Natural history of caesarean scar pregnancy on prenatal ultrasound the cross-over sign Cali 2016
-
Upload
vo-ta-son -
Category
Health & Medicine
-
view
134 -
download
4
Transcript of Natural history of caesarean scar pregnancy on prenatal ultrasound the cross-over sign Cali 2016
Acc
epte
d A
rticl
eNatural history of caesarean scar pregnancy on prenatal ultrasound:
the cross-over sign
Giuseppe Cali1, Francesco Forlani1, Ilan Timor-Trisch2, Jose Palacios-Jaraquemada3, Gabriella Minneci1, Francesco D’Antonio4
1: Department of Obstetrics and Gynaecology, Arnas Civico Hospital, Palermo, Italy
2: Department of Obstetrics and Gynaecology, Division of Maternal-Fetal Medicine, New York University SOM, New York, NY, USA
3: Centre for Medical Education and Clinical Research (CEMIC), University Hospital, Buenos Aires, Argentina
4: Department of Clinical Medicine, Faculty of Health Sciences, UiT-The Arctic University of Norway, Tromsø, Norway
Corresponding Author: Francesco D’Antonio, MD, PhD, Women's Health and Perinatology Research Group Department of Obstetrics and Gynaecology Department of Clinical Medicine Faculty of Health Sciences, UiT - The Arctic University of Norway Hansine Hansens veg 18 9019 Tromsø, Norway [email protected]
This article is protected by copyright. All rights reserved.
This article has been accepted for publication and undergone full peer review but has not been through the copyediting, typesetting, pagination and proofreading process, which may lead to differences between this version and the Version of Record. Please cite this article as doi: 10.1002/uog.16216
Acc
epte
d A
rticl
eABSTRACT
Objective: Advances in prenatal imaging techniques have led to an increase in the diagnosis of
caesareans scar pregnancy (CSP). However, antenatal counselling when CSP is diagnosed is
challenging and the current evidence mainly derived from small series reporting high rates of
adverse maternal outcomes. The aim of this study was to ascertain the performance of prenatal
ultrasound in anticipating the natural history of CSP introducing a new sonographic sign, the cross-
over sign (COS).
Methods: Retrospective analysis of early first trimester (6-8 weeks of gestation) ultrasound images
of women with morbidly adherent placenta (MAP) treated in the third trimester of pregnancy. We
tested the hypothesis the relationship between the gestational sac of the CSP, anterior uterine wall
and previous caesarean scar (COS insertion) can be used to predict the evolution of these cases. The
odd ratios (OR) and logistic regression analysis were carried out in order to ascertain the association
between different types of COS insertion and the occurrence of MAP.
Results: Sixty-eight pregnancies were included in the study. The risk of placenta percreta was
significantly higher in pregnancies with COS1 compared to COS2 insertion (OR: 6.67, 95% CI 1.3-
33.3; p= 0.001); There was a significant difference in the occurrence of placenta percreta between
COS1 and COS 2+ (10/12 vs 18/39, p= 0.04), with an OR of 5.83, 95% CI 1.1-30.2 (p=0.036);
furthermore, this risk was even higher when comparing cases with COS1 with COS2- (OR: 12.0,
95% CI 1.9-75.7; p= 0.003). Women with COS2- had a lower risk of developing placenta percreta
compared to those with COS1 (OR: 0.069, 95% CI 0.01-0.4). Logistic regression analysis showed
that COS1 insertion was independently associated with the occurrence of severe forms of MAP
such as placenta percreta and increta (OR: 12.85, 95% CI 2.0-84.0), while COS2+ insertion was
associated independently associated with the occurrence of placenta accreta (OR: 4.37, 95% CI 1.1-
17.0).
Conclusion: Ultrasound assessment of the relationship between the ectopic gestational sac and the
endometrial line (COS insertion) may anticipate the natural history of CSP. CSP may theoretically
progress towards less severe forms of MAP amenable of post-natal treatment and successful
pregnancy outcome. Large prospective studies are needed in order to confirm these finding and to
elucidate the natural history of this disease.
