multicommutated stepwise injection analysis of biodiesel with classical least squares method
-
Upload
kelmy-thomas-maria -
Category
Documents
-
view
216 -
download
0
Transcript of multicommutated stepwise injection analysis of biodiesel with classical least squares method

8/10/2019 multicommutated stepwise injection analysis of biodiesel with classical least squares method
http://slidepdf.com/reader/full/multicommutated-stepwise-injection-analysis-of-biodiesel-with-classical-least 1/7
Determination of silicon, phosphorus, iron and aluminum in biodiesel
by multicommutated stepwise injection analysis with classical least
squares method
Christina S. Vakh ⇑, Andrey V. Bulatov, Andrey Y. Shishov, Andrey V. Zabrodin, Leonid N. Moskvin
Department of Analytical Chemistry, Institute of Chemistry, Saint Petersburg State University, Saint-Petersburg, pr. Universitetskij 26, 198504, Russia
h i g h l i g h t s
The flow-based procedure for Si, P, Fe,
Al determination in biodiesel is
proposed.
The proposed procedure is simple,
fast and flexible.
Applied to biodiesel samples obtained
from castor bean, sunflower, canola
and corn.
g r a p h i c a l a b s t r a c t
a r t i c l e i n f o
Article history:
Received 8 May 2014
Received in revised form 19 June 2014
Accepted 25 June 2014
Available online 8 July 2014
Keywords:
Biodiesel
Multicommutated stepwise injection
analysis-classical least squares method
Inorganic elements
a b s t r a c t
Recently suggested multicommutated stepwise injection analysis (MCSWIA) coupled with classical least
squares method (CLS) has been developed for silicon, phosphorus, iron and aluminum determination in
biodiesel samples after a microwave digestion. This method allows to carry out four color-forming reac-
tions simultaneously: aluminum (III) and iron (III) with chromazurol S and phosphate and silicate ions
with ammonium molybdate, in two mixing chambers which are commutated into MCSWIA manifold.
The concentrations of analytes were determined by two data matrixes using CLS method. Each of exper-
imental calibration matrixes for CLS were designed with 10 calibration set solutions of 1–10 mg kg1 for
aluminum and 2–20 mg kg1 for iron, silicon and phosphorus. The appropriate experimental conditions
for microwave digestion and automated MCSWIA spectrophotometric determination of analytes were
investigated. Recovery tests show results between 98% and 107% for all type of biodiesel samples. The
limits of detection (LOD) obtained using the MCSWIA-CLS system were 0.3 mg kg
1
for aluminum and0.6 mg kg1 for iron, silicon and phosphorus. The proposed method was successfully applied for the
determination of analytes in different type of biodiesel. The analytical results obtained by MCSWIA-
CLS were compared with the results obtained by reference methods.
2014 Elsevier Ltd. All rights reserved.
1. Introduction
Biodiesel is a relatively new source of renewable energy the
value of which has recently obtained increasing attention
http://dx.doi.org/10.1016/j.fuel.2014.06.059
0016-2361/ 2014 Elsevier Ltd. All rights reserved.
⇑ Corresponding author. Tel./fax: +7 9291100987.
E-mail address: [email protected] (C.S. Vakh).
Fuel 135 (2014) 198–204
Contents lists available at ScienceDirect
Fuel
j o u r n a l h o m e p a g e : w w w . e l s e v i e r . c o m / l o c a t e / f u e l

8/10/2019 multicommutated stepwise injection analysis of biodiesel with classical least squares method
http://slidepdf.com/reader/full/multicommutated-stepwise-injection-analysis-of-biodiesel-with-classical-least 2/7
worldwide [1]. Biodiesel is composed by the mixture of mono-alkyl
esters of long chain fatty acids, obtained through a transesterifica-
tion reaction of vegetable oils or animal fats with short-chain alco-
hols in the presence of catalysts, such as sodium hydroxide or
potassium [2]. Nevertheless, the final mixture of biodiesel produc-
tion process may contain different kind of contaminants which
have negative impacts for both motor and environment. During
the transesterification process, such undesired elements as silicon,phosphorus, iron and aluminum may also present in a biodiesel
product. The presence of these elements in biodiesel can have a
great damage the engines. Thus, during the combustion of biodie-
sel the organic compounds of silicon, iron and aluminum are trans-
formed into insoluble oxides, which can deposit in combustion
chamber. The raised content of phosphorus in biodiesel causes to
significant harmful influence to the environment and prevent the
operation of catalytic convectors [3].
For determination of silicon, phosphorus, iron and aluminum in
biodiesel the atomic absorption and atomic emission spectroscopy
have been proposed using the procedure described in [4–6] and
[7,8] respectively. For determination of other metals (Cu and Pd)
in biodiesel the electrochemical methods have been proposed
[9,10]. For reducing of matrix influence the atomic absorption
and atomic emission procedures recommend dissolving the biodie-
sel sample in organic solvents, such as xylene, kerosene and other.
