) Schematic figure of the production of O-GlcNAc modified TDP-43. The plasmids encoding TDP-43 with His6 tag and OGT with MBP tag were co-transformationed into E. coli bacteria cells, induced by IPTG, then the O-GlcNAc modified TDP-43 was purified by Ni-NTA chromatography. (C) Commassie blue staining indicated equal purified protein loaded on the gel, and equal TDP-43 loading was demonstrated by probing with the anti-TDP-43 antibody. (D-E) Immunoblots using anti-O-GlcNAc CTD110.6, RL2 and 1F5.D6 (14) antibodies demonstrate that recombinant human TDP-43 is O-GlcNAc modified only when co-expressed with wtOGT.

123
Sample (Run #) Site Modified by
O‐GlcNAc p‐value Sequence
Trypsin (1) S292 8.01E‐08 PGGFGNQdGGFGNSgRGGGAGLGNNdQ
Trypsin (2) S292 4.15E‐05 GGNPGGFGNdQGGFGNdSgR
Trypsin (2) S292 8.01E‐08 PGGFGNQdGGFGNSgRGGGAGLGNNdQ
Trypsin (2) S342 9.55E‐08 WGMMGMMGMLASg
Trypsin (1) S389 2.91E‐05 WGSASgNdAG
Lys‐C S389 5.90E‐06 WGSASgNdAGSGSGFNG
Trypsin (2) S393 7.61E‐06 WGSASNdAGSg
g = O‐GlcNAc d = deamidation
Table 4- 1 O-GlcNAc modification sites mapped from digestion of recombinant human TDP-43.

124
4.7 Reference List
[1] T. Arai, M. Hasegawa, H. Akiyama, K. Ikeda, T. Nonaka, H. Mori, D. Mann, K. Tsuchiya, M. Yoshida, Y. Hashizume, T. Oda, TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Biochem Biophys Res Commun 351 (2006) 602-611.
[2] C.S. Arnold, G.V. Johnson, R.N. Cole, D.L. Dong, M. Lee, G.W. Hart, The microtubule-associated protein tau is extensively modified with O-linked N-acetylglucosamine, The Journal of biological chemistry 271 (1996) 28741-28744.
[3] O.A. Brady, P. Meng, Y. Zheng, Y. Mao, F. Hu, Regulation of TDP-43 aggregation by phosphorylation and p62/SQSTM1, Journal of neurochemistry 116 (2011) 248-259.
[4] E. Buratti, F.E. Baralle, Characterization and functional implications of the RNA binding properties of nuclear factor TDP-43, a novel splicing regulator of CFTR exon 9, The Journal of biological chemistry 276 (2001) 36337-36343.
[5] E. Buratti, F.E. Baralle, The multiple roles of TDP-43 in pre-mRNA processing and gene expression regulation, RNA biology 7 (2010) 420-429.
[6] E. Buratti, A. Brindisi, M. Giombi, S. Tisminetzky, Y.M. Ayala, F.E. Baralle, TDP-43 binds heterogeneous nuclear ribonucleoprotein A/B through its C-terminal tail: an important region for the inhibition of cystic fibrosis transmembrane conductance regulator exon 9 splicing, The Journal of biological chemistry 280 (2005) 37572-37584.
[7] R.N. Cole, G.W. Hart, Cytosolic O-glycosylation is abundant in nerve terminals, J Neurochem 79 (2001) 1080-1089.
[8] F.I. Comer, K. Vosseller, L. Wells, M.A. Accavitti, G.W. Hart, Characterization of a mouse monoclonal antibody specific for O-linked N-acetylglucosamine, Analytical biochemistry 293 (2001) 169-177.
[9] Y. Deng, B. Li, F. Liu, K. Iqbal, I. Grundke-Iqbal, R. Brandt, C.X. Gong, Regulation between O-GlcNAcylation and phosphorylation of neurofilament-M and their dysregulation in Alzheimer disease, FASEB J 22 (2008) 138-145.
[10] R.J. Dennis, E.J. Taylor, M.S. Macauley, K.A. Stubbs, J.P. Turkenburg, S.J. Hart, G.N. Black, D.J. Vocadlo, G.J. Davies, Structure and mechanism of a bacterial beta-glucosaminidase having O-GlcNAcase activity, Nature structural & molecular biology 13 (2006) 365-371.
[11] D.L. Dong, G.W. Hart, Purification and characterization of an O-GlcNAc selective N-acetyl-beta-D-glucosaminidase from rat spleen cytosol, J Biol Chem 269 (1994) 19321-19330.
[12] D.L. Dong, Z.S. Xu, M.R. Chevrier, R.J. Cotter, D.W. Cleveland, G.W. Hart, Glycosylation of mammalian neurofilaments. Localization of multiple O-linked N-acetylglucosamine moieties on neurofilament polypeptides L and M, The Journal of biological chemistry 268 (1993) 16679-16687.
[13] D.L. Dong, Z.S. Xu, G.W. Hart, D.W. Cleveland, Cytoplasmic O-GlcNAc modification of the head domain and the KSP repeat motif of the neurofilament protein neurofilament-H, The Journal of biological chemistry 271 (1996) 20845-20852.

125
[14] Y. Gao, L. Wells, F.I. Comer, G.J. Parker, G.W. Hart, Dynamic O-glycosylation of nuclear and cytosolic proteins: cloning and characterization of a neutral, cytosolic beta-N-acetylglucosaminidase from human brain, J Biol Chem 276 (2001) 9838-9845.
[15] M.A. Gitcho, R.H. Baloh, S. Chakraverty, K. Mayo, J.B. Norton, D. Levitch, K.J. Hatanpaa, C.L. White, 3rd, E.H. Bigio, R. Caselli, M. Baker, M.T. Al-Lozi, J.C. Morris, A. Pestronk, R. Rademakers, A.M. Goate, N.J. Cairns, TDP-43 A315T mutation in familial motor neuron disease, Ann Neurol 63 (2008) 535-538.
[16] C. Guinez, A.M. Mir, Y. Leroy, R. Cacan, J.C. Michalski, T. Lefebvre, Hsp70-GlcNAc-binding activity is released by stress, proteasome inhibition, and protein misfolding, Biochem Biophys Res Commun 361 (2007) 414-420.
[17] G.W. Hart, C. Slawson, G. Ramirez-Correa, O. Lagerlof, Cross Talk Between O-GlcNAcylation and Phosphorylation: Roles in Signaling, Transcription, and Chronic Disease, Annu Rev Biochem (2011).
[18] M. Hasegawa, T. Arai, T. Nonaka, F. Kametani, M. Yoshida, Y. Hashizume, T.G. Beach, E. Buratti, F. Baralle, M. Morita, I. Nakano, T. Oda, K. Tsuchiya, H. Akiyama, Phosphorylated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Ann Neurol 64 (2008) 60-70.
[19] Y. Inukai, T. Nonaka, T. Arai, M. Yoshida, Y. Hashizume, T.G. Beach, E. Buratti, F.E. Baralle, H. Akiyama, S. Hisanaga, M. Hasegawa, Abnormal phosphorylation of Ser409/410 of TDP-43 in FTLD-U and ALS, FEBS letters 582 (2008) 2899-2904.
[20] E. Kabashi, P.N. Valdmanis, P. Dion, D. Spiegelman, B.J. McConkey, C. Vande Velde, J.P. Bouchard, L. Lacomblez, K. Pochigaeva, F. Salachas, P.F. Pradat, W. Camu, V. Meininger, N. Dupre, G.A. Rouleau, TARDBP mutations in individuals with sporadic and familial amyotrophic lateral sclerosis, Nat Genet 40 (2008) 572-574.
[21] F. Kametani, T. Nonaka, T. Suzuki, T. Arai, N. Dohmae, H. Akiyama, M. Hasegawa, Identification of casein kinase-1 phosphorylation sites on TDP-43, Biochem Biophys Res Commun 382 (2009) 405-409.
[22] N. Khidekel, S.B. Ficarro, P.M. Clark, M.C. Bryan, D.L. Swaney, J.E. Rexach, Y.E. Sun, J.J. Coon, E.C. Peters, L.C. Hsieh-Wilson, Probing the dynamics of O-GlcNAc glycosylation in the brain using quantitative proteomics, Nat Chem Biol 3 (2007) 339-348.
[23] N. Khidekel, S.B. Ficarro, E.C. Peters, L.C. Hsieh-Wilson, Exploring the O-GlcNAc proteome: direct identification of O-GlcNAc-modified proteins from the brain, Proc Natl Acad Sci U S A 101 (2004) 13132-13137.
[24] L.K. Kreppel, M.A. Blomberg, G.W. Hart, Dynamic glycosylation of nuclear and cytosolic proteins. Cloning and characterization of a unique O-GlcNAc transferase with multiple tetratricopeptide repeats, J Biol Chem 272 (1997) 9308-9315.
[25] B.D. Lazarus, D.C. Love, J.A. Hanover, O-GlcNAc cycling: implications for neurodegenerative disorders, The international journal of biochemistry & cell biology 41 (2009) 2134-2146.

126
[26] N.F. Liachko, C.R. Guthrie, B.C. Kraemer, Phosphorylation promotes neurotoxicity in a Caenorhabditis elegans model of TDP-43 proteinopathy, J Neurosci 30 16208-16219.
[27] F. Liu, K. Iqbal, I. Grundke-Iqbal, G.W. Hart, C.X. Gong, O-GlcNAcylation regulates phosphorylation of tau: a mechanism involved in Alzheimer's disease, Proc Natl Acad Sci U S A 101 (2004) 10804-10809.
[28] F. Liu, J. Shi, H. Tanimukai, J. Gu, J. Gu, I. Grundke-Iqbal, K. Iqbal, C.X. Gong, Reduced O-GlcNAcylation links lower brain glucose metabolism and tau pathology in Alzheimer's disease, Brain 132 (2009) 1820-1832.
[29] W.A. Lubas, D.W. Frank, M. Krause, J.A. Hanover, O-Linked GlcNAc transferase is a conserved nucleocytoplasmic protein containing tetratricopeptide repeats, J Biol Chem 272 (1997) 9316-9324.
[30] T. Luthi, R.S. Haltiwanger, P. Greengard, M. Bahler, Synapsins contain O-linked N-acetylglucosamine, J Neurochem 56 (1991) 1493-1498.
[31] M.S. Macauley, X. Shan, S.A. Yuzwa, T.M. Gloster, D.J. Vocadlo, Elevation of Global O-GlcNAc in rodents using a selective O-GlcNAcase inhibitor does not cause insulin resistance or perturb glucohomeostasis, Chemistry & biology 17 (2010) 949-958.
[32] M.S. Macauley, G.E. Whitworth, A.W. Debowski, D. Chin, D.J. Vocadlo, O-GlcNAcase uses substrate-assisted catalysis: kinetic analysis and development of highly selective mechanism-inspired inhibitors, J Biol Chem 280 (2005) 25313-25322.
[33] C. Martinez-Fleites, M.S. Macauley, Y. He, D.L. Shen, D.J. Vocadlo, G.J. Davies, Structure of an O-GlcNAc transferase homolog provides insight into intracellular glycosylation, Nat Struct Mol Biol 15 (2008) 764-765.
[34] A. Nandi, R. Sprung, D.K. Barma, Y. Zhao, S.C. Kim, J.R. Falck, Y. Zhao, Global identification of O-GlcNAc-modified proteins, Analytical chemistry 78 (2006) 452-458.
[35] M. Neumann, L.K. Kwong, E.B. Lee, E. Kremmer, A. Flatley, Y. Xu, M.S. Forman, D. Troost, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Phosphorylation of S409/410 of TDP-43 is a consistent feature in all sporadic and familial forms of TDP-43 proteinopathies, Acta neuropathologica 117 (2009) 137-149.
[36] M. Neumann, D.M. Sampathu, L.K. Kwong, A.C. Truax, M.C. Micsenyi, T.T. Chou, J. Bruce, T. Schuck, M. Grossman, C.M. Clark, L.F. McCluskey, B.L. Miller, E. Masliah, I.R. Mackenzie, H. Feldman, W. Feiden, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Science (New York, N.Y) 314 (2006) 130-133.
[37] S.H. Ou, F. Wu, D. Harrich, L.F. Garcia-Martinez, R.B. Gaynor, Cloning and characterization of a novel cellular protein, TDP-43, that binds to human immunodeficiency virus type 1 TAR DNA sequence motifs, J Virol 69 (1995) 3584-3596.
[38] G.S. Pesiridis, V.M. Lee, J.Q. Trojanowski, Mutations in TDP-43 link glycine-rich domain functions to amyotrophic lateral sclerosis, Human molecular genetics 18 (2009) R156-162.

127
[39] X. Shan, D. Vocadlo, C. Krieger, Mislocalization of TDP-43 in the G93A mutant SOD1 transgenic mouse model of ALS, Neuroscience letters 458 (2009) 70-74.
[40] C.M. Snow, A. Senior, L. Gerace, Monoclonal antibodies identify a group of nuclear pore complex glycoproteins, J Cell Biol 104 (1987) 1143-1156.
[41] J. Sreedharan, I.P. Blair, V.B. Tripathi, X. Hu, C. Vance, B. Rogelj, S. Ackerley, J.C. Durnall, K.L. Williams, E. Buratti, F. Baralle, J. de Belleroche, J.D. Mitchell, P.N. Leigh, A. Al-Chalabi, C.C. Miller, G. Nicholson, C.E. Shaw, TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis, Science 319 (2008) 1668-1672.
[42] C.F. Teo, S. Ingale, M.A. Wolfert, G.A. Elsayed, L.G. Not, J.C. Chatham, L. Wells, G.J. Boons, Glycopeptide-specific monoclonal antibodies suggest new roles for O-GlcNAc, Nat Chem Biol 6 (2010) 338-343.
[43] C.R. Torres, G.W. Hart, Topography and polypeptide distribution of terminal N-acetylglucosamine residues on the surfaces of intact lymphocytes. Evidence for O-linked GlcNAc, J Biol Chem 259 (1984) 3308-3317.
[44] H.Y. Wang, I.F. Wang, J. Bose, C.K. Shen, Structural diversity and functional implications of the eukaryotic TDP gene family, Genomics 83 (2004) 130-139.
[45] S.A. Yuzwa, M.S. Macauley, J.E. Heinonen, X. Shan, R.J. Dennis, Y. He, G.E. Whitworth, K.A. Stubbs, E.J. McEachern, G.J. Davies, D.J. Vocadlo, A potent mechanism-inspired O-GlcNAcase inhibitor that blocks phosphorylation of tau in vivo, Nat Chem Biol 4 (2008) 483-490.
[46] S.A. Yuzwa, A.K. Yadav, Y. Skorobogatko, T. Clark, K. Vosseller, D.J. Vocadlo, Mapping O-GlcNAc modification sites on tau and generation of a site-specific O-GlcNAc tau antibody, Amino acids 40 857-868.
[47] N.E. Zachara, N. O'Donnell, W.D. Cheung, J.J. Mercer, J.D. Marth, G.W. Hart, Dynamic O-GlcNAc modification of nucleocytoplasmic proteins in response to stress. A survival response of mammalian cells, J Biol Chem 279 (2004) 30133-30142.
[48] B.W. Zaro, Y.Y. Yang, H.C. Hang, M.R. Pratt, Chemical reporters for fluorescent detection and identification of O-GlcNAc-modified proteins reveal glycosylation of the ubiquitin ligase NEDD4-1, Proc Natl Acad Sci U S A 108 (2011) 8146-8151.
[49] Y.J. Zhang, T.F. Gendron, Y.F. Xu, L.W. Ko, S.H. Yen, L. Petrucelli, Phosphorylation regulates proteasomal-mediated degradation and solubility of TAR DNA binding protein-43 C-terminal fragments, Molecular neurodegeneration 5 33.

