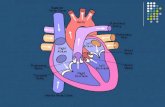
HEART
description
Transcript of HEART

HEART
ByMary Yvonnette C. Nerves, MD, FPSP

Once I had brains, and a heart also; so having tried them both, I should much rather have a heart.
--The Tin Woodsman of Oz

NORMAL

NORMALWeight: 250 to 300 g in females
300 to 350 g in males
Thickness of the free wall:
RV = 0.3 to 0.5 cm.
LV = 1.3 to 1.5 cm.


Hypertrophy: greater heart weight or ventricular thickness
Dilation: an enlarged chamber size
Cardiomegaly: an increase in cardiac weight or size (owing to hypertrophy and/or dilation)


Layers of the heart:
1. Epicardium
2. Myocardium
3. Endocardium
Pericardium: fibrous covering around the heart


Myocardiumcomposed primarily of a collection of specialized muscle cells called cardiac myocytes
Ventricular muscle contracts during systole and relaxes during diastole
Sarcomere: functional intracellular contractile unit of cardiac muscle


Specialized excitatory and conducting myocytes

BLOOD SUPPLY(3) Major Epicardial Coronary arteries:a. Left Anterior Descending Artery
- Anterior wall- Anterior two thirds of septum- Entire apex of heart, circumferentially
b. Left Circumflex Coronary Artery- posterior, lateral left aspect of heart.
c. Right Coronary Artery- posterior one third of septum, inferior
aspect, and posterior wall of heart.


VALVES1. Semilunar valves
a. Aortic valveb. Pulmonary valve
2. Atrioventricular valvesa. Mitral valveb. Tricuspid valve




PATHOLOGY

PATHOLOGY Failure of the pump
An obstruction to flow
Regurgitant flow
Disorders of cardiac conduction.
Disruption of the continuity of the circulatory system

Congestive Heart Failure (CHF)
pathophysiologic state resulting from impaired cardiac function that renders the heart muscle unable to maintain an output sufficient for the metabolic requirements of the tissues & organs of the body

Congestive Heart Failure (CHF)
the physiology of HF involves an interplay between 2 factors:
a) inability of the failing heart tomaintain sufficient cardiac output to support body functions
b) recruitment of compensatorymechanisms to maintain cardiacreserve

Cardiac output: amt of blood that the heart pumps each minute
- reflects how often the heart beats each minute (HR) and how much blood the heart pumps with each beat (SV)
CO = HR x SV

Causes of CHF Pump Failure: Failure that is intrinsic to
the myocardium. Two types:
Systolic Failure: Failure to pump blood out of heart.
Diastolic Failure: Failure to distend the heart to fill the ventricles, as in constrictive pericarditis.
Most common reason for pump failure is from myocardial hypertrophy, usually sec. to HPN.

Conduction System Failure: Secondary to
MI
Valvular Failure: Inflammatory (endocarditis), autoimmune, or congenital.
Cardiac Malformations: Congenital
Blood Loss / Obstruction of Blood Flow: Extracardiac causes. Pulmonary
emboli or bleeding.

Mechanisms that maintain arterial pressure and perfusion of vital organs in the presence of excessive hemodynamic burden or disturbance in myocardial contractility:
1. Frank-Starling mechanism- increased preload of dilation helps to
sustain cardiac performance by enhancing contractility
2. Myocardial structural changes - augmented muscle mass w/ or w/o cardiac chamber dilation, in whichthe mass of contractile tissue isaugmented

3. Activation of neurohumoral systems release of the neurotransmitter
norepinephrine by adrenergic cardiac nerves
activation of the renin-angiotensin-aldosterone system, and
release of atrial natriuretic peptide.

Heart’s compensatory changes Hypertrophy Ventricular dilatation Blood volume expansion by salt &
water retention Tachycardia

Extent of hypertrophy varies for different underlying causes: 350 – 600 gms: pulmonary HPN / IHD 400 – 800 gms: systemic HPN / aortic
stenosis / mitral regurgitation / dilated cardiomyopathy
600- 1000 gms: aortic regurgitation /hypertrophic
cardiomyopathy


The pattern of hypertrophy reflects the nature of the stimulus:1. Pressure-overload (concentric)
hypertrophy Pressure-overloaded ventricles (e.g., in
hypertension or aortic stenosis) develop pressure-overload hypertrophy of the LV, with an increased wall thickness
LV: the augmented muscle may reduce the cavity diameter
the predominant deposition of sarcomeres is parallel to the long axes of cells; cross-sectional area of myocytes is expanded (but cell length is not).

2. Volume-overload hypertrophy volume overload stimulates deposition
of new sarcomeres and cell length (as well as width) is increased
characterized by dilation with increased ventricular diameter
muscle mass and wall thickness are increased approximately in proportion to chamber diameter



Left-sided CHF
Failure of the left side of the heart to pump sufficient blood
Common causes of left-sided failure Ischemia (old or recent myocardial infarct,
ischemic muscle disease) Aortic or mitral valve disease Systemic hypertension Myocardial disease / cardiomyopathy

Clinical manifestations: Pulmonary congestion and edema Reduced cardiac output also causes reduced
renal perfusion, leading to Further salt & water retention Ischemic acute tubular necrosis Impairment of waste excretion, causing prerenal
azotemia
CNS perfusion is reduced, often resulting to hypoxic encephalopathy

Right-sided CHF
Failure of the right side of the heart to pump enough blood
Common causes of right-sided failure Pulmonary emboli (acute or chronic) Any disease interfering with lung ventilation
Emphysema / Cystic fibrosis Left-sided heart failure! Cardiac defects with left-to-right shunts

Clinical manifestations: Portal, systemic and dependent peripheral
congestion & edema & effusions (pleural & peritoneal)
Hepatomegaly w/ centrilobular congestion & atrophy of central hepatocytes nutmeg liver (chronic passive congestion)
Centrilobular necrosis Cardiac sclerosis
Congestive splenomegaly Renal congestion




