MSC Biology Mechanism and Clinical Design...were transplanted under the skin of nude mice. (D) At 21...
Transcript of MSC Biology Mechanism and Clinical Design...were transplanted under the skin of nude mice. (D) At 21...

MSC Biology Mechanism and Clinical Design
Robert Deans, Athersys
ISCT NA Regional Meeting Philadelphia
9.8.13
The statements and discussions contained in this presentation that are not historical facts constitute forward-looking statements, which can beidentified by the use of forward looking words such as “believes,” “expects,” “may,” “intends,” “anticipates,” “plans,” “estimates” and analogousor similar expressions intended to identify forward-looking statements. These forward-looking statements and estimates as to futureperformance, estimates as to future valuations and other statements contained herein regarding matters that are not historical facts, are onlypredictions, and that actual events or results may differ materially. We cannot assure or guarantee you that any future results described in thispresentation will be achieved, and actual results could vary materially from those reflected in such forward-looking statements.
Information contained in this presentation has been compiled from sources believed to be credible and reliable. However, we cannot guaranteesuch credibility and reliability. The forecasts and projections of events contained herein are based upon subjective valuations, analyses andpersonal opinions. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy any securities. Such an offer orsolicitation, if made, will only be made pursuant to an offering memorandum and definitive subscription documents

Clinical Development Pipeline
2013
MultiStem®Immunological
IBD / Ulcerative ColitisHematopoietic Stem Cell Transplant / GVHD
Solid Organ TransplantDiabetes
CardiovascularAMI
Congestive Heart FailurePeripheral Artery Disease
NeurologicalStroke
Traumatic Brain InjuryMultiple SclerosisSpinal Cord Injury
OtherBone Allograft
5HT2c Agonists (obesity, other)
IMMUNOLOGICALCARDIOVASCULAR
NEUROLOGICAL
2013-4 data
2013 Data
FDA approved P2
FDA discussions re: P2/3 design
German P1 approval
2

Phases of Commercialization (Circa NOW)
Minimally manipulated products regulated under tissue transplant guidelines and bypass
pre-market clinical testing
In some Regulatory environments, market entry of autologous products generates
revenue stream and enables development of allogeneic therapy

Clinical Proof of Concept

Running on All Cylinders
• Translational community addresses need for (industry) executing well designed studies and securing broad open access data registries
• Promoting an educated patient base with access to treatment without having to go underground
• Bioprocessing industry realizes business opportunity and invests in technology to bring manufacturing costs down
• Pharma and Healthcare see validated business model and invest with capital and development experience
• Regulatory policy accelerates access and responds to global alternatives
• Universities develop training programs tailored to cell therapy and bring next generation scientists into medicine


What Impact Can We Have As A Community?
• Better understand and control PK/PD profiles for cell therapy
• Collectively solve downstream cell processing technology improvements
• Educate Regulatory agencies around potency assays and cell comparability assays
• Influence reimbursement decisions and bring product COGS under realistic healthcare ceilings

Enhancing Healing Through Multiple Mechanisms
IMMUNOMODULATION
INFLAMMATION
REDUCTION
NEUROPROTECTION, CYTOPROTECTION
ANGIO- / VASCULOGENESIS
CELLULAR REGENERATION / REPLACEMENT BY HOST
PROGENITORS
Certain Secreted Proteins
Cytokines / Chemokines
Growth Factors
Other Proteins
SHIFTING BALANCE IN REPAIR PROCESSES
MultiStem® cells express multiple therapeutic proteins to enhance healing and tissue
repair in multiple ways
8Multimodal mode of action compared to single mode of biologic/drug

Pharmacokinetics
Infused cells reside in reticuloendothelial system with a
minority trafficking to site of injury

MultiStem Homing in Mouse GvHD Model
MultiStem Intravenous Infusion in Mouse GvHD Model
6 hours post-i.v. 24 hours post-i.v.
10

Limited MultiStem Retention Over Time
Day 1 Day 2 Day 3 Day 4 Day 5 Day 6 Day 7
Model:• LAD ligation and brachiocephalic vein
injection of luciferase labeled rat MultiStem® (5x105)
• Bioluminesent Imaging over one week
11Confidential

Whole Body Imaging Provides Improved Mass Balance Accounting

Whole Body Imaging

Quantifying Biodistribution

3-D Rendering of Cell Distribution in Liver, Femur
MAPC found in marginal zone of white pulp in spleen

