Name: ODD Period: Week: 21–22 Dates: 1/18–1/29 Unit:...
Transcript of Name: ODD Period: Week: 21–22 Dates: 1/18–1/29 Unit:...
-
clementaged.weebly.com Name: _______________________________________________ ODD Period: _______
Week: 21–22 Dates: 1/18–1/29 Unit: Atmosphere
�1
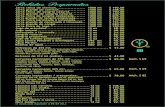
Assignment Your Score Total Points Possible
Good to Know Vocabulary Words 40
Atmosphere Notes 80
Atmospheric Layers Internet Worksheet 20
Atmospheric Layers Foldable 40
Solar Energy and the Atmosphere Notes 40
Oxygen in the Atmosphere Lab 40
Atmosphere Crossword 20
Study Guide 60
TOTAL 340
Monday Tuesday Wednesday Thursday Friday
18 No School 19 E 20 O *Atmosphere Notes *Vocabulary *Atmospheric Layers Internet Worksheet
21 E 22 O *Solar Energy Notes *Atmospheric Layers foldable
clement gone
25 E 26 O *Oxygen in the Atmosphere Lab *Study Guide *Crossword
27 E
*Mandatory Fair Meeting 6pm
28 O *Unit Quiz *Packet Due
29 E
Ag Earth Science Fact of the Week
Did you know...The mixture of gases in the atmosphere has taken over 4.5 billion years to evolve.About 99% of the atmosphere is made up of oxygen and nitrogen. The remainder is made up of argon, carbon dioxide and very small amounts of hydrogen, ozone, methane, carbon monoxide, helium, neon, krypton and xenon.
-
Good to Know Vocabulary WordsWord Definition Picture or Example
Ozone
Troposhpere
Stratosphere
Mesosphere
Thermosphere
Heat
Temperature
Conduction
�2
-
Radiation
Reflection
Greenhouse Effect
Atmosphere
Chromosphere
Photosphere
Word Definition Picture or Example
�3
-
Cue Column(titles, vocab, big ideas,
test questions)Aatmosphere notes
�4
-
Cue Column(titles, vocab, big ideas,
test questions)Aatmosphere notes
�5
-
Cue Column(titles, vocab, big ideas,
test questions)Aatmosphere notes
�6
-
Summary(briefly describe the main concepts, major points)
Cue Column(titles, vocab, big ideas,
test questions)Aatmosphere notes
�7�
-
atmospheric layers internet worksheet
Please type in the following website:
http://www.windows.ucar.edu/tour/link=/earth/Atmosphere/layers.html
At the top, please click on “Advanced” and answer the following questions. After you read the paragraph, you will also have to click on the words in blue to answer some of the questions.
1. Where is the atmosphere the thickest?
2. The troposphere contains how much of the Earth’s atmosphere?
3. What occurs in the troposphere?
4. Heat is mixed through the troposphere because on average that layer is usually what?
5. Why do most jets fly in the stratosphere?
6. What is absorbed from the Sun in the stratosphere?
7. What is the stratosphere also known as?
8. Which two layers is the stratosphere between?
9. What burns up in the mesosphere?
10. Why do these burn in the mesosphere?
11. Atmospheric temperatures reach to what value (low) in the mesosphere?
12. Which two layers is the mesosphere between?
13. The thermosphere is a layer with what?
14. What else orbits in the thermosphere?
15. The air is so thin, that what happens with temperature changes?
16. What is the ionosphere made of?
17. In what layer does the atmosphere merge into space?
18. The Exosphere is what limit to our atmosphere?
When you are finished, please go to the website below in the Atmospheric Arcade.