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
eINTRODUCTION
Morbidly adherent placenta (MAP) encompasses a spectrum of conditions characterized by an
abnormal adherence of the placenta to the implantation site. MAP is one of the major cause of
maternal mortality and morbidity worldwide and is associated with the occurrence of several major
complications such as severe maternal hemorrhage, need for blood transfusion, peri-partum
hysterectomy, damage to adjacent organs and post-operative complications1.
Placenta previa and previous uterine surgery represent the main risk factors for the occurrence of
this condition and the incidence of MAP has been shown to rise in the last decade most likely as the
consequence of the increase in caesarean section rate2-4.
Despite this, the underlying mechanisms leading to MAP are not well understood yet. A defective
development of the decidua basalis allowing the invasion of the myometrium by the trophoblastic
tissue constitutes the pathophysiological explanation for the occurrence of invasive placentation2-4.
Recent evidences pose peculiar attention on caesarean scar pregnancy (CSP) as the precursor of
MAP. CSP5-8. Timor-Tritsch et al evaluated the natural history of CSP in 10 cases diagnosed before
10 weeks of gestation, whose parents opted to continue pregnancy. All cases had sonographic signs
suggestive of MAP in the second trimester and all underwent hysterectomy at the time of CD.
Histopathological diagnoses confirmed placenta percreta in all 10 cases6.
Advances in prenatal imaging technique has led to an increase in the diagnosis of CSP7-9. However,
antenatal counselling once CSP is diagnosed is challenging10-12. The association between CPS and
MAP poses the prognostic dilemma of how women with a prenatal diagnosis of CPS should be
counselled and whether termination of pregnancy should be the only therapeutic option offered to
these women. Although evolution towards early severe haemorrhage or uterine rupture may seem
the most likely scenario, the reported association with MAP raises doubts about the proportion of
CSP progressing to viability and how to identify those cases amenable of post-natal treatment and
successful pregnancy outcome.
The aim of this study was to ascertain the performance of prenatal ultrasound in predicting the
natural history of CSP introducing a new sonographic sign, the cross-over sign (COS).
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
eMETHODS
This is a retrospective case series of all women referred to our centre for the ultrasound suspicion of
MAP, from January 2007 to December 2015. These women were identified from an electronic
database of the fetal medicine unit. The delivery details were retrieved from hospital maternity
records. Operative notes were checked for details of operative findings and interventions performed.
All cases underwent pathology examination to confirm the presence and the degree of placental
invasion.
Early first trimester ultrasound images from 6 to 8 weeks of gestations were retrieved from the
computerized databases of each referring hospital. All the stored images showed the gestational sac
or trophoblast within the previous caesarean section scar and thus were labelled as CSP13. Other
ultrasound criteria used to confirm the diagnosis were:
• Visualization of an empty uterine cavity as well as an empty endocervical canal.
• A closed and empty cervical canal.
• The presence of embryonic/fetal pole and/or yolk sac with or without heart activity.
• The presence of a prominent and at times rich vascular pattern at or in the area of a scar in
the
We tested the hypothesis the relationship between the gestational sac of the CSP, previous
caesarean scar and the anterior uterine wall can be used to predict the evolution of these cases. In
order to do this, we propose a new sonographic sign, the cross-over sign (COS).
Briefly, in a sagittal view of the uterus, a straight longitudinal line was drawn connecting the
internal cervical os and the uterine fundus trough the endometrium (endometrial line) (Figure 1).