Cell Migration to Spinal Cord Injury

Cardiovascular Disease

5 sec 30 sec 60 sec Vessel patency
Delivery of MultiStem® in AMI patient
Rapid, efficient delivery of cells to ischemic area
Retention of cells in relevant region of heart
Well tolerated
MultiStem®: Acute Myocardial Infarction
Blockage causingischemia
Angioplasty toclear blockage
Delivery of MultiStem

Biodistribution Following Transarterial Delivery in Pig Ischemia Model
19Medicetty et al, Cell Transplantation in press
Injection sites
Transarterial catheter delivery of pig MultiStem® cells, 2 weeks, animal 132

MultiStem Increases Ejection Fraction and Reduces Scar Size in Pig AMI Model
Source: Wang et al, Circulation 2009
20

MultiStem®: Acute Myocardial Infarction
Phase 1 Clinical Study completed- open label, dose escalation
DesignAdministered through coronary artery 2-5 days after AMI
Multiple clinical sites (Cleveland Clinic, Henry Ford and others)
Evaluation of safety
ResultsClean safety profile over 4 month period following treatment
Heart function benefit also observed: ~25% relative improvement in LVEF relative to baseline,
improvements in wall motion, and LV end systolic volume
0.00
4.00
8.00
12.00
16.00
20.00
Reg 20M 50M 100M
D E
F%
21Mean ± SEMPatients with Baseline LVEF of >30 – <45 at time of MultiStem administration

Context Setting – Cell Therapy for AMILVEF Comparison at Early Time Point (3-6 Months)
2.51.3 1.1 1.9
4.1
8.7
0.43
12.6
10
0
2
4
6
8
10
12
14
REPAIR-AMI
∆ 4m
ASTAMI∆ 6m
Janssens∆ 4m
Osiris∆ 3m
ATHX-20MM
∆ 4m
ATHX-50MM
∆ 4m
ATHX-100MM
∆ 4m
ATHX-45-20MM
ATHX-45-50MM
ATHX-45-100MM
Difference between Treated and (Control ) Patients
Change in mean
LVEF from baseline
compared to
(control)
*
PhasePatientsControl
II204
Placebo
II97
SOC
II67
Placebo
I53
Placebo
I25
Registry
Baseline LVEF ≤ 45 %
22
Note: ATHX data presented here not statistically significant

MultiStem v. HSC v. MSCPhase I / II Cardiovascular Cell Therapy Studies
23
AthersysMultiStem
Amorcyte (Neostem)HSC/CD34+
OsirisMSC
MesoblastMPC
Indication AMIAllogeneic
AMIAutologous
AMIAllogeneic
CHFAllogeneic
Delivery Transarterial Intracoronary Intravenous Endocardial
Study parameters
25 patients3 dose groups (20, 50, 100 MM cells)
Registry control
31 patients3 dose groups (5, 10,
15 MM cells)Placebo control
53 patients3 dose groups (0.5,
1.6, 5.0 MPK)Placebo control
60 patients3 dose groups (25, 75,
150 MM cells)Placebo control
∆ LVEFBL→Xmo
treatment v. control
4 months5.7% (pooled)
9.0% (LVEFBL ≤ 45%)12.6% (LVEFBL ≤ 45%,
50MM dose)
3 months2.5% (pooled)
3 months1.9% (pooled)
3 months3.4% (pooled)
Otheroutcomes
Improve stroke volume, wall motion,
ESV
Improve perfusion (in higher dose group)
Improve FEV1 Improve clinical outcomes, perfusion
Confidential

Angiogenesis Potency Assay
• Pre-clinical models show neo-angiogenesis in parallel with recovery benefit
• Screening of conditioned media shows expression of angiogenic factors, increased when exposed to inflammatory environment
• Confirmed by in vivo tissue microarrays
VEGF, CXCL5, IL-8

Neo-Angiogenesis Correlates with Response to Ischemia
Wang et al, Circulation 2009
AMIPVD
CHF
PBS
MultiStem
0
200
400
600
800
1000
1200
2 million 5 million 10 million PBS
vess
els
/mm
2
Vessel Density

Minimum Levels of VEGF, CXCL5 and IL-8 Required for Induction of Angiogenesis
IL-8VEGF
0 pg/ml
250 pg/ml
1000 pg/ml
2000 pg/ml
5 pg/ml
10 pg/ml
0 pg/ml
25pg/ml
0 pg/ml
20 pg/ml
60 pg/ml
80 pg/ml
CXCL5
0
10
20
30
40
50
60
Ave
# o
f Tu
be
s Fo
rme
d P
er
Fie
ld
0
10
20
30
40
50
60
70
EBM EGM SFM SFCM ISO 0pg/ml
IL8
20pg/ml
IL8
60pg/ml
IL8
80pg/ml
IL8
100pg/ml
IL8
Ave
# o
f Tu
be
s Fo
rme
d P
er
Fie
ld
Pass-Fail Criteria: Knockdown and Add-back Sets Critical Threshold Required for Activity