HEART DISEASE
Congenital heart disease
Ischemic heart disease
Hypertensive heart disease (systemic & pulmonary)
Valvular heart disease
Nonischemic (primary) myocardial disease
Cardiac Neoplasms

CONGENITAL HEART
DISEASE

CONGENITAL HEART DISEASE Abnormalities of the heart andgreat vessels present at birth Most common type of heart disease among chldrenOccurs during 3rd to 8th week of gestation Incidence: 1% of livebirths Higher incidence among prematures
and stillborn

Heart at 18 to 22 days
22 days
Angioblasts (heart formingcells) first appear at 18 days
Two dorsal aortae developand subsequently fuse in themid-line to form a primitiveheart tube.
Heart first forms rostral tothe neural plate
As the brain grows, the heartmoves caudally into the neckand then into the chest
Origin of the Heart
Heart
Heart
Angioblasts
Latepresomiteembryo
18 days




EMBRYOLOGIC HEART DERIVATIVES:
Embryonic Structure Adult Structure
Bulbus Cordis Right Ventricle and Aortic Outflow Track
Primitive Ventricle Left Ventricle
Truncus Arteriosus Ascending Aorta, Pulmonary Trunk
Primitive Atria Auricular Appendages
Sinus Venosus Left Horn: Coronary Sinus Right Horn: Smooth part of the Right Atrium
Right Common and Anterior Cardinal Veins
Superior Vena Cava

Malformation
Incidence per Million Live
Births %
Ventricular septal defect 4482 42
Atrial septal defect 1043 10
Pulmonary stenosis 836 8
Patent ductus arteriosus 781 7
Tetralogy of Fallot 577 5
Coarctation of aorta 492 5
Atrioventricular septal defect 396 4
Aortic stenosis 388 4
Transposition of great arteries 388 4
Truncus arteriosus 136 1
Total anomalous pulmonary venous connection
120 1
Tricuspid atresia 118 1
TOTAL 9757

Etiology 1. Genetics - Trisomy 13, 15, 18 , 21, Turner
Syndrome
2. Environmental Factors- congenital rubella infection /
teratogens / alcohol / amphetamines / chemotx / anticonvulsants / thalidomide / retinoic acid
3. Multifactorial

Classification
1. Left to right shunt anomaly
2. Right to left shunt anomaly
3. Obstructive anomaly
SHUNT: an abnormal communication between chambers or blood vessels


LEFT TO RIGHT SHUNT
1. ASD –Atrial septal defect
2. VSD – ventricular septal defect
3. PDA – Patent ductus arteriosus
4. AVSD – Atrio-ventricular septal defect

ATRIAL SEPTAL DEFECT
direct communication between the atrial chambers
Abnormal opening in the atrial septum Most common CHD that is asymptomatic until adulthood Acyanotic at birth Symptoms may manifest in third decade

ATRIAL SEPTAL DEFECT
Major Types:1. Ostium secundum - 90% of ASD (most common)
- defect in the fossa ovale
- may be solitary or with other anomaly

ATRIAL SEPTAL DEFECT cont. Major Types:
2. Ostium primum - 5 % of ASD
- defect occurs near the AV valve
- assoc with a clefted anterior MV valve (partial ASD)

ATRIAL SEPTAL DEFECT cont. Major Types:
3. Sinus venosus defect- 5 % of ASD
- located near the entrance of SVC
- associated with TAPVR



Effects of ASD
RAH and enlargement RVH and enlargement Increase pulm blood flow pulm
HPN Murmur due to inc blood flow to pulm valve

VENTRICULAR SEPTAL DEFECT
direct communication between theventricular chambers
Incomplete closure of the ventricularseptum associated with other structural defects,such as tetralogy of Fallot 30% occur as isolated anomalies

VENTRICULAR SEPTAL DEFECT
Major Types:1. Membranous VSD
- 90 % of VSD
2. Infundibular VSD
- located below the PV or within the muscular septum (Muscular VSD)



ATRIOVENTRICULAR SEPTAL DEFECT
Complete AV canal defect Abn development of AV canal Incomplete closure of AV septum and inadequate formation of TV and MV Failure of fusion of sup and inf
endocardial cushions

AVSD: Types1. Partial AVSD - primum ASD, cleft ant mitral
leaflet2. Complete AVSD - large combined AVSD and a
large common AV valve - free communication of all 4
chambers




PATENT DUCTUS ARTERIOSUS
Persistence of communication between PA and aorta via ductusarteriosus 90 % as isolated anomaly 10 % assoc with VSD, coarctation, pulmonary or aortic stenosis “Machinery murmur “



RIGHT TO LEFT SHUNT
1. Tetralogy of Fallot (TOF)2. Transposition of the Great Vessels3. Truncus arteriosus4. Tricuspid atresia5. Total Anomalous Pulmonary Venous
Return

Components:
TETRALOGY OF FALLOT

cont. TOF: Morphology heart is often enlarged “ boot – shaped “ ("coeur en sabot“) RVH
Large VSD, overriding the aorta Subpulmonic valve stenosis obs to
RV outflow Assoc lesions: ASD, right aortic arch

TOF : clinical features
Right to left shunt “ Pink tetralogy “ – mild Pulm
Stenosis , LR shunt “ classic tetralogy “ severe PS ,
RL shunt Survival dependent on PS


TRANSPOSITION OF GREAT VESSELS
abnormal development of the truncoconal septum that results in inversion of the aorta and pulmonary arteries with respect to the ventricles (Ventriculo-arterial discordance) Aorta arises from RV, Pulm artery from LV Incompatible with life Assoc lesions: VSD (stable shunts); PDA (unstable shunts); RVH; LVhypoplasia




TRUNCUS ARTERIOSUS Failure to develop a dividing septum bet the aorta and pulmonary artery, resulting in a common trunk single great artery Failure of separation of truncus arteriosus into aorta and pulmonary
artery Assoc lesions: VSD Mixing of blood early cyanosis,
Pulm HPN


