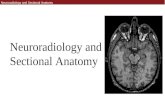
Neuroradiology and Sectional Anatomy Neuroradiology and Sectional Anatomy.
Neuroradiology Lecture Aug2007
-
Upload
api-3743483 -
Category
Documents
-
view
122 -
download
4
description
Transcript of Neuroradiology Lecture Aug2007

Diagnostic Imaging Methods in Central Nervous System Disorders
P. Danilo J. Lagamayo, MD

Headache:Primary:
MigraineClusterTension
Secondary:Increased Intracranial Pressure:
NeoplasmsAbscessGranulomas
Meningeal Irritations:MeningitisSubarachnoid Hemorrhage
Vascular Disorders:StrokeMalformationsArteritis
Head Trauma:ConcussionHematoma
Other Cranio-Facial Pains:Trigeminal Neuralgia

Incidence of Primary Brain Tumors:
- 6 persons / 100,000 population / year
- about 1 in 12 primary brain tumors occur in children under 15 years old.

Clinical Presentation of Brain Tumors:
Focal neurologic deficitIncrease intracranial
pressure: - Headache that is more severe in AM
- Nausea / Vomiting- Diplopia - Papilledema
- Ontundation & Lethargy (ominous) Focal neurologic signs and symptoms:
- seizure, seen in about ½ of patients

Clinical Presentation of Brain Tumors: (II) Non-localizing findings: - fatigue
- malaise- impotense
- glactorrhea - growth failure
- macrocephaly in young children

Pathological Classification of Intracranial Tumors:
Neuroepithelial:Astrocytes -
AstrocytomaOligodendrocytes - Oligodendroglioma
Ependymal cells & Choroid Plexus-
Ependymoma Choroid Plexus Papilloma Neurons -
Gangliomas, Gangliocytomas,
NeuroblastomasPineal cells - Pineocytomas
PineoblastomasPoorly differentiated
Glioblastoma multiforme and embryonic cells Medulloblastoma

Pathological Classification of Intracranial Tumors: (cont.)
Meninges - MeningiomaNerve sheet cells -
Neuroma- Neurofibroma Blood
vessels - HemangioblastomasGerm cells - Germinoma
- TeratomaTumors of malde-velopmental origin -
Craniopharyngioma- Epidermoid/Dermoid
cyst- Colloid cysts

Pathological Classification of Intracranial Tumors:
Anterior pituitary gland- Pituitary adenoma- Adenocarcinoma
Local extension - Chordoma from adjacent - Glomus jugulare tumors - Chondroma
- Chondrosarcoma - Cylindroma

Incidence of Tumors:Glioblastoma - - - - - - - - -
55% Astrocytoma - - - - - - - - - 20.5% Ependymoma - - - - - - - - 6% Medulloblastoma - - - - - - 6% Oligodendroglioma _ - - -
5% Choroid Plexus Papilloma 2%
Other less common entities:Neuronal tumors –
Gangliocytomas, ganglioglioma Embryonal – PNET Pineal Region – germ cellPrimary CNS Lymphoma

Primary Imaging Methods for Diagnosis of CNS tumors:
- MRI
- CT scan
- Angiography

Advantages of CT Scan
- Wide availability;
- Can accommodate life support systems;
- Fast imaging methods
- Can show bone structures and their pathologic changes like fractures;
- Cheap.

Disadvantages of CT Scan
• Cannot demonstrate soft tissue detail of the sella turcica, the brain stem and the cerebellum;
• Not very sensitive to white matter lesions;
• Cannot differentiate encephalomacic lesions of hemorrhage from infarcts;
• Uses ionizing radiation and cannot be used on pregnant patients.

Advantages of MRI
• Unaffected by the thick bone encasement of the calvarium in the posterior fossa and the sellar turcica;
• Accurate determination of the age and evidence of hemorrhage;
• More sensitive for detection of white matter lesion;
• Ability to perform multiplanar imaging;• Does not use radiation and can be safely used
on pregnant patients.

Disadvantages of MRI
• Longer time needed to complete an examination;
• Needs patient cooperation, e.g. patient must not move during an imaging sequence that can take anywhere from 1 minute to as long as 7 minute;
• Any metal implement is not allowed into the MR room.