128
5: O-GlcNAc stabilizes tau againsts aggregation and slows neurodegeneration in an AD mouse model
Manuscript in preparation.
Authors: Scott A. Yuzwa (1), Xiaoyang Shan (1), Matthew S. Macauley, Yulia
Skorobogatko, Keith Vosseller, and David J. Vocadlo.
(1) Contributed equally to this work
Author contribution: D.J.V. directed the study. S.A.Y., X.S., M.S.M. and D.J.V., designed the experiments. X.S. conducted the rotarod assessment and all of the JNPL3 immunohistochemistry. S.A.Y. conducted the cage hang test and the JNPL3 tau biochemistry and the tau in vitro aggregation experiments. M.S.M. conducted the sTAB1 experiments. Y.S. and K.V. conducted the tau mass spectrometry. S.A.Y., D.J.V., and X.S., wrote the manuscript.
5.1 Abstract
The aggregation of the microtubule-associated protein tau to form paired helical
filaments (PHFs) and larger neurofibrillary tangles (NFTs) is a key pathological process
contributing to the progressive death of neurons in Alzheimer disease (AD) and related
tauopathies. Recently, serine and threonine residues of tau have been shown to compete
for modification by phosphate and N-acetylglucosamine (O-GlcNAc) groups. O-GlcNAc
is derived from cellular glucose pools, thus observations that impaired glucose transport
in AD brains correlates with enhanced tau aggregation may stem at the molecular level
from decreased tau O-GlcNAc levels enabling its inappropriate hyperphosphorylation.

129
Here we find that treating hemizygous JNPL3 mice, which harbor the human FTDP-17
P301L tau transgene, with an OGA inhibitor over a period of months increased tau O-
GlcNAc, hindered tau aggregation to form NFTs, and protected mice against neuronal
cell loss and consequent weight loss. Surprisingly, though OGA inhibition increased O-
GlcNAc on both soluble and the proposed pathological tau species, the extent of tau
phosphorylation was indistinguishable between treated and control animals. Using
biochemical assays we find that O-GlcNAc modification of tau, on its own, blocks tau
aggregation in vitro. These findings suggest a basic biochemical function of O-GlcNAc is
to promote stability of proteins against aggregation, which could contribute to the
protective effects of increased O-GlcNAc. These results also provide strong support for
OGA and O-GlcNAc processing as viable therapeutic targets that might offer the
opportunity to alter disease progression in AD and offer benefit in other disease states
involving aggregation of O-GlcNAc modified proteins.
5.2 Introduction
The looming personal and societal burdens associated with the increasing incidence of
Alzheimer disease (AD) within the aging population has spurred considerable interest in
both the basic biological processes contributing to AD, as well as in potential targets that
could be exploited to develop approaches to slow disease progression. The aggregation of
the microtubule-associated protein tau is receiving increasing attention as a key
pathological process contributing to the progressive death of neurons in AD and
associated tauopathies. Consistent with tau playing a key role in neurodegeneneration, the
number of NFTs within human brain correlate closely with disease severity[2]. The
discovery that point mutations in the MAPT gene are associated with the

130
neurodegenerative disorder known as frontotemporal dementia with parkinsonism linked
to chromosome 17 (FTDP-17) [17] reveals that tau malfunction, on its own, can drive
neurodegeneration both in humans and in animal models harboring FTDP-17 mutant
transgenes [22, 35]. These mutations lead to abnormal aggregation of tau to form paired
helical filaments (PHFs) that subsequently assemble to form neurofibrillary tangles [13,
41] (NFTs). NFTs are a key pathological hallmark of AD and the development of animal
models harboring FTDP-17 mutant transgenes has enabled studies to probe the cascade of
events leading their formation.
Two early molecular events have been proposed to be critical for the aggregation of tau.
The hyperphosphorylation of tau appears to be an essential step, which then enables
adoption of a conformation of tau that favors its subsequent aggregation[43].
Accordingly, efforts to block tau phosphorylation have been a topic of intense interest as
a means to alter the progression of AD [16]. Tau, however, is also subject to many other
post-translational modifications, a number of which have also been suggested to be
involved in its pathological aggregation [15]. Bovine tau has been shown to be
extensively modified by N-acetylglucosamine residues linked to serine and threonine
residues (O-GlcNAc) [1] and this modification has been more recently found to decorate
human and rat tau [23, 42]. O-GlcNAc is installed by an enzyme known as uridine
diphosphate-N-acetyl-D-glucosamine: polypeptidyl transferase (OGT) using the donor
sugar uridine 5'-diphospho-N-acetylglucosamine (UDP-GlcNAc) [19, 26], which is
derived from glucose via the hexosamine biosynthetic pathway (HBP) [27]. The removal
of O-GlcNAc from proteins is mediated by a glycoside hydrolase known as O-

131
GlcNAcase [8, 10] (OGA). The cycling of O-GlcNAc by these two enzymes can occur
more quickly than the lifetime of the protein backbone, which makes O-GlcNAc a
reversible post-translational modification [33]. Interestingly, O-GlcNAc and
serine/threonine phosphorylation have been found to compete at certain sites of
modification on various proteins including tau, where O-GlcNAc has been shown to
reduce phosphorylation at pathologically relevant sites in culture [21], ex vivo [23], and
in vivo in healthy rodents [45]
Recently, it has been shown that O-GlcNAc levels on tau decrease in response to
decreased glucose availability and this correlates with a concomitant increase in tau
phosphorylation [24, 29]. The observation that impaired glucose metabolism is an early
hallmark of Alzheimer disease [36], that NFTs appear to lack O-GlcNAc [23], and the
observed reciprocal relationship between O-GlcNAc and phosphorylation on tau [23],
suggests that impaired O-GlcNAc modification could contribute to the pathological
aggregation of tau in AD. Consistent with this view, genome-wide association (GWAS)
studies and immunoblot analysis of samples from AD patients has linked decreased levels
of glucose transporters to AD pathology [25, 37]. Furthermore, cellular stresses
contribute to the pathological hyperphosphorylation of tau [12, 18, 30] but are also
known to increase O-GlcNAc levels [47]. Although the molecular rationale for increased
O-GlcNAc levels in response to cellular stress remains unknown it seems possible that O-
GlcNAc is an adaptive protective response that could limit pathological protein
phosphorylation under such conditions. Accordingly, O-GlcNAc may serve a protective
function in the healthy mammalian brain by limiting tau hyperphosphorylation and the

132
lower levels of O-GlcNAc observed in the AD brain, and within NFTs, could reflect a
failure of this protective mechanism.
Based on these considerations we set out to test the hypothesis that increased tau O-
GlcNAc levels offer protective benefit by hindering tau hyperphosphorylation and/or its
subsequent aggregation to form NFTs. Here we use a pharmacological approach to
increase O-GlcNAc levels in the JNPL3 transgenic mouse model of AD, which harbors
the human FTDP-17 P301L tau transgene. In a long term study we find that increased O-
GlcNAc hinders tau aggregation and protects against neuronal cell loss. We further
discover that O-GlcNAc modification of tau, on its own, blocks tau aggregation in vitro,
providing strong support for O-GlcNAc processing as a viable therapeutic target that
might offer the opportunity to alter disease progression in AD. These findings suggest a
basic protective function of O-GlcNAc is to promote stability of proteins against
aggregation.
5.3 Methods
5.3.1 Animal dosing, handling, and motor testing
Forty 9 -12 week old hemizygous female JNPL3 mice (TgN(MAPT)JNPL3HlmcFemale)
were obtained from Taconic farms. All animal studies, described below, were approved
by the SFU University Animal Care Committee. Animals were divided into two groups
each containing 20 animals, co-housed in groups of five in each cage, and allowed to
acclimatize for one week prior to beginning the dosing regiment. The Thiamet-G treated
group was dosed by including 3.75 mg / mL Thiamet-G in the drinking water bottles.
This concentration was based on an average animal weight of 30 g and an average water

133
consumption of 4 mL / day / animal. Both groups were allowed ad libitum access to food
and water. Animals were dosed with Thiamet-G for 36 weeks. Animals were monitored
daily and assessed for motor impairment once per week by an investigator blinded to the
study groups. Absence of escape extension during tail elevation and spontaneous back
paw clenching during standing were used to assess each animal’s level of motor
impairment. When an animal displayed any of the aforementioned motor symptoms the
animal was classified as having a motor phenotypic stage of one. When significant
reductions in weight in addition to the above mentioned motor symptoms were detected
the animal was classified as having a motor phenotypic stage of two. Finally, when an
animal was unable to complete a wire cage hang (described below) or showed signs of
being unable to right itself the animal was sacrificed and also given a motor phenotypic
stage of two.
The wire cage hang test was conducted by allowing the animal to grasp a wire-cage lid,
inverting the cage lid over an empty cage (about 30 cm in height) and timing how long
the animal could remain hanging. A maximum length of the test was set at 60 sec at
which point the animal was removed from the wire-cage lid. Wire cage hang data was
collected unblinded to the investigators. Data was collected by averaging two trials per
animal.
5.3.2 Rotarod methods
The rotarod test was performed to evaluate motor function and symptoms of possible
motor neuron degeneration in both Thiamet-G treated and control JNPL3 mice. All mice
were pre-trained twice a day (am and pm) for 4 consecutive days. After pre-training, mice

134
were placed on the rod at speed of 10 rpm and latency to fall was recorded. Mice that fell
in less than 10 sec were given a second trial. A value of 120 sec was recorded for mice
that stayed on the rotarod for the assigned maximum duration of rotation. Mice were
tested twice (am and pm) on test days. Rotarod data was collected unblinded to the
investigators. Data were collected by averaging two trials.
5.3.3 Tissue collection and blinding
Animal were quickly sacrificed with CO2 and perfused transcardially with 30 mL of
saline solution. The brain was then quickly removed and separated between the two
hemispheres. The left hemisphere was placed into a solution of 4 % paraformaldehyde
(PFA). A ~5 mm section of the cervical spinal cord was also removed and fixed in PFA.
The right hemisphere was dissected on ice into the forebrain, cerebellum and brainstem
regions and quickly frozen in liquid nitrogen. The remaining portion of the spinal cord
was dissected down to roughly the end of the thoracic region and also frozen in liquid
nitrogen and stored at –80 °C until required. At this point, all of the samples were
relabeled with numbers in order to blind the investigators carrying out the analysis from
the study groups.
5.3.4 Tissue homogenization
Tissues were homogenized in six volumes of tissue homogenization buffer (THB)
containing 50 mM Tris-HCl pH 8, 274 mM NaCl, 5 mM KCl, 2 mM EDTA, 2 mM
EGTA, one complete-mini protease inhibitor tablet (Roche) per 50 mL, 5 mM sodium
pyrophosphate, 30 mM -glycerophosphate, 30 mM sodium fluoride and 1 mM
phenylmethylsulfonyl fluoride (PMSF) and then spun at 13, 000 x g in an Eppendorf

135
5417C centrifuge for 20 min. The resulting pellet was then re-extracted with three more
volumes of THB and spun again at 13,000 x g for 20 min. The supernatants were then
combined and referred to as the low-speed supernatant (LSsup). The LSsup was then
spun at 100,000 x g in a Beckman TLA-45 in a Beckman TL-100 centrifuge at 4 ºC for
45 min. The supernatant was removed and referred to as the high-speed supernatant
(HSsup). The resulting pellet was resuspended in 50 µL of 1 x sodium dodecyl sulfate
polyacrylamide gel (SDS-PAGE) loading buffer per 125 µL of LSsup that was spun at
100,000 x g. For the sarkosyl extraction, 1% N-laurylsacrosinate (Sigma) was added to
the LSsup and allowed to incubate at 37 °C for one hour with shaking. The samples were
then spun at 100, 000 x g in the TLA-45 rotor. The supernatant was removed and the
sarkosyl insoluble pellet was resuspended in SDS-PAGE loading buffer.
5.3.5 Fluorescent immunohistochemistry (IHC)
Following fixation of the brain and cervical spinal cord in 4% PFA for 24 hr the samples
were transferred to 20% (w/v) sucrose overnight for cryoprotection. Brains and spinal
cords were embedded in O.C.T. (Optimal Cutting Temperature) embedding medium
(Sakura Finetek USA Inc). Brains were sectioned in the sagittal plane, and spinal cords
were sectioned in the transverse plane at 30 µm on a Leica cryostat. Free floating sections
were permeabilized with 1 x PBS (pH 7.4) containing 0.3% Triton X-100 (PBST) for 15
min. After blocking with 10% normal goat serum (NGS) and 2.5% BSA in PBST for 60
min, sections were incubated with appropriate primary antibodies at 4 ºC for 24 hr. After
washing with PBST for 45 min, sections were incubated with appropriate secondary
antibodies conjugated with Alexa 488, Alexa 568 and Alexa 647 (Invitrogen) for 90 min.
After 45 min washing, the sections were mounted on slides (Superfrost/Plus, Fisher), and

136
coverslipped with Vectashield Mounting Medium with DAPI (H-1200, Vector Labs).
Sections examined in parallel but without being exposed to primary antibody served as
experimental controls. In some double or triple lableling experiments, staining was
performed with species- and isotype-specific secondary antibodies applied
simultaneously after incubating in cocktails containing two or three primary antibodies in
various combinations.
5.3.6 Fluorescent microscopy and densitometry analysis
Stained tissue sections were analyzed by an upright Leica DM4000B fluorescent
microscope equipped with four filter sets (A4, excitation at 340-380 nm, and emission at
470 nm; L5, excitation at 480 nm, and emission at 527 nm; TX2, excitation at 560 nm,
and emission at 645 nm; Y5, excitation at 620 nm, and emission at 700 nm,
respectively.). Images were acquired at a full frame size of 1392 x 1040 pixels by Leica
DFC 350 FC digital camera and processed by Leica LAS imaging acquisition and
analysis system. Densitometry analyses were conducted using modified methods
previously used by others in similar transgenic mice for Alzheimer’s disease [4]. In
detail, for AT8 densitometry analysis, grey scale images were acquired from the same six
nonoverlapping neuroanatomic regions (three in brainstem, two from hypothalamus, and
one in cortex, respectively) at size of 713 x 532 µm from each brain section using 20x
objective lens (200x magnification), and three nonoverlapping neuroanatomic regions
(one each from ventral, intermedial, and dorsal regions in grey matter, respectively) at
size of 356 x 266 um from each spinal cord section using a 40 x objective lens (400 x
magnification). For pSer422 densitometry analysis, grey scale images were acquired from
three nonoverlapping neuroanatomic regions in brainstem at size of 713 x 532 um from