The use of these organic solvents requires special attention
because of their toxicity. The electrochemical methods require
the use of less toxic solvents such ethanol or isopropanol but they
have not been proposed for determination of silicon, phosphorus,
iron or aluminum Thus, though atomic absorption and atomic
emission described procedures are highly sensitive and relatively
simple, however, they involve some disadvantages, such as large
consumption of toxic organic solvents and high cost of the analysis.
Taking into account these disadvantages, more preferable for
determination of silicon, phosphorus, iron and aluminum are auto-
mated procedures which are based on principles of flow analysis
and allowed to work with small volumes, saving samples and
reagents and therefore contributing to a green analytical chemistry[11–15]. Despite the simplicity of flow injection analysis (FIA) the
automated flow procedures were developed only for phosphorus
determination in biodiesel. The most of these procedures are based
on the well-known molybdenum blue reaction. The fully auto-
mated flow injection procedure for phosphorus determination in
biodiesel [11] includes the samples digestion by mixture of acids
in a heating block of FIA system. The procedure described in [14]
proposed to determined phosphorus in biodiesel using a micro-
flow-batch analyzer with solenoid micro-pumps. The samples in
this case were mineralized using an ashing procedure at 550 C fol-
lowed by dissolution of the residue.
This present work describes a new procedure for simultaneous
determination of silicon, phosphorus, iron and aluminum in bio-
diesel after a microwave digestion using recently suggested multi-commutated stepwise injection analysis [16].
The MCSWIA is an automated system which integrates the con-
cepts of batch and fl0ow methods. The MCSWIA manifold consists
of several mixing chambers and programmed multi-commutated
valves, peristaltic pumps and detector. The mixing chambers are
often used for mixing of the sample with solutions of color-forming
reagents by gas bubbles. The number of mixing chambers is deter-
mined by the number of analytes and the number of corresponding
color-forming reactions, which are necessary for their determina-
tion. Previously MCSWIA has been successfully implemented for
determination of zinc (II), copper (II) and nickel (II) in wet aerosols
[14]. In this procedure three mixing chambers were used for per-
forming of three selective color-forming reactions. Nevertheless,
the MCSWIA offers a number of disadvantages such as the neces-sity to increase the number of mixing chambers for performing
of corresponding color-forming reactions for each new analyte.
This significantly increased the time of analysis, memory effects
and complicated the analysis. Moreover the number of mixing
chambers is limited by the number of ports of a valve.
These disadvantages can be overcome by coupling of MCSWIA
with classical list squares method (MCSWIA-CLS). The CLS method
has been successfully implemented for multicomponent flow-
injection [17,18] and sequential injection [19] analysis. In thepresent work, CLS method has been used for data analysis of simul-
taneously MCSWIA determination of four analytes. For this pur-
pose, MSWIA manifold includes two similar mixing chambers
where the color-forming reactions were performed: aluminum
(III) and iron (III) with chromazurol S and phosphate and silicate
ions with ammonium molybdate.
2. Experimental
2.1. Reagents and solutions
All solutions were prepared from analytical reagent grade. Ultra
pure water from Millipore Milli-Q RG (Millipore, USA) was used for
preparation of solutions and dilution. The aqueous stock solutionsof 0.04 mmol L 1 silicon (IV) and phosphorus (V) were prepared by
diluting of 1 gL 1 standard solutions of SiO32 and PO4
3 (Merk,
Germany) with deionized water. To prepare the stock solutions
of organic forms of silicon and phosphorus (10 mg kg1) the appro-
priate amount of silicon oil and triphenylphosphine were dissolved
in the biodiesel. The solutions of ammonium molybdate
(0.2 mol L 1), oxalic acid (0.01 mol L 1), ascorbic acid
(0.05 mol L 1) and sulfuric acid (0.02 mol L 1) were prepared by
dissolving the corresponding amount in deionized water.
The stock aqueous solutions of 0.04 mmol L 1 of aluminum (III)
and iron (III) were prepared by dissolving of corresponding amount
of Al(NO3)3 and FeCl3 in 0.1 mol L 1 of HCl. To prepare the stock
solutions (10 mg kg1) of organic forms of iron (III) and aluminum
(III) in biodiesel 2-ethylhexanoates of Al and Fe (ABCR GmbH &
CO.KG) were dissolved in the biodiesel. The 0.2 mmol L 1 of the
chromazurol S solution was prepared immediately before the
experiment by mixing the solution of 0.4 mmol L 1 of color-form-
ing reagent and acetate buffer (pH 5) solution in a ratio of 1:1,
respectively. All chemicals of analytical reagent grade were used
without further purifications.
2.2. Samples
Four different samples of biodiesel were obtained from different
sources: castor bean, sunflower, canola and corn. These samples
were previously prepared using alkaline catalyzer sodium hydrox-
ide (NaOH) [18,20].
2.3. Sample preparation
The sample preparation was performed by microwave diges-
tion. Biodiesel sample (1 g) was placed into a digestion vessel.
Then, 5 mL of 10 mol L 1 HNO3 and 2 mL of 9 mol L 1 H2O2 were
added to this vessel. The sample was heated according to the opti-
mized program presented in Table 1. After cooling the solution was
Table 1
Optimized program for microwave-assisted acid digestion of biodiesel samples.