Disavantages of MRI
• Cannot image patients with:– Pacemakers– Neurostimulators– Newly applied vascular clips – Vacular clips with ferromagnetic materials
(steel or iron)– Metal foreign bodies in the orbit– Claustrophobic patients– Patients who need extensive life support

Causes of Low Signal Intensity in Tumors in T2WI:
Paramagnetic effects- Iron with dystrophic Ca or
necrosis - Ferritin/hemosiderin from prior bleed
- Deoxy hgb in acute bleed- Intracellular met hgb in
early subacute bleed - Melanin (or other free radicals)

Causes of Low Signal Intensity in Tumors in T2WI (cont.):
Low spin density - Calcifications
- Scant cytoplasm (high nucleus:cytoplasm ratio)
- Dense CellularityFibrocollagenous stroma
Macromolecule content Very high (non-paramagnetic) protein content
Intratumoral vessels - Signal void from
rapid flow

Causes of High Signal Intensity in Tumors in T1WI:
Paramagnetic effects from hemorrhage- Subacute – chronic
blood (met hgb) Paramagnetic materials w/out hemorrhage - Melanin - Naturally occuring ions associated with
necrosis of calcification:Manganese, Iron, CopperNon-paramagnetic effects
- Very high (non-paramagnetic) proteins - Fat - Flow related enhancement in tumor vessels

Requirements for contrast enhancement:
- absence of blood-brain barrier- adequate delivery of
contrast material - extracapillary interstitial space to accommodate contrast
- appropriate contrast dosage- spatial resolution
- imaging parameters to allow contrast detection
- time for contrast to accumulate in the region
of interest

Mechanism for contrast enhancement in CNS tumors:
Formation of capillaries with deficient blood-brain barrier rather than the destruction of blood-barrier is presumed as the mechanism for tumor enahcement.
The capillaries of metastatic tumors to the brain has no blood-brain barrier since these tumors come from elsewhere and not from the brain.

Type of enhancement:- immediate or delayed
- evanescent or persistent - dense and homogenous - minimal or irregular
Note: Lack of tumor enhancement do not signify lack of tumor.

Effects of Tumor Necrosis on Signal Intensity:
Short relaxation times HemorrhageLiberation of
cellular iron Release of free radicalsProteinaceous debris
Prolong relaxation Cystic change with increased times water

Frequently Cystic TumorsColloid cystCraniopharyngiomaDesmoplastic infantile gangliomaDermoidEpendymoma (supratentorial and spinal)EpidermoidGanglion cell tumorsGlioblastoma (cystic necrosis)HemangioblastomaPilocytic astrocytomaPleomorphic xanthoastrocytomaRathke cleft cyst

Magnetic Resonance Criteria for Cystic LesionsMorphology Sharply demarcated, round
smooth
Signal Intensity Isointense to cerebrospinal fluid on all spin echo images (tumor cysts can be hyperintense due to ↓ T1)
Fluid-debris levels (bleed into necrotic or cystic regions)
Intracellular blood-cyst fluid Intracellular blood-extracellular blood
Motion of intra- lesional fluid
Lesion emanates ghost images along phase-encoding axis
Intralesional signal loss (especially on steady-state sequences)

Hemorrhagic TumorsPrimary brain tumors
Glioblastoma/anaplastic asctrocytomaAnaplastic oligodendroglioma/oligodendrogliomaEpendymomaTeratoma
Metastatic diseaseMelanomaRenal cell carcinomaChoriocarcinomaLung carcinomaBreast carcinomaThyroid carcinoma

Intratumoral Hemorrhage vs. BenignIntracranial Hematomas
Intratumoral hemorrhage:
- Markedly heterogenous, related to: Mixed stages of blood Debris-fluid (intracellular-extracellular blood) levels Edema + tumor + necrosis with blood
- Identification of nonhemorrhagic tumor component- Delayed evolution of blood breakdown products- Absent, diminished, or irregular ferritin/hemosiderin -Persistent surrounding high intensity on long TR images (i.e., tumor/edema) and mass effect, even in late stages

Intratumoral Hemorrhage vs. Benign Intracranial HematomasBenign hemorrhage:
- Shows expected signal intensities of acute, subacute or chronic blood, depending on stage of hematoma
- No abnormal nonhemorrhagic mass- Follows expected orderly progression- Regular complete ferritin/hemosiderin rim- Complete resolution of edema and mass effect in chronic stages

Intratumoral Melanin vs. Hemorrhage
Signal intensity
(relative to gray matter)
T1-weighted
Image
T2-weighted
Image
Amelanotic tumor
Melanotic tumor
Early subacute blood
(intracellular methemoglobin)
Late subacute blood
(extracellular methemoglobin)
↓
↑↑
↑↑
↑↑
sl. ↑
= or sl. ↓
↓↓
↑↑