Then the gestational sac was identified and its superior-inferior (S-I) diameter perpendicular to the
endometrial line traced. According to the relationship between the endometrial line and the S-I
diameter of the ectopic sac, we divided the patients into two different groups:
1. COS1 insertion: when the gestational sac was implanted within the previous CS and ≥ two
thirds of the S-I diameter of the gestational sac was above the endometrial line towards the
anterior uterine wall (Figures 1,2)
2. COS2 insertion: when the gestational sac was implanted within the previous CS and < two
thirds of the S-I diameter of the gestational sac was above the endometrial line. Furthermore,
these cases were divided into two different categories according to the presence (COS2+) or
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
enot (COS2-) of an intersection between the S-I diameter of the ectopic sac and the
endometrial line (Figure 1,2).
Two examiners, blinded to pregnancy outcome and pathology reports, analysed all the images
independently and labelled them according to their COS insertion.
Furthermore, COS was ascertained in stored pictures from 38 consecutive patients referred to our
centre with a prenatal diagnosis of CSP ended in abortion or termination of pregnancy.
Final diagnosis of the type of MAP was made after surgery and hysterectomy on the basis of the
pathological examination of the removed uterus. Placenta accreta when diagnosed when anchoring
placental villi were attached to myometrium rather than decidua, but without completely invading
it.
Placenta increta when diagnosed when chorionic villi penetrate into the myometrium, while
placenta percreta when chorionic villi penetrate through the myometrium to the uterine serosa or
adjacent organs)14.
Study groups were compared using Student’s t-test for normally distributed continuous variables or
the Mann–Whitney U-test for non-normally distributed continuous variables, and the chi-square or
Fisher’s exact test (for expected cell size less than five) for dichotomous variables. The odd ratios
(OR) and 95% CI for the occurrence of MAP in each group were calculated. Furthermore, logistic
regression analysis was carried out in order to look for maternal, pregnancy or ultrasound
characteristics, including the type of COS insertion, independently associated with the occurrence
of different types of MAP.
The statistical analysis was performed using Stat Direct (StatsDirect statistical software,
http://www.statsdirect.com; England: StatsDirect Ltd. 2013) and SPSS version 15.0 (SPSS Inc.,
Chicago, IL, USA). Statistical significance was set at p<0.05. All p values were two-tailed.
STROBE guidelines were followed15.
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
eRESULTS
During the study period, 102 women with a prenatal diagnosis of MAP confirmed at
histopathological examination were referred to our center. Early first trimester ultrasound images
were retrieved from the local computerized databases in 68 cases.
At histopathological analysis, 34 cases were diagnosed as placenta percreta, 13 increta and 21
accreta.
General characteristics of the women included in the study are reported in Table. The two groups
did not show any significantly difference as regard for maternal age, parity and number of previous
cesarean section. Gestational age at birth was significantly higher in COS2 and this was due to the
higher number of placenta percreta cases in COS1 group. There was full agreement in the labelling
of all CPS images as regard for the type of COS sign between the two researchers.
The proportion of cases with placenta percreta was significantly higher in COS1 compared to COS2
group (83.3% vs 42.9%; p= 0.002); all pregnancies with COS1 implantation resulted in severe
forms of MAP, such as placenta increta or percreta, while there was no case of placenta accreta in
this group.
COS2 cases were then subdivided into two different categories on the basis of the intersection (39)
or no intersection (17) with the endometrial line (Figures 1,2). The risk of placenta percreta was
significantly higher in pregnancies with COS1 compared to COS2 insertion (OR: 6.67, 95% CI 1.3-
33.3; p= 0.001); furthermore, there was a significant difference in the occurrence of placenta
percreta between COS1 and COS 2+ (10/12 vs 18/39, p= 0.04), with an OR of 5.83, 95% CI 1.1-
30.2 (p=0.036). Finally, the risk of placenta percreta was even higher when comparing cases with
COS1 with COS2- (OR: 12.0, 95% CI 1.9-75.7; p= 0.003). COS2- cases had significantly more
cases of less severe forms of MAP (placenta accreta) compared to COS2+ (64.71% vs 28.21%; p=
0.017) and had a significantly lower risk of developing placenta percreta compared to those with
COS1 (OR: 0.069, 95% CI 0.01-0.4). Logistic regression analysis showed that COS1 insertion was
independently associated with the occurrence of severe forms of MAP such as placenta percreta and
increta (OR: 12.85, 95% CI 2.0-84.0), while COS2+ insertion was independently associated with
the occurrence of placenta accreta (OR: 4.37, 95% CI 1.1-17.0).