TRICUSPID ATRESIA
absence of communication between the right atrium and ventricle due to developmental failure to form the tricuspid valve (complete absence of TV)
Results from unequal devision of AV canal
Assoc lesions: Large MV, underdeveloped RV, ASD, VSD or PDA
Early cyanosis with high MR

TOTAL ANOMALOUS PULM VENOUS CONNECTION (TAPVC)
No pulmonary veins in LA Pulm veins drain into the coronary
sinus or left innominate veins Assoc ASD or PDA
RAD, RVD, pulm trunk dilation, hypoplastic LA



Comparison of Left vs. Right Shunt Congenital Disease
Right Left Shunt Early cyanosis (blue
babies) Blood shunted past the
lungs
TOFTOGVTricuspid atresiaTruncus arteriosus
Left Right Shunt Late cyanosis (blue
kids) Pulmonary HPN
reversal of shunt (Eisenmenger syndrome)
VSD, ASD, PDA

OBSTRUCTIVE ANOMALIES
1. Coartation of aorta
2. Pulmonic stenosis / atresia
3. Aortic stenosis / atresia

COARTATION OF AORTA Types :
1. Infantile Coarctation
- tubular hypoplasia of aortic arch proximal to PDA
2. Adult Coarctation
- ridgelike coaptation of aorta at the level of ductus
arteriosus

Clinical manifestations depend almost entirely on the severity of the narrowing and the patency of the ductus arteriosus.






PULMONARY STENOSIS / ATRESIA
Common S/S dependent on size of PV May occur as isolated event or part of complex anomalyAssoc lesions: RVH; PA hypoplasia;
RV hypoplasia inPV atresia; ASD


AORTIC STENOSIS / ATRESIA
Narrowing/ obstruction of AV
Three types:
a. Valvular AS
b. Subaortic stenosis
c. Supravalvular stenosis


ISCHEMIC HEART
DISEASE

ISCHEMIC HEART DISEASE
Group of related cardiac diseases resulting from imbalance bet. blood supply to the heart and its demand for oxygen (myocardial ischemia)
Aka: Coronary artery disease or coronary heart disease

SYNDROMES OF IHD
1. Acute Myocardial Infarction
2. Angina Pectoris
3. Chronic Ischemic Heart Disease
4. Sudden Cardiac Death

IHD: Pathogenesis Decrease coronary perfusion relative
to blood demand Causes :
fixed stenosing atherosclerosis fissure, rupture, ulceration, hge
(acute plaque change) thrombosis vasoconstriction/ vasospasm

IHD : Role of fixed coronary obstruction
90 % of IHD with fixed CA atherosclerosis (fixed obstruction)
may affect single, dual or triple CA
Onset of symptoms depend on extent, severity of occlusion and acute plaque change
Associated with typical angina pectoris

Fixed coronary atherosclerosis

IHD: Role of acute plaque change
Assoc with acute coronary syndromes (unstable angina, AMI, sudden cardiac death) Acute change: rupture/fissuring,erosion/ulceration, hge into the atheroma Ischemia ppted by acute plaque change followed by thrombosis High in moderate stenosis and lipid rich atheroma

Acute plaque change

Acute Plaque Change: Fissuring, hemorrhage

ACUTE PLAQUE CHANGE : HGE Acute Plaque Change: Hemorrhage

IHD: Role of inflammation initial lesion: interaction bet.
endothelial cells & circulating leukocytes later stages: secretion of
metalloproteinases bymacrophages

IHD : role of thrombosis
Occlusive thrombi assoc with acute transmural MI
Partial occlusive thrombi assoc with unstable angina, subendocardial infarction and SCD

Acute Plaque Change Thrombosis


Role of vasoconstriction
Reduction of lumen size increases local mechanical forces increase risk of plaque rupture


ANGINA PECTORIS
paroxysmal, recurrent precordial orsubsternal chest pain, caused bytransient myocardial ischemia
which falls short of infarction 3 types :
a. Stable or typical AP b. Prinzmetal or variant AP c. Unstable or crescendo AP

Angina pectoris: Etiology Increased myocardial demand and decreased myocardial perfusion secondary to :
Chronic stenosing coronary atherosclerosis
Disrupted artherosclerotic plaques Vasospasm Thrombosis Coronary artery embolization

Stable Angina Pectoris
Most common form of AP Reduction of perfusion caused by chronic stenosing CA atherosclerosis Chest pain: transient, ppted by exertion, emotion, relieved by rest and vasodilators

Prinzmetal angina pectoris
Episodic pain at rest
Secondary to vasospasm
ECG changes suggestive of transmural ischemia (ST segment elevation)
Responds to nitroglycerin, Ca channel blockers and vasodilators

Unstable angina pectoris
Pain progressively increasing in frequency, duration, intensity, occurs at rest Assoc with acute plaque change,with
superimposed partial thrombosis or vasospasm Precursor to AMI (pre-infarction AP)

MYOCARDIAL INFARCTION
Localized area of cardiac muscle necrosis due to ischemia
Risk factors:
Major: HPN, Cigarette smoking, DM, Hyperlipidemia
Minor: obesity, sex, age, stress, physical inactivity

AMI: PathogenesisCoronary arterial occlusion: atherosclerosis with acute plaque change, thrombosis, vasospasm
Increase myocardial demand
Hemodynamic compromise
Emboli
unknown

Irreversible myocardial damage
After severe ischemia , lasting from 20 – 40 min or longer
Assoc with less than 10 % reduction of blood flow
Initial damage to sarcolemmal membrane

Myocardial changes in AMI:
Ischemic coagulation necrosis , initially at subendocardial region,
with wavefront progression
Morphologic changes evolve in time

Factors affecting severity of AMI
Location , severity of obstruction
Rate of development of occlusion
Duration of occlusion
Extent of collateral vessels
Metabolic demands of myocardium
Other factors : BP, HR, cardiacrhythm