137
each brain section using a 20 x lens. All images were thresholded at the pre-determined
levels to contain positive immunoreactivities (IR) exclusively. Threshold, area
measurement of IRs and densitometric quantifications were performed using the ImageJ
(1.43u) software package (NIH). Scion Image was used to for densitometry of
immunoblot data.
5.3.7 Motor neuron counts
Motor neuron counts were performed using modified methods previously used by others
for counting neurons in animal preparations [22]. Briefly, every third 30 µm transverse
cervical enlargement sections from TG treated and non-treated JNPL3 stained with NeuN
antibody and countstained with DAPI. Under 40 x objective lens, motor neurons meeting
the following criteria were counted: 1) size > 25 µm; 2) possession of at least one thick
process; 3) location in ventral grey matter regions below a horizontal level through the
center of central canal. Motor neurons with a visible nucleolus were also counted if they
were not in the initial optical plane of focus, but came into focus as the optical plate
moved through the tissue. Data were collected by averaging six ventral horn regions per
animal, total 120 sections were counted.
5.3.8 AT8 IHC before and after btOGA digestion in tissue
To determine whether elevated O-GlcNAc modification levels on tau by TG mask the
AT8 antibody binding in tissue sections, we compared AT8 IR before and after the
sections digested by a bacteria OGA (btOGA) [7]. In a preliminary experiment, we
observed AT8 fluorescent IR was not diminished by btOGA digestion. Therefore, in the
study for the same section comparision, free floating sections were first single stained

138
with AT8 antibody, and followed by incubation with Alexa 568 secondary antibody.
Without acquiring images, the same sections were digested in PBS (pH7.0) solution
containing btOGA at 1:10 (mg/ml) concentration in 25 °C water bath for 2 hr. After
washing, the btOGA digested sections were double stained with AT8 and CTD110.6
antibodies, followed by incubation with secondary antibodies of Alexa 488 for AT8
(differ from the secondary used for AT8 before btOGA digestion) and Alexa 647 for
CTD110.6. Tissue sections digested with an enzymatic disfunction mutant btOGA [5]
were carried out in parallel served as experimental controls.
5.3.9 AT8 IHC by using ABC method
For AT8 labeling with an avidin biotin peroxidase (ABC) method (Vector Labs,
Burlingame, CA), the free-floating sections were first incubated with 0.3% hydrogen
peroxide for 30 min. After blocking with 10% normal goat serum (NGS) in PBS for 1 hr,
sections were incubated with 1:500 AT8 antibody at 4 °C overnight. Sections were then
incubated with biotinylated anti-mouse IgG secondary antibody (BA9200, Vector Labs)
for 1 hr at room temperature, followed by incubation with ABC reagent (PK6100, Vector
Labs) for 30 min. Color development was performed using diaminobenzidine
tetrahydrochloride (DAB, SK4105 from Vector Labs) as a substrate for peroxidase and
the reaction was stopped by flooding with distilled water. Then sections were dehydrated
through an ascending alcohol bath series (70%, 80%, 90%, 100%, 1 minute each), cleared
in xylene 2 times for 1 min each. Slides were coverslipped with Permount (Sigma), and
air dried overnight. Sections were first examined by a digitalized Leica DM4000B
microscope using its setting for bright field. Then bright field images were acquired by

139
Leica DFC 350 FC digital camera and processed by Leica LAS imaging system by
applying pseudo-colour to the images.
5.3.10 Gallyas silver staining
Gallyas silver staining was carried out using a modified method developed by Braak et al.
[3]. In brief, 30 µm free floating cryo-sections were washed in PBS, then mounted onto
slides and air dried overnight in room temperature. The sections on slides were
rehydrated and placed in 5% periodic acid for 5 min. After washing in distilled water,
slides were placed in alkaline silver iodide solution for 1 min, washed in 0.5% acetic acid
for 3min, then place in developer solution for 6-10 min. Washed in 0.5% acetic acid for
3min again, rinsed with tap water for 5min. Then sections were dehydrated through an
ascending alcohol bath series (70%, 80%, 90%, 100%, 1 minute each), cleared in xylene
2 times for 1 min each. Slides were coverslipped with Permount (Sigma), and air dried
overnight. For purposes of same section comparisons in this study, the Gallyas silver
staining was performed after fluorescent images had been acquired from triple labeling
sections of AT8, pSer422 and CTD110.6 antibodies using methods described above.
5.3.11 Immunoblotting
Samples were electrophoeresed through 10% sodium dodecyl sulfate polyacrylamide gels
(SDS-PAGE) in either the Bio-rad mini-protean system or a C.B.S scientific system
capable of running up to 50 samples at once. Gels were then transferred to nitrocellulose
(Bio-rad) membranes. Membranes were then blocked for 1 hr at room temperature (RT)
with 2 % bovine serum albumin (BSA) in PBS containing 0.1 % Tween-20 (Sigma)
(PBS-T) and then subsequently probed with appropriate primary antibody delivered in 2

140
% BSA in PBS-T overnight at 4 ºC. Membranes were then extensively washed with PBS-
T, blocked again for 30 min with 2 % BSA in PBS-T at RT and then probed with the
appropriate HRP conjugated secondary antibody for 1 hr at RT delivered in 2 % BSA in
PBS-T. Finally, the membranes were washed extensively with PBS-T and then developed
with SuperSignal West Pico Chemiluminesence substrate (Pierce) and exposed to CL-
XPosure Film (Pierce). For blots pretreated with alkaline phosphatase (Roche), the blots
were incubated with 100 Units of calf intestinal alkaline phosphatase in 50 mM Tris-HCl,
pH 8.5, 0.1 mM EDTA, 0.5 mM MgCl2 for 4.5 hr at 37 ºC following the first blocking
step. The blots were then washed briefly in PBS-T and the rest of the immunoblot
protocol was followed as described above.
5.3.12 Statistical analyses
Statistical analyses were carried out using Graphpad Prism 5.03. The data from the
control and treatment groups was not always accurately estimated by a normal
distribution as judged by the D’Agostino & Pearson omnibus normality test. For values
of n > 10, if the data was accurately estimated by a normal distribution the unpaired
student’s t-test was employed. If the data was not accurately estimated by a normal
distribution the non-parametric Mann-Whitney U-test was used. For values of n ≤ 10 the
student’s t-test was used. Single-tailed tests were used to test for reductions in tau
phosphorylation or increases in O-GlcNAcylation because we have demonstrated
previously that Thiamet-G results in the appropriate differences in these parameters. In
the case of measurements which we have not measured previously, such as motor neuron
counts, body weights and tau aggregation data, two-tailed tests were used. In the case of

141
the hang-test data an Analysis of Covariance (ANCOVA) was used. For all statistical
tests p < 0.05 was used as the threshold for statically significant data.
5.3.13 Antibodies used in this study
Mouse monoclonal -Tau-46, which recognizes the C-terminal region of tau in a
phosphorylation-independent manner was purchased from Abcam. Mouse monoclonal -
human tau antibody, HT7, recognizes amino acids 159-163 of tau in a phosphorylation
independent manner was from Pierce. Mouse monoclonal antibody, AT8 recognizes
phospphorylation of tau at Ser202/Ser205 was from Innogenetics. Rabbit polyclonal -
tau antibodies, pSer400, pSer231 and pSer422, recognize their respective phosphorylated
serine residue and were purchased from Invitrogen. Mouse monoclonal antibody, PHF-1,
recognizes tau phosphorylated at Ser396/Ser404 and mouse monoclonal, CP27, which
recognizes human tau were a kind gift from Dr. Peter Davies. Mouse monoclonal -tau
antibodies, nY29 and TauC3, recognize nitrated tau and tau truncated at Ser422 were
from Millipore and Invitrogen, respectively. Finally, the 3925 -O-GlcNAc tau at Ser400
is a custom rabbit polyclonal antibody described previously [46].
Mouse monoclonal -NeuN antibody, A60, was purchased from Millipore. Mouse
monoclonal -O-GlcNAc antibodies, CTD110.6 and RL2, were purchased from Covance
and Abcam, respectively. Mouse monoclonal -GAPDH antibody was purchased from
Invitrogen. Goat -mouse IgG, goat -rabbit IgG, and goat -mouse IgM horseradish
peroxidase conjugated secondary antibodies were obtained from Santa Cruz
Biotechnology. See the IHC sections (above) for secondary antibodies used in that work.

142
5.3.14 Molecular cloning
The human OGT gene was cloned into the pMal-c2X vector (New England Biolabs)
using the following primers: 5'-gccgccggatccaagcgtcttccgtgggcaacgtgg-3’ (BamHI cut
site shown in bold) and 5’-gccgccgtcgacctatgctgactcagtgacttcaac-3' (SalI cut site shown
in bold). The H558A point mutation of OGT was created using the following primers:
5’-gagttccgactttgggaatgctcctacttctcaccttatgc-3’ and 5’-
gcataaggtgagaagtaggagcattcccaaagtcggaactc-3’. These primers were used to introduce the
point mutation using QuikChange site-directed mutagenesis. The Tau244-441 construct was
cloned into the pET28a vector (Novagen) using the following primers: 5’-
gccgcccatatgatgcagacagcccccgtg-3’ (NdeI cut site shown in bold) and 5’-
gccgccctcgagttacaaaccctgcttggccaggg-3’ (XhoI cut site shown in bold). The S400A point
mutation in Tau244-441 was created with the following primers: 5’-
gcaagtcgccagtggtggctggggacacgtctccc-3’ and 5’- gggagacgtgtccccagccaccactggcgacttgc-
3’.
5.3.15 Production of recombinant O-GlcNAc modified tau
The gene encoding Tau244-441 or S400A Tau244-441, in pET28a, were co-transformed with the
gene encoding wild-type OGT (wtOGT) or H558A OGT (mutOGT), in pMal-c2X, into
E. coli Tuner cells (Stratagene). In order to increase the probability of obtaining
cotransformant colonies, LB plates with 1/3rd the concentration of ampicillin and
kanamycin (33 g/mL and 16.6 g/mL, respectively) were used. Once colonies were
obtained, ampicillin and kanamycin concentrations of 100 g/mL and 50 g/mL,
respectively, were used to grow the bacteria in solution. For expression, Tau244-441 or
S400A Tau244-441, co-transformants were induced with IPTG (0.5 mM) overnight at 22 C

143
(~20 hr). The bacterial cells were harvested by centrifugation at 5000 rpm for 10 min in a
Sorvall RC-6 plus centrifuge in a FIBERLite F9S-4x1000y rotor. The bacterial pellets
were then resuspended in 30 mL of Ni-NTA column binding buffer (20 mM sodium
phosphate, 500 mM NaCl, 5 mM imidazole, pH 7.4). wtOGT / Tau244-441 and mutOGT /
Tau244-441 or wtOGT / S400A Tau244-441 and mutOGT / S400A Tau244-441 co-transformants
were lysed by the addition of 2 mg / mL lysozyme (Bioshop) in the presence of one
Roche protease complete tablet per 30 mL of resuspended bacterial pellet. Sonication was
carried out at ~30 % power on a Fischer Scientific sonic dismembrator (model 500) for
six cycles consisting of 20 sec on followed by a 40 sec rest period. Cellular debris was
removed by centrifugation at 13,000 rpm in an SS-34 rotor in a Sorvall RC-6 plus
centrifuge. The supernatants were then loaded onto HisTrap FF Ni-NTA columns (GE
Healthcare) using a peristaltic pump. The columns were washed with 90 mL of Ni-NTA
column wash buffer (20 mM sodium phosphate, 500 mM NaCl, 60 mM imidazole, pH
7.4) and then eluted with 25 mL of Ni-NTA column elution buffer (20 mM sodium
phosphate, 500 mM NaCl, 250 mM imidazole, pH 7.4). The eluates were then
exhaustively dialyzed against 20 mM sodium phosphate buffer, pH 6.7 and concentrated
to 2 mL. From here forward the Tau244-441 or S400A Tau244-441 expressed in either the
presence of wtOGT or mutOGT will be referred to as control and modified Tau244-441 or
control and modified S400A Tau244-441.
5.3.16 Purification of control and modified Tau244-441 or S400A Tau244-441 by reversed-phase HPLC
The control or modified Tau244-441 or S400A Tau244-441 protein (0.25 mg / run) was acidified
using 10 % trifluoroacetic acid (TFA) and loaded onto an Agilent Zorbax 300SB-C8 (9.4

144
mm x 250 mm) semi-preparative HPLC column housed in an 1100 series Agilent HPLC.
The column was held at 30% buffer B (acetonitrile (ACN), 0.1% TFA) for the first five
min after which the proteins were purified using a linear gradient of 30 % buffer B to 75
% buffer B over 45 min operating at 1 mL / min. Fractions were collected using a Foxy
Jr. fraction collector set to collect 1 mL fractions over the entirety of the HPLC run. The
major peak eluted at approximately 24.2 min and was collected in fractions 24-27. These
fractions were pooled and lyophilized to dryness. This procedure typically results in ~1
mg of pure control or modified Tau244-441 or S400A Tau244-441 from 6 L of bacterial culture.
The control or modified Tau244-441 or S400A Tau244-441 samples are taken up in 1 mL of 20
mM sodium phosphate buffer, pH 7. Samples were assessed for purity by analysis on 15
% SDS-PAGE gels.
5.3.17 In vitro aggregation of control and modified Tau244-441 or S400A Tau244-441
Aggregation of the tau constructs was carried out at 0.25 mg / mL tau protein, 1 mM
dithiothreitol (DTT), 0.5 mM PMSF, and 0.01 mg / ml thioflavin-S (ThS) in a final
volume of 100 µL. Reactions were initiated by the addition of 0.06 mg / mL heparin
(5600-6400 Average mol. wt., Int Labs USA) to the reaction mixtures contained in wells
of a 96-well fluorescence microplate (Nunc). The aggregation process was followed by
reading the microplate (at the indicated times) in a Molecular Devices Fmax
spectrofluorometer using an excitation filter of 440 nm and an emission filter of 520 nm.
The microplate was incubated at 37 ºC in a sealed humidified box to prevent evaporation
of the aggregation reactions. Data was corrected by subtraction of control samples which
contained all of the reaction mixture components except for the tau protein. Samples were

145
analyzed in replicates of at least three and data is presented as the ThS fluorescence
average ± standard error of the mean (s.e.m.). Data presented in Figure 4 is presented as
relative ThS fluorescence where the control tau sample end point value is set to 100 %.
For aggregation with arachidonic acid (AA), the aggregation reaction was carried out as
described above except AA (75 m) was substituted for heparin. Because AA was
dissolved in ethanol, the appropriate amount of ethanol was also included in the control
reactions.
5.3.18 Filtertrap assay and quantitiation of the sarkosyl insoluble pellet tau by Slot blot
The filtertrap assay was essentially carried out as descried previously [31]. Briefly,
following aggregation, samples were diluted 50-fold using phosphate buffered saline
(PBS). 0.2 m Nitrocellulose (Bio-rad) was wetted using PBS and loaded into the Bio-
rad slot blot apparatus. 50 µL of each sample was then filtered through the nitrocellulose
and then each well was washed three times with 100 µL of PBS. The membrane was then
removed from the apparatus washed briefly with PBS-T and then blocked for one hour
with 5% non-fat milk (Bio-rad) the rest of the immunoblot protocol was then followed as
above except non-fat milk was substituted for BSA. The Tau-46 antibody was used at a
dilution of 1:5000. For quantitation of human tau in the sarkosyl pellet samples in SDS-
PAGE loading buffer were diluted 10-fold in PBS and then 50 µL of each sample was
filtered through 0.45 m nitrocellulose (Bio-rad), washed three times with PBS and then
the HT7 immunoblotting was carried out exactly as above.

