Mh Year 5 linical Skills Session Arterial lood Gas Sampling€¦ · Arterial blood gases (AGs) are...
Transcript of Mh Year 5 linical Skills Session Arterial lood Gas Sampling€¦ · Arterial blood gases (AGs) are...

MBChB Year 5
Clinical Skills Session
Arterial Blood Gas Sampling
Authors:
The Clinical Skills Lecturer Team
Reviewed & ratified by:
2019

Arterial Blood Gas (ABG) Sampling
Aim: For the student to be able to take an Arterial Blood Gas Sample from the radial artery
Objective: For the student to know what an ABG test is and to have a basic understanding of the chemistry
Objective: For the student to be able to recognise the indications and contraindications for ABG sampling
Objective: For the student to be able to recognise, interpret and report abnormal results
Objective: For the student to understand how to perform the radial artery puncture safely using ANTT
principles.
What is Arterial Blood Gas analysis?
This is a procedure of taking a sample of arterial blood for analysis.
This is a direct vascular puncture usually performed on the radial artery, although you may see the brachial or
femoral artery used in practice.
The information in this study guide and in the teaching presentation relate to radial artery puncture only.
ABG sampling is a blood test that looks at the amount of certain gases dissolved in arterial blood, the test can
then examine the pH of the patient’s blood along with the amount of oxygen, carbon dioxide, bicarbonate.
Many areas have the ability to examine the blood further, testing for concentrations of lactate, electrolytes
and or haemoglobin.
Arterial blood gases (ABGs) are an important routine investigation to monitor the acid-base balance of
patients, and they may help make a diagnosis. They can also indicate the severity of a condition and help to
assess treatment.
What does the ABG test?
Listed below are the main components tested in an ABG;
pH > the acidity/ alkalinity of the blood.

Pa02 > The partial pressure of oxygen in the arterial blood, measured in kPa or mmHg (kPa will be used in this
guide), allowing staff to check if the patient is being sufficiently oxygenated.
PaC02 > The partial pressure of carbon dioxide in arterial blood measured in kPa or mmHg, which can indicate
to staff the efficacy of ventilation.
HC03- > The bicarbonate ion measured in mEq/L. The HC03- is used when assessing the metabolic component
of acid-base disorders.
BE> Base excess measured in mmol/L. The base excess is used when assessing the metabolic component of
acid-base disorders. Contrasted with the bicarbonate levels, the base excess is a calculated value intended to
completely isolate the non-respiratory portion of the pH change
Other tests are often available on the results including;
Lactate
Magnesium
Haemoglobin (Hb)
Calcium
Potassium
Blood glucose
Please be aware that the above may not always be 100% accurate, so please check with the area/ Trust that
you are working in, to confirm accuracy of tests.
Biochemistry of ABG
Carbonic Acid Equation
This equation is the basis for the acid/ base regulation of the body;
CO2 + H2O ↔ H2CO3 ↔ H+ + HCO3-
Bicarbonate (HC03-) and Hydrogen (H+) are produced from the dissociation of carbonic acid (H2C03), which is a
result of a reaction from carbon dioxide (C02) and water (H2O).

Increased carbon dioxide would shift the balance on this equation to the right, therefore there would be more
Hydrogen ions increasing acidity and reducing the pH.
The Henderson-Hasselbalch equation
This equation is useful for approximating the pH of a buffer solution, and can relate the pH of blood to the
bicarbonate buffering system.
This equation tends to be used in more specialist areas, and if you want further information please see the
references or additional reading.
(Larson D)
HA is the molar concentration of the undissociated acid and A- is the molar concentration of this acid’s base
and pKa is -logKa where Ka is the acid dissociation constant.
Modifying the previous calculation; this can be used to relate the pH of blood to the concentration of
bicarbonate and the partial pressure of carbon dioxide from ABG’s.
where, 6.1 is the pKa (negative logarithm of the acid dissociation constant) for carbonic acid, and 0.0307 is the
factor relating the partial pressure of C02 to the amount of C02 dissolved in plasma. Bray et al (1999)
Kassirer-Bleich equation
[H+] = 24 × PCO2 / [HC03-]
The Kassirer-Bleich equation can be used to calculate the concentration of any part of the bicarbonate buffer
system as long as the concentrations of the other two components are known ie.; if the PaC02 and HC03- are
known, the pH can be determined.
All of these equations can be difficult to use at the bedside, but the equations represent a very important
relationship. They predict that the ratio of dissolved CO2 to HCO3- , determines H+ concentration and
consequently pH.
A drop or rise in PCO2 will result in a drop or rise in H+ respectively.

HCO3- on the other hand, is inversely related to H+ concentration, a drop in bicarbonate levels will result in an
increase in H+ concentration while a rise in bicarbonate levels result in a reduction in H+ concentration.
This buffer system is of physiological importance because both the respiratory and renal mechanisms for
regulating pH work by adjusting this ratio.
The PaCO2 can be modified by changes in alveolar ventilation, while plasma HCO3- can be altered by regulating
its generation and excretion by the kidneys, these can be used as considerations for practice.
Interpretation
Before you take an ABG, you should consider the patient, there are several reasons why an ABG may be
indicated. ABGs are taken often taken routinely for acutely unwell patients, patients with acute pancreatitis,
patients with diabetic ketoacidosis, patients evidencing signs of cyanosis, or respiratory problems.
It is possible that venous gases may be taken depending on the patient’s condition, please ensure that this is
taken into consideration before attempting to interpret the result.
Try to ensure that you have enough information about the patient, including any recent observations, full
renal profile, full blood count, blood glucose and any other relevant blood results. You will also need to know
the patients temperature and the amount of oxygen that they are inspiring prior to analysing the sample. All
of the above can be used to aid diagnosis in conjunction with the ABG result.
Understanding the individual components for interpretation
pH
Many reactions within the cells are controlled by enzymes that can only function between rigid parameters.
The pH is inversely proportional to the number of hydrogen ions (H+) in the blood. The more H+ present in the
blood the lower the pH and the more acidic the blood will be.
The body produces 1,450,000,000 H+ daily. If unrestricted these ions would cause a massive acidaemia. The
body prevents this through a series of buffers that mop up H+; proteins, haemoglobin, plasma proteins and
bicarbonate (within plasma).

Buffering is a transient process as the buffering agents will ultimately be consumed and acids will regenerate.
The lungs and kidneys are therefore required to eliminate acids and restore buffers.
Normal pH;
Partial pressure of Carbon Dioxide (PaC02)
PaCO2 is the partial pressure of
carbon dioxide in arterial blood.
When CO2 is dissolved in the
blood it is converted to carbonic
acid and releases a H+. Therefore,
the more CO2 the more H+.
Buffers
Bicarbonate (HC03-)
is often referred to as the
metabolic component, rising levels
of HC03- make the blood more
alkaline and a reduction of HC03-
makes the blood more acidic.
It is known as a buffer: if the pH rises the kidneys respond by excreting HC03- in the urine. Likewise if the pH
becomes acidotic the kidneys retain HC03-.

Base Excess (BE)
This is the amount of strong base
which would need to be added or
subtracted from a substance in
order to return the pH to normal
It indicates how much the body is
buffering to maintain neutral pH,
therefore, infers the amount of
alkali base that needs to be added to or taken away from blood to restore a neutral pH
Partial Pressure of Oxygen (Pa02)
This measures the amount of oxygen dissolved in the arterial blood.
There is a lot of information on this, please see Driscoll et al (2008). Ultimately in health the ideal Pa02 is
within the range of 12.0–14.6 kPa, (Driscoll 2008) but this figure decreases with age, chronic lung disease,
altitude, position of patient, (lower if patient supine), and site that ABG taken (a capillary arterial sample from
an earlobe can be up to 1kPa lower than a radial ABG).
Also anaemia, pH, temperature and raised C02 levels can affect the Pa02, for more information please see
references and look at the Oxygen Dissociation curve and the Bohr effect, (Driscoll et al, 2008)
All the factors above should be taken into consideration. If a patient has a low Pa02, supplementary oxygen
may be required for an acutely unwell patient. If the patient is on supplementary oxygen and the Pa02 is still
low, further intervention is needed to deal with the hypoxia, and senior help should be sort.
Determining the cause of abnormal ABG’s
There is a need to calculate if the abnormality is respiratory or metabolic in origin.
Then it should be determined whether the body has tried to compensate for the abnormality and how well it
has compensated.
Compensation
This is the body’s defence system, it will automatically try to correct an abnormal pH.

If a patient has a metabolic acidosis, (such as Diabetic keto acidosis (DKA) the body will compensate to try to
decrease the acidosis, therefore the patient will have an increased respiratory rate as they try to breathe out
more C02.
Respiratory compensation is quick but hard to maintain.
Whereas is a patient has an acute exacerbation of chronic respiratory condition the body would try to
compensate, however metabolic compensation takes longer, up to 3 days.
Fully compensated arterial blood gas results are when the acidosis/ alkalosis has been corrected so that the
pH is in normal limits.
Partial compensation- when compensatory mechanisms have occurred but the pH remains abnormal.
Systematic approach to interpreting ABG
In order to interpret the results, it is simplest if you break it down into small steps.
1st – Check the patient to get an overall picture
2nd – Check the Pa02 in conjunction with Sp02 and the Fi02 of supplementary oxygen.
3rd- What is the pH of the gas?
4th- What is the PaC02?
5th- What are the HC03- and the Base Excess
6th- What is the ABG and is there any compensation?
A potentially useful tip is;
Draw arrows next to the result- is the result high or low?
If the pH and the C02 are abnormal, but going in different directions, then the origin of the abnormality will
tend to be respiratory.
If the pH is low but not excessively so (acidotic) and the PaC02 is high (probably respiratory) and if the
Bicarbonate and the Base excess are slightly raised, this would indicate that this is a partially compensated
respiratory acidosis.
Please see the table below.

Anion Gap
The anion gap (AG) can be calculated and used to differentiate different disease pathways causing imbalances
of the pH.
It is the difference between primary measured cations (sodium, Na+ and potassium, K+) and the primary
measured anions (chloride Cl- and bicarbonate HCO3-) in serum. This test is most commonly performed in
patients who present with altered mental status, unknown exposures, acute renal failure, and acute illnesses,
maybe as a result of poisoning.
Indications, Contraindications and Complications
Indications
Arterial blood gases (ABGs) are an important routine investigation to monitor the acid-base balance of
patients.
They may help make a diagnosis.
They can indicate the severity of a condition and help to assess treatment.
o Acutely unwell patients
o Exacerbation of asthma/ COPD/ Hypercapnia
o Cyanosis
o Pancreatitis
o DKA
o If there is suspicion of Carbon Monoxide poisoning.
Other tests are often available on the ABG results including;

o Lactate
o Magnesium
o Hb
o Calcium
o Potassium
o Blood glucose
Please check with your Trust and confirm accuracy of these results. But an ABG may be taken to identify some
of the above.
Be aware that venous gases may be taken instead depending on Trust policies.
Contraindications for using the radial artery
Do not perform the procedure on that arm if:
o The Allen’s test is negative.
o The patient has an arterio-venous shunt/ fistula.
o The patient has peripheral vascular disease.
o There is underlying skeletal trauma.
o There is infection at the puncture site.
Use caution if:
o The patient is on anti-coag therapy.
o They have a history of clotting or bleeding disorder.
Complications
Some complications that can occur following ABG sampling;
o Thrombus
o Arteriospasm
o Pain
o Haematoma
o Haemorrhage
o Infection
o Distal ischaemia
o Air embolism
o Sharps injury to staff
o Necrosis leading to potential amputation

Please consider whether the patient is suitable to have this procedure performed, and be aware of Trust
policy.
Patient Safety
On first meeting a patient introduce yourself, confirm that you have the correct patient with the name and date
of birth, if available please check this with the name band and written documentation such as the prescription
chart and the NHS/ hospital number/ first line of address. Some Trusts may have electronic ordering, please
check your Trust.
Check the patient’s allergy status, being aware of the skin preparation and if it is trust policy to infiltrate
lignocaine first. Ensure the procedure is explained to the patient in terms that they understand, gain informed
consent and ensure that you are supervised, with a chaperone available as appropriate. Allow the patient to ask
any questions that they may have and discuss any past problems (e.g. fainting/ bleeding/ anti-coagulant
medication history). Assist patient into a comfortable position, with arm supported. If the patient has a history
of fainting, they may be positioned better on a trolley, ask the relatives if they have any problems with needles.
Also consider the patient’s own personal preference (e.g. choice of arm) or issues preventing the use of 1 arm,
eg; lymphoedema. Don personal protective equipment as you are coming into contact with bodily fluids
(Loveday 2014), some Trusts recommend sterile glove use, please follow Trust policy and use sharps in
accordance with HSE (2013).
Be aware of hand hygiene and preventing the spread of disease, WHO (2009) https://www.who.int/infection-
prevention/tools/hand-hygiene/en/

Other Safety Considerations;
Check patient for allergies eg, Chloraprep® / Latex / Lidocaine
Do not remove 02 to assess ABG’s, this potentially can cause profound hypoxaemia.
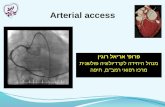
Vascular anatomy of the lower arm
By Henry Vandyke Carter - Henry Gray (1918) Anatomy of the Human Body (See "Book" section below)Bartleby.com: Gray's Anatomy, Plate 528, Public
Domain, https://commons.wikimedia.org/w/index.php?curid=541389
Modified Allen’s test
Radial arterial puncture is contraindicated when there is a known deficiency of collateral circulation to the
distal upper extremity.
A Modified Allen Test can be performed to assess the adequacy of the collateral circulation of the radial artery
by the ulnar artery.
It is recommended that this test be performed prior to sampling.
To perform the test:
Instruct the patient to make a fist to force the blood from the hand; this should be done for approximately 30
seconds. Occlude both the ulnar and radial arteries. Then instruct the patient to unclench the fist. The
Radial Artery
Ulnar Artery

patient’s palm should appear blanched or pale. Now, release pressure only from the ulnar artery. Adequate
collateral circulation is indicated by the return of normal colour within 10 seconds (Ruengsakulrach et al 2001).
Modified Allen’s test
The test is classed as positive if collateral circulation is present, and this should be documented accordingly.

Procedure
Locate the radial artery, gently supinate the patient’s
wrist.
Personal Protective Equipment (PPE).
Don appropriate PPE (see trust policy),
apron and gloves should be a minimum.
Clean skin- Product is dependent on trust
policy, ChloraPrep needs 30 seconds to
put on and then 30 seconds to dry as per
manufacturers advice. Do ensure the skin
is dry before commencing procedure.
ANTT suggests that the skin should not be re-palpated after cleaning, otherwise sterile gloves should be worn-
please refer to individual trust policy
Syringe
Some syringes are pre-set and self-fill, some have safety needles.
Please check the manufacturer’s instructions prior to carrying out the procedure.
Sampling
o Hold the needle like a pen/ dart,
different people develop different
techniques
o Beware of sharps injury at all times
o Slowly insert the needle into the
radial artery maintaining an angle of
45-60 degrees.

o On most patients the needle should not be
advanced more than 4-6mm (caution with
peripheral swelling/obesity)
o Some patients can find this procedure to be
extremely painful, so warn patients
accordingly.
o Self-filling syringes will allow blood to flow
into the syringe
o If you are palpating the artery once the syringe is filling gently release the pressure on the artery
otherwise it can occlude and you will not get the sample
o Obtain 1-2mls blood
o Remove the needle and immediately
apply pressure to the puncture site
with gauze.
o Pressure should be applied for at least
5 minutes and caution should be taken
with any patients on anti-coagulants.
o Activate the needle safe device if
appropriate
o Gently roll the sample to ensure that the blood and heparin are mixed.
o Dispose of the needle in the sharps bin.
o Consider use of filter cap if available to expel air.
o Send the sample (with/without ice) straight to the lab or the analyser- depending on where you are
working.
Documentation
Ensure that the procedure is documented clearly, this should include;
o Was informed consent gained and if not why not?
o Was the Modified Allen’s test performed and what was the result?
o Record the time and date that the sample was taken and if necessary what time it was sent to the
laboratory.
o Record the percentage of oxygen the patient is on and the temperature of the patient

o Any complications during the procedure
o Who the results should be reported to, or who will review the results, if the reviewer is not likely to be
present when the results return.
References
o Bray, John J. et al. (1999) Lecture notes on human physiology, 4th Edition. page 556, section
"Estimating plasma pH" in:. Malden, Mass.: Blackwell Science. ISBN 978-0-86542-775-4.
o Cho KC. (2012) Chapter 21. Electrolyte & Acid-Base Disorders. McPhee SJ, Papadakis MA, Rabow MW,
eds. CURRENT Medical Diagnosis & Treatment 2012. New York: McGraw-Hill.
o Criner, GJ. (2010) "Metabolic Disturbance of Acid-Base and Electrolytes." Critical Care Study Guide:
Text and Review. 2nd ed. Philadelphia, PA: Springer; 696.
o Driscoll et al (2008). BTS guidelines for emergency oxygen use in adult patients. Thorax an
international journal of respiratory medicine Vol 63 supplement VI.
o Emmet, M. (2010) The anion gap/HCO3 ratio in patients with a high anion gap metabolic acidosis.
Stern RH (ed). UpToDate. Waltham, MA: UpToDate; June 17 2010.
o Grogono,A. Retrieved 2016-04-04 "Acid-Base Tutorial - History" accessed 5/01/2017. www.acid-
base.com. accessed 3/9/2019
o Graham (2006) “Acid Base Online Tutorial” University of Connecticut
https://www.merlot.org/merlot/viewMaterial.htm?id=345419 accessed 3/9/2019
o Health and Safety Executive. Health and Safety (Sharp Instruments in Healthcare) Regulations 2013.
London: Crown Copyright, 2013.
o Jun Sik Kang et al (2014) Finger necrosis after accidental radial artery puncture. Clinical and
experimental emergency medicine Volume 1(2) Accessed 3/9/19
http://ceemjournal.org/journal/view.php?number=23
o Kang, J. S., Lee, T. R., Cha, W. C., Shin, T. G., Sim, M. S., Jo, I. J., . . . Jeong, Y. K. (2014). Finger necrosis
after accidental radial artery puncture. Clinical and Experimental Emergency Medicine, 1(2), 130-133.
doi:10.15441/ceem.14.045 [doi]
o Kaufman, David A. Interpretation of Arterial Blood Gases (ABGs) accessed 3/01/2017
https://www.thoracic.org/professionals/clinical-resources/critical-care/clinical-education/abgs.php
o Larsen, D. reviewed 05/06/2019; "Henderson-Hasselbalch Approximation". Chemwiki. University of
California. Accessed 3/9/19

http://chem.libretexts.org/Core/Physical_and_Theoretical_Chemistry/Acids_and_Bases/Buffers/Hend
erson-Hasselbalch_Approximation
o Loveday, H P., Wilson, J A., Pratt, R J., Golsorkhi, M., Tingle, A., Bak, A., Browne, J., Prieto, J., and Wilcox,
M. (2014) ‘epic3: National Evidence-Based Guidelines for Preventing Healthcare-Associated Infections
in NHS Hospitals in England’, Journal of Hospital Infection, 86(S1), pp. S1–S70.
o Mehta (2012) Henderson Hasselbalch Equation: Derivation of pKa and pKb ; accessed 03/09/2019
http://pharmaxchange.info/press/2012/10/henderson%E2%80%93hasselbalch-equation-derivation-
of-pka-and-pkb/
o Nicolaou and Kelen (2015) in Chapter 19, “Acid Base Disorders” in Tintinalli’s Emergency medicine 7th
Edition A comprehensive study guide; American College of emergency physicians; McGraw-Hill Global
Education Holdings, LLC.
o Ruengsakulrach P, Brooks M, Hare DL, Gordon I, Buxton BF. (2001) Preoperative assessment of hand
circulation by means of doppler ultrasonography and the Modified Allen Test. J Thorac Cardiovasc
Surg;121:526-31.
o Rice, B., Tomkins, S., & Ncube, F. (2015). Sharp truth: Health care workers remain at risk of
bloodborne infection. Occupational Medicine, 65(3), 210-214.
o WHO (2009); WHO guidelines on hand hygiene in Health Care; https://www.who.int/infection-
prevention/tools/hand-hygiene/en/ accessed September 2019.
o World Health Organization. (2010). WHO guidelines on drawing blood: Best practices in
phlebotomy World Health Organization.
Accessed September 2019;
o Basic Metabolic Panel: ARUP Lab Tests. ARUP Laboratories: National Reference Laboratories. 2006-
2012. Available at http://www.aruplab.com/.
o "Anion gap - Wikipedia, the free encyclopedia." N.p., 18 May 2012. Web. 20 May 2012. Wikipedia, the
free encyclopedia. Available at http://en.wikipedia.org/wiki/Anion_gap.
o https://en.wikipedia.org/wiki/Henderson%E2%80%93Hasselbalch_equation