Classification of Astrocyctic Brain Tumors
Diffuse (infiltrative) Localized (circumscribed)
Astrocytoma
Anaplastic astrocytoma
Glioblastoma multiforme
Pilocytic astrocytoma
Pleomorphic xanthoastrocytoma
Subependymal giant cell astrocytoma

Diffuse Astrocytic Brain NeoplasmsAstrocytoma Anaplastic astrocytoma
Typical site(s) of origin
Signal intensity
characteristics(on
T2-weighted image)
Vascular flow voids
Contrast
enhancement
Prognosis (median survival, if available)
Cerebral hemisphere
(adult)
Brainstem (child)
Cerebellum (young adult)
Homogeneous; high
intensity
Not seen
Variable; irregular
7-8 yr
Cerebral hemisphere
(adult)
Brainstem (child)
Some heterogeneity
Unusual
Common; irregular
2-3 yr

Glioblastoma Gliomatosis
Typical site(s) of origin
Signal intensity
characteristics(on
T2-weighted image)
Vascular flow voids
Contrast
enhancement
Prognosis (median survival, if available)
Cerebral hemisphere
(adult)
Markedly heterogeneous;
hemorrhage and
necrosis common
Common
Common; irregular
12 mo
Cerebral hemisphere (young
or middle-aged adult)
Ill-defined; high intensity
Rare
Uncommon
Estimated as months
Diffuse Astrocytic Brain Neoplasms

Pediatric Supratentorial Hemispheric Neoplasms
Juvenile pilocytic astrocytoma
Ganglioglioma
Signal intensity
characteristics (on
T2-weighted image)
Contrast
enhancement
Hemorrhage
Calcification
Prognosis
Sharply demarcated;
commonly cystic
Common; dense
Rare
Uncommon
Excellent
Sharply demarcated;
commonly cystic
Common; irregular
Rare
Common
Excellent

Pleomorphic xanthoastrocytoma
Embryonal tumor
(e.g., cerebral neuroblastoma)
Signal intensity
characteristics (on
T2-weighted image)
Contrast
enhancement
Hemorrhage
Calcification
Prognosis
Sharply demarcated
with subjacent cyst
Common in solid
portion
Rare
Uncommon
Variable
Markedly
heterogeneous
Common; irregular
Common
Common
Poor
Pediatric Supratentorial Hemispheric Neoplasms

DNT
Signal intensity
characteristics (on
T2-weighted image)
Contrast
enhancement
Hemorrhage
Calcification
Prognosis
Sharply demarcated
heterogeneous
Unknown
Rare
Common
Excellent
Pediatric Supratentorial Hemispheric Neoplasms

Tumor type Typical location
Central neurocytoma
Ependymoma
Subependymoma
Oligodendroglioma
Pilocytic astrocytoma
Meningioma
Choroid plexus tumor
Epidermoid
Subependymal giant cell
astrocytoma
Colloid cyst
Arachnoid cyst
Lateral (attached to septum pellucidum)
Fourth, lateral
Lateral, fourth
Lateral
Lateral, third, or fourth
Lateral (atrium)
Lateral (atrium) or third in children, fourth in adults
Any ventricle
Lateral
Third
Any ventricle
Intraventricular Masses

Tumor type Intensity characteristics on T2-weighted images
Contrast enhancement
Central neurocytoma
Ependymoma
Subependymoma
Oligodendroglioma
Pilocytic astrocytoma
Meningioma
Choroid plexus tumor
Epidermoid
Subependymal giant cell astrocytoma
Colloid cyst
Arachnoid cyst
Isointense to gray matter
Heterogeneous
Hyperintense to gray matter
Heterogeneous
Hyperintense to gray matter
Isointense to gray matter
Heterogeneous
Slightly hyperintense to CSF
Hyperintense to gray matter
Hyperintense to gray matter
Usually dense
Heterogeneous
None
Variable; irregular
Dense
Dense
None
Generally enhance
Limited enhancement at periphery
None
Isointense to CSF
Intraventricular Masses