146
5.3.19 Production of O-GlcNAc modified sTAB1 and in vitro aggregation
The gene encoding sTAB1 or CaMKIV, in pET28a, were cotransformed with the gene
encoding WT or H558A OGT, in pMal, into E. coli Tuner expression cells. To induce
expression of sTAB1, IPTG (0.5 mM) was added to culture in exponential phase and the
cells were grown overnight at 25 C. Bacteria were harvested by centrifugation, lysed,
and protein purified by nickel column chromatography (as above).
All in vitro aggregation assays were carried out using a Cary 3E UV-VIS
spectrophotometer equipped with a Peltier temperature controller at either 40 or 45 C.
Reactions were monitored continuously at 500 nm. Other wavelengths produced similar
rates indicating that it was not absorbance of light that was being monitored but rather
scattering by the aggregates. A protein concentration of 40 M was used for reactions
carried out at 40 C, whereas a protein concentration of 10 M was used for reactions
carried out at 45 C to slow down the rate and quantity of aggregated protein.
5.4 Results and discussion
Thiamet-G (TG) is a potent (KI = 20 nM) and allele-selective inhibitor of the -N-
acetylglucosaminidase OGA previously described by us [45]. TG crosses the blood-brain
barrier where it blocks the removal of O-GlcNAc from modified proteins by OGA while
OGT activity is unaffected. OGA inhibition thus elevates O-GlcNAc modification of
nucleocytoplasmic proteins[45]. Short term administration of TG also reduces
phosphorylation of tau in healthy animals, presumably through competition between O-

147
GlcNAc and phosphorylation [45]. We therefore reasoned that long term inhibition of
OGA in JNPL3 mice by TG might block pathological hyperphosphorylation of tau,
prevent its aggregation, and slow downstream neuronal cell loss.
The JNPL3 model is an aggressive transgenic model with fairly variable penetrance in
which tau overexpression is several fold higher than endogenous tau in affected regions
of the brain. To evaluate the effects of increased O-GlcNAc in this aggressive model we
carried out a eight month long blinded study using forty hemizygous female JNPL3 mice
of ages ranging from 9-12 weeks. The mice were divided into two groups of twenty
animals and TG was delivered in the drinking water of the treated group to provide a
daily dose of 500 mg/kg/day. JNPL3 mice show impaired motor function at 40 weeks of
age and this is associated with the formation of NFTs that are most extensive in spinal
cord, hindbrain, and to a lesser extent in the midbrain and hypothalamus.
We therefore subjected animals at 40 weeks of age to two standard quantitative measures
of motor impairment; the wire cage hang test and the rotarod test. To evaluate disease
progression we carried out testing once a week for 5 consecutive weeks. No differences
were observed in the performance in the rotarod test (analysis of covariance (ANCOVA),
p = 0.74) (Figure 5-2b), however, the TG treated group had a 3-fold slower decline in the
cage hang test (-1.6 sec / week vs. -0.52 sec / week, (ANCOVA), p = 0.021) compared to
the control group (Figure 5-1a). During this testing period we remarked that the body
weights of the animals in each group differed significantly; the TG-treated group were
heavier on average than the control group (41.7 g vs. 35.7 g, two-tailed, unpaired t-test, p

148
< 0.005, Figure 5-2a). We attribute the average shorter cage hang latency in the TG
treated group to this difference. Because it is known that JNPL3 mice lose body mass
during disease progression due to neurogenic atrophy of skeletal muscle[22, 40], we
speculate that this difference in body weight stems from a prevention of weight loss
arising from treatment by TG; the control group showed a trend toward losing weight
more quickly than the treated group (Figure 5-2a). A blinded investigator also judged
qualitatively that after the 8 month treatment period more animals in the TG group had no
motor impairment, whereas more control animals showed mild motor impairment (Figure
5-1b). These collective results and the known almost 2-fold decrease in neuron counts
occurring in cervical spinal cord prompted us to evaluate if TG treatment would increase
survival of motor neurons.
We obtained motor neuron counts from the cervical spinal cord from each animal and
found the TG treated group had on average 1.4-fold more motor neurons than the control
group (p = 0.014, two tailed, unpaired t-test) (Figure 5-1c, 5-3 and 5-4). These higher
motor neuron counts in this region of the spinal cord may explain the slower decline in
the wire cage hang test as muscles in the forelimbs are innervated by motor neurons in
the cervical spinal cord[28] and could well account for the prevention of weight loss in
the TG treated group as compared to control group. This result suggested that TG
treatment might be providing a protective benefit through increased O-GlcNAc
modification of tau and decreased tau pathology

149
To evaluate if increased O-GlcNAc levels correlate with decreased tau
hyperphosphorylation, tau aggregation, and motor neuron protection, we first examined
the effect of treating three 14 weeks old hemizygous female JNPL3 mice with 500 mg /
kg / day of TG for one week. Previously, we showed that tau in healthy rat brain is O-
GlcNAc modified at Ser-400[42]. Here we find (Figure 5-1d) that TG increases Ser400
O-GlcNAc levels by 1.5-fold (p = 0.036, one-tailed unpaired t-test) as well as 9.3-fold
globally on cellular proteins (p < 0.0001, one-tailed unpaired t-test). We next examined
whether increased O-GlcNAc modification in JNPL3 mice treated with TG for 8 months
leads to decreased pathological phosphorylation and aggregation of tau. Major AD-
related phosphorylation sites in human tau also occur in the JNPL3 model, including
those phosphorylated by proline directed kinases, such as glycogen synthase kinase
(GSK3) and cdk5, or by microtubule affinity regulating kinase (MARK)[20, 34]. By
immunohistochemistry (IHC), using the AT8 antibody that specifically detects
pathological tau phosphorylated at Ser202 and Thr205, and to a lesser degree at Ser199
and Ser208, we compared TG treated mice to control mice and found, as hypothesized,
that AT8 immunoreactivity with NFT-like morphology decreased by 2.6-fold in
brainstem (p = 0.0039, one tailed, Mann-Whitney U test), 1.3-fold in spinal cord (p =
0.021, one tailed, Mann-Whitney U test), 2.0-fold in the hypothalamus (p = 0.025, one
tailed, Mann-Whitney U test), and 2.0-fold in the cortex (p = 0.0062, one tailed, Mann-
Whitney U test) (Figure 5-5, 5-6, 5-7, 5-8 and 5-9). We used DAB staining on cervical
spinal cord using the AT8 and pSer422 antibodies to verify by light microscopy the
nature of the staining observed by fluorescence (Figure 5-10). As expected, we also
found by IHC significantly increased O-GlcNAc levels in the brain and spinal cord

150
tissues from TG treated mice using an anti-O-GlcNAc antibody (CTD110.6) (Figure 5-5,
5-7, 5-8 and 5-9).
During the course of these studies we considered that O-GlcNAc present on tau could
obscure sites of phosphorylation and this might give rise to apparent decreases that could
be misinterpreted as decreased levels of aggregates. We therefore evaluated O-GlcNAc
modification of tau using a recombinant expression methodology that we have used
previously to map O-GlcNAc to Thr123, Ser400, and one of Ser409, Ser412 or
Ser413[46]. Using this approach we were able to establish an additional site of
modification at Ser208 (Figure 5-11). We next aimed to evaluate whether Ser-208 O-
GlcNAc could obscure the AT8 epitope by performing enzymatic digestion of O-GlcNAc
in tissue sections. We find O-GlcNAc levels were efficiently reduced but that AT8
immunoreactivity was not significantly altered (Figure 5-12), strongly suggesting Ser208
O-GlcNAc does not complicate the AT8 analysis. To further evaluate this issue, we
performed IHC using a pSer422 antibody, which detects the phosphorylation of Ser422
on tau in a region where no O-GlcNAc site was detected. We find that the pSer422
immunoreactivity shows trends 2.0-fold lower level in TG treated mice (p = 0.067, one
tailed, Mann-Whitney U test) (Figure 5-13). During our IHC analyses we observed that
both AT8 and pSer422 antibodies stained identical structures that had typical NFT
morphology. However, to confirm that the AT8 and pSer422 antibodies predominantly
detect NFTs, we used the PHF-1 antibody which is a commonly used antibody to detect
NFTs. We found that the PHF-1 antibody labeled similar structures as the AT8 and
pSer422 antibodies and reductions in PHF-1 immunoreactivity, similar to those observed

151
for AT8, were noted in the TG treated mice (Figure 5-14). We also performed triple
staining by making use of Gallyas-Braak staining on the same brain sections that had
previously been immunostained with AT8 and pSer422. In this way we find that AT8 and
pSer422 immunoreactivity overlap with Gallyas-Braak positive structures (Figure 5-15,
5-16). Finally, nitrated tau fibrillar lesions are known to correlate with NFTs in the AD
brain[32] and we found that the nY29 antibody, which recognizes nitrated tyrosine 29 of
tau, labeled similar structures as the pSer422 antibody and showed decreased
immunoreactivity in brain and spinal cord tissues from TG treated mice (Figure 5-17, 5-
18, 5-19, 5-20). Thus four separate markers all appear to detect similar structures having
NFT morphology and all show a similar reduction in TG treated mice compared to
controls. Taken together, these results indicate that the neuronal protective effects that we
observed in the TG treated mice correlate with a reduction in NFT formation, which
likely results from increased O-GlcNAc modification of tau and reciprocal decreases in
tau phosphorylation. Having shown that TG is able to reduce NFT formation in the
JNPL3 brain and spinal cord we carried out biochemical analyses of extracted tissue from
these animals. We first evaluated whether TG treatment causes the expected increased O-
GlcNAc levels and decreased phosphorylation of soluble tau. We find that global levels
of O-GlcNAc in these animals are increased by 4-5 fold on average (p = 0.0007, one-
tailed, Mann-Whitney U test) (Figure 5-21d) and tau O-GlcNAc levels are increased by
1.6 fold (p = 0.0077, one-tailed, unpaired t-test) in the brainstem (Figure 5-21e,f).
However, when we evaluated the AT8 tau phospho-epitope (data not shown) from
complete homogenates we were surprised to find that TG treatment had no significant
effect on the levels. We therefore considered whether tau O-GlcNAc might act to reduce

152
the amount of a 64 kDa pathologically hyperphosphorylated form of tau that is
sedimentable by high-speed centrifugation. This 64 kDa form of tau has been shown to
correlate with the severity of motor impairment in JNPL3 animals[20]. Again, quite
unexpectedly, we found that TG treatment reduced neither the amount of 64 kDa tau
species in high-speed pellets from the brainstem (Figure 5-21a) nor their phosphorylation
(Figure 5-21b,c, 5-22). Given previous findings that tau contained in sarkosyl extracts of
brain tissue from JNPL3 mice comprises mainly fibrillar tau[34], we evaluated sarkosyl
extracts and found the TG treated group had 1.6-fold less sarkosyl insoluble tau (p = 0.05,
one tailed, unpaired t-test). The fact that we observe differences in the amount of fibrillar
tau in these animals but see no difference in tau hyperphosphorylation, as measured by
immunoblots, suggested to us that increased O-GlcNAc levels on tau may not hinder
hyperphosphorylation in this model. Instead, increased O-GlcNAc may yield tau species
that are less prone to aggregation into NFTs, regardless of their degree of
phosphorylation. Given that we see no effect of O-GlcNAc on tau phosphorylation and
the formation of the hyperphsophorylated 64 kDa species, for O-GlcNAc to have a
protective function we expected that it must be found on the hyperphosphorylated form
of tau. Consistent with this view, we find that O-GlcNAc at Ser400 is found on the 64
kDa form of tau (Figure 5-21g) and that the Ser400 O-GlcNAc levels are increased in TG
treated mice.
To demonstrate that O-GlcNAc on tau serves a protective role in vivo using the JNPL3
model is challenging due to the complexity of the system; there are many interacting
proteins and other post-translational modifications present on tau. Therefore, to address

153
the role of O-GlcNAc in aggregation in a clear manner, using biochemical methods, we
used an in vitro tau aggregation assay. The in vitro aggregation of tau can be induced
using anionic molecules such as heparin or arachadonic acid[14, 44] and can be followed
by fluorescence using the preferential Thioflavin-S dye binding to the aggregates[9].
Using a recombinant method that yields tau modified at the physiologically relevant sites,
we generated an O-GlcNAc modified tau construct (Tau244-441) that lacks the N-terminal
domain (Figure 5-23)[46]. We verified that the unmodified N-terminal construct
aggregates in vitro over useful time frames [14, 39] and that the O-GlcNAc modified
Tau244-441 is indeed O-GlcNAc modified (Figure 5-24a). We monitored the aggregation of
unmodified (Ctrl) and O-GlcNAc modified (Mod) Tau244-441 (Figure 5-24b) and found that
the Ctrl Tau244-441 aggregates both more quickly and to a greater extent than O-GlcNAc
modified Tau244-441. To further test whether this observed effect is attributable to the O-
GlcNAc moieties we prepared Ctrl and Mod Tau244-441 bearing a S400A mutation. This
mutation leads to a ~2 fold reduction in the O-GlcNAc levels as detected by CTD110.6
and shows no reactivity with the 3925 antibody (Figure 5-24b). Aggregation of Ctrl and
Mod S400A Tau244-441 (Figure 5-24c) occurs at the same rate and to the same extent,
suggesting that O-GlcNAc at the C-terminus of tau inhibits its aggregation. We
confirmed this result using arachidonic acid in place of heparin as the inducer of
aggregation (Figure 5-25) as well by filter trap assay (Figure 5-24d), which enables
trapping and quantitation of tau aggregates. These in vitro observations provide a good
biochemical rationale for the fewer NFTs observed in the TG treated JNPL3 animals, the
increased motor neuron counts, and the prevention of weight loss.

154
On the basis of these findings we speculate that one general biochemical role for O-
GlcNAc could be that it contributes to the stability of modified cellular proteins, thereby
hindering their aggregation. To evaluate whether O-GlcNAc modification of proteins
contributes to their stabilization against aggregation we decided to test this idea in an
entirely different context; we recombinantly produced several O-GlcNAc modified
proteins including a construct of TAK1-binding protein (sTAB1) [6, 11] (Figure 5-26a).
We evaluated the aggregation potential of these proteins and found sTAB1 aggregates in
vitro at elevated temperatures. Furthermore, at both 40 ºC and 45 ºC, O-GlcNAc modified
sTAB1 aggregates significantly more slowly than the unmodified sTAB1 (Figure 5-26b).
These results suggest O-GlcNAc hinders sTAB1 aggregation and support the hypothesis
that O-GlcNAc acts more generally to stabilize proteins to prevent their aggregation. In a
related vein, we note that a recent study reported that higher O-GlcNAc modification of
keratins 8 and 18 within cells correlated with higher levels of monomeric soluble
cytoplasmic keratins and their decreased inclusion into keratin intermediate
filaments[38]. Although other processes within cells might be contributing to this
behavior of keratins 8 and 18, the observations are consistent with the hypothesis that O-
GlcNAc could serve a generally protective role against protein aggregation.
5.5 Conclusion
In summary, we show that small molecule inhibition of OGA by TG and consequent
increases in O-GlcNAc slow motor impairment and motor neuron loss in JNPL3 animals
over a period of eight months without any apparent adverse effects. We further show that
preservation of motor neurons likely stems from O-GlcNAc hindering tau aggregation to

155
form NFTs in vivo. Surprisingly, we find that O-GlcNAc is sufficient to prevent tau
aggregation in vitro on its own though modification of Ser400. These findings
collectively suggest that O-GlcNAc acts directly on tau to prevent its aggregation
downstream of phosphorylation. Accordingly, OGA inhibition may be a promising
therapeutic approach for the treatment of AD and associated tauopathies. OGA inhibitors
therefore also appear to be among a very select class of compounds able to slow tau-
driven neurodegeneration. These findings are also consistent with the view that impaired
glucose flux in the brains of AD patients could compromise a protective function of O-
GlcNAc normally operative in healthy brain. Notably, this protective phenomenon is not
unique to tau protein; we speculate that a general biochemical function of the O-GlcNAc
modification is to protect proteins from aggregation and contribute to their cellular
stability. Studies are underway to evaluate the potential protective effects of O-GlcNAc
on proteins in different experimental paradigms.