TRANSMURAL INFARCTION
Transmural infarct necrosis involves the full or nearly the full
thickness of wall
Assoc with chronic atherosclerotic obstruction , acute plaque change and superimposed complete thrombosis

SUBENDOCARDIAL INFARCTION
Necrosis limited to inner 1/3 or ½ ofventricle
Assoc with diffuse stenosing coronaryatherosclerosis w/o thrombosis
and acute plaque change

AMI: MorphologyGROSS Early recognition: Triphenyl tetrazolium
chloride (unstained area) Coagulation necrosis of myocardium Gross changes evident after 4-12
hours 18-24 hrs: pale to cyanotic 7-10 days: circumferential rim of
granulation tissue 6 weeks: fibrous scar

AMI : morphology Microscopic: within 1 hr: intercellular edema, wavy fibers
at periphery, coagulation necrosis not yetevident
12-72 hrs: neutrophilic infiltration into necrotictx, myocyte coag necrosis, dead myocytes hypereosinophilic w/ loss of nuclei
between 3-7 days: disintegration of dead myofibers
After 7-10 days: granulation tx replaces necrotic tx fibrous scar

TimeGross
Features Light Microscope Electron Microscope
Reversible Injury
0-½ hr
None None Relaxation of myofibrils; glycogen loss; mitochondrial swelling
Irreversible Injury
½-4 hr
None Usually none; variable waviness of fibers at border
Sarcolemmal disruption; mitochondrial amorphous densities
4-12 hr
Occasionally dark mottling
Beginning coagulation necrosis; edema; hemorrhage
12-24 hr
Dark mottling Ongoing coagulation necrosis; pyknosis of nuclei; myocyte hypereosinophilia; marginal contraction band necrosis; beginning neutrophilic infiltrate

Time Gross Features Light Microscope EM
1-3 days Mottling with yellow-tan infarct center
Coagulation necrosis, with loss of nuclei and striations; interstitial infiltrate of neutrophils
3-7 days Hyperemic border; central yellow-tan softening
Beginning disintegration of dead myofibers, with dying neutrophils; early phagocytosis of dead cells by macrophages at infarct border
7-10 days
Maximally yellow-tan and soft, with depressed red-tan margins
Well-developed phagocytosis of dead cells; early formation of fibrovascular granulation tissue at margins

Time Gross Features Light Microscope EM
10-14 days
Red-gray depressed infarct borders
Well-established granulation tissue with new blood vessels and collagen deposition
2-8 wk Gray-white scar, progressive from border toward core of infarct
Increased collagen deposition, with decreased cellularity
>2 mo Scarring complete Dense collagenous scar

CA Atherosclerosis with Thrombosis


Acute Plaque Change: Fissures /
Ulceration

AMI : Wavy fibers
AMI : ½ to 4 hrs

AMI : Coagulation necrosis, hge, edema at 4-12 hrs


AMI : 12-24 hrs

AMI at 12-24 hrs: Contraction band necrosis

Contraction band necrosis

AMI at 12-24 hrs: Myocyte hypereosinophilia, contraction band, leukocytes

AMI : 1-3 days

AMI at 1-3 days: Coagulation necrosis, loss of striations,
leukocytes

AMI at 1-3 days: Necrosis, loss of nuclei/striations, leukocytic infiltration

AMI at 3-7 days

AMI at 3-7 days: Disintegration of myocyte, PMNs, phagocytosis

AMI at 3-7 days: Disintegration of myocytes, neutrophils, early
phagocytosis


AMI at 7-10 days: Well developed phagocytosis, fibrovascular granulation tissue



AMI at 10-14 days: Well developed granulation tissue



AMI at 2-4 weeks: Well developed fibrosis

Fibrosis


Clinical Features Symptoms: rapid weak pulse &
diaphoretic; dyspnea
ECG: new Q waves
Labs: myoglobin, cardiac troponins I & T (TnI, TnT), creatine kinase, LDH, CRP, etc.
Absence of a change in the levels of CK and CK-MB during the 1st 2 days of chest pain & of troponin in the days ff essentially excludes the diagnosis of MI

Serum Markers Used to Diagnose MI
Elevated by
Peak Returns to Normal by
CK-MB 4-8 hrs 18 hrs 2-3 days
Troponin I & T 3-6 hrs 16 hrs 7-10 days
LDH 24 hrs 3-6 hrs 8-14 days

Complications Contractile dysfunction
Arrhythmias
Myocardial rupture
Pericarditis
RV infarction
Infarct extension
Infarct expansion

Complications Mural thrombus
Ventricular aneurysm
Papillary muscle dysfunction
Progressive late heart failure

Complications The propensity towards specific complications & the prognosis after MI depend primarily on infarct size, site & fractional thickness of the myocardial wall that is damaged.
Large transmural infarcts: cardiogenic shock, arrhythmias, & late CHF
Anterior transmural: free wall rupture, expansion, mural thrombi, & aneurysm
Posterior transmural infarcts: conduction blocks, RV involvement

CHRONIC ISCHEMIC HEART DISEASE
Cardiac findings in patients, often but not exclusively elderly, who develop progressive heart failure as a consequence of ischemic myocardial damage
Aka: ischemic cardiomyopathy

Morphology:Heart is enlarged & heavy, sec. to LVH & dilation
Discrete gray white scars of healed infarcts
Micro: myocardial hypertrophy, diffuse subendocardial vacuolization, scars of previous healed infarcts

SUDDEN CARDIAC DEATHUnexpected death from cardiac causes early after symptom onset (usually within 1 hr) or without the onset of symptoms

Nonatherosclerotic causes of SCD:Congenital structural or coronary arterial abnormalities
Aortic valve stenosis
Mitral valve prolapse
Dilated or hypertrophic cardiomyopathy
Myocarditis
Pulm HPN
Hereditary or acquired abn of the cardiac conduction system
Isolated hypertrophy

The ultimate mechanism of SCD is most often a lethal arrythmia (eg. asystole, ventricular fibrillation)