Step Applied power (W) Time (min) Pressure (MPa)
1 400 5 0.5
2 400 5 1
3 400 25 15
C.S. Vakh et al. / Fuel 135 (2014) 198–204 199

8/10/2019 multicommutated stepwise injection analysis of biodiesel with classical least squares method
http://slidepdf.com/reader/full/multicommutated-stepwise-injection-analysis-of-biodiesel-with-classical-least 3/7
transferred to a 10 mL volumetric flask, made up to the pH 5.0 by
addition of ammonia solution (1:1) and diluted to 10 mL with
deionized water. The prepared solution was analyzed by
MCSWIA-CLS.
2.4. Manifold and apparatus
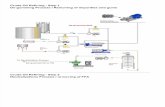
The MCSWIA-CLS manifold (Fig. 1) includes: two solenoidvalves (Cole–Parmer, USA), two peristaltic pumps MasterFlex L/S
(Cole–Parmer, USA) ensuring a reverse flow (flow rate is from 0.5
to 5 mL/min), two identical thermostated mixing chambers (MC)
(cylindrical-shaped glass tubes with a funnel-shaped inlet at the
bottom, 20 mm in height and 10 mm in i.d.) and communication
tubes (PTFE, 0.5 mm in i.d.). It is equipped with a source of visible
light LS-1 and fiber-optic spectrometer USB 4000 (Ocean Optics,
USA) with a 50 mm path length flow cell FIA-Z-SMA-50-TEF (Fia-
lab, USA) and a single-channel membrane separator with a PTFE
membrane. Mathematical operations were performed by the pro-
gram Excel (Microsoft Office).
Acidity of the solution was measured by a pH meter pH-410
(Aquilon, Russia). MDS-6 microwave digestion system (Shandong,
China) was used for samples digestion.
ICPE-9000 Inductively Coupled Plasma Emission Spectrometer
(Shimadzu, Japan) was used for silicon determination by a refer-
ence method. AA-7000 Atomic Absorption Spectrophotometer
(Shimadzu, Japan) was used for phosphorus, aluminum and iron
determination by a reference method.
2.5. The MCSWIA-CLS procedure
At the first stage of the measurement, sample portions of 0.1 mL
each (a) were sequentially delivered to the thermostated under
60 C MC (1, 2) using valves (1) and (2). Then, for aluminum and
iron determination the 0.1 mL of 0.2 mmol L 1 chromazurol S solu-
tion (b) was delivered to the MC (1). To stir the reaction mixture, a
flow of air was passing through the port (d) at a rate of 5 mL min1
into the MC (1) for 30 s. To achieve the completeness of the color-
forming reaction the homogeneous solution was stay in the MC (1)
for 240 s.
At the second stage of measurement, while the colored com-
plexes were formed in MC (1), the components of reacting mixture
for determination of phosphorus and silicon were sequentially
delivered to the MC (2) through the port of valve (1) in following
order: 0.1 mL of 0.2 mol L 1 solution of ammonium molybdate
(e), 0.3 mL of 0.02 mol L 1 sulfuric acid ( f ) and 0.1 mL of
0.01 mol L 1 oxalic acid solution ( g ). The reacting mixture was
stirred in the MC (2) by the flow of air ( d) for 60 s. After that the
0.1 mL of 0.05 mol L 1 ascorbic acid solution (h) was also delivered
to MC (2). Then, pause was kept to complete the color-forming
reactions for 120 s.
At the third stage of measurement, the reaction mixtures from
MC (1) and MC (2) using valve (1, 2) and peristaltic pump (2) were
sequentially delivered to the spectrophotometric detector where
the analytical signals were measured in the flow-stop conditionfor 20 s at the wavelength range from 510 to 570 nm (for alumi-
num and iron determination) and at the wavelength range from
820 to 910 nm (for phosphorus and silicon determination). Then,
the solutions from detector were discharged to waste. To avoid
the memory effect the manifold was washed out with deionized
water.
2.6. Procedure for the determination silicon, phosphorus, iron and
aluminum in biodiesel by a reference method
For the determination of silicon [7] and phosphorus [5] in
biodiesel by a reference method the pre-treatment stage was per-
formed by dilution of the samples in ethanol or xylene and farther
analysis by microwave-induced plasma optical emission spectrom-
etry (MIP OES) and atomic absorption spectroscopy (AAS), respec-
tively. Aqueous solutions of silicon and phosphorus were used for
plotting the analytical curve.
The determination of iron and aluminum in biodiesel was per-
formed by high-resolution continuum source electrothermal
atomic absorption spectrometry [4]. Sample preparation includes
simply dilution the biodiesel samples by ethanol at room temper-
ature. Aqueous solutions of iron were used for plotting the analyt-
ical curve. For the calibration of aluminum the solutions were
prepared using ethanol.