Juvenile pilocytic astrocytoma
Medulloblastoma
Signal intensity
characteristics (on T2-WI)
Contrast enhancement
Calcification
Hemorrhage
Tendency to seed
CSF pathways
Prognosis (estimated survival)
Sharply demarcated;
commonly cystic
Common in solidportion (mural nodule)
Uncommon
Rare
Extremely low
>90% 10-yr survival
Homogeneous; low to
moderate intensity
Common; dense
Uncommon
Uncommon
High
50% 5-yr survival
Posterior Fossa Tumors in Childhood

Ependymoma Diffuse pontine glioma
Signal intensity
characteristics
(on T2WI)
Contrast
enhancement
Calcification
Hemorrhage
Tendency to seed
CSF pathways
Prognosis (estimated
survival)
Markedly heterogeneous
Common; irregular
Common
Common
Low to moderate
65-70% 5-yr survival
Ill-defined; high intensity
Variable
Rare
Common
Low
<1-2% 5-yr survival
Posterior Fossa Tumors in Childhood

Pineal Region TumorsGerminoma Teratoma Pineoblastoma
Age; sex predilection
Pineal vs.
parapineal
Signal intensity
(heterogeneous
vs.
homogeneous)
Hemorrhage
Calcification
Brain edema or
invasion
Tendency to
metastasize
Enchancement
Prognosis
Child; male
Pineal
Homogeneous
(but often
hemorrhagic)
Common
Rare
Common
Yes
Dense
Excellent
Child; male
Pineal
Strikingly
heterogeneous
Typical
Typical
Variable
Variable
Variable
Variable
Child; none
Pineal
Homogeneous
(unless
hemorrhagic)
Common
Common
Common
Yes
Dense
Poor

Pineal Region TumorsPineocytoma Glioma Meningioma
Age; sex
predilection
Pineal vs.
parapineal
Signal intensity
(heterogeneousvs.
homogeneous)
Hemorrhage
Calcification
Brain edema or
invasion
Tendency to
metastasize
Enchancement
Prognosis
Adult; none
Pineal
Variable
Common
Common
Uncommon
No
Dense
Variable
Child; none
Parapineal
(usually)
Homogeneous
(usually)
Rare
Common
Primarily
midbrain
Variable
Variable
Variable
Adult; none
Parapineal
(usually)
Homogeneous
Rare
Common
Occasional
No
Dense
Excellent

Magnetic Resonance Findings in Extraaxial Mass Lesions
Suggestive Definitive
Peripheral, broadly based
along calvarium
Overlying bone changes
Enhancement of adjacent
meninges
Displacement of brain from
skull
Cerebrospinal fluid cleft between
brain and lesion
Vessels interposed between brain
and lesion
Cortex between mass and
(edematous) white matter
Dura (meninges) between
(epidural) mass and brain

Stroke: a new, often acute, loss of
neurologic function secondary
to parenchymal ischemia or hemorrhage.

Main Etiologies for Symptomatology of Stroke:
1. Cerebral Infarction 2. Intraparenchymal
Hemorrhage 3. Subarachnoid
Hemorrhage

Role of Imaging in Stroke:
1. Rule out hemorrhage 2. Rule other causes of stroke syndrome 3. Help assess etiology in known ischemic infarction

The Normal Brain:
To sustain the normal brain, a normal mean regional cerebral blood flow (rCBF) must be maintained at about
54 (± 12 ml) / 100 g / min
The Normal Brain:
To sustain the normal brain, a normal mean regional cerebral blood flow (rCBF) must be maintained at about
54 (± 12 ml) / 100 g / min

The Normal Brain:
The threshold for cerebral ischemia is approximately at:
23 ml / 100 g /min.
The Normal Brain:
The threshold for cerebral ischemia is approximately at:
23 ml / 100 g /min.

Autoregulation plays a very important role in maintaining intracerebral blood flow.
This mechanism can be temporarily lost in ischemia leaving the control of blood flow to peripheral flow volumes.

Ischemic Strokes:
1. Large Artery or Atherosclerotic Infarction
2. Cardioembolic Infarction 3. Small Vessel Infarction 4. Venous Infarction

The Abnormal Brain:Between cerebral blood flow rate of:
15 & 20 ml / 100 gm / min.,
ischemic brain injury begins w/ loss of neurologic function, noted as flattening of the electroencephalogram.
This may still be reversible.
The Abnormal Brain:Between cerebral blood flow rate of:
15 & 20 ml / 100 gm / min.,
ischemic brain injury begins w/ loss of neurologic function, noted as flattening of the electroencephalogram.
This may still be reversible.