156
5.6 Figures
Figure 5 - 1 Treatment of JNPL3 animals slows motor impairment and reduces neurodegeneration.
a, Wire cage hang test of control and Thiamet-G (TG) treated JNPL3 mice beginning at age 40 weeks reveals a significantly faster decline in the control group compared to the TG treated group (error bars indicate +/- S.E.M). b, more TG treated JNPL3 mice show no motor impairment, while more mice from the control group show mild motor impairment as judged by a blinded independent investigator. c, Motor neurons per ventral horn in the cervical spinal cord. TG treated JNPL3 mice show 1.4 fold more motor neurons per ventral horn. d, Short term treatment of 14 week old JNPL3 mice with 500 mg / kg / day TG for one week increases global and tau O-GlcNAc levels in the brain. Global O-GlcNAc levels as detected by CTD110.6 (upper left panel) are increased while actin indicates equal protein loading (lower left panel). O-GlcNAc levels on tau at Ser400 are also increased as detected by 3925 (upper right panel) while the HT7 antibody indicates equal total human tau (lower right panel).

157
Figure 5 - 2 JNPL3 body weights and Rotarod Performance.
a, Control and Thiamet-G treated JNPL3 animals of 40 weeks of age were weighed once a week for 5 consecutive weeks. Thiamet-G treated animals were significantly heavier on average (error bars represent +/ - S.E.M.). b, Beginning at the age of 40 weeks Control and Thiamet-G animals were also subjected to the rotarod test. No difference was found between the two groups on the rotarod test as indicated by the analysis of covariance (ANCOVA, p = 0.74). Error bars indicate (+/- S.E.M.).

158
Figure 5 - 3 Motor neuron counting method.
30 m cervical enlargement sections from control and Thiamet-G treated JNPL3 mice were stained with a -NeuN antibody (green) and then counterstained with DAPI. Under the 40x objective motor neurons of size > 25 m and having one thick process found within the ventral horn (indicated here by the dotted and solid white line) were counted. Scale bar indicates 200 m.

159
Figure 5 - 4 O-GlcNAc levels are increased in motor neurons of the ventral horn.
Cervical enlargement sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with a -NeuN antibody (green) and a -O-GlcNAc antibody (CTD110.6, red) (middle two panels) and counter stained with DAPI (left most panels). Overlay of NeuN, O-GlcNAc, and DAPI signals (right most panels) indicates that the O-GlcNAc levels are increased in motor neurons upon treatment of Thiamet-G. Scale bar indicates 50 m.

160
Figure 5 - 5 Immunohistological analysis of TG treated JNPL3 animals reveal significant reductions in AT8 immunoreactivity in the brainstem.
(A) AT8 immunohistochemistry (IHC) analysis of the brainstem region of sagittal sections from control and TG treated JNPL3 mice indicates a significant reduction in the immunostaining of this antibody. (B) Representative data from the quantitation in (A) reveals a general reduction in AT8 staining (left panels, in red) while global O-GlcNAc levels (CTD110.6) are greatly increased in the TG treatment group (upper right panel, in green). Scale bar indicates 100 m.

161
Figure 5 - 6 AT8 immunoreacitivity densitometry analysis method for region of interest (ROI) selection
Stained brain and cervical spinal cord sections were analyzed by Leica DM4000B fluorescent microscope. Grey scale images were acquired by Leica DFC 350 FC digital camera. (A) On each brain sections examined, the six nonoverlapping neuroanatomic ROI were selected (three in brainstem – solid line, two from hypothalamus – dash line, and one in cortex – round dot line, respectively) at the same size of 713 x 532 µm using 20x objective lens (200x magnification), and in (B) reference lines for the ROIs were provided. (C) On each cervical spinal cord sections examined, the three nonoverlapping neuroanatomic regions (one each from ventral – solid line, intermedial – dash line, and dorsal regions – round dot line in grey matter, respectively) at the same size of 356 x 266 um using a 40 x objective lens (400 x magnification), and in (D) reference lines for the ROIs were provided. Scale bars: 500 µm.

162
Figure 5 - 7 AT8 tau phosphorylation is decreased and global O-GlcNAc levels are increased in the cervical spinal cord.
Cervical enlargement sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with a AT8 antibody (red) and a -O-GlcNAc antibody (CTD110.6, green) (middle two panels) and counter stained with DAPI (left most panels). Overlay of AT8, O-GlcNAc, and DAPI signals (right most panels) indicates that the O-GlcNAc levels are increased in the cervical spinal cord, whereas AT8 levels are decreased upon treatment with Thiamet-G. Scale bar indicates 50 m.

163
Figure 5 - 8 AT8 tau phosphorylation is decreased and global O-GlcNAc levels are increased in the hypothalamus.
Sagittal brain sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with a antibody (red) and a -O-GlcNAc antibody (CTD110.6, green) (middle two panels) and counter stained with DAPI (left most panels). Overlay of AT8, O-GlcNAc, and DAPI signals (right most panels) indicates that the O-GlcNAc levels are increased in the hypothalamus, whereas AT8 levels are decreased upon treatment with Thiamet-G. Scale bar indicates 50 m.

164
Figure 5 - 9 AT8 tau phosphorylation is decreased and global O-GlcNAc levels are increased in the cortex.
Sagittal brain sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with a AT8 antibody (red) and a -O-GlcNAc antibody (CTD110.6, green) (middle two panels) and counter stained with DAPI (left most panels). Overlay of AT8, O-GlcNAc, and DAPI signals (right most panels) indicates that the O-GlcNAc levels are increased in the cortex, whereas AT8 levels are decreased upon treatment with Thiamet-G. Scale bar indicates 50 m.

165
A
B
Figure 5 - 10 Immunohistological analysis of TG treated JNPL3 animals reveals significant reductions in NFTs.
(A-B) DAB staining of cervical spinal cord sections using the AT8 (A) and pSer422 (B) antibodies visualized using light microscopy labels structures with typical NFT morphology. Scale bar indicates 50 m.

166
Figure 5 - 11 MS/MS and MS/MS/MS spectra from an in vitro O-GlcNAc modified Tau tryptic digest identifies a site of glycosylation at Ser208.
y+ and b series fragment ions unmodified or retaining O-GlcNAc are shown for (a) MS/MS of 799.1 [M+2H]2+ and for (b) MS/MS/MS of the O-GlcNAc neutral loss ion in MS/MS at 697.5 [M+2H]2+ . c, Mulitple reaction monitoring (MRM) of the Ser400 O-GlcNAc modified tau peptide from the Thiamet-G treated mouse brain. d, MRM experiment of the Ser400 O-GlcNAc modified and Ser404 phosphorylated tau peptide. 17 MRM transitions were detected for (c) and 14 transitions were detected for (d) to correctly identify the locations of O-GlcNAc and phosphorylation on the peptide: SPVVSGDTSPR. The unique nature of this peptide results in its retention on both the C18 and graphitous carbon stationary phases, resulting in the detection of two separate sets of peaks at two distinct retention times.

167

168
Figure 5 - 12 Increased O-GlcNAc modification does not block binding of the AT8 antibody binding in the JNPL3 mouse brain.
Sagittal brain sections from 8-month Thiamet-G treated and control JNPL3 mice. Upper panel, representative images of AT8 staining before (red) and after (green) the sections were treated with a bacterial homologue of OGA (btOGA) show AT8 immunoreactivity was not altered, while the remaining O-GlcNAc staining (purple) was no longer observed by staining with the CTD110.6 antibody. Lower panel, adjacent sections digested with a catalytically inactive mutant of btOGA carried out in parallel served as experimental controls, in which representative images showing AT8 staining before (red) and after (green) were not significantly altered while the intense O-GlcNAc staining (purple) was observed in the tissue section from the Thiamet-G treated mouse. The last column of images in each panel contains merged red, green, purple signals, and overlaid with DAPI (blue). Scale bar indicates 50 m.

169
Figure 5 - 13 Immunohistological analysis of TG treated JNPL3 animals reveal significant reductions in pSer422 immunoreactivity.
(A) pSer422 immunohistochemistry (IHC) analysis reveals a similar reduction in the brainstem region of sagittal sections from TG treated JNPL3 mice. (B), Representative data from the quantitation in (A) reveals a general reduction in pSer422 staining (left panels, in red) while global O-GlcNAc levels are greatly increased in the TG treatment group (upper right panel, in green). Scale bar indicates 100 m.

170
Figure 5 - 14 PHF-1 tau immunoreactivity is decreased and global O-GlcNAc levels are increased in the brainstem of treated mice.
Sagittal brain sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with the PHF-1 antibody (red) and an -O-GlcNAc antibody (CTD110.6, green) (middle two panels) and counter stained with DAPI (left most panels). Overlay of PHF-1, O-GlcNAc, and DAPI signals (right most panels) indicates that the O-GlcNAc levels are indeed increased in the brainstem, whereas PHF-1 immunoreactivity is decreased upon treatment with Thiamet-G. Scale bar indicates 50 m.

171
Figure 5 - 15 Immunohistological analysis of TG treated JNPL3 animals reveals antibodies recognize NFTs, which have classical morphologies.
Triple immunstaining with AT8 (in red), pSer422 (in green) and CTD110.6 (global O-GlcNAc, in purple) followed by Gallyas-Braak silver staining indicates all four of these measures label typical NFT morphology in the brainstem of JNPL3 mice. Scale bar indicates 50 m.

172
Figure 5 - 16 Gallyas-Braak silver staining reveals typical NFT morphology.
Sagittal brain sections from control and 8-month Thiamet-G treated JNPL3 animals were stained by Gallyas-Braak silver staining and the brainstem region was viewed under x4 (left most panel) and x40 (right two panels, two different regions of interest) optical objective. The typical agyrophillic globose and flame shaped NFT structures that are observed in the brainstem are noticeably fewer in the 8-month Thiamet-G treated JNPL3 animals. Scale bars indicate 500 m (x4) and 50 m (x40).

173
Figure 5 - 17 Immunohistological analysis of TG treated JNPL3 animals reveals NFT immunoreactivity measured using phosphor-depedent antibody overlaps with phosphor-independent antibody known to detect tau pathology in the brainstem.
Triple immunstaining with pSer422 (in green), CTD110.6 (global O-GlcNAc, in purple), and nY29 (in red) indicates that nitrotyrosine at Tyr29 also labels typical NFT morphology in the brainstem. Scale bar indicates 50 m.

174
Figure 5 - 18 Nitrotyrosine on Y29 of tau (nY29) is reduced in the hypothalamus.
Sagittal brain sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with the pSer422 antibody (green) and a nitrotyrosine Y29 antibody (nY29, green) (middle two panels) and counter stained with DAPI (left most panels). Overlay of pSer422, nY29, DAPI, and -O-GlcNAc signals (CTD110.6, purple) (right most panels) indicates that the O-GlcNAc levels are indeed increased in the hypothalamus and pSer422 and nY29 stain similar structures with NFT morphology that are decreased upon treatment with Thiamet-G. Scale bar indicates 50 m.

175
Figure 5 - 19 Nitrotyrosine on Y29 of tau (nY29) is reduced in the cortex.
Sagittal brain sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with the pSer422 antibody (green) and a nitrotyrosine Y29 antibody (nY29, green) (middle two panels) and counter stained with DAPI (left most panels). Overlay of pSer422, nY29, DAPI, and -O-GlcNAc signals (CTD110.6, purple) (right most panels) indicates that the O-GlcNAc levels are increased in the cortex and pSer422 and nY29 stain similar structures with NFT morphology that are decreased upon treatment with Thiamet-G. Scale bar indicates 50 m.

176
Figure 5 - 20 Nitrotyrosine on Y29 of tau (nY29) is reduced in the cervical spinal cord.
Sagittal brain sections from control and 8-month Thiamet-G treated JNPL3 animals were stained with the pSer422 antibody (green) and a nitrotyrosine Y29 antibody (nY29, green) (middle two panels) and counter stained with DAPI (left most panels). Overlay of pSer422, nY29, DAPI, and -O-GlcNAc signals (CTD110.6, purple) (right most panels) indicates that the O-GlcNAc levels are increased in the cervical spinal cord and pSer422 and nY29 stain similar structures with NFT morphology that are decreased upon treatment with Thiamet-G. Scale bar indicates 50 m.

177
Figure 5 - 21 Biochemical analysis of TG treated JNPL3 animals.
a, b, Relative levels of Ser422 (a) and Ser202/Ser205 (b, AT8) phosphorylation on the high speed sedimentable 64-kDa species of tau corrected for human tau expression (HT7) and total protein (GAPDH) reveals no significant difference in phosphorylation at these epitopes in the brainstem of control and TG treated JNPL3 animals. c, Relative amounts of the 64-kDa species of human tau (detected by HT7) corrected for total protein (GAPDH) show no difference between the control and TG treated animals. d, Relative global O-GlcNAc levels (judged by CTD110.6) are increased in the brainstem of the TG treated animals compared to control. e, O-GlcNAc at Ser400 of tau is increased by 1.6 fold (judged by ELISA using the 3925 antibody). f, Relative amount of sarkosyl insoluble tau from the brainstem (as detected by the HT7 human tau antibody) are decreased in the TG treated JNPL3 animals.

178
Figure 5 - 22 Total and phosphorylated tau levels on the 64-kDa human tau species.
The brainstem region from control and 8-month Thiamet-G treated JNPL3 animals was extracted and centrifuged at 100,000 x g (see methods for further details). The pellet was then resuspended and immunoblots using a human tau antibody (HT7) as well as various phosphorylation specific tau antibodies we carried out. All of these antibodies detect exclusively the 64-kDa species of human tau and reveal no difference in the total amount of human tau (HT7) or the level of phosphorylation on this tau species.

179
Figure 5 - 23 Tau Isoforms and Constructs.
The longest human isoform of tau is generated from the microtubule associated protein tau (MAPT) gene by alternative splicing of exons 2, 3, and 10. This results in an isoform which is 441 amino acids in length and contains two N-terminal inserts and four microtubule binding repeats (MTBRs). The Tau244-441 construct used in this study contains the four MTBRs and the C-terminal region but lacks the majority of the N-terminus.