Morphology:Marked coronary atherosclerosis with critical (> 75%) stenosis involving one or more of the 3 major vessels is present in 80-90% of SCD
Healed MI is present in 40%
Subendocardial myocyte vacuolization indicative of severe chronic ischemia is common

HYPERTENSIVE HEART DISEASE

HYPERTENSIVE HEART DISEASE
Systemic (left-sided) hypertensiveheart disease
Pulmonary (right-sided) hypertensive heart disease (Cor pulmonale)

Systemic HHD
Diagnostic Criteria: LVH, concentric
- Absence of other lesions that might induce cardiac hypertrophy
Hx or pathologic evidence of HPN

Morphology: GROSS: thickened LV wall w/
increased heart wt MICRO:
- earliest change: increase in transverse diameter of
the myocytes
- myocytes & nuclei enlarged
- diffuse interstitial fibrosis


Pulmonary HHDconsists of RVH, dilation, & potentially failure secondary to pulmonary HPN caused by dso of the lungs or pulmonary vasculature

Pulmonary HHD Acute cor pulmonale:
- RV dilation after massive pulmonary embolization
Chronic cor pulmonale
- Result of chronic RV pressure overload

Table 12-6. Disorders Predisposing to Cor Pulmonale
Diseases of the Pulmonary Parenchyma
Chronic obstructive pulmonary disease
Diffuse pulmonary interstitial fibrosis
Pneumoconioses
Cystic fibrosis
Bronchiectasis
Diseases of the Pulmonary Vessels
Recurrent pulmonary thromboembolism
Primary pulmonary hypertension
Extensive pulmonary arteritis (e.g., Wegener granulomatosis)
Drug-, toxin-, or radiation-induced vascular obstruction
Extensive pulmonary tumor microembolism

Disorders Affecting Chest Movement
Kyphoscoliosis
Marked obesity (pickwickian syndrome)
Neuromuscular diseases
Disorders Inducing Pulmonary Arterial Constriction
Metabolic acidosis
Hypoxemia
Chronic altitude sickness
Obstruction to major airways
Idiopathic alveolar hypoventilation
Table 12-6. Disorders Predisposing to Cor Pulmonale

Morphology: GROSS:
- Acute: marked RV dilation w/o hypertrophy
- Chronic: RV wall thickens RV dilation may lead to tricuspid
regurgitation



VALVULAR HEART DISEASE




valvular involvement by dse causingstenosis, insufficiency, or both
STENOSIS: failure of a valve toopen completely, impeding forward flow INSUFFICIENCY: failure of a valve to close completely, allowing reversed flow
Valvular Heart Disease

Mitral Valve Disease Aortic Valve Disease
Mitral Stenosis Aortic Stenosis
Postinflammatory scarring (rheumatic heart disease)
Postinflammatory scarring (rheumatic heart disease)
Senile calcific aortic stenosis
Calcification of congenitally deformed valve
Mitral Regurgitation Aortic Regurgitation
Abnormalities of Leaflets and Commissures
Intrinsic Valvular Disease
Postinflammatory scarring Postinflammatory scarring (rheumatic heart disease)
Infective endocarditis Infective endocarditis
Mitral valve prolapse
Fen-phen-induced valvular fibrosis
MAJOR ETIOLOGIES OF ACQUIRED HEART VALVE DISEASE

Abnormalities of Tensor Apparatus Aortic Disease
Rupture of papillary muscle Degenerative aortic dilation
Papillary muscle dysfunction (fibrosis) Syphilitic aortitis
Rupture of chordae tendineae Ankylosing spondylitis
Rheumatoid arthritis
Marfan syndrome
Abnormalities of Left Ventricular Cavity and/or Annulus
LV enlargement (myocarditis, dilated cardiomyopathy)
Calcification of mitral ring
Mitral Valve Disease Aortic Valve Disease

Most frequent causes of major functional valvular lesions:
Aortic stenosis: calcification ofanatomically normal & congenitally bicuspid aortic valve
Aortic insufficiency: dilation of theascending aorta
Mitral stenosis: RHD
Mitral insufficiency: myxomatous degeneration (MVP)

Valvular Degeneration caused by Calcification
Calcific Aortic Stenosis
Calcific Stenosis of Congenitally Bicuspid Aortic Valve
Mitral Annular Calcification

Calcific Aortic Stenosis most common of all valvular
abnormalities
age-related dystrophic calcification, degeneration & stenosis of the AV
Acquired aortic stenosis: calcif owing to progressive & advanced age-assoc “wear & tear” of either previously anatomically normal AV or congenitally bicuspid valves

Morphology:
- Hallmark: heaped-up calcified masses w/in the aortic
cusps
- commissural fusion is NOT a usual feature
- MV generally normal
- concentric LVH from chronicpressure overload


Calcific Stenosis of Congenitally Bicuspid Aortic Valve
Frequency: 1.4% of live births neither stenotic nor symptomatic atbirth or throughout early life Congenitally Bicuspid Aortic Valve: only 2 functional cusps of unequal size, the larger having a median raphe
MV is normal


Mitral Annular Calcification
degenerative, noninflammatory, calcific deposits w/in the mitral annulus, usually in the elderly GROSS: irreg, stony hard, occlly
ulcerated nodules behind the leaflets
Regurgitation occur owing to inadeq systolic contraction of the MV ring

Mitral Annular Calcification
stenosis by impairing opening of the mitral leaflets
Calcific nodules may provide a site for thrombi that can embolize
Nodular calcific deposits may cause arrythmias by impinging on the conduction pathways
rare focus of infective endocarditis

Calcific valvular degeneration. C and D, Mitral annular calcification, with calcific nodules at the base. C, Left atrial view. D, Cut section of myocardium.