The Abnormal Brain:
Blood flow values below:
10 ml / 100 gm / min.,
may lead to infarction within a few minutes.
The Abnormal Brain:
Blood flow values below:
10 ml / 100 gm / min.,
may lead to infarction within a few minutes.

The Ischemic Brain:
There are two ischemic changes thresholds, one occurring at blood
flow range of 15-20 ml / 100 gm / min., resulting to loss of electrical function and another one at 10ml /
100 gm / min. , resulting to loss of cell polarizaton.

PENUMBRAHeterogeneity in brain injury has been documented in an infarcted zone.
Blood flow to an infarcted zone is said to have: A. a central region or core of very low flow that results in rapid cell demise and B. a peripheral penumbra where decline in flow is more moderate and cell death is not immediate.

PENUMBRAThe penumbra is thought to
represent salvageable tissues that may go on to infarction.
If blood flow is normalized at an adequate time, the brain cells will normalize.

Imaging in stroke:Most commonly used imaging method non-contrast CT scan but MRI is fast catching-up.
CT scan is commonly used in stroke due to:
- Widespread and ready availability;- Ease of hemorrhage detection;- Compatibility with monitoring
equipment;- Rapid scanning techniques for unstable patients.

Emergent evaluation in Acute Stroke:
Goals: - Confirm cause of deficit is stroke
related. - Assess possible reversibility of
the lesion.- Determine most likely etiology.- Predict likelihood of immediate
complications.- Begin appropriate treatment.

Emergent Evaluation in Acute Stroke:
Opportunities for Intervention:
- Before any clinical symptoms.- After transient ischemic
attack or minor stroke.- During acute ischemic
stroke. - Before a recurrence.

Imaging Signs of Hyperacute Infraction:
1. Hyperdense LMCA sign2. Loss of gray-white matter differentiation3. Sulcal effacement.4. Loss of insular ribbon.5. Obscurred lentiform nucleus.

Lacunar Infarction:- Not larger than 1.5 cm - Deep gray matter - Brain stem - Deep hemispheric white matter - Supplied by perforators

Cardioembolic Infarction- Relative stasis resulting to mural thrombus, ex.: M.I., atrial fib., ventricular aneurysm - Valvular heart disease resulting to vegetation or from prosthesis - Cardiac tumors - Congenital HD, ex.: right to left shunt

Watershed Infarction:
- Boundary zone infarct - Internal carotid stenosis or occlusion - Systemic hypotension - Embolic events

Hemorrhagic Infarction:
- Hemorrhagic transformation results to petechial hemorrhage or frank hematoma - Anticoagulant therapy
- Thrombolytic agents - More common in cardioembolic strokes - Larger cardioembolic strokes are more
likely to bleed

Temporal Evolution of Infarction on CT Scan: 0 – 4 hrs. Normal to subtle hypodensity
± sulcal effacement
1 – 7 days Mass effect peaks at 3 – 4 days
1 – 8 weeks Contrast enhancement
Days to Hypodensity months/ yrs
Weeks to years Atrophy

Acute to Subacute Infarction Changes:
1. Vasogenic Edema that later on wanes
2. Enhancement - (Luxury perfusion)
3. Petechial hemorrhage

Hypertensive Hemorrhage
In hypertensives, hyalinization within the walls of small cerebral vessels results in
microaneurysms that are less than 1.0 mm in size,
(Charcot & Bouchard), that tend to arise from perforating vessels that will later on bleed.

Some of the Causes of ICH:HypertensionAmyloid VasculopathyAneurysmA-V malformationNeoplasmCoagulation disorders, e.g.
hemophiliaAticoagulantsVasculitisDrug abuse e.g. cocaineTraumaIdiopathic

Hypertension accounts for 40-50% of deaths from non-traumatic hemorrhage in an autopsy series.
In young (less than 40 y/o) normotensive patients, cause remains unknown but cryptic AVM is a suspect.

Why is there a need to measure hemorrhage size?
Volume of the hemorrhage is a strong indicator of the 30 day survival of the patient.

Methods of measuring ICH Volume:
A. Direct volume measurement in the CT Scan system or in a work station;
B. PlanimetryC. Application of the formula for
the sphere:
Volume = 4/3 (r)3
D. ABC/2 method

Among different methods of volume measurements, the direct volume measurement in the CT scanner is the most accurate but this would depend on the cooperation of the facility operators.
Once the patient data is deleted from the memory file of the system, the direct volume measurement can no longer be applied on the data in the hard copy (film).