180
Figure 5 - 24 O-GlcNAc on Tau244-441 slows aggregation in vitro.
a, Coomassie Blue (CB) stained SDS-PAGE gel. Ctrl Tau244-441 (lane 1), Mod Tau244-441 (lane 2), Ctrl S400A Tau244-441 (lane 3) and Mod S400A Tau244-441 (lane 4) species all have apparent molecular weight of ~25 kDa. Only the Mod Tau244-441 and not the Mod S400A Tau244-441 are detected by the O-GlcNAc at Ser400 of tau antibody (3925) immunoblot while both are detected by the global O-GlcNAc antibody (CTD110.6). Tau-46 immunoblot indicates equal loading of each of these species. b, Relative thioflavin-S (ThS) fluorescence of both Ctrl and Mod Tau244-441 (5 M ) over time. The Mod Tau244-441 shows a slower rate and extent of polymerization (error bars indicate +/- S.E.M.) using heparin as the inducer. c, Relative thioflavin-S (ThS) fluorescence of both Ctrl and Mod S400A Tau244-441 (5 M) aggregate at the same rate and to the same extent over time using heparin as the inducer (error bars indicate +/- S.E.M.). d, Filter trap assay of Ctrl and Mod Tau244-441 (5 M) aggregated for 40 hours. The Mod Tau244-441 showed less retained tau aggregates as detected by the Tau46 antibody. A representative blot is shown to the right and the quantitation of two such experiments is shown to the left.

181
Figure 5 - 25 In vitro Tau244-441 aggregation using arachidonic acid as the inducer.
a, Relative thioflavin-S (ThS) fluorescence of both Ctrl and Mod Tau244-441 (5 M ) over time. The Mod Tau244-441 shows a slower rate and extent of aggregation (error bars indicate +/- S.E.M.) than the Ctrl using 75 M arachidonic acid as the inducer. b, A representative filter trap blot is shown for the aggregation of Ctrl and Mod wt Tau244-441 (5 m) using heparin as the inducer.

182
Figure 5 - 26 Production of O-GlcNAc modified sTAB1 inhibits its aggregation in vitro.
Coexpression of a deletion construct consisting of residues 7-402 of short TAB1 (sTAB1) or with WT or mutant (H558A) OGT. (Upper panels) Coomassie stained gel of sTAB1 obtained after purification by nickel chelate chromatography shows that the proteins are of high purity (>95% purity). (Lower panels) immunoblot analyses using an anti-O-GlcNAc antibody (RL2) demonstrate that only sTAB1 coexpressed with WT OGT is modified. b, A turbidity assay was carried out whereby the absorbance of solutions containing sTAB1 was monitored continuously over time at 500 nm in 50 mM tris, 100 mM sodium chloride, and 5 mM -mercaptoethanol (pH 7.5). Assays were carried out with O-GlcNAc-modified (blue) and unmodified sTAB1 (red). As indicated in the figure, the assays were repeated at two different temperature and protein concentrations: 40 M at 40 C or 10 M at 45 C. Data was acquired every five seconds over the course of the experiment and curves represent the average of three replicates. The standard deviation at each time point ranges from 1-5%.

183
5.7 Reference List
[1] C.S. Arnold, G.V. Johnson, R.N. Cole, D.L. Dong, M. Lee, G.W. Hart, The microtubule-associated protein tau is extensively modified with O-linked N-acetylglucosamine, J Biol Chem 271 (1996) 28741-28744.
[2] L.M. Bierer, P.R. Hof, D.P. Purohit, L. Carlin, J. Schmeidler, K.L. Davis, D.P. Perl, Neocortical neurofibrillary tangles correlate with dementia severity in Alzheimer's disease, Archives of neurology 52 (1995) 81-88.
[3] H. Braak, E. Braak, T. Ohm, J. Bohl, Silver impregnation of Alzheimer's neurofibrillary changes counterstained for basophilic material and lipofuscin pigment, Stain technology 63 (1988) 197-200.
[4] J.C. Carroll, E.R. Rosario, L. Chang, F.Z. Stanczyk, S. Oddo, F.M. LaFerla, C.J. Pike, Progesterone and estrogen regulate Alzheimer-like neuropathology in female 3xTg-AD mice, J Neurosci 27 (2007) 13357-13365.
[5] N. Cetinbas, M.S. Macauley, K.A. Stubbs, R. Drapala, D.J. Vocadlo, Identification of Asp174 and Asp175 as the key catalytic residues of human O-GlcNAcase by functional analysis of site-directed mutants, Biochemistry 45 (2006) 3835-3844.
[6] A.J. Clarke, R. Hurtado-Guerrero, S. Pathak, A.W. Schuttelkopf, V. Borodkin, S.M. Shepherd, A.F.M. Ibrahim, D.M.F. van Aalten, Structural insights into mechanism and specificity of O-GlcNAc transferase, EMBO J 27 (2008) 2780-2788.
[7] R.J. Dennis, E.J. Taylor, M.S. Macauley, K.A. Stubbs, J.P. Turkenburg, S.J. Hart, G.N. Black, D.J. Vocadlo, G.J. Davies, Structure and mechanism of a bacterial beta-glucosaminidase having O-GlcNAcase activity, Nature structural & molecular biology 13 (2006) 365-371.
[8] D.L. Dong, G.W. Hart, Purification and characterization of an O-GlcNAc selective N-acetyl-beta-D-glucosaminidase from rat spleen cytosol, J Biol Chem 269 (1994) 19321-19330.
[9] P. Friedhoff, A. Schneider, E.M. Mandelkow, E. Mandelkow, Rapid assembly of Alzheimer-like paired helical filaments from microtubule-associated protein tau monitored by fluorescence in solution, Biochemistry 37 (1998) 10223-10230.
[10] Y. Gao, L. Wells, F.I. Comer, G.J. Parker, G.W. Hart, Dynamic O-glycosylation of nuclear and cytosolic proteins: cloning and characterization of a neutral, cytosolic beta-N-acetylglucosaminidase from human brain, J Biol Chem 276 (2001) 9838-9845.
[11] R. Godemann, J. Biernat, E. Mandelkow, E.M. Mandelkow, Phosphorylation of tau protein by recombinant GSK-3beta: pronounced phosphorylation at select Ser/Thr-Pro motifs but no phosphorylation at Ser262 in the repeat domain, FEBS Lett 454 (1999) 157-164.
[12] M. Goedert, M. Hasegawa, R. Jakes, S. Lawler, A. Cuenda, P. Cohen, Phosphorylation of microtubule-associated protein tau by stress-activated protein kinases, FEBS Lett 409 (1997) 57-62.
[13] M. Goedert, R. Jakes, R.A. Crowther, Effects of frontotemporal dementia FTDP-17 mutations on heparin-induced assembly of tau filaments, FEBS Lett 450 (1999) 306-311.

184
[14] M. Goedert, R. Jakes, M.G. Spillantini, M. Hasegawa, M.J. Smith, R.A. Crowther, Assembly of microtubule-associated protein tau into Alzheimer-like filaments induced by sulphated glycosaminoglycans, Nature 383 (1996) 550-553.
[15] C.X. Gong, F. Liu, I. Grundke-Iqbal, K. Iqbal, Post-translational modifications of tau protein in Alzheimer's disease, J Neural Transm 112 (2005) 813-838.
[16] D.P. Hanger, B.H. Anderton, W. Noble, Tau phosphorylation: the therapeutic challenge for neurodegenerative disease, Trends in molecular medicine 15 (2009) 112-119.
[17] M. Hutton, C.L. Lendon, P. Rizzu, M. Baker, S. Froelich, H. Houlden, S. Pickering-Brown, S. Chakraverty, A. Isaacs, A. Grover, J. Hackett, J. Adamson, S. Lincoln, D. Dickson, P. Davies, R.C. Petersen, M. Stevens, E. de Graaff, E. Wauters, J. van Baren, M. Hillebrand, M. Joosse, J.M. Kwon, P. Nowotny, L.K. Che, J. Norton, J.C. Morris, L.A. Reed, J. Trojanowski, H. Basun, L. Lannfelt, M. Neystat, S. Fahn, F. Dark, T. Tannenberg, P.R. Dodd, N. Hayward, J.B. Kwok, P.R. Schofield, A. Andreadis, J. Snowden, D. Craufurd, D. Neary, F. Owen, B.A. Oostra, J. Hardy, A. Goate, J. van Swieten, D. Mann, T. Lynch, P. Heutink, Association of missense and 5'-splice-site mutations in tau with the inherited dementia FTDP-17, Nature 393 (1998) 702-705.
[18] A. Korneyev, L. Binder, J. Bernardis, Rapid reversible phosphorylation of rat brain tau proteins in response to cold water stress, Neurosci Lett 191 (1995) 19-22.
[19] L.K. Kreppel, M.A. Blomberg, G.W. Hart, Dynamic glycosylation of nuclear and cytosolic proteins. Cloning and characterization of a unique O-GlcNAc transferase with multiple tetratricopeptide repeats, J Biol Chem 272 (1997) 9308-9315.
[20] S. Le Corre, H.W. Klafki, N. Plesnila, G. Hubinger, A. Obermeier, H. Sahagun, B. Monse, P. Seneci, J. Lewis, J. Eriksen, C. Zehr, M. Yue, E. McGowan, D.W. Dickson, M. Hutton, H.M. Roder, An inhibitor of tau hyperphosphorylation prevents severe motor impairments in tau transgenic mice, Proc Natl Acad Sci U S A 103 (2006) 9673-9678.
[21] T. Lefebvre, S. Ferreira, L. Dupont-Wallois, T. Bussiere, M.J. Dupire, A. Delacourte, J.C. Michalski, M.L. Caillet-Boudin, Evidence of a balance between phosphorylation and O-GlcNAc glycosylation of Tau proteins--a role in nuclear localization, Biochim Biophys Acta 1619 (2003) 167-176.
[22] J. Lewis, E. McGowan, J. Rockwood, H. Melrose, P. Nacharaju, M. Van Slegtenhorst, K. Gwinn-Hardy, M. Paul Murphy, M. Baker, X. Yu, K. Duff, J. Hardy, A. Corral, W.L. Lin, S.H. Yen, D.W. Dickson, P. Davies, M. Hutton, Neurofibrillary tangles, amyotrophy and progressive motor disturbance in mice expressing mutant (P301L) tau protein, Nat Genet 25 (2000) 402-405.
[23] F. Liu, K. Iqbal, I. Grundke-Iqbal, G.W. Hart, C.X. Gong, O-GlcNAcylation regulates phosphorylation of tau: a mechanism involved in Alzheimer's disease, Proc Natl Acad Sci U S A 101 (2004) 10804-10809.
[24] F. Liu, J. Shi, H. Tanimukai, J. Gu, J. Gu, I. Grundke-Iqbal, K. Iqbal, C.X. Gong, Reduced O-GlcNAcylation links lower brain glucose metabolism and tau pathology in Alzheimer's disease, Brain 132 (2009) 1820-1832.

185
[25] Y. Liu, F. Liu, I. Grundke-Iqbal, K. Iqbal, C.X. Gong, Brain glucose transporters, O-GlcNAcylation and phosphorylation of tau in diabetes and Alzheimer's disease, J Neurochem 111 (2009) 242-249.
[26] W.A. Lubas, D.W. Frank, M. Krause, J.A. Hanover, O-Linked GlcNAc transferase is a conserved nucleocytoplasmic protein containing tetratricopeptide repeats, J Biol Chem 272 (1997) 9316-9324.
[27] S. Marshall, V. Bacote, R.R. Traxinger, Discovery of a metabolic pathway mediating glucose-induced desensitization of the glucose transport system. Role of hexosamine biosynthesis in the induction of insulin resistance, J Biol Chem 266 (1991) 4706-4712.
[28] J.E. McKenna, G.T. Prusky, I.Q. Whishaw, Cervical motoneuron topography reflects the proximodistal organization of muscles and movements of the rat forelimb: a retrograde carbocyanine dye analysis, The Journal of comparative neurology 419 (2000) 286-296.
[29] M. Neumann, D.M. Sampathu, L.K. Kwong, A.C. Truax, M.C. Micsenyi, T.T. Chou, J. Bruce, T. Schuck, M. Grossman, C.M. Clark, L.F. McCluskey, B.L. Miller, E. Masliah, I.R. Mackenzie, H. Feldman, W. Feiden, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Science (New York, N.Y 314 (2006) 130-133.
[30] S.C. Papasozomenos, Y. Su, Altered phosphorylation of tau protein in heat-shocked rats and patients with Alzheimer disease, Proc Natl Acad Sci U S A 88 (1991) 4543-4547.
[31] M. Pickhardt, Z. Gazova, M. von Bergen, I. Khlistunova, Y. Wang, A. Hascher, E.M. Mandelkow, J. Biernat, E. Mandelkow, Anthraquinones inhibit tau aggregation and dissolve Alzheimer's paired helical filaments in vitro and in cells, J Biol Chem 280 (2005) 3628-3635.
[32] M.R. Reynolds, J.F. Reyes, Y. Fu, E.H. Bigio, A.L. Guillozet-Bongaarts, R.W. Berry, L.I. Binder, Tau nitration occurs at tyrosine 29 in the fibrillar lesions of Alzheimer's disease and other tauopathies, J Neurosci 26 (2006) 10636-10645.
[33] E.P. Roquemore, M.R. Chevrier, R.J. Cotter, G.W. Hart, Dynamic O-GlcNAcylation of the small heat shock protein alpha B-crystallin, Biochemistry 35 (1996) 3578-3586.
[34] N. Sahara, J. Lewis, M. DeTure, E. McGowan, D.W. Dickson, M. Hutton, S.H. Yen, Assembly of tau in transgenic animals expressing P301L tau: alteration of phosphorylation and solubility, J Neurochem 83 (2002) 1498-1508.
[35] K. Santacruz, J. Lewis, T. Spires, J. Paulson, L. Kotilinek, M. Ingelsson, A. Guimaraes, M. DeTure, M. Ramsden, E. McGowan, C. Forster, M. Yue, J. Orne, C. Janus, A. Mariash, M. Kuskowski, B. Hyman, M. Hutton, K.H. Ashe, Tau suppression in a neurodegenerative mouse model improves memory function, Science 309 (2005) 476-481.
[36] D. Schubert, Glucose metabolism and Alzheimer's disease, Ageing research reviews 4 (2005) 240-257.
[37] J.M. Shulman, P. Chipendo, L.B. Chibnik, C. Aubin, D. Tran, B.T. Keenan, P.L. Kramer, J.A. Schneider, D.A. Bennett, M.B. Feany, P.L. De Jager, Functional

