Neonatal Encephalopathykfafhconferences.com/neonate/images/2-Neonatal... · MRI and Prognosis...
Transcript of Neonatal Encephalopathykfafhconferences.com/neonate/images/2-Neonatal... · MRI and Prognosis...

Neonatal Encephalopathy
Hannah C. Glass, MDCM, MAS Professor Neurology & Pediatrics University of California, San Francisco
February 2020
15th Hot Topics in Neonatal Medicine
Jeddah, Saudi Arabia


San Francisco

Objectives
List the causes of neonatal
encephalopathy
Explain the evidence for hypothermia
therapy
Understand prognosis after hypothermia.
therapy



Arterial/Venous Stroke Hemorrhage
Infection
HIE
Inborn Error of
Metabolism Malformation Epileptic
Encephalopathy
Many Causes of Encephalopathy

Hypoxic-Ischemic Brain Injury
• Neonatal encephalopathy due to peripartum asphyxia
• Perinatal asphyxia affects 3-5 per 1000 live births – 0.5-1 per 1000
will have HIE

HIE - Etiology
• Impaired placental or pulmonary gas exchange
• Placental abruption, cord rupture, uterine rupture, nuchal cord, true knot

Primary energy failure
(minutes) Early cell death -
necrosis
Hypoxic-Ischemic
Injury
Depletion of glucose & ATP
Anaerobic metabolism
Failure of ATP dependent pump
Membrane depolarization
Na+
Intracellular Ca++
Extracellular glutamate
Therapeutic
window for
hypothermia
Secondary energy failure
(hours to days) Late cell death -
apoptosis
Reperfusion
Chronic brain injury
(weeks to months)
Delayed cerebral
atrophy and cell
loss
Partial recovery of oxidative
metabolism
Inflammation
Excitatory amino acids
Extracellular Ca++ NO, H2O2

Cell death
Inflammation Oxidative Stress R
esp
on
se
Hours Days Weeks
Timing: Injury and Repair
Excitotoxicity
Ferriero DM, NEJM
Repair

HIE - Clinical Features
• Low Apgar scores
• Acidotic cord gas
• Advanced resuscitation (ventilation, chest compressions, epinephrine)
• Multi-organ failure – hypotension, high liver enzymes, coagulopathy
• Abnormal mental status, abnormal neurological examination, seizures

Clues that NE is not HIE
Condition
• No event/normal delivery
• Seizure onset at birth
• Seizures don’t stop within 4 days
• Difficulty extubating
What to consider
• Genetic, inborn error of
metabolism, infection
• SSRI
• Genetic epilepsy, inborn
error of metabolism
• Neuromuscular

Sarnat Staging 1976

Barkovich et al. AJNR 2006
T1
ADC
NAA
Cho
Cr
Lactate
Basal Ganglia/
Thalamus
Injury
HIE - Neuroimaging
Slide courtesy of Dr. Sonia Bonifacio

Watershed
Injury
T2
ADC
NAA
Cho
Cr
NAA
Cho Cr
Lactate
Barkovich AJ AJNR 2001
Slide courtesy of Dr. Sonia Bonifacio

HIE - Outcomes
• 10-20% mortality
• Additional 25% develop severe impairment
• Cerebral palsy, intellectual disability, epilepsy

Hypothermia
Hypothermia optimization
Add-on therapies/alternate agents
Neurocritical care – “brain focused care”
Neuroprotective Strategies

Hypothermia
Hypothermia optimization
Add-on therapies/alternate agents
Neurocritical care – “brain focused care”
Neuroprotective Strategies

• Core temperature of 33.5°C x 72 hours – Passive cooling
initiated at referral center
Hypothermia Overview
Slide courtesy of Sonia Bonifacio
• Brain Monitoring – with aEEG/cEEG - Provides information about prognosis
- Seizures are common (34 – 65%)
• MRI at completion of treatment
• Most discharged at 7-15 days

Favors hypothermia for death/disability at 18-22 months
Increased rate of normal survival
Lower death/severe disability at school age
Number needed to treat = 6-9
The Evidence: Moderate/Severe HIE
Tagin et al. JAMA Pediatrics 2013
Shankaran S et al, NEJM 2012

The Conundrum of “Mild” HIE
Shankaran S et al. N Engl J Med 2005
NICHD Trial: Moderate/Severe HIE
>3 criteria at <6 hours after birth

Shankaran S et al. N Engl J Med 2005
“Mild” excluded as these infants were thought to have a good outcome
The Conundrum of “Mild” HIE

Sarnat Stages are Not Static
21 participants
Stage 1 Stage 2 Stage 3
7 (33%)
9 (43%)
4 (19%)
1 (5%)
Stage 1 lasted >6 hours for >70% Sarnat & Sarnat 1976

MRI in “Mild” HIE
• Injury in 20-40%
– Includes severe
lesions that are
highly predictive of
poor outcome
– Watershed is the
predominant
pattern
Walsh B & Inder T, Early Hum Dev 2018

Neurodevelopment in “Mild” HIE
• PRIME study – International cohort
– 16% with disability
• Systematic review
– 16 observational studies
• 22% had an abnormal outcome
– 4 RCT
• Abnormal outcome - cooled 29% vs not cooled
37% (p=0.6)
Chalak LF et al, Pediatri Res 2018
Conway JM et al, Early Hum Dev 2018
“Mild” HIE does not have a uniformly good outcome!