Consistency of Angiogenic Factor Production
• Tested culture supernatants using in vivo matrigelangiogenesis assay
• Determined levels of secreted angiogenic factor in spent media at time of clinical harvest
Statistical pass fail criteria

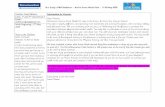
MultiStem (MAPC) promotes More Stable Blood Vessels than MSC and mesoangioblasts (Mab) in vivo
Roobrouck et al, Stem Cells 2011
Undifferentiated MSC, mesoangioblasts (Mab) and MAPC mixed with matrigel and VEGF165/bFGFwere transplanted under the skin of nude mice. (D) At 21 days, matrigel plugs were removed and examined macro- and microscopically (1nd and 2nd row) (2.5x and 6.6x respectively)
Human MSC and Mab-containing Matrigelsharbored leaky vessels, indicated with white arrows, which could be also seen on H&E stained cross-sections (last row) (40x)
28

Peripheral Vascular Disease (PVD)• Chronic occlusion of the lower extremity arteries
• Loss of blood flow results in pain, ischemic lesions, sepsis and can lead to limb amputation
• Can range from mild ( intermittent claudication) to severe ( critical limb ischemia: 5-10% of patients)
• Globally, up to 12% of global population is affected by PVD. Direct and indirect cost of treating PVD estimated > $2 billion.
• Risk factors: age, hypercholesterolemia, diabetes, smoking, obesity
• Formation of collaterals around site of stenosis can offer protection against ischemia and help restore blood flow
• Unmet need: current therapies have severe side effects, counterindicated for heart failure, many patients are unfit candidates for revascularization
29

Proximal Hindlimb Adductor Muscle Perfusion andFlow Reserve After MultiStem (MAPC) Injection
Day
BL 1 3 21
Blo
od
Flo
w (
mL
/min
/g)
0.0
0.2
0.4
0.6
0.8
1.0
Control(PBS)
Mic
rovascu
lar
Flo
w R
eserv
e
0.0
0.5
1.0
1.5
2.0
2.5
3.0
3.5
MAPC
*
Perfusion Flow Reserve(day 21)
ControlPBSMAPC
Inject
*
*
* p<0.05 vs control and PBS
30

MultiStem treatment to increase transverse arterioles
31
Control day 21 Control day 21
MAPC day 21 MAPC day 21

Examination of Arteriolar networks and distribution
32
Day 1 DAY 21
Untreated Controls
MultiStem

Cell Therapy as Disruptive Technology in Inflammation and Immunity

Dose-Dependent Modulation in T-Cell Assay
34
Stimulation
Classic MLR – reflects indirect antigen presentation by macrophages and naïve response
CD3/CD28 stimulation – reflects TCR signalling (direct Ag)
Flu Ag in MLR – reflects memory cell stimulation
Cytokine (IL-2, 7, 15) stimulation –reflects homeostatic T cell expansion
Readout
Inhibition of T cell proliferation
Prevention of T cell activation (FACS)
Inhibition of cytokine secretion

MultiStem’s Dynamic Regulation of Activated T-Cells
Activated T Cells
13 chemokines
5 cytokines
>25 secreted factors
>10 secreted factors
>40 receptors
3 receptors
>40 intracellular factors
>10 intracellular factors
3 chemokines
3 cytokines
>10 receptors
>20 intracellular proteins
3 intracellular proteins
> 5-fold difference in expression
MultiStem
35

MultiStem Interferes with Trafficking of Activated Immune Cells (Leukocyte Extravasation)
CD15s
36
MultiStem associated with limiting cell surface expression of CD15s on inflammatory cells and e-selectin and adhesion receptors on endothelial cells,
and inhibits leukocyte extravasation in multiple ways
Leukocyte extravasation:• Contributes to inflammation
and tissue damage (e.g., in regions of ischemia)
• Occurs mainly in post-capillary venules (minimized hemodynamic shear forces)
• Includes several steps, including chemo-attraction, rolling adhesion, tight adhesion, (endothelial) transmigration
• Process halted whenever any of these steps is suppressed