186
Screening of Alzheimer Pathology Genome-wide Association Signals in Drosophila, The American Journal of Human Genetics 88 (2011) 232-238.
[38] B. Srikanth, M.M. Vaidya, R.D. Kalraiya, O-GlcNAcylation determines the solubility, filament organization, and stability of keratins 8 and 18, J Biol Chem 285 (2010) 34062-34071.
[39] T. Taniguchi, M. Sumida, S. Hiraoka, K. Tomoo, T. Kakehi, K. Minoura, S. Sugiyama, K. Inaka, T. Ishida, N. Saito, C. Tanaka, Effects of different anti-tau antibodies on tau fibrillogenesis: RTA-1 and RTA-2 counteract tau aggregation, FEBS Lett 579 (2005) 1399-1404.
[40] Y. Uno, H. Iwashita, T. Tsukamoto, N. Uchiyama, T. Kawamoto, M. Kori, A. Nakanishi, Efficacy of a novel, orally active GSK-3 inhibitor 6-Methyl-N-[3-[[3-(1-methylethoxy)propyl]carbamoyl]-1H-pyrazol-4-yl]pyridi ne-3-carboxamide in tau transgenic mice, Brain Res 1296 (2009) 148-163.
[41] V. Vogelsberg-Ragaglia, J. Bruce, C. Richter-Landsberg, B. Zhang, M. Hong, J.Q. Trojanowski, V.M. Lee, Distinct FTDP-17 missense mutations in tau produce tau aggregates and other pathological phenotypes in transfected CHO cells, Molecular biology of the cell 11 (2000) 4093-4104.
[42] Z. Wang, N.D. Udeshi, M. O'Malley, J. Shabanowitz, D.F. Hunt, G.W. Hart, Enrichment and site mapping of O-linked N-acetylglucosamine by a combination of chemical/enzymatic tagging, photochemical cleavage, and electron transfer dissociation mass spectrometry, Mol Cell Proteomics 9 (2010) 153-160.
[43] C.L. Weaver, M. Espinoza, Y. Kress, P. Davies, Conformational change as one of the earliest alterations of tau in Alzheimer's disease, Neurobiology of aging 21 (2000) 719-727.
[44] D.M. Wilson, L.I. Binder, Free fatty acids stimulate the polymerization of tau and amyloid beta peptides. In vitro evidence for a common effector of pathogenesis in Alzheimer's disease, The American journal of pathology 150 (1997) 2181-2195.
[45] S.A. Yuzwa, M.S. Macauley, J.E. Heinonen, X. Shan, R.J. Dennis, Y. He, G.E. Whitworth, K.A. Stubbs, E.J. McEachern, G.J. Davies, D.J. Vocadlo, A potent mechanism-inspired O-GlcNAcase inhibitor that blocks phosphorylation of tau in vivo, Nat Chem Biol 4 (2008) 483-490.
[46] S.A. Yuzwa, A.K. Yadav, Y. Skorobogatko, T. Clark, K. Vosseller, D.J. Vocadlo, Mapping O-GlcNAc modification sites on tau and generation of a site-specific O-GlcNAc tau antibody, Amino acids 40 (2011) 857-868.
[47] N.E. Zachara, N. O'Donnell, W.D. Cheung, J.J. Mercer, J.D. Marth, G.W. Hart, Dynamic O-GlcNAc modification of nucleocytoplasmic proteins in response to stress. A survival response of mammalian cells, J Biol Chem 279 (2004) 30133-30142.

187
6: Summary, conclusions, and future studies
6.1 Summary
The objective of this work was to investigate the presence and potential function of the
O-GlcNAc post-translational modification on various proteins implicated in
neurodegeneration. This research specifically focussed on (i) examining changes in O-
GlcNAc levels in the spinal cord of the G93A mSOD mouse model of ALS as compared
to control animals; (ii) characterizing the pathology and identifying O-GlcNAc
modification sites on the protein TDP-43 using the G93A mSOD mouse model and
proteomics studies; and (iii) exploring the potential neuronal protective effects of
increasing O-GlcNAc levels by using the highly selective OGA inhibitor Thiamet-G in
the transgenic JNPL3 tau mouse model of AD.
In Chapter 2, the normal distribution of O-GlcNAc in the mSOD spinal cord was studied
along with changes arising from disease progression. The following observations were
made. (1) O-GlcNAc levels were reduced in the spinal cord of mSOD mice as compared
to controls. Specifically, this reduction in O-GlcNAc levels was prominent in the
surviving motor neurons located in the ventral horn region of the spinal cord suggesting
the neurodegeneration found in mSOD mice might be associated with a reduction of O-
GlcNAc levels in motor neurons. (2) Three days of acute oral treatment of NButGT
dramatically enhanced O-GlcNAc levels in the spinal cord of both mSOD and control
mice. In addition, NButGT increased levels of O-GlcNAc modified NFM in spinal cords

188
of control mice indicating that O-GlcNAc levels can be modulated in vivo to increase
levels of O-GlcNAc on NFM, however, alterations in the levels of O-GlcNAc modified
NFM or levels of phosphorylated NFM were not observed in mSOD mice. We speculate
that the inability of OGA inhibition to alter NFM phosphorylation and glycosylation in
mSOD mice may stem from the aggregated form of hyperphosphorylated NFs in these
end-stage mice [3], which may prevent access of enzymes that act to dephosphorylated
and subsequently modify O-GlcNAc.
As detailed in Chapter 3, my work using G93A mSOD mice demonstrated that
mislocalization of TDP-43 occurred in aged mice having an extended life span, thereby
providing new insight into the potential association of SOD1 and TDP-43. This idea has
gained further support from a recent study showing that mutant SOD1, but not wild-type
SOD1, physically interacts with TDP-43, as demonstrated by co-immunoprecipitation
assays in G93A mSOD1 transgenic mice [10]. Furthermore, in this new study, cell
fractionation assays using cultured cells showed that mutant SOD1 is localized in the
cytosolic fraction but not in the nuclear fraction, suggesting that mutant SOD1 interacts
with TDP-43 in the cytoplasm [10].
In Chapter 4, I demonstrated that mouse brain TDP-43 is O-GlcNAc modified.
Furthermore, in collaboration with my colleague, Tom Clark, we mapped four O-GlcNAc
modification sites on recombinant full-length human TDP-43 that are localized to its C-
terminal region, which has been shown to be a region involved in disease specific
phosphorylation [2, 27]. Notably, all four O-GlcNAc sites are also sites that can be

189
phosphorylated by CK1 [14]. This observation suggests that O-GlcNAc modification has
the potential to influence the abnormal phosphorylation of TDP-43 in ALS and other
neurodegenerative diseases [1, 9, 22].
In Chapter 5, in collaboration with my colleague, Scott Yuzwa, we demonstrated that
treating a JNPL3 mouse model of AD with Thiamet-G for 36 weeks increased tau O-
GlcNAc modification, hindered tau aggregation, and protected mice against neuronal cell
loss. However, we did not observe differences in the extent of tau phosphorylation
between treated and control animals. To explore the possible mechanisms that underlie
these observations, we used in vitro biochemical assays to show that O-GlcNAc
modification of tau slows its aggregation.
6.2 Conclusions
Protein hyperphosphorylation and protein aggregation are common features found in
many neurodegenerative diseases including ALS and AD [7, 15, 16, 24]. Many disease
associated proteins, such as NF and TDP-43 linked to ALS, and tau linked to AD are
found to be hyperphosphorylated and aggregated in both patients and animal models,
including the G93A mSOD mouse model for ALS and the JNPL3 mouse model for AD
[1, 8, 18, 22, 24]. However, the exact molecular mechanisms contributing to progression
of these devastating neurodegenerative diseases are not clear, nor are the processes that
might lead to abnormal protein phosphorylation and/or aggregation.

190
Previous evidence suggests that there is either aberrant protein kinase expression or
activity in ALS and AD, such as CdK5 [23] and GSK3 [12], or other altered post-
translational modifications, such as O-GlcNAc, that may contribute to abnormal protein
modification states that are associated with disease progression [19, 20]. Both, NF and
tau proteins have previously been shown to be O-GlcNAc modified which stimulated our
interest in these proteins [5, 19]. Hyperphosphorylation of TDP-43 is one of the
characteristic features of TDP-43 pathology [11, 21]. In Chapter 4, we showed that
mouse brain TDP-43 was also O-GlcNAc modified, and four O-GlcNAc mapped sites on
recombinant TDP-43 overlapped with known phosphorylated sites. A reciprocal
relationship between phosphorylation and O-GlcNAc modification has been suggested on
some proteins, including NF and tau [4, 19]. Thus, decreases in O-GlcNAc levels of these
proteins may be one factors contributing to abnormal protein phosphorylation and
aggregation in these neurodegenerative diseases. In Chapter 2, I observed a reduction in
both global and NF O-GlcNAc modified protein levels in G93A mSOD mice, which
providesis consistent with the above idea.
The literature offers considerable support for the idea that hyperphosphorylation of tau
plays a role in its aggregation. O-GlcNAc modification of tau has been shown to be
reciprocal to its phosphorylation in certain models, and increasing O-GlcNAc levels leads
to reductions in tau phosphorylation in healthy animals [4, 26]. It has also been shown
that increasing O-GlcNAc levels using the OGA inhibitor Thiamet-G correlates with
decreased tau phosphorylation at pathologically relevant sites in the brains of healthy rats
[25]. Thus, as discussed in Chapter 5, we conducted a long-term study in which we

191
treated JNPL3 mice, a model of the tauopathic component of AD, with Thiamet-G and
observed increased tau O-GlcNAc modification, reduced levels of tau aggregation, and
decreased levels of neurodegeneration. Together with in vitro biochemical assays that
showed O-GlcNAc modification of tau slowed its aggregation, these findings suggested
that a basic biochemical role of O-GlcNAc might be to promote the stability of proteins
against aggregation, and hence increased O-GlcNAc may contribute to an overall
protective effect. These results also provide strong support for OGA and its role in O-
GlcNAc processing as viable therapeutic targets that might offer the opportunity to alter
disease progression in AD and offer benefits in other disease states involving aggregation
of proteins that can be O-GlcNAc modified.
These studies support and extend previous observations showing that proteins known to
aggregate in the neurodegenerative diseases, including the tauopathies and ALS as
studied here, are O-GlcNAc modified. Global O-GlcNAc levels are decreased during the
process of neurodegeneration in the mSOD1 G93A model, consistent with observations
made previously in multiple models. We find that O-GlcNAc levels can be modulated in
vivo in the brain of animal models, offering a route to evaluating the value of increasing
O-GlcNAc levels and the resulting impact on the progression of neurodegeneration [25].
In the JNPL3 mouse model we find such treatment slows neurodegeneration, likely
mediated at least in part by the ability for O-GlcNAc to protect tau against aggregation to
form oligomers and downstream tangles. Collectively, these data provide encouraging
data that should stimulate further studies evaluating the role of O-GlcNAc in various
neurodegenerative diseases.

192
6.3 Future studies
To further explore a possible role for O-GlcNAc on TDP-43 in ALS, additional
functional studies of O-GlcNAc on TDP-43 need to be investigated. I have established a
cellular model system that will enable the pursuit of these functional studies (Appendices
A-B). In this cellular model, wild-type and mutant full-length TDP-43 were subcloned
into a pCMV vector containing a Myc tag. After independent transient transfection of the
resulting vectors into SK-N-SH cells (a neuroblastoma cell line), diffused cytosolic TDP-
43 was observed to gradually accumulate normally in the nucleus. In contrast, the N390D
mutant TDP-43 predominantly accumulated in the cytoplasm and appeared to form
aggregates in transfected cells. Using this cellular system, we expect that we can now
monitor cellular viability, spatial and temporal distribution or aggregation of TDP-43 in
cells under conditions of cellular stress and make comparisons between wild-type TDP-
43, TDP-43 with the O-GlcNAc serine residues mutated, as well as a mutant TDP-43 that
is known to aggregate, such as the N390D mutant I have already generated.
A preliminary in vitro examination of the role of O-GlcNAc on the stabilization of TDP-
43 protein has also been carried out (Appendices C-D). Using the method described in
Chapter 4, recombinant His6-tagged TDP-43 was produced and partially purified in both
its O-GlcNAc modified and unmodified forms. The effect of O-GlcNAc on the stability
of TDP-43 was determined by a turbidity assay with the help of Matthew Macauley, a
former colleague in our laboratory. The aggregation rate of TDP-43 was monitored using
absorbance spectrophotometry at a wavelength of 500 nm. At 45 C, the unmodified
TDP-43 aggregated at a markedly faster rate than the O-GlcNAc modified protein

193
(Appendices C-D). These results suggests that the presence of the O-GlcNAc
modification on TDP-43 might affect the kinetics of aggregation, thus supporting the idea
that O-GlcNAc modification of TDP-43 can enhance its stability in vitro and suggesting
this may occur in cells. Though the protein concentrations were standardized to avoid the
possibility that differences in initial protein concentration contributed to the result, a
major limitation of this study is the presence of some degradation products, which have
may contributed to the differences observed. Therefore, to confirm this result, future
turbidity assays need to be carried out using highly purified TDP-43 protein, perhaps
generated by using size exclusion gel filtration chromatography or ion-exchange
chromatography. In addition, examining the aggregation of mutant forms of TDP-43
lacking glycosylation sites, yet expressed using the same recombinant system in the
presence of active or inactive OGT, should be informative.
One obvious limitation of the study performed in Chapter 5 is that the use of the JNPL3
animal model does not fully recapitulate the neuropathology in AD. APP has been shown
to be O-GlcNAc modified [6], and a recent study suggested that O-GlcNAc levels affect
APP processing [13]. Thus, in addition to tau, APP could be another potential target of
Thiamet-G treatment. Therefore, to further explore the potential beneficial effects from
Thiamet-G treatment, future studies should employ the TAPP bi-transgenic mouse model
of AD, which expresses both a human P301L tau mutation and the human APP with a
double Swedish mutation that exhibits both NFT and AP [17]. This model could be used
for testing the therapeutic potential of OGA inhibition by monitoring neuronal loss, NFT,
and Aβ production.

194
6.4 Reference List
[1] T. Arai, M. Hasegawa, H. Akiyama, K. Ikeda, T. Nonaka, H. Mori, D. Mann, K. Tsuchiya, M. Yoshida, Y. Hashizume, T. Oda, TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Biochem Biophys Res Commun 351 (2006) 602-611.
[2] O.A. Brady, P. Meng, Y. Zheng, Y. Mao, F. Hu, Regulation of TDP-43 aggregation by phosphorylation and p62/SQSTM1, J Neurochem 116 (2011) 248-259.
[3] M.C. Dal Canto, M.E. Gurney, Neuropathological changes in two lines of mice carrying a transgene for mutant human Cu,Zn SOD, and in mice overexpressing wild type human SOD: a model of familial amyotrophic lateral sclerosis (FALS), Brain research 676 (1995) 25-40.
[4] Y. Deng, B. Li, F. Liu, K. Iqbal, I. Grundke-Iqbal, R. Brandt, C.X. Gong, Regulation between O-GlcNAcylation and phosphorylation of neurofilament-M and their dysregulation in Alzheimer disease, FASEB J 22 (2008) 138-145.
[5] D.L. Dong, Z.S. Xu, M.R. Chevrier, R.J. Cotter, D.W. Cleveland, G.W. Hart, Glycosylation of mammalian neurofilaments. Localization of multiple O-linked N-acetylglucosamine moieties on neurofilament polypeptides L and M, J Biol Chem 268 (1993) 16679-16687.
[6] L.S. Griffith, M. Mathes, B. Schmitz, Beta-amyloid precursor protein is modified with O-linked N-acetylglucosamine, J Neurosci Res 41 (1995) 270-278.
[7] I. Grundke-Iqbal, K. Iqbal, M. Quinlan, Y.C. Tung, M.S. Zaidi, H.M. Wisniewski, Microtubule-associated protein tau. A component of Alzheimer paired helical filaments, J Biol Chem 261 (1986) 6084-6089.
[8] I. Grundke-Iqbal, K. Iqbal, Y.C. Tung, M. Quinlan, H.M. Wisniewski, L.I. Binder, Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology, Proc Natl Acad Sci U S A 83 (1986) 4913-4917.
[9] M. Hasegawa, T. Arai, T. Nonaka, F. Kametani, M. Yoshida, Y. Hashizume, T.G. Beach, E. Buratti, F. Baralle, M. Morita, I. Nakano, T. Oda, K. Tsuchiya, H. Akiyama, Phosphorylated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Ann Neurol 64 (2008) 60-70.
[10] S. Higashi, Y. Tsuchiya, T. Araki, K. Wada, T. Kabuta, TDP-43 physically interacts with amyotrophic lateral sclerosis-linked mutant CuZn superoxide dismutase, Neurochemistry international 57 (2010) 906-913.
[11] Y. Inukai, T. Nonaka, T. Arai, M. Yoshida, Y. Hashizume, T.G. Beach, E. Buratti, F.E. Baralle, H. Akiyama, S. Hisanaga, M. Hasegawa, Abnormal phosphorylation of Ser409/410 of TDP-43 in FTLD-U and ALS, FEBS Lett 582 (2008) 2899-2904.
[12] K. Iqbal, C. Alonso Adel, S. Chen, M.O. Chohan, E. El-Akkad, C.X. Gong, S. Khatoon, B. Li, F. Liu, A. Rahman, H. Tanimukai, I. Grundke-Iqbal, Tau pathology in Alzheimer disease and other tauopathies, Biochim Biophys Acta 1739 (2005) 198-210.