Myxomatous Degeneration of the Mitral Valve (MV Prolapse)
one or both MV leaflets are “floppy” & prolapse, or balloon back into the LA during systole
Mitral Valve Prolapse: affect 3% or more of adults, most often women
Myxomatous degeneration of the MV: pathologic

MORPHOLOGY
Gross: interchordal ballooning (> 4mm) of the MV leaflets & elongated, attenuated, or occlly ruptured chordae tendinae

Myxomatous degeneration of the mitral valve. A, Long axis of left ventricle demonstrating hooding with prolapse of the posterior mitral leaflet into the left atrium (arrow).

Myxomatous degeneration of the mitral valve.

MORPHOLOGY
Micro: thinning & degeneration ofthe fibrosa layer w/ myxomatous thickening of the spongiosa
- Secondary changes:
a. fibrous thickening of the valve leaflets
b. thickened LV endocardium
c. atrial thrombosis
d. calcification of the mitral annulus

Pathogenesis
underlying developmental defect of CT
common feature of Marfan syndrome (caused by mutations inthe gene encoding fibrillin-1 – formation of elastic fiber)

Clinical Features
MVP: asymptomatic; midsystolic click on auscultation
- Complications:
a. infective endocarditis
b. mitral insufficiency
c. atrial & ventricular arrhythmias
d. stroke or other systemic infarcts

Rheumatic Fever & RHD
RF: an acute, immunologicallymediated, multisystem, inflamm
dse triggered by pharyngeal infection w/ Group A beta- hemolytic strep
Most important consequence: chronic valvular deformities


MORPHOLOGY
Micro: Myocarditis: Aschoff bodies foci of swollen
eosinophilic collagen surrounded by lymphos, occasional plasma cells, and plump macrophages called Anitschkow cells (pathognomonic for RF); some of the larger macrophages become
multinucleated to form Aschoff giant cells Found in muscle layer
Fibrinous pericarditis – “bread & butter” Endocarditis



MORPHOLOGY
Micro: Subendocardial collections of Aschoff
nodules, usually in the LA, induce thickenings called MacCallum plaques
Chronic RHD: Cardinal anatomic changes
1. Fibrous thickening of the leaflets
2. Commissural fusion & shortening
3. Thickening and fusion of the tendinous cords


Pathogenesis ARF is a hypersensitivity reaction induced by group A streptococci
Abs directed against the M proteins of certain strains of streptococci cross-react with glycoprotein Ags in the heart, joints, & other tissues


Clinical Features Diagnosis rests on the clinical hx & the presence of 2 of 5 major (Jones) criteria:
(1) migratory polyarthritis of the large joints
(2) carditis
(3) subcutaneous nodules
(4) erythema marginatum of the skin,
(5) Sydenham chorea: neurologic dso w/ involuntary purposeless, rapidmovements.


Infective Endocarditis
colonization of the heart valves w/ microbiologic organisms that leads to
fotn of friable, infected vegetations & freq valve injury
most cases are bacterial
Classification:1. Acute Infective Endocarditis
2. Subacute Infective Endocarditis

Acute Endocarditis caused by highly virulent organisms,
often seeding a previously normal valve & producing a necrotizing, ulcerative & invasive infection
Subacute Endocarditis caused by an organism of mod to low
virulence seeding previously injured valve, causing less valvular destruction


Etiology & Pathogenesis
bacteremia are prerequisites
Risk factors:
- Cardiac congenital anomalies
- Chronic RHD
- MVP, degenerative calcific stenosis, Bicuspid aortic valve, Prosthetic
valves
- Host factors: neutropenia, malignancy
- sterile platelet-fibrin deposits

Etiology & Pathogenesis Causative organisms: S. viridans: endocarditis of native but
previously damaged or otherwise abn valves
S. aureus: attack either healthy or deformed valves; IV drug users
Enterococci & HACEK grp (Haemophilus, Actinobacillus,Cardiobacterium, Eikenella, Kingella)
S. epidermidis: prosthetic valve endocarditis

MORPHOLOGY Gross: friable, bulky, & destructive vegetations containing fibrin, inflam cells & bacteria or other organisms present
Most common sites of infection: AV & MV
Vegetations sometimes erode into theunderlying myocardium to produce an abscess cavity (ring abscess)




MORPHOLOGY Systemic emboli may occur at any time because of the friable nature of the vegetations
Subacute endocarditis: less valvular destruction; granulation tx at their bases suggesting chronicity

Clinical Features
fever is the most consistent sign
Subacute: fever is slight or absent
Acute: fever, chills, weakness & lassitude
Duke’s Criteria: stdized assessment of pts w/ IE

Pathologic Criteria
Microorganisms, demonstrated by culture or histologic examination, in a vegetation, embolus from a vegetation, or intracardiac abscess
Histologic confirmation of active endocarditis in vegetation or intracardiac abscess
Clinical Criteria
Major
Positive blood culture(s) indicating characteristic organism or persistence of unusual organism
Echocardiographic findings, including valve-related or implant-related mass or abscess, or partial separation of artificial valve
New valvular regurgitation
Minor
Predisposing heart lesion or intravenous drug use
Fever
Vascular lesions, including arterial petechiae, subungual/splinter hemorrhages, emboli, septic infarcts, mycotic aneurysm, intracranial hemorrhage, Janeway lesions†
Immunologic phenomena, including glomerulonephritis, Osler nodes,‡ Roth spots,§ rheumatoid factor
Microbiologic evidence, including single culture showing uncharacteristic organism
Echocardiographic findings consistent with but not diagnostic of endocarditis, including new valvular regurgitation, pericarditis

Non-infected Vegetations
Nonbacterial ThromboticEndocarditis (NBTE)
Endocarditis of SLE (Libman-Sacks Disease

marantic endocarditis char by deposition of small masses of
fibrin, platelets, & other bld components on the leaflets of the cardiac valves Valve lesions are sterile & do not contain microorg
Nonbacterial ThromboticEndocarditis (NBTE)

Gross: vegetations are sterile, nondestructive and small & occur singly or multiply along the line of closure of the leaflets Micro: bland thrombus w/o accompanying inflam reaction or induced valve damage
Morphology

hypercoagulable state w/ systemic activation of blood coagulation
such as DIC related to some underlying dse eg. CA
Pathogenesis



mitral amd tricuspid valvulitis w/ small, sterile vegetations Micro: verrucae consists of a finely
granular, fibrinous eosinophilic material that may contain
hematoxylin bodies
Endocarditis of SLE (Libman-Sacks Dse.)