Inhibition of Inflammatory Cell Extravasation in AMI, Stroke
MultiStem
0
5
10
15
20
25
30
35
40
45
50
PBS (n=5) MAPC (n=4)
neu
tro
ph
ils/h
igh
po
wer f
ield
Neutrophil count in infarcted hearts Reduction in neutrophil tissue elastase
Rat Ischemia Model - direct injection in infarct zone; sacrifice after 3 days
p=.005 PBS Multistem
37
Tissue Microarray Evaluation in Ischemic Stroke Model Confirms Role
in Leukocyte Extravasation

Ki67
CD
4
No
MAPC
1:4No M
APC
1:4
0
1
2
3
4
% K
I67
+V
ENo M
APC
1:4
0
1
2
3
4
% K
I67
+V
E
No M
APC
1:4
0
2
4
6
8
% K
I67
+V
E
No M
APC
1:4
0
2
4
6
8
% K
I67
+V
E
No M
APC
1:4
0
5
10
15
20
25
% K
I67
+V
E
No M
APC
1:4
0
5
10
15
20
25
% K
I67
+V
E
* * *
IL-2 IL-7 IL-15
IL-2 IL-7 IL-15
Teff
Treg
0
20
40
60
80
100
% S
up
pre
ssio
n
IL-2
Teff
Treg
0
20
40
60
80
100
% S
up
pre
ssio
n
IL-7
Teff
Treg
50
60
70
80
90
100
% S
up
pre
ssio
n
IL-15
IL-2 IL-7 IL-15
*NS
*
FoxP
3
CD25
Treg
Teff
MAPCs facilitate expansion of Tregs during homeostatic proliferation

Solid Organ Transplant
Liver, Lung

AMS50
Graft >d50 after spleen cell application from
„tolerant“ animal
Heterotopic Heart Transplant
Allogeneic heart is grafted to adominalartery and venous
circulation – keeps on beating

MAPCs
(2x)Myco-
phenolate
(d0-7)
Lewis rat
ACI rat
Heterotopic Cardiac Allotransplant Model
0 20 40 60 80 1000
20
40
60
80
100
rejection day
Gra
ft S
urv
ival
(%
)Donor-type MAPC
+ MPA
3rd party MAPC+ MPA
MPA alone
41Confidential

LEW to ACI model (secondary skin Tx)
d0 d7 d9 d13 d15 d20
Lew ACI
Result from nine long term survivors: no acceptance of LEW skin.
representative picture
Heart Accepted / Skin Rejected – No Systemic Tolerance
Remarks
Even when skin graft is rejected in allogeneic response, heart remains unaffected!
Heterotopic heart can be transplanted to naïve allogeneic recipient - and is accepted without drug!
42Confidential

Secondary Tx:
Heart from LT acceptance animals
into ACI animals
NO IMMUNO-SUPPRESSION
0 20 40 60 80 1000
20
40
60
80
100
rejection day
Gra
ft S
urv
ival
(%
)
2 x 5 Mio LEWISMAPC + MPA is
Control
CsA therapy
0 20 40 60 80 1000
20
40
60
80
100
rejection day
Gra
ft S
urv
ival
(%
)
ControlHeart from LEWISMAPC therapyHeart from CsA therapy
100%
40%
CsA Treated
MAPC Treated
foxP3-APC
CD
25-P
E
CD4CD25highfoxP3
0
2
4
6
8ns.
*
Treg
s (%
of
lym
ph
ocy
tes)
ctr CsA MAPC
primary hearts
Primary Tx:
Lewis heart into ACI
animals + CsA or MAPC
MultiStem Provides Distinctive Benefit in Secondary Transplant
43Confidential

MiSOT-I Study Protocol Review – Dose Escalation

Summary
• Understanding mechanistic pathways and building PK/PD profile in disease models is key to clinical proof of mechanism
• Societies increasingly engaged in standards setting for cell characterization and potency– ISCT, ARM, CIRM, MSC Connect
• Nomenclature, reference standards, characterization assays– FDA, FACT, USP, DIA, Toxicology Society
• ISCT can be a core strength in advancing adherent cell therapies– Commercialization Committee / MSC Scientific Committee– Commercialization Committee – institutional membership
• WFIRM, UC Davis, Univ Loughborough, Karolinska, Catapult, UnivPenn

Immunology, GVHD – Richard Maziarz, Oregon Health Sciences University
AMI – Marc Penn, Summa Healthcare
Critical Limb Ischemia – Johnathan Lindner, Oregon Health Sciences University
Solid Organ Transplant – Marc Dahlke, Univ Regensburg DE
thanks again