Electronic Behavior of Atoms. LO Describe how light is created. Explain how wavelengths of light...
-
Upload
melissa-marilyn-stone -
Category
Documents
-
view
223 -
download
0
Transcript of Electronic Behavior of Atoms. LO Describe how light is created. Explain how wavelengths of light...

Chapter 1 Section 5
Electronic Behavior of Atoms

January 27, 2012HW: 1.4 CTG p. 43 #1-11 (Due Wed)
LODescribe how light is created.Explain how wavelengths of light
relate to energy levels in the atom.
SCView the spectrum of hydrogen.Interpret changes in electron
energies in the hydrogen atom to develop an explanation for where the colored light in the hydrogen spectrum comes from.
Use Bohr’s model of the atom to predict parts of the hydrogen-atom spectrum.
Compare the wavelengths, energies, and frequencies of light of different colors.
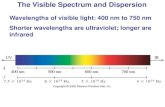
Identify regions in the electromagnetic spectrum.
Explain the photoelectric effect.
Do NowGrade 1.4 QuizCopy LO and SCWDYS, WDYT p. 35
AgendaWDYS, WDYTInvestigationChem TalkSummary

Investigate
Read through and complete numbers 1 and 2You MUST draw diagrams with what you see that include the colors


Investigation
Complete numbers 3 and 4 on pg. 36Be ready to discuss as a class!

Investigation
Complete numbers 5-9, reading through each of the explanations and answering the questions.Pay close attention to the diagram on p. 38

Date:HW: CTG p. 43 #1-11
LODescribe how light is created.Explain how wavelengths of light
relate to energy levels in the atom.
SCView the spectrum of hydrogen.Interpret changes in electron
energies in the hydrogen atom to develop an explanation for where the colored light in the hydrogen spectrum comes from.
Use Bohr’s model of the atom to predict parts of the hydrogen-atom spectrum.
Compare the wavelengths, energies, and frequencies of light of different colors.
Identify regions in the electromagnetic spectrum.
Explain the photoelectric effect.
Do NowCopy LO and SCWDYS, WDYT p. 35
AgendaWDYS, WDYTInvestigationChem TalkSummary

Chem Talk
Skim through the chem talk on p. 39Do not take notes!

http://player.discoveryeducation.com/index.cfm?guidAssetId=0D2CCF17-8C4E-4101-A1FF-D11305F6878C
Watch first 5 minutes

Chem Notes
Who was Niels Bohr?
What did he discover?

Chem Notes
Who was Niels Bohr?
What did he discover?
A Danish Physicist
He thought that electrons took a circular orbit around an atom and that they jump between energy levels while taking in energy and forming light


From Bohr’s Model

Chem Notes
What is light
What is frequency?

Chem Notes
What is light
What is frequency?
Photons, or packets of energy, that travel in waves.Come from energy level transfers

Chem Notes
What is light
What is frequency?
Photons, or packets of energy, that travel in waves.Come from energy level transfers
The number of cycles per second, measured in Hz usually