195
[13] K.T. Jacobsen, K. Iverfeldt, O-GlcNAcylation increases non-amyloidogenic processing of the amyloid-beta precursor protein (APP), Biochem Biophys Res Commun 404 (2011) 882-886.
[14] F. Kametani, T. Nonaka, T. Suzuki, T. Arai, N. Dohmae, H. Akiyama, M. Hasegawa, Identification of casein kinase-1 phosphorylation sites on TDP-43, Biochem Biophys Res Commun 382 (2009) 405-409.
[15] E. Kopke, Y.C. Tung, S. Shaikh, A.C. Alonso, K. Iqbal, I. Grundke-Iqbal, Microtubule-associated protein tau. Abnormal phosphorylation of a non-paired helical filament pool in Alzheimer disease, J Biol Chem 268 (1993) 24374-24384.
[16] C. Krieger, J.H. Hu, S. Pelech, Aberrant protein kinases and phosphoproteins in amyotrophic lateral sclerosis, Trends Pharmacol Sci 24 (2003) 535-541.
[17] J. Lewis, D.W. Dickson, W.L. Lin, L. Chisholm, A. Corral, G. Jones, S.H. Yen, N. Sahara, L. Skipper, D. Yager, C. Eckman, J. Hardy, M. Hutton, E. McGowan, Enhanced neurofibrillary degeneration in transgenic mice expressing mutant tau and APP, Science (New York, N.Y 293 (2001) 1487-1491.
[18] J. Lewis, E. McGowan, J. Rockwood, H. Melrose, P. Nacharaju, M. Van Slegtenhorst, K. Gwinn-Hardy, M. Paul Murphy, M. Baker, X. Yu, K. Duff, J. Hardy, A. Corral, W.L. Lin, S.H. Yen, D.W. Dickson, P. Davies, M. Hutton, Neurofibrillary tangles, amyotrophy and progressive motor disturbance in mice expressing mutant (P301L) tau protein, Nat Genet 25 (2000) 402-405.
[19] F. Liu, K. Iqbal, I. Grundke-Iqbal, G.W. Hart, C.X. Gong, O-GlcNAcylation regulates phosphorylation of tau: a mechanism involved in Alzheimer's disease, Proc Natl Acad Sci U S A 101 (2004) 10804-10809.
[20] N. Ludemann, A. Clement, V.H. Hans, J. Leschik, C. Behl, R. Brandt, O-glycosylation of the tail domain of neurofilament protein M in human neurons and in spinal cord tissue of a rat model of amyotrophic lateral sclerosis (ALS), J Biol Chem 280 (2005) 31648-31658.
[21] M. Neumann, L.K. Kwong, E.B. Lee, E. Kremmer, A. Flatley, Y. Xu, M.S. Forman, D. Troost, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Phosphorylation of S409/410 of TDP-43 is a consistent feature in all sporadic and familial forms of TDP-43 proteinopathies, Acta neuropathologica 117 (2009) 137-149.
[22] M. Neumann, D.M. Sampathu, L.K. Kwong, A.C. Truax, M.C. Micsenyi, T.T. Chou, J. Bruce, T. Schuck, M. Grossman, C.M. Clark, L.F. McCluskey, B.L. Miller, E. Masliah, I.R. Mackenzie, H. Feldman, W. Feiden, H.A. Kretzschmar, J.Q. Trojanowski, V.M. Lee, Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis, Science (New York, N.Y 314 (2006) 130-133.
[23] M.D. Nguyen, R.C. Lariviere, J.P. Julien, Deregulation of Cdk5 in a mouse model of ALS: toxicity alleviated by perikaryal neurofilament inclusions, Neuron 30 (2001) 135-147.
[24] M.J. Strong, S. Kesavapany, H.C. Pant, The pathobiology of amyotrophic lateral sclerosis: a proteinopathy?, J Neuropathol Exp Neurol 64 (2005) 649-664.
[25] S.A. Yuzwa, M.S. Macauley, J.E. Heinonen, X. Shan, R.J. Dennis, Y. He, G.E. Whitworth, K.A. Stubbs, E.J. McEachern, G.J. Davies, D.J. Vocadlo, A potent

196
mechanism-inspired O-GlcNAcase inhibitor that blocks phosphorylation of tau in vivo, Nat Chem Biol 4 (2008) 483-490.
[26] S.A. Yuzwa, D.J. Vocadlo, O-GlcNAc modification and the tauopathies: insights from chemical biology, Curr Alzheimer Res 6 (2009) 451-454.
[27] Y.J. Zhang, T.F. Gendron, Y.F. Xu, L.W. Ko, S.H. Yen, L. Petrucelli, Phosphorylation regulates proteasomal-mediated degradation and solubility of TAR DNA binding protein-43 C-terminal fragments, Molecular neurodegeneration 5 (2010) 33.

197
Appendices

198
Appendix A
Characterization of the cellular localization of WT and N390d mutant TDP-43 in SK-N-SK human neuroblastoma cells. Cells were transfected for 6h, 12h, and 24h with pCMV vectors containing Myc-tagged wild-type (WT) and N390D mutant TDP-43. Cells were visualized using mouse anti-Myc (green, for exogenous TDP-43), rabbit anti-TDP-43 (red, for both of endogenous and exogenous TDP-43), and DAPI (blue, nuclear marker). 24 hr after transefction, WT TDP-43 mainly localized into the nucleus with a diffuse cytoplasmic distribution. N390D mutant TDP-43 mainly accumulated and/or aggregated within the cytoplasm.

199
Appendix B
Methods for SK-N-SH cellular model Subcloning the full-length human TDP-43 into the pCMV-Myc vector
The gene encoding TDP-43 was amplified using essentially the same set of primers used
to clone it into pET28a except the primers contained a 5 SalI cut site and a 3 NotI cut
site to enable ligation into the pCMV-myc mammalian expression vector (Clontech,
Mountain View, CA, USA). In addition, the primers also contained one extra nucleotide
between the cut site and the set of complementary nucleotides in order to place the gene
in frame with the vector-encoded myc tag that is 5 to the insert. pCMV-myc was
digested using SalI and NotI according to the instructions provided by the manufacturer
(New England Biolabs, Ipswich, MA, USA) as was the PCR amplified TDP-43 DNA
fragments. Double DNA digestions were carried out in a 50 µL reaction volume
consisting of 40 µL of TDP-43 PCR products and 10 µL of plasmid DNA, and were
incubated at 37 C for 1 hr. The digested DNA fragments were isolated by
electrophoresis through agarose gels (0.8 % for TDP-43, 1.2 % for plasmid, respectively)
for 45 min at 90 V in 1x TAE buffer (40 mM Tris-Acetate, 1mM EDTA) buffer.
Ethidium bromide (5 µL, 10 mg/mL, Bioshop, Burlington, ON, Canada) was added into
100 mL agarose before it sets, which allows visualization of the DNA at 360 nm. By
comparison to a DNA ladder (Fermentas, Glen Burnie, MD, USA), the correct size of
DNA bands were excised from the gels, and the DNA was extracted and purified from
the gel pieces using a DNA extraction kit (Qiagen, Valencia, CA, USA). The purified
DNA, 1 µL of the digested plasmid DNA and 5 µL of the digested TDP-43 PCR product,
were ligated together using T4 DNA ligase (New England Biolabs, Ipswich, MA, USA)
at RT for 1 hr. The ligation product was transformed into XL-10 gold E. coli (Stratagene,

200
Santa Clara, CA, USA). 1 µL of the ligation product was added into 20 µL of XL-10
cells, incubated on ice for 20 min, then heat shocked at 42 C for 20 sec, then incubated
on ice for another 2 min. After incubation at 37 C with shaking for 1 hr, 300 µL of the
mixture was plated onto an LB plate containing 20 g/L agar, 50 µg/mL kanamycin, and
100 µg/mL ampicillin. The plate was kept at RT for 30 min, then incubated at 37 C for
12-16 hr. Three colonies were picked and cultured, and their plasmid DNA were
extracted by a mini-prep kit (Qiagen, Valencia, CA, USA). The plasmid DNA was then
digested with the same two restriction enzymes that were used previously to determine if
the appropriately sized insert was present. One verified plasmid DNA sample was sent
for DNA sequencing validation (NAPS, University of British Columbia).
Site directed mutagenesis of TDP-43
The following set of primers was used to generate the N390D point mutant in TDP-43.
N390D forward: 5-GGGGATCAGCATCCGATGCAGGGTCGGGCAG-3; N390D
reverse: 5-CTGCCCGACCCTGCATCGGATGCTGATCCC-3. Mutated nucleotides
are shown in bold case.
SK-N-SH cell culture
SK-N-SH cells, a human neuroblastoma cell line, was obtained from the American Type
Culture Collection (ATCC). Cells were grown in DMEM media with high glucose
(Invitrogen) and supplemented with 5% FBS (Invitrogen). Cells were maintained at 37
C in 5% CO2 and split approximately 1:10 every two to three days as required. Splitting
the culture was carried out by first washing the cells twice with PBS and then removing
the cells by incubation with trypsin in a solution of PBS (0.05 % (w/v), Gibco) for 3-5

201
min at 37 C. The trypsinized cells were then mixed well, and were seeded onto new
plates containing fresh media at a 1:10 (v/v) ratio.
Transfection and immunocytochemistry
SK-N-SH cells were seeded on 18 mm circular coverslips (Fisher Scientific) in 12-well
tissue culture plates (Fisher) and allowed to grow for 24 hr before transfection. At the
time of transfection, the cells were approximately 60% confluent. SK-N-SH cells were
transfected with the appropriate expression plasmids using Lipofectamine-2000
(Invitrogen) according to manufacturer’s instruction. Each transfection took place in the
12-well culture plates and was carried out in duplicate. The transfections consisted of 0.5
g of plasmid and 1.5 l Lipofectamine-2000 in a total of 400 l of serum-free media.
The cells were incubated in this mixture for 10 hr and the media was then replaced with
DMEM containing 10% FBS. Using these conditions, the transfection efficiency was
estimated to be approximately 20-30%. Cells were incubated for 6 hr, 12 hr, and 24 hr,
respectively, before fixation.
The cells were washed with PBS (pH 7.4) twice, fixed with 0.5 mL of pre-warmed (37
C) 4% paraformaldehyde (PFA, Anachemia) in PBS for 12 min at 37 C. Each well
contained a coverslip at the base and were washed three times with PBS, after which the
cells were permeabilized using 0.3% Triton X-100 in PBS (PBST) for 30 min at room
temperature. Each well was then treated with blocking solution (5% BSA and 10%
normal goat serum (NGS) in PBS) for 30 min. The appropriate primary antibodies
(diluted in 2% NGS) were added and the samples were incubated overnight at 4 C. Anti-
myc (mouse IgG, Invitrogen) was used at a dilution of 1:200 and the anti-TDP-43

202
antibody (rabbit IgG, Proteintech, 10782-2-AP) was used at a dilution of 1:1000. After
washing with PBS, cells were incubated with the appropriate secondary antibodies
(diluted in 2% NGS) for 1 hr at room temperature in dark, including Alexa 488 (green)
conjugated to goat anti-mouse IgG (Invitrogen) and Alexa 568 (red) conjugated to goat
anti-rabbit IgG (Invitrogen). Secondary antibodies were used at a dilution of 1:1000.
After washing with PBS, coverslips were mounted using Vectashield Mounting Medium
containing DAPI (H-1200, Vector Laboratories) onto Superfrost/Plus slides (Fisher
Scientific). Stained cells were visualized using a Leica fluorescent microscope
(DM4000B). The filter sets used to image were as follows: DAPI (excitation peak: 360
nm, emission peak: 460 nm, Leica), Alexa 488 (excitation peak: 480 nm, emission peak:
520 nm, Leica), Alexa 568 (excitation filter: 530-550 nm, emission filter: 570 nm, Leica).
Images were acquired using a Spot digital camera (Diagnostic Instruments) and
processed using LAS software (Leica).

203
Appendix C
O-GlcNAc modification of TDP-43 slows its rate of aggregation. A turbidity assay was carried out with O-GlcNAc-modified (red) and unmodified TDP-43 (blue). Protein concentrations were measured by the Bradford assay, and a protein concentration of 5 µM in 175 µL of reaction volume was used for turbidity assay. Curves represent the average of three replicates.

204
Appendix D
Methods for O-GlcNAc modified TDP-43 aggregation assay O-GlcNAc modified and unmodified TDP-43 protein expression and purification
The same protocol was used as described in Chapter 3, section 3.3.6.
Turbidity Assay
All assays were carried out using a Cary 3E UV-VIS spectrophotometer equipped with a
Peltier temperature controller. O-GlcNAc-modified and unmodified TDP-43 protein
samples were measured in parallel. The spectrophotometer was maintained at 45 C and
the reactions were monitored continuously over time at 500 nm in 50 mM tris, 100 mM
sodium chloride, and 5 mM -mercaptoethanol (pH 7.5). The Bradford assay (BioRad)
was used to determine the protein concentrations. A series of protein standards ranging
from 0-12 µg/µL BSA (Bioshop) was prepared. Protein assay reagent (50 µL) was added
to each of the protein samples (50 µL) and standards (50 µL) and mixed gently. After
incubation for 10 min, the absorbances were measured at 595 nm in a plate reader
spectrophotometer (Molecular Devices, SpectraMAX 340). Then the protein samples
were diluted in the same dialysis buffer (PBS), and a protein concentration of 5 M in
175 µL reaction volume was used for the turbidity assays. Data was acquired every five
seconds over the course of the experiment and three replicate experiments were
performed.