3. Results and discussion
3.1. Theoretical aspects
Color-forming reagent chromazurol S was chosen for simulta-
neous spectrophotometric determination of aluminum and iron
in biodiesel. This reagent forms stable intensively colored com-
plexes with aluminum (III) and iron (III) in acetate buffer medium
[21]. The absorbance spectra of color-forming reagent chromazurol
S and its complexes with aluminum (III) and iron (III), obtained in
MCSWIA manifold, are shown on Fig. 2. The absorbance spectra of
iron (III) and aluminum (III) complexes with chromazurol S have
maxima at wavelengths 530 and 560 nm respectively and they
Fig. 1. The MCSWIA manifold for determination of silicon, phosphorus, iron and aluminum in biodiesel.
200 C.S. Vakh et al./ Fuel 135 (2014) 198–204

8/10/2019 multicommutated stepwise injection analysis of biodiesel with classical least squares method
http://slidepdf.com/reader/full/multicommutated-stepwise-injection-analysis-of-biodiesel-with-classical-least 4/7
are strongly overlapped. So, the simultaneous spectrophotometric
determination of aluminum (III) and iron (III) by individual calibra-
tion curves is impossible.
The molybdenum blue reactions were chosen for determination
of silicon and phosphorus. The absorbance spectra of molybdo-
phosphoric and molybdosiliconheteropoly acids solutions mea-
sured in MSWA manifold (Fig. 2) are strongly overlapped. So, the
simultaneous spectrophotometric determination of silicon and
phosphorus by individual calibration curves is also impossible.
3.2. The MCSWIA manifold
The MCSWIA manifold includes two similar eight-way solenoidvalves and two peristaltic single-channel pumps (Fig. 1). The first
valve is used for sequential injection of samples, reagent solutions
and a gas phase into the flow system. The gas phase is used for
mixing of solutions in the mixing chambers which are input to
the second valve.
To provide the effective mixing of solution in the MC, the cylin-
drical-shaped glass tube with a funnel-shaped inlet at the bottom
was preferred. The optimal volumes of samples and reagent solu-
tions were studied. A volume of 0.1 mL provides minimum con-
sumption of samples and reagent solutions as well as the
maximum reproducibility (RSD 0.25%). In order to prevent the mix-
ing of color-forming reactions at their consistent movement from
the MCs to the detector, these zones were segmented by gas bub-
bles, which were injected into the flow system for mixing of thesolutions in MCs. To eliminate the air bubbles from the flow
stream, the membrane separator was installed in front of the
detector.
3.3. Optimization of chemical variables
3.3.1. Optimization the MCSWIA spectrophotometric determination of
iron (III) and aluminum (III)
To optimize the condition of MCSWIA spectrophotometric
determination of iron (III) and aluminum (III) the effect of concen-
tration of color-forming reagent has been studied. For this purpose,
a portion (0.1 mL) of 0.04 mmol L 1 aluminum (III) or iron (III)
solution, 0.1 mL of chromazurol S were delivered into the thermo-
static MC (1) (60C). The concentration of color-forming reagentwas varied from 0.05 to 0.5 mmol L 1. The reacting mixture was
stirred by the flow of air for 300 s, and then stopped for 300 s.
When the completeness of the coloring reaction was achieved
the reacting mixture was delivered to the spectrophotometric
detector. Absorbance of solution was measured in the stop-flow
conditions at 530 and 560 nm for iron (III) and aluminum (III) ions
respectively. It was found that the optimal concentration of color-
forming reagent for aluminum (III) and iron (III) determination is
0.08 mmol L 1 which ensures the formation of complexes in a ratio
of 2:1 (reagent:analyte). To provide the simultaneously determina-
tion of aluminum (III) and iron (III) with chromazurol S the concen-
tration of 0.2 mmol L 1 of color-forming reagent was chosen as
optimal.
Also the influence of temperature and time on the completeness
of color-forming reactions in MCSWIA was investigated. The tem-
perature in the MC (1) varied from 30 C to 60 C, as well as the
thermostating time in the MC (1) was varied from 1 to 10 min. It
was found that the completeness of complexes formation wereachieved by thermostating the reacting mixture during 4 min at
60 C for both aluminum and iron.
3.3.2. Optimization the MCSWIA spectrophotometric determination of
silicon and phosphorus
The molybdenum blue reactions are depended from the acidity
of medium and the concentration of molybdenum ions and oxalic
acid. To obtained the optimal acidity of MCSWIA-CLS spectropho-
tometric determination of silicon and phosphorus the effect of
the concentration of sulfuric and oxalic acid has been studied.
For this purpose, 0.1 mL of 0.04 mmol L 1 phosphate ions or sili-
cate ions solution or water, 0.1 mL of 0.2 mol L 1 solution of
molybdenum ions, 0.3 mL of sulfuric acid and 0.1 mL of solution
of oxalic acid were delivered into a MC (2). The concentration of sulfuric acid was varied from 0.005 to 0.05 mol L 1. The concentra-
tion of oxalic acid was varied from 0.005 to 0.05 mol L 1. The react-
ing mixture in the MC (2) was stirred by the flow of air for 60 s (f).