Diagrammatic comparison of the lesions in the four major forms of vegetative endocarditis.


CARDIOMYOPATHIES


Inflammatory disorders (myocarditis) Immunologic & systemic metabolic
disorders Genetic abnormalities in cardiac muscle cells
Myocardial Diseases

Cardiomyopathy: heart disease resulting from a primary abnormality in the myocardium
(3) Categories of cardiomyopathy:a. Dilated – most commonb. Hypertrophicc. Restrictive
Dx: Endomyocardial biopsy


char by progressive cardiac dilation & contractile (systolic)
dysfunction Aka: Congestive cardiomyopathy 25-35% have familial form
Dilated Cardiomyopathy

Gross: heart is heavy & flabby, w/ dilation of all chambers
Micro: histologic abnormalities arenonspecific & usually do not
reflect a specific etiologic agent Most muscle cells are hypertrophied
with enlarged nuclei, but many are attenuated, stretched, and irregular
Interstitial and endocardial fibrosis of variable degree
Morphology



Myocarditis: viral nucleic acids (coxsackievirus B & other enterovirus) Alcohol or other toxicity Pregnancy-associated: peripartum CM Genetic influences: autosomal dominant/autosomal recessive/x-linked recessive/mitchondrial defect affects the cytoskeleton
Pathogenesis

commonly affects individuals between ages 20-50 slowly progressive signs & symptoms of CHF
Clinical Features

Aka: idiopathic hypertrophicsubaortic stenosis / hypertrophic obstructive cardiomyopthy
char by myocardial hypertrophy,abnormal diastolic filling, &
intermittent ventricular outflow obstruction heart is thick-walled, heavy, &
hypercontracting
Hypertrophic Cardiomyopathy

Gross: massive myocardial hypertrophy without ventricular dilation- classic pattern: disproportionate thickening of the ventricular septum as compared with the free wall of the LV (assymetrical septal hypertrophy)- C/S: ventricular cavity compressed
Morphology


Micro: most impt histologic features- extensive myocyte hypertrophy w/
transverse myocyte diameters > 40um
- myofiber disarray- interstitial & replacement fibrosis
Morphology


caused by a mutation in any one of several genes that encode proteins that are part of the sarcomere, the contractile unit of cardiac & skeletal muscle most cases are familial, transmitted as autosomal dominant 12 sarcomeric genes: B-MHC gene
Pathogenesis


impaired diastolic filling of the massivelyhypertrophied LV reduced chambersize & poor compliance with reduced stroke volume decreased CO & increase in pulm venous
pressure exertional dyspnea harsh systolic ejection murmur
Clinical Features

anginal pain Major clinical problems:
- atrial fibrillation w/ mural thrombus fotn
- infective endocarditis of MV- intractable cardiac failure
- ventricular arrythmias- sudden death

char by primary decrease in ventricular compliance, resulting inimpaired ventricular filling during diastole contractile (systolic) function of the
LV is not affected
Restrictive Cardiomyopathy

Gross: ventricles of normal size or sl enlarged, cavities are no dilated & the myocardium is firm
biatrial dilation commonly observed
Micro: patchy or diffuse interstitial fibrosis
Morphology


idiopathic or associated with distinct diseases that affect the myocardium:
- radiation fibrosis- amyloidosis- sarcoidosis- metastatic tumor- products of inborn errors of metabolism
Pathogenesis

other restrictive conditions:- endomyocardial fibrosis- Loeffler endomyocarditis- endocardial fibroelastosis
Pathogenesis


Functional Pattern
LV Ejection Fraction*
Mechanisms of Heart Failure Causes
Indirect Myocardial Dysfunction (Not Cardiomyopathy)
Dilated <40% Impairment of contractility (systolic dysfunction)
Idiopathic; alcohol; peripartum; genetic; myocarditis; hemochromatosis; chronic anemia; doxorubicin (Adriamycin); sarcoidosis
Ischemic heart disease; valvular heart disease; hypertensive heart disease; congenital heart disease
Cardiomyopathy and Indirect Myocardial Dysfunction: Functional Patterns and Causes

Hypertrophic 50-80% Impairment of compliance (diastolic dysfunction)
Genetic; Friedreich ataxia; storage diseases; infants of diabetic mothers
Hypertensive heart disease; aortic stenosis
Restrictive 45-90% Impairment of compliance (diastolic dysfunction)
Idiopathic; amyloidosis; radiation-induced fibrosis
Pericardial constriction
Cardiomyopathy and Indirect Myocardial Dysfunction: Functional Patterns and Causes

Cardiac Infections
Viruses
Chlamydia
Rickettsia
Bacteria
Fungi
Protozoa
Toxins
Alcohol
Cobalt
Catecholamines
Carbon monoxide
Lithium
Hydrocarbons
Arsenic
Cyclophosphamide
Doxorubicin (Adriamycin) and daunorubicin
Conditions Associated with Heart Muscle Diseases

Metabolic
Hyperthroidism
Hypothyroidism
Hyperkalemia
Hypokalemia
Nutritional deficiency (protein, thiamine, other avitaminoses)
Hemochromatosis
Neuromuscular Disease
Friedreich ataxia
Muscular dystrophy
Congenital atrophies
Storage Disorders and Other Depositions
Hunter-Hurler syndrome
Glycogen storage disease
Fabry disease
Amyloidosis
Conditions Associated with Heart Muscle Diseases


include inflammatory diseases of the myocardium that result in injury to cardiac myocytes the inflammatory process is thecause rather than a response
to myocardial injury
Myocarditis

Major Causes of Myocarditis
Infections
Viruses (coxsackievirus, ECHO, influenza, HIV, CMV)
Chlamydiae (eg. C. psittaci)
Rickettsiae (eg. R. typhi, typhus fever)
Bacteria (eg. C. diphtheriae, N. meningitidis, Borrelia)
Fungi (eg. Candida)
Protozoa (eg. T. cruzi, toxoplasmosis)
Helminths (eg. Trichinosis)
Immune-mediated reactions
Postviral
Poststreptococcal (rheumatic fever)
SLE
Drug hypersensitivity
Transplant rejection
Unknown
Sarcoidosis
Giant cell myocarditis

Gross: normal or dilated;lesions diffuse or patchy
ventricular myocardium flabby & mottled by either pale foci or minute hemorrhagic lesions
mural thrombi present in any chamber
Micro: interstitial inflam infiltrate &focal necrosis of myocytes adjacent to the inflam cells
Morphology




PERICARDIAL DISEASE


Normal: 30-50 ml; thin, clear, straw-colored fluid in the pericardial sac
parietal pericardium undergoes distention by
- fluid: pericardial effusion- blood: hemopericardium- pus: purulent pericarditis
Pericardial Effusion & Hemopericardium

Types of Pericardial effusion Serous: most common form
- Causes: CHF, hypoproteinemia Serosanguinous: usually the result
of blunt chest trauma (eg. CPR) Chylous: lymphatic obstruction

Hemopericardium: accum of pure,often clooted blood in the pericardium w/o an inflam component
Cause: traumatic perforation myocardial rupture after a transmural
MI rupture of the intrapericardial aorta, hge from an abscess or tumor
metastasis

PericarditisInfectious Agents
Viruses
Pyogenic bacteria
Tuberculosis
Fungi
Other parasites
Presumably Immunologically Mediated
Rheumatic fever
Systemic lupus erythematosus
Scleroderma
Postcardiotomy
Postmyocardial infarction (Dressler) syndrome
Drug hypersensitivity reaction
Miscellaneous
Myocardial infarction
Uremia
Following cardiac surgery
Neoplasia
Trauma
Radiation

usually secondary to a variety of cardiac diseases, thoracic or systemic dso, metastases from neoplasms arising in remote site or a surgical procedure in the heart most often of viral origin
Pericarditis

Serous pericarditis: RF, SLE,scleroderma, tumors & uremia
Morph: inflam reaction in the epicardial &pericardial surfaces w/
scant numbers of PMNs, lymphocytes, & macrophages
Acute Pericarditis

Fibrinous / Serofibrinous pericarditis: - most frequent type of pericarditis
composed of serous fluid mixed w/ a fibrinous exudate
Causes: AMI, Dressler syndrome, uremia, chest radiation, RF,
SLE,trauma Clinical: pericardial friction rub

Morph:
Fibrinous: surface is dry w/ a fine granular roughening
Serofibrinous: increased inflam process induces more & thicker fluid (yellow & cloudy) & often fibrin




Purulent or Suppurative pericarditis: denotes invasion of the pericardial
space by infective organisms Routes:
a) direct extension fr neighboring inflam
b) seeding from the blood c) lymphatic extension d) direct extension during cardiotomy

Purulent pericarditis: Exudate ranges from a thin to
creamy pus of up to 400 to 500ml- serosal surfaces are
reddened, granular, & coated w/ the exudate
Micro: acute inflam reaction- organization is the usual
outcome


Hemorrhagic pericarditis: composed of blood mixed w/ a fibrinous
or suppurative effusion most commonly caused by malignant
neoplastic involvement of the pericardial space
Other causes: bacterial infections, patients w/ an underlying bleeding diathesis, TB, cardiac surgery
Caseous pericarditis


Healing of acute lesions resolution or pericardial fibrosis ranging from a thick, pearly, nonadherent epicardial plaque (soldier’s plaque), to thin delicate adhesions to massive adhesionsAdhesive mediastinopericarditis:
pericardial sac is obliterated & the parietal layer is tethered to mediastinal tissue
Chronic or Healed Pericarditis

Constrictive pericarditis: marked by thick, dense, fibrousobliteration, often w/ calcification of the pericardial sac that encases the heart, limiting diastolic expansion & restricting cardiac output
- most common cause: TB
Chronic or Healed Pericarditis

Adhesive Mediastinopericarditis - follow a suppurative or caseous pericarditis, previous cardiac surgery, or irradiation to the mediastinum. - pericardial sac is obliterated, and adherence of the external aspect
of the parietal layer to surr structures produces a great strain on cardiac function.

CARDIAC NEOPLASMS

CARDIAC NEOPLASMS
Rare
Metastatic tumors: 5%
Benign tumors: 80-90% of primary tumors of the heart

Myxoma
Most common primary cardiac tumor in adults
90 % located in the left atrium Complication :
ball-valve obstruction embolization

Myxoma Gross: almost always single
- favored site of origin: region ofthe fossa ovalis in the atrial septum
- Size: small to large (up to 10 cm.)- sessile or pedunculated masses that vary from globular hard masses mottled w/ hge to soft, translucent, papillary,or villous lesions having a gelatinous appearance


Micro: stellate or globular myxoma (“lepidic”) cells, endothelial cells, smooth muscle cells and undifferentiated cells embedded within an abundant acid mucopolysaccharide ground substance & covered on the surface by endothelium Carney’s syndrome: autosomal dominant
transmission, multiple cardiac & extracardiac myxomas, spotty pigmentation, & endocrine overactivity


Rhabdomyoma
Most frequent primary cardiac tumor in children
Most arise from ventricular chamber
cause valvular or outflow tract obstruction

Rhabdomyoma Gross: small, gray-white myocardial
masses up to several cm located on either the left or the right side of the heart & protruding into the ventricular chambers



Micro: large, rounded, or polygonal cells containing numerous glycogen-laden vacuoles separated by strands of
cytoplasm running from the plasma membrane to the more or less centrally located nucleus (spider cells)



Metastases to the heart

Have a nice day!