Ultrasound images of 38 cases of CSP referred to our centre in the same time period and which
underwent termination of pregnancy were evaluated for the presence of COS; assessment of this
ultrasound sign was possible in all cases; 79.0% (95% CI 62.7-90.4; 30/38) showed COS1 and
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
e21.1% (95% CI 9.6-37.3; 8/68) COS2+, while there was no case of COS2- in this sub-group of
patients.
DISCUSSION
The findings from this study showed that the assessment of the relationships between the ectopic
gestational sac, previous caesarean scar and anterior uterine wall (COS insertion) may represent a
reliable tool able to stratify the obstetric risk of women with a first trimester diagnosis of CSP. The
risk of placenta percreta was significantly higher in pregnancy with COS1 compared to COS 2+
(OR: 5.83, 95% CI 1.1-30.2). Furthermore, women with COS2- insertion had a significantly lower
risk of developing placenta percreta compared to those with COS1 (OR: 0.069, 95% CI 0.01-0.4).
Finally, COS1 insertion was an independent risk factor for placenta percreta.
Antenatal counselling of women with a prenatal diagnosis of CSP is challenging, especially in
asymptomatic cases. The current evidence is sparse and mainly derived from small retrospective
series including only highly symptomatic patients which report high rates of adverse maternal
outcomes such as profuse bleeding, uterine rupture and need for hysterectomy16. This has led in turn
to the common practice of offering termination of pregnancy to all this women10-12.
Evaluation of gestational sac implantation using COS criteria is a simple a reproducible tool to
ascertain the relationship between the ectopic sac, caesarean scar and anterior uterine wall. In the
present series, all cases with COS1 implantation ended in severe variants of MAP such as placenta
percreta or increta, while there was no case of placenta accreta in this group. It might be speculated
that COS1 implantation represents the most severe variant of CSP characterized by the implantation
of the ectopic sac within the previous caesarean scar and close to the anterior uterine wall, thus
potentially resulting in higher risk of bladder or parametrial invasion, massive haemorrhage and
intra-surgical complications.
The findings from this study highlight the fact that the relationship between the previous caesarean
scar and the ectopic gestational sac are not the only determinants in predicting the natural history of
CSP; the distance between the upper pole of the ectopic sac and the anterior uterine wall may
determine whether CSP will evolve towards anterior invasion eventually ending in acute symptoms
before viability or less severe form amenable of elective post-natal treatment.
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
eThe majority of MAP included in the present study showed COS2+ implantation at first trimester
ultrasound; furthermore, when looking at the ultrasound images of the 38 cases with CPS which
ended in termination of pregnancy, there was no case of COS2- implantation. COS2- implantation
may represent the milder variant of CPS which does not completely fit the classical proposed
ultrasound criteria for CSP; in this scenario, the implantation of the upper pole of ectopic
gestational sac relatively away from the uterine wall might result in a lower risk of further placental
invasion, with a reduced risk of developing severe types of MAP. Furthermore, the peculiar
insertion of COS2- cases relatively away from the anterior uterine wall, might contribute to the high
rates of false negative diagnoses during the early first trimester of pregnancy. At the time of the 11-
14 weeks’ scan, the relative migration of the uterine sac towards the uterine cavity, would make this
sub-type of CSP difficult to differentiate from a normal intra-uterine pregnancy. On the other hand,
insertion of the ectopic sac in close proximity to the anterior uterine wall, such as in case of COS1
and COS2+, would make CPS easier to diagnosis.
It might be speculated that the high rates of adverse maternal outcomes reported in women with
CSP, would be the results of COS1 implantation, which is relatively easy to diagnose, while most of
the COS2- cases will be missed at the scan, thus potentially leading to over-estimate the burden of
co-morbidities associated with this anomaly.