After that, the 0.1 mL of 0.05 mol L 1 of ascorbic acid solution was
delivered into the MC (2) and a pause was kept for 120 s for
Fig. 2. Absorbance spectra of chromazurol S (a) and its complexes with aluminum
(III) (b), iron (III) (c) (CR = 0.04 mmol L 1, C Al = 0.02 mmol L 1, C Fe = 0.02 mmol L 1);
the mixture of iron (III) and aluminum (III) (d) (C R = 0.04 mmol L 1, CAl = 0.01 -
mmol L 1, CFe = 0.01 mmol L 1); phosphomolybdic (e), silicomolybdic (f) and
ammonium molybdic (g) heteropoly acids (CR = 30 mmol L 1, CSi = 0.03 mmol L 1,
CP = 0.03 mmol L 1, pH = 2).
Fig. 3. The RSD dependence on the number of set calibration solutions.
Table 2
Concentrations of iron, aluminum, silicon and phosphorus in the calibration set
solutions (mg kg1) (matrix of concentrations C).
Solution 1 2 3 4 5 6 7 8 9 10
Al (III) 1 2 3 4 5 6 7 8 9 10
Fe (III) 20 18 16 14 12 10 8 6 4 2
Si (IV) 20 18 16 14 12 10 8 6 4 2
P (V) 2 4 6 8 10 12 14 16 18 20
C.S. Vakh et al. / Fuel 135 (2014) 198–204 201

8/10/2019 multicommutated stepwise injection analysis of biodiesel with classical least squares method
http://slidepdf.com/reader/full/multicommutated-stepwise-injection-analysis-of-biodiesel-with-classical-least 5/7
achieving the completeness of color-forming reactions. Then, the
colored solution was delivered from the MC (2) using the valve into
the spectrophotometric detector (5). The analytical signals were
measured in the stop-flow conditions at 882, 860 and 870 nm for
phosphate, silicate ions and water, respectively. It was found that
the optimal concentrations of sulfuric and oxalic acids for determi-
nation of phosphate and silicate ions are 0.02 mol L 1 and
0.01 mol L 1, respectively. These concentrations provide the lowest
absorbance of background solution and the greatest absorbance of
phosphate and silicate ions solution.
3.4. Multivariate calibration
For data analysis of silicon, phosphorus, iron and aluminum
determination the multivariate calibration CLS method have been
used. The procedures of multivariate calibration applied for deter-
mination of all analytes were similar. The 10 calibration set solu-
tions with different ratios of the concentrations of Al (III) and Fe
(III) or phosphate and silicate ions were prepared. According to
[22] number of calibration set solutions must not be less than
the number of determined components. In order to find the opti-
mal number of calibration set solutions, RSD dependence on the
number of set calibration solutions (Fig. 3) has been studied. The
optimum number of calibration set solutions is 10.
To design a matrixes of concentrations (C-matrix) the concen-
trations of analytes are include into Table 2. The calibration set
solutions were analyzed according to MCSWIA-CLS procedureincluding the stage of digestion. For determination of Al (III) and
Fe (III) following wavelengths were chosen as working: 510, 520,
530, 540, 550, 560 and 570 nm. For phosphate and silicate determi-
nation the chosen working wavelengths were 820, 830, 840, 850,
860, 870, 880, 890 900 and 910 nm.
The working wavelengths were chosen by the following crite-
ria: the number of wavelengths must be more than the amount
of analytes; the value of the absorbance at the chosen wavelength
should not be less than ½ the absorbance at the maximum of the
absorption spectra; with each selected wavelength linear depen-
dence of the absorption on the concentration of the analytes, must
be observed [22].
Absorbance, which were measured at the selected wavelengths
were placed into Table 3 with purpose to receive a matrixes A. The
experimental calibration matrixes (P -matrixes) (Table 4) were cal-
culated according to the formula P = CAT ( AAT )1. These matrixes
were used for the analysis of model and real samples. Computa-
tions of the P -matrixes were performed by Microsoft Excel.
The P -matrixes were checked by MCSWIA determination of Al(III), Fe (III), phosphate and silicate ions in test samples. The absor-
bance of complexes of Al (III) and Fe (III) with chromazurol S and
molybdenum blue complexes in the test samples were placed in
table A*, and the concentrations of aluminum (III), iron (III), phos-
phate and silicate ions were found by the formula C = PA*. The
results are presented in Table 5.
3.5. Interference study
The effect of potentially interfering ions on the determination of
silicon, phosphorus, iron and aluminum in biodiesel was investi-
gated. It was performed by addition of known concentration of
each ion to fixed concentrations of analytes. The tolerable concen-
tration of each taken foreign species is considered to be less than5% of relative error in the signal.