http://calipsooutreach.hamptonu.edu/arcade.html
Please play or watch the following in this order if time permits: 1. Layers of the Atmosphere 2. Atmospheric Crossword Puzzle 3. Atmospheric Trivia Game
�8
http://www.windows.ucar.edu/tour/link=/earth/Atmosphere/layers.htmlhttp://calipsooutreach.hamptonu.edu/arcade.html
-
atmospheric layers foldable
�9
-
Cue Column(titles, vocab, big ideas,
test questions)solar energy and the atmosphere
�10
-
Cue Column(titles, vocab, big ideas,
test questions)solar energy and the atmosphere
�11
-
Cue Column(titles, vocab, big ideas,
test questions)solar energy and the atmosphere
�12
-
�
Summary(briefly describe the main concepts, major points)
Cue Column(titles, vocab, big ideas,
test questions)solar energy and the atmosphere
�13
-
Finding the Amount of Oxygen in the Atmosphere: Lab
Background Information: The atmosphere of the earth is composed of a mixture of gases. The two most abundant gases are nitrogen and oxygen. Oxygen also appears in the earth’s crust combined with other elements to form minerals. By measuring volume, you can determine the percentage of oxygen in the air.
Objective In this investigation, you will study how much of the air is
consumed during combustion.
Problem: What percentage of earth’s atmosphere is oxygen?
Materials:
! Birthday candle ! 250-mL beaker ! 100-mL beaker
! Matches ! Wet-erase marker ! Large test tube
! Penny ! Water
Procedure: 1. Light the candle and let a few drops of wax drop onto the penny. Blow out the candle, and
place the candle upright on the penny in the wax. The candle should stick to the penny. 2. Fill the test tube with water. Using the graduated cylinder, measure this volume of water and
record it in the Data Table, #1. (The volume is also the volume of air in the test tube!) 3. Pour the water into the beaker. Carefully place the penny and the candle in the center of the
beaker. The penny should keep the candle upright. 4. Carefully light the candle, and rapidly invert the test tub over the lighted candle. Make sure
that the open end of the test tube is well under the surface of the water but not touching the bottom of the beaker.
5. As the candle uses the oxygen in the test tube, the candle will go out and water will be drawn into the tube to replace the oxygen.
6. When the candle goes out, carefully mark the level of the water in the test tube with the wet-erase marker. Remove the test tube.
7. Fill the test tube with water to this line. Using the graduated cylinder, measure this volume of water and record it in the Data Table, #2. (The volume is the volume of air in the test tube minus the oxygen that the candle used!)
8. To find the volume of oxygen originally in the test tube, subtract the volume of air after the candle goes out from the original volume. Record the volume in the Data Table, #3.
9. Determine the percentage of oxygen in the air by using the formula below. Record this percentage in the Data Table, #4.
% oxygen in air = x 100
�14
Volume of oxygen in test tube . Total volume of air in test tube at start
-
�15
Nam
eD
ate
(Key
# 1
- 91
0090
)
Ear
th S
cien
ce -
Cha
pter
17
Com
plet
e th
e cr
ossw
ord
puzz
le.
78
S E
1
5
S
14 T
16
13 P
2 4
E Q N
10 O
17
3
6
F
9
M
11
T
12
E
E
15
I
18
UT
19 S
CONDUCTION
OZONE
STRATOSPHERE
MESOSPHERE
SUMMERSOLSTICE
WINTERSOLSTICE
THERMOSPHERE
RADIATION
HEAT
ISOTHERM
GREENHOUSEEFFECT
CONVECTION
SPRINGEQUINOX
SCATTERING
TROPOSPHERE
AUTUMNALEQUINOX
ALBEDO
REFLECTION
TEMPERATURE
Across
4The change in direction that
occurs when a light ray
bounces off of a surface.
6the heating of Earth's surface
and atmosphere from solar
radiation being absorbed and
emitted by the atmosphere,
mainly by water vapor and
carbon dioxide (2 words)
9the third layer in the
atmosphere; the atmospheric
layer between the stratosphere
and the thermosphere. (coldest
layer)
11
a measure of the average
kinetic energy of individual
atoms or molecules in a
substance
12
The time of the year when the
sun is directly over the
Southern Hemisphere marking
the beginning of winter around
Dec. 21. (2 words)
15
The movement of thermal
energy through liquids and
gases.
16
the fraction of total radiation
that is reflected back by a
surface
18
The transfer of thermal energy
by direct contact between
objects.
19
the redirecting (in all directions)
of light by small particles and
gas molecules in the
atmosphere.
Down
1A line that connects points of
equal temperature
2This is when the earth tilts so
that we get an equal amount of
daylight and night time. Spring
Equinox happens after the
season of Winter. (2 words)
3A threeatom form of oxygen
which absorbs ultraviolet light,
protecting earth's life forms.
5The time of year in the northern
hemisphere when the noon sun
appears to be farthest north. It
occurs around June 21 and
marks the beginning of the
summer season in the northern
hemisphere. (2 words)
7thermal energy transferred from
one object to another
8the outer most layer of earth's
atmosphere.
10
The time of year when day and
night are of equal length;
beginning of autumn in the
Northern hemisphere. The sun
is on the equator. (2 words)
13
the lowest layer of Earth's
atmosphere, where weather
occurs
14
the second lowest layer of
Earth's atmosphere; the ozone
layer is located in the upper
stratosphere.
17
Thermal energy that travels in
waves and can pass through
empty space as well as some
objects.
-
Data & Results:
DATA TABLE
Conclusions:
1. Would the same result for the percentage of oxygen in air be obtained if a larger test tube was used? What about if a larger candle was used?
2. Why does the water rise in the test tube as the candle goes out?
3. Nitrogen is the other major component of air (78.1%). What property of nitrogen have you discovered as a result of this experiment?
4. How much oxygen is present in 5 L of air?
5. Why is oxygen such an important part of the earth’s atmosphere?
6. Based on your observations, what is an effective method of putting out a small fire
7. “As the altitude of an area increases, the density of the atmosphere decreases.” How can this statement be used to explain why it is more difficult to breathe in Denver, which has an altitude of more than 1500 meters, than in Houston, which is at sea level?
1. Volume of Air in the Test Tube at Start mL
2. Volume of Air in Test Tube After Candle Goes Out mL
3. Volume of Oxygen (#1 - #2) mL
4. Percentage of Oxygen in Air %
�16
-
atmosphere study guide Answer the questions in complete sentences or phrases on a SEPARATE piece of paper. Attach pages to packet when done.
1. What is weather? 2. What is climate? 3. Which layer in the atmosphere does weather change occur in? 4. The atmosphere is pulled toward earth’s surface by ___________________. 5. What are the most abundant elements in the atmosphere?
a. b.c.d.
6. The compounds carbon dioxide and water vapors are not listed on the atmosphere make up because
7. Why is there no longer an abundant about of hydrogen and helium in our atmosphere any more?
8. Most of the ozone in located in the ________________________. 9. Most weather changes take place in what layer of the atmosphere? 10. The ozone layer assists with filtering ____________________. 11. Most air pollution come from the burning of ______________________. 12. About 50% of the solar energy is absorbed by _____ ____________
__________. 13. The process by which the earth’s atmosphere traps infrared rays is called 14. Increases in atmospheric levels of carbon dioxide would probably cause the
atmosphere to __________________ __________________. 15. Is air a good conductor of heat? 16. How is water vapors added to the atmosphere? 17. What would happen over time if there was no ozone layer? 18. Which layer is the coldest, what is its temperature? 19. Which layer is ozone primarily found in? 20.Which atmospheric layer is closest to the earth’s surface? 21. Which layer do we live in? 22.The lower boundary of the mesosphere extends upward from the
_______________. 23.The lower boundary of the stratosphere extends upward from the
_______________. 24.Which gas is most responsible for the greenhouse effect __________________. 25. What are the two must abundant compounds on the atmosphere? 26.Describe the green house effect: 27. List the four layers of the atmosphere and give one distinguishing characteristic
of each layer: a.b.c.
�17
-
28.Be able to look at this image and answer questions:
�18