“Not fulfilling NICHD moderate-severe”
Shankaran S et al. N Engl J Med 2005.
The Conundrum of “Mild” HIE

Which newborns will evolve from mild moderate?
How should we define “mild”
encephalopathy?
When should we perform the encephalopathy exam?
What is the role of ancillary testing?
The Conundrum
Can we accurately determine the degree of
encephalopathy <6 hours? Can clinical signs determine who will or will not
benefit from cooling?

UCSF Cooling Criteria

Thoresen et al, Pediatrics 2010
Nash et al, Neurology 2011
Normalreassuring
Persistent abnormal
brain injury, death, disability
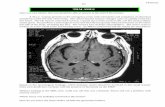
EEG/aEEG and Prognosis

MRI and Prognosis
Rutherford, Lancet Neurol, 2009
•Decreased lesions - Basal ganglia & thalamus > white matter
• Excellent accuracy - Normothermia 0·81 (0·71 – 0·91)
- Hypothermia 0·84 (0·74 – 0·94)
Normal Mild injury Moderate-severe injury

Predicting Outcome:
Counseling Families
203 Cooled neonates from 3 centers internationally
Outcome = death/severe disability
Birth/
Resuscitation Factor
1.0 (0.4-2.6) P=0.97
Persistent Abnormal
aEEG at 24 hours
5.4 (1.4-21.3)
P = 0.015
Moderate-Severe MRI
Injury
8.2 (2.9-32.4)
P<0.0001
Day 1 Day 2 Day 4-5
Bonifacio SL et al, PAS 2013

Summary
• Hypothermia is standard of care for neonatal encephalopathy due to perinatal asphyxia (HIE) – Always consider the differential diagnosis of NE
– Lower risk of death and disability
• Risk/benefit of treating “mild” encephalopathy not known
• Prognosis – Neonates with normal/near normal aEEG/EEG and normal
MRI have a good outcome
– Neonates with basal ganglia injury spastic/dyskinetic quadriparesis
– Watershed injury usually do very well
– Severely abnormal EEG/MRI death/severe disability

Acknowledgements Neurology
Donna M. Ferriero, UCSF
Dawn Gano, UCSF
Sharon Wietstock, UCSF
Yvonne Wu, UCSF
Steven Miller, Hospital for Sick Children
Vann Chau, Hospital for Sick Children
Emily Tam, Hospital for Sick Children
Taeun Chang, DC National Children’s Hospital Janet Soul, Boston Children’s Hospital Faye Silverstein, U Michigan
Kevin Staley, Mass General Hospital
Monica Lemmon, Duke
Cameron Thomas, Cincinnati Children’s
Neonatology/Pediatrics
Sonia Bonifacio, UCSF/Stanford
Elizabeth Rogers, UCSF
Michael Kuzniewicz, Kaiser Permanente
Patrick McQuillen, UCSF
Neuroradiology
A. James Barkovich, UCSF
Duan Xu, UCSF
Olga Tymofiyeva, UCSF
Yi Li, UCSF
Neurophysiology Joseph E. Sullivan, UCSF Maria Roberta Cilio, UCSF Adam Numis, UCSF Renee A. Shellhaas, U Michigan
Nicholas Abend, CHOP Courtney Wusthoff, Stanford Tammy Tsuchida, DC National Children’s Hosp Catherine Chu, Mass General Hospital Shavonne Massey, CHOP
Biostatistics and Epidemiology Charles McCullough, UCSF David Glidden, UCSF Nursing Linda Franck, BSN, PhD Susan Peloquin, Elizabeth Papp, Jeannie Chan NICN Nurses, UCSF
Psychology Shannon Lundy, UCSF Bridget Johnson, UCSF Research Assistants Laurel Haeusslein
Manogna Manne Jessica Kan Vedder Isheeta Madeka Bria Bailey Rebecka Craig Olivia Girvan
Children
&
Families









![Thalamus Hypothalamus [Repaired].pdf](https://static.fdocuments.in/doc/165x107/577cd6b41a28ab9e789d06fd/thalamus-hypothalamus-repairedpdf.jpg)