The influence of some inorganic compounds (Mn2+, Pb2+, Cd2+,
Zn2+, Co2+, Ca2+, Mg2+, Ni2+ and Cu2+ and organic compounds
(methanol, acetone, ethanol and glycerol) were studied. It was
investigated that interferences were not found for Cu2+ at 20-fold
Table 3
Absorbance matrixes A.
k (nm) Solution
1 2 3 4 5 6 7 8 9 10
Absorbance of calibration solutions for aluminum and iron determination
510 0.706 0.697 0.678 0.697 0.712 0.577 0.656 0.646 0.593 0.546
520 0.78 0.779 0.771 0.802 0.824 0.687 0.788 0.78 0.749 0.704
530 0.833 0.842 0.85 0.898 0.936 0.8 0.938 0.94 0.946 0.919
540 0.849 0.879 0.915 0.984 1.043 0.927 1.096 1.108 1.169 1.162
550 0.86 0.892 0.931 1.003 1.06 0.938 1.106 1.106 1.166 1.138
560 0.835 0.848 0.845 0.885 0.879 0.721 0.798 0.731 0.725 0.599
570 0.742 0.729 0.698 0.718 0.689 0.554 0.605 0.544 0.534 0.44
Absorbance of calibration solutions for silicon and phosphorus determination
820 0.388 0.440 0.532 0.284 0.320 0.488 0.484 0.422 0.440 0.409
830 0.432 0.492 0.588 0.320 0.356 0.532 0.524 0.465 0.472 0.453
840 0.464 0.532 0.636 0.352 0.388 0.568 0.552 0.499 0.500 0.490
850 0.488 0.564 0.672 0.372 0.416 0.588 0.576 0.524 0.516 0.517
860 0.496 0.576 0.688 0.384 0.424 0.596 0.592 0.535 0.520 0.527
870 0.496 0.576 0.684 0.384 0.424 0.592 0.596 0.534 0.516 0.526
880 0.484 0.560 0.668 0.376 0.412 0.580 0.592 0.522 0.504 0.513
890 0.464 0.536 0.644 0.360 0.396 0.564 0.576 0.504 0.488 0.494
900 0.440 0.508 0.608 0.340 0.372 0.548 0.556 0.480 0.464 0.469
910 0.412 0.476 0.572 0.320 0.348 0.528 0.532 0.454 0.444 0.443
Table 4
Experimental calibration matrixes (P -matrixes).
Experimental calibration matrix (P-matrix) for aluminum and iron determination
155.91 313.45 199.13 86.27 14.47 1.42 35.22
7.53 291.82 425.29 96.58 74.96 107.59 114.11
Experimental calibration matrix (P-matrix) for silicon and phosphorus determination
106.00 1088.00 1213.00 498.00 3336.00 3104.00 1304.00 2288.00 3647.00 2231.14
1323.00 1385.00 31.00 886.00 994.00 1364.00 1328.00 308.00 53.00 270.47
202 C.S. Vakh et al./ Fuel 135 (2014) 198–204

8/10/2019 multicommutated stepwise injection analysis of biodiesel with classical least squares method
http://slidepdf.com/reader/full/multicommutated-stepwise-injection-analysis-of-biodiesel-with-classical-least 6/7
(at high copper content it is necessary to use thiourea), Mn2+, Pb2+,
Cd2+, Zn2+, Co 2+, Ni2+ at 100-fold excess and for Ca2+, Mg2+ at 2000-
fold excess for determination of aluminum (III) iron (III). In case of
silicon and phosphorus determination the Mn2+, Pb2+, Cd2+, Zn2+,
Co2+, Ni2+, Ca2+, Mg2+ at 1000-fold excess have no interference to
the reaction.
Methanol, acetone, ethanol and glycerol do not interfere, as
their interfering effect is eliminated during sample preparation.
3.6. Analytical performance
The limits of detection (LOD) obtained using the MCSWIA-CLS
system were 0.3 mg kg1 for aluminum and 0.6 mg kg1 for iron,
silicon and phosphorus (Table 6). The experimental calibration
matrixes for CLS were designed with 10 calibration set solutions
of 1–10 mg kg1 for aluminum and 2–20 mg kg1 for iron, silicon
and phosphorus. The weight of sample was 1 g.
3.7. Application
The developed automated procedure was applied for determi-
nation of silicon, phosphorus, iron and aluminum in biodiesel sam-
ples (castor bean, sunflower, canola and corn). Samples were
digested by microwave and then analyzed by MCSWIA-CLS. The
correctness of the proposed method was carried out by the com-
parison with the reference methods. Analytical results measured
by MCSWIA and reference methods for each analyte are presented
in Table 7. The recoveries obtained for each of the samples was
within of 98–107% range which is considered acceptable for this
type of samples. The RSD does not exceed 10% for each element.
Analytical results measured for the samples by MCSWIA and refer-
ence methods were compared using the F -test. F -values 68.2 are
obtained indicating insignificant difference in precision between
both methods at the 95% confidence level (F -critical = 8.8).
Table 5
Added and found concentrations of Fe, Al, Si, and P in model samples (n = 5, P = 0.95).
Analyte Added, mg kg1 Found, mg kg1 RSD (%)
Fe 5 5.1 ± 0.1 2
6 6.1 ± 0.2 2
2 2.1 ± 0.1 5
3 2.9 ± 0.1 3
1 1.0 ± 0.1 1
Al 1 1.1 ± 0.1 10
3 2.9 ± 0.2 4
8 8.1 ± 0.2 1
4 4.1 ± 0.1 2
7 7.2 ± 0.3 3
Si 4 4.1 ± 0.1 2
7 7.0 ± 0.1 1
1 0.9 ± 0.1 10
5 4.8 ± 0.2 4
9 9.2 ± 0.2 2
P 3 3.1 ± 0.1 3
9 9.1 ± 0.1 1
2 2.0 ± 0.2 1
8 8.2 ± 0.1 2
4 4.1 ± 0.1 2
Table 6
Analytical performance data.