In the present series, a significant proportion of CSPs with COS2 implantation ended in milder
forms of MAP such as placenta accreta; furthermore, although the majority of CPS cases terminated
during the first trimester had COS1 implantation, almost one third (28%) had COS2+ insertion.
These findings raise doubts about whether women with CSP showing the implantation of the
ectopic sac away from the anterior uterine wall (COS2) should be offered termination of
pregnancies as the only available therapeutic option. It might be speculated that at least a small
proportion of these cases may progress towards milder forms of MAP which can be successfully
identified and treated. This concept should be mentioned during parental counselling, pointing out,
on the other hand, that the most likely evolution of CSP is towards severe invasion eventually
ending in acute life-threatening symptoms requiring immediate surgical intervention.
The small number of included cases, retrospective design of the study are the main limitations of
this study. Another limitation was the lack of information about the evolution of CPS cases
diagnosed in the first trimester which ended in termination of pregnancy.
Large multicentre studies prospectively looking at women with previous caesarean section from the
early first trimester onwards are needed in order to quantify the actual incidence and the evolution
of CSP in order to more appropriately counsel patients with a prenatal diagnosis of this anomaly.
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
eREFERENCES
1. Oyelese Y, Smulian JC. Placenta previa, placenta accreta, and vasa previa. Obstet Gynecol
2006; 107: 927-941.
2. Miller DA, Chollet JA, Goodwin TM. Clinical risk factors for placenta previa-placenta accreta.
Am J Obstet Gynecol 1997; 177:210-214.
3. E Jauniaux, D Jurkovic Placenta accreta: pathogenesis of a 20th century iatrogenic uterine
disease. Placenta 2012; 33; 244-251
4. Wu S, Kocherginsky M, Hibbard JU. Abnormal placentation: twenty-year analysis. Am J Obstet
Gynecol 2005; 192: 1458-1461.
5. Timor-Tritsch IE, Monteagudo A, Cali G, Palacios-Jaraquemada JM, Maymon R, Arslan AA,
Patil N, Popiolek D, Mittal KR. Cesarean scar pregnancy and early placenta accreta share
common histology. Ultrasound Obstet Gynecol 2014; 43: 383-395.
6. Timor-Tritsch IE, Monteagudo A, Cali G, Vintzileos A, Viscarello R, Al-Khan A, Zamudio S,
Mayberry P, Cordoba MM, Dar P. Cesarean scar pregnancy is a precursor of morbidly adherent
placenta. Ultrasound Obstet Gynecol 2014; 44: 346-353.
7. Zosmer N, Fuller J, Shaikh H, Johns J, Ross JA. Natural history of early first-trimester
pregnancies implanted in Cesarean scars. Ultrasound Obstet Gynecol 2015; 46: 367-375.
8. D'Antonio F, Palacios-Jaraquemada J, Lim PS, Forlani F, Lanzone A, Timor-Tritsch I, Cali G.
Counseling in fetal medicine: evidence-based answers to clinical questions on morbidly
adherent placenta. Ultrasound Obstet Gynecol 2016;47: 290-301.
9. Timor-Tritsch IE, Monteagudo A, Cali G, El Refaey H, Kaelin Agten A, Arslan AA. Easy
sonographic differential diagnosis between intrauterine pregnancy and cesarean delivery scar
pregnancy in the early first trimester. Am J Obstet Gynecol 2016 Feb 17. pii: S0002-
9378(16)00316-1.
10. Seow KM, Huang LW, Lin YH, Lin MY, Tsai YL, Hwang JL. Cesarean scar pregnancy: issues
in management. Ultrasound Obstet Gynecol 2004; 23: 247-253.
11. Timor-Tritsch IE, Monteagudo A, Bennett TA, Foley C, Ramos J, Agten Kaelin A. A new
minimally invasive treatment for cesarean scar pregnancy and cervical pregnancy. Am J Obstet
Gynecol 2016 Mar 12. pii: S0002-9378(16)00472-5.