In older model CT Scan where volume measurement is not available, an alternative method is possible by using the area of the hemorrhage:
Volume in cubic cm =
Area x slice thickness (millimeters) 1000

ABC/2 Method:
Kothari, et. al., has developed a simple bedside method of ICH volume determination with the following formula:
ICH volume = A x B x C2

ABC/2 Method (continued):Step 1: The largest dimension of the
hemorrhage is determined in the series of CT slices, then the largest diameter of the hematoma is measured and labeled - A;
Step 2: On the same slice, the largest diameter of hemorrhage 90o to A is determined and labeled – B.

ABC/2 Method (continued):Step 3: “C” or the cephalocaudal
dimention of the hemorrhage is determined by comparing the rest of the CT slices to the largest hemorrhage on the scan.
If the hemorrhage area is 75 % of the largest hemorrhage area = one (1) slice for determining C;

ABC/2 Method (continued):Step 3: If the area was 25 to 75% of the
slice where the hemorrhage was largest, the slice is considered as one-half a hemorrhage slice;
If the area was less than 25 % of the largest hemorrhage, this is not considered as a hemorrhage slice.

When the CT slice thickness is smaller than the table movement, as will be commonly encountered in CT slices of the posterior fossa, there will necessarily be the presence of inter-slice gaps.
To remedy this, use the table movement measurement for thickness of the slice instead of the actual slice thickness to calculate for volume.

1 2
3 4

A
B
(2)
“1” slice

ABC/2 Method:
(A x B x C ) ÷ 2 = Volume in ccA = 4.0 cmB = 2.6 cmC = 2.5 cm
(4.0 x 2.6 x 2.5) ÷ 2 = 13 cc
Actual computation directly done in the CT scan = 13.3 cc

Reader No. Intraclass Correlatio
n
Difference From Planimetric,*
cm3
P† Mean Time per Measurement,‡
s
1 (Neurosurgery faculty)
20 .99 -2.0 ± 1.2 .11 35
2 (Neurosurgery resident)
20 .99 0.6 ± 3.0 .85 40
3 (Emergency physician)
20 .99 0.8 ± 1.3 .55 33
4 (Nurse) 20 .99 -2.5 ± 1.5 .07 31Interrater reliability (readers 1-4): Interclass correlation = .99Intrarater reliability (reader 3): Interclass correlation = .99 (P=.19)
* Mean±SE difference from planimetric measurement.† Difference from planimetric measurement.‡ Mean time to determine hemorrhage volume per CT scan with the ABC/2 technique
Reliability & Reproducibility of the ABC/2 Method of Measuring Intraparenchymal Hemorrhage Volume

Hemorrhage Volume, cm3
Location No. Planimetric ABC/2 R2
Deep 83 23.0 ± 2.7 23.5 ± 2.9 .94
Lobar 21 44.6 ± 8.4 49.9 ± 9.9 .96
Brain Stem
8 13.6 ± 7.2 12.3 ± 6.3 .99
Cerebellar 6 19.6 ± 4.3 24.4 ± 5.9 .78
Total 118 26.0 ± 2.6 27.5 ± 2.9 .96
Hemorrhage volumes are mean ± SE.
Mean Hemorrhage Volumes

Temporal Evolution of ICH
Biochemical Form
Clinical Stage Approximate Time of Appearance
OxyHg in RBCs HyperacuteImmediately to first several hours
DeoxyHg in RBCs
Acute Hours to days
MetHg in RBCs Early subacute First several days
Extracellular MetHg
Subacute to chronic
Days to months
Ferritin and Hemosiderin
Remote Days to indefinitely

Biochemical Form
Intensity on T1WI
Intensity on T2WI
OxyHg in RBCs
DeoxyHg in RBCs
,
MetHg in RBCs Extracellular metHg
Ferritin and hemosiderin
,
Temporal Evolution of ICH

Acute Infarction findings in MRI:
1. Lesion in arterial distribution 2. High intensity in Proton density or in T2 FLAIR
3. Gyral swelling / sulcal effacement 4. Absent arterial flow void 5. Subcortical white matter hypointensity 6. Intravascular contrast enhancement

Diffusion weighted imaging:-Signal attenuation is noted in areas of free diffusion - Signal intensity is increased in areas of restricted diffusion with decrease in apparent diffusion coefficient in brain tissue
- Decrease in diffusion of water in early ischemia is due to shift of water from extracellular to intracellular