Parameter Fe Al S i P
pH 5 5 2 2
Sample weight (g) 1
Time of MCSWIA analysis (min) 4
Linear range (mg kg1) 2–20 1–10 2–20 2–20
Detection limit (mg kg1) 0.6 0.3 0.6 0.6
Quantification limit (mg kg1) 2 1 2 2
RSD (%) (n = 10) 5 10 10 3
S ample freque nc y ( pe rhour) 6
Table 7
The results of the determination of iron, aluminum, silicon and phosphorus in biodiesel samples (n = 10, P = 0.95).
Sample Analyte (mg kg1) Ad de d (mg kg1) Found by MCSWIA-CLS (mg kg1) Found by reference method (mg kg1) Recovery (%) F -value
Sunflower Al 2.0 4.2 ± 0.4 4.1 ± 0.3 105 1.9
3.0 5.1 ± 0.5 5.3 ± 0.3 102 2.5
Fe 2.0 3.2 ± 0.2 3.1 ± 0.2 107 1.1
3.0 3.9 ± 0.3 4.1 ± 0.1 98 8.1
Si 2.0 3.2 ± 0.2 3.1 ± 0.2 106 1.1
3.0 4.1 ± 0.3 4.3 ± 0.1 103 8.2
P 2.0 10.2 ± 0.5 10.1 ± 0.2 102 6.4
3.0 11.0 ± 0.3 11.3 ± 0.4 100 2.0
Canola Al 2.0 5.2 ± 0.5 5.1 ± 0.5 104 1.1
3.0 5.9 ± 0.6 6.1 ± 0.4 98 2.1
Fe 2.0 3.1 ± 0.2 3.1 ± 0.2 103 1.0
3.0 4.0 ± 0.2 4.1 ± 0.1 100 3.8
Si 2.0 4.2 ± 0.2 4.1 ± 0.2 106 1.1
3.0 5.1 ± 0.3 5.2 ± 0.2 102 2.2
P 2.0 12.3 ± 0.5 12.1 ± 0.2 103 6.53.0 12.9 ± 0.3 13.0 ± 0.3 99 1.1
Corn Al 2.0 4.0 ± 0.4 4.1 ± 0.5 100 1.6
3.0 5.2 ± 0.5 5.1 ± 0.6 104 1.4
Fe 2.0 3.2 ± 0.2 3.1 ± 0.2 106 1.1
3.0 3.9 ± 0.3 4.1 ± 0.1 98 8.1
Si 2.0 3.0 ± 0.2 3.1 ± 0.2 100 1.0
3.0 4.1 ± 0.3 4.3 ± 0.1 103 8.2
P 2.0 13.2 ± 0.5 13.1 ± 0.2 102 6.3
3.0 14.0 ± 0.3 14.3 ± 0.1 100 2.2
Castor bean Al 2.0 5.0 ± 0.5 5.1 ± 0.5 100 1.0
3.0 6.1 ± 0.5 6.2 ± 0.5 102 1.0
Fe 2.0 5.2 ± 0.2 5.1 ± 0.2 106 1.0
3.0 6.0 ± 0.3 6.1 ± 0.2 100 2.2
Si 2.0 4.0 ± 0.2 4.1 ± 0.2 100 1.1
3.0 5.2 ± 0.3 5.3 ± 0.2 104 2.2
P 2.0 13.1 ± 0.5 13.1 ± 0.2 101 6.3
3.0 13.9 ± 0.3 14.3 ± 0.2 99 2.2
C.S. Vakh et al. / Fuel 135 (2014) 198–204 203

8/10/2019 multicommutated stepwise injection analysis of biodiesel with classical least squares method
http://slidepdf.com/reader/full/multicommutated-stepwise-injection-analysis-of-biodiesel-with-classical-least 7/7
4. Conclusions
The MCSWIA-CLS provides the new possibility for multicompo-
nent analysis. The MCSWIA-CLS allows to achieve the completeness
of color-forming reaction for multivariate spectrophotometric
determination of several analytes. The CLS method was chosen for
multivariate calibration because of its simple mathematical tools
(EXCEL program is enough) and less time spent for calibration.Unlike FIA and SIA methods, MCSWIA-CLS simplifies greatly the cal-
ibration procedure, because it is not necessary to control the move-
ment of the sample in the carrier stream. Moreover, because of the
absence of dispersion of colored complex in the carrier stream
while its delivering to detector it is possible to receive the high sen-
sitivity of analysis.The MCSWIA-CLS determination of silicon, phos-
phorus, iron and aluminum in biodiesel is simple, fast and flexible.
It uses small quantities of samples and reagents, therefore generat-
ing less waste and especially can be provided at a lower cost.
Acknowledgements
This work was supported by the Russian Foundation for Basic
Research (Project No. 13-03-00031-a). Scientific researches wereperformed at the Center for chemical analysis and materials
research of St. Petersburg State University.