12. Timor-Tritsch IE, Cali G, Monteagudo A, Khatib N, Berg RE, Forlani F, Avizova E. Foley
balloon catheter to prevent or manage bleeding during treatment for cervical and Cesarean scar
pregnancy. Ultrasound Obstet Gynecol 2015; 46: 118-123.
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
e13. Timor-Tritsch IE, Monteagudo A, Santos R, Tsymbal T, Pineda G, Arslan AA. The diagnosis,
treatment, and follow-up of cesarean scar pregnancy. Am J Obstet Gynecol 2012; 207: 44.e1-13.
14. Benirschke K, Kaufmann P, Baergen RN. Pathology of the human placenta. 5th ed. New York:
Springer-Verlag; 2006.
15. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE
Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology
(STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 2008;
61:344-349.
16. Timor-Tritsch IE, Cali G, Monteagudo A, Khatib N, Berg RE, Forlani F, Avizova E. Foley
balloon catheter to prevent or manage bleeding during treatment for cervical and Cesarean scar
pregnancy. Ultrasound Obstet Gynecol 2015; 46: 118-123.
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
eTable 1: general characteristics of the population analysed in the study.
Characteristics Overall population (n=68) COS 1 (n=12) COS 2 (n=56) p-value Maternal age 34.32±4.24 34.50±3.32 34.29±4.44 0.877 GA at delivery 34.6±2.06 31.86±2.37 35.18±1.43 0.0001 Parity 2 (1-3) 3 2 (2-3) 0.629 Previous CS 2 (1-2) 2 2 (1-2) 0.826 Histopathology
Placenta accreta Placenta increta Placenta percreta
33 13 34
0 (0)
2 (16.7) 10 (83.3)
21 (37.5) 11 (19.6) 24 (42.9)
0.012
1 0.02
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
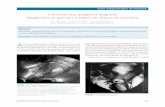
eFIGURE LEGEND
Normal COS1
COS2-COS2+
a) b)
c) d)
Figure 1 (a-d). Diagrammatic representation showing the relationships between ectopic gestational
sac, previous CS and anterior uterine wall (COS insertion). In a sagittal view of the uterus, a straight
longitudinal line is drawn connecting the internal cervical os and the uterine fundus trough the
endometrium (endometrial line) (yellow line). Then the gestational sac is identified and its S-I
diameter perpendicular to the endometrial line traced. According to the relationship between the
endometrial line and the S-I diameter of the ectopic sac, patients can be divided into two different
groups: normal pregnancy (a), the gestational sac implant away from the CS, in close proximity to
the uterine fundus; COS1 insertion (b), gestational sac is implanted within the previous CS and ≥
two thirds of the S-I diameter of the gestational sac was above the endometrial line towards the
anterior uterine wall; COS2+ insertion (c), gestational sac is implanted within the previous CS and
< two thirds of the S-I diameter of the gestational sac is above the endometrial line; COS2- insertion
(d), gestational sac is implanted within the previous CS, < two thirds of the S-I diameter of the
gestational sac is above the endometrial line but there is no intersection between the S-I diameter of
the ectopic sac and the endometrial line.
This article is protected by copyright. All rights reserved.
Acc
epte
d A
rticl
eCS
B
GS
EL
E
A Ut W
Ut F
CSO12a
C
2b CSO2+ CSO2-2c
A Ut WC
EL
Ut F
CS
GS CS
Ut F
EL
GS
CA Ut W
Figure 2 (a-c). Ultrasound images showing the different types of COS insertion (A. Ut. W: anterior
uterine wall; B: bladder; C: cervix, CS: caesarean scar; E: endometrium; EL: endometrial line; GS:
gestational sac; S-I d: superior-inferior diameter; Ut. F: uterine fundus).
This article is protected by copyright. All rights reserved.