Heating Curve evruC gnitaeH. Explain the plateau of heating and cooling curves. Include: IMFs,...
-
Upload
roberta-murphy -
Category
Documents
-
view
217 -
download
2
Transcript of Heating Curve evruC gnitaeH. Explain the plateau of heating and cooling curves. Include: IMFs,...

Heating CurveevruC gnitaeH

• Explain the plateau of heating and cooling curves.
Include: IMFs, exothermic, endothermic • Define melting and boiling point in terms of
intermolecular forces and particle size.
Additional KEY Terms


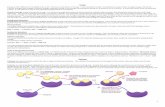
Heating Plateau:Minimum amount of energy is needed to overcome the intermolecular forces – endothermic process.
All added heat is used to overcome these forces, NOT increase kinetic energy (temperature).
Plateau ends when all particles have phase
changed.


Cooling Plateau:Particles lose energy and slow down - pulled together by the IMFs.
Organizing into restrictive phases causes the release of stored energy – exothermic process
Freezing of Water
Freezing Point
Plateau ends when all particles have phase
changed.

Freezing of Water
Freezing Point



Melting and boiling points are a characteristic physical property.
Generally, as mass increases, melting/boiling point also increases.
(more intermolecular forces to work against)

Ionic compounds typically have higher melting point than molecular compounds.
NaCl
Sugar
160oC
801oC

IMFs must be overcome in order for a substance to change states/phases.
The greater the forces between the particles, the more energy needed to overcome these forces.
H
O
H Cl -Na +
H
O
H
Cl-
Na+
So, the greater the IMFs, the higher the melting and boiling point.

CAN YOU / HAVE YOU?
• Explain the plateau of heating and cooling curves.
Include: IMFs, exothermic, endothermic • Define melting and boiling point in terms of
intermolecular forces and particle size.
Additional KEY Terms