References
[1] Fangrui M, Milford AH. Biodiesel production: a review. Bioresource Technol1999;70:1–15.
[2] Sekhar MCh, Mamilla VR, Reddy KV, Rao GN. Production of biodiesel fromneem oil. Int J Eng Sci Technol 2009;1:295–302.
[3] Monteiro MR, Ambrozin AP, Liao LM, Ferreira AG. Critical review on analyticalmethods for biodiesel characterization. Talanta 2008;77:593–605.
[4] Quadros PD, Rau M, Idrees M, Chaves ES, Curtius AJ, Borges DG. A simple andfast procedure for the determination of Al, Cu, Fe and Mn in biodiesel usinghigh-resolution continuum source electrothermal atomic absorptionspectrometry. Spectrochim Acta Part B 2011;66:373–7.
[5] Campos RC, Correia CT, Vieira F, Saint-Pierre TD, Oliveira AC, Goncalves R.
Direct determination of P in biodiesel by high-resolution continuum sourcegraphite furnace atomic absorption spectrometry. Spectrochim Acta Part B2011;66:352–5.
[6] Ghisi M, Chaves ES, Quadros DC, Marques EP, Curtius AJ, Marques AB. Simplemethod for the determination of Cu and Fe by electrothermal atomic
absorption spectrometry in biodiesel treated with tetramethylammoniumhydroxide. Microchem J 2011;98:62–5.
[7] Amais RS, Donati GL, Schiavo D, Nobrega JA. A simple dilute-and-shootprocedure for Si determination in diesel and biodiesel by microwave-inducedplasma optical emission spectrometry. Microchem J 2013;106:318–22.
[8] Korn MA, Santos DS, Welz B, Vale MR, Teixeira AP, Lima DC, et al. Atomicspectrometric methods for the determination of metals and metalloids inautomotive fuels – a review. Talanta 2007;73:1–11.
[9] Almeida ES, Monteiro MA, Montes RH, Mosquetta R, Coelho NM, Richter EM,et al. Direct determination of copper in biodiesel using stripping analysis.
Electroanalysis 2010;22:1846–50.[10] Martiniano LC, Abrantes VR, Neto SY, Marques EP, Fonseca TC, Paim LL, et al.
Direct simultaneous determination of Pb(II) and Cu(II) in biodiesel by anodicstripping voltammetry at a mercury-film electrode using microemulsions. Fuel2013;103:1164–7.
[11] Lira LB, Santos DB, Guida MB, Stragevitch L, Korn GA, Pimentel MF, et al.Determination of phosphorus in biodiesel using FIA with spectrophotometricdetection. Fuel 2011;90:3254–8.
[12] Río V, Larrechi MS, Callao MP. Sequential injection titration method usingsecond-order signals: determination of acidity in plant oils and biodieselsamples. Talanta 2010;81:1572–7.
[13] Río V, Larrechi MS, Callao MP. Determination of sulphate in water andbiodiesel samples by a sequential injection analysis – multivariate curveresolution method. Anal Chim Acta 2010;676:28–33.
[14] Lima MB, Barreto IS, Andrade SE, Neta MS, Almeida LF, Araujo MU. Photometricdetermination of phosphorus in mineralized biodiesel using a micro-flow-batch analyzer with solenoid micro-pumps. Talanta 2012;98:118–22.
[15] Diniz PD, Almeida LF, Harding DP, Araujo MU. Flow-batch analysis. TrendsAnal Chem 2012;35:39–49.
[16] Fulmes ChS, Bulatov AV, Yasakova OG, Freze EA, Moskvin AN, Dedkov YM, et al.Multicommutated stepwise injection analysis as new approach forsimultaneous determination of nickel (II), copper (II) and zinc (II) in wetaerosols. Microchem J 2013;110:649–55.
[17] Saurina J, Hernandez-Cassou S. Quantitative determinations in conventionalflow injection analysis based on different chemometric calibration strategies:a review. Anal Chim Acta 2001;438:335–52.
[18] Whitman DA, Seasholtz MB, Christian GD, Ruzicka J, Kowalski BR. Double-injection flow-injection analysis using multivariate calibration formulticomponent analysis. Anal Chim Acta 1991;63:775–81.
[19] Pasamontes A, Callao MP. Optimization by means of responses surface of ananalytical sequence using a sequential injection system. Talanta2006;68:1617–22.
[20] Uddin MR, Ferdous K, Uddin MR, Khan MR, Islam MA. Preparation andoptimization of biodiesel production from mixed feedstock oil. Chem Eng Sci2013;1:22–6.
[21] Dong H, Jiang M, Zhao G, Wang M. Simultaneous spectrophotometricdetermination of Aluminum(III), Iron(III) and Beryllium(III) in rainwater by a
matrix method. Anal Sci 1991;7:69–75.[22] Wang Z, Li J, Shen H. Criteria for selecting analytical wavelengths for
multicomponent mixtures by the CPA matrix method and simultaneousspectrophotometric determination of niobium and tantalum. Anal Chim Acta1988;212:145–53.
204 C.S. Vakh et al./ Fuel 135 (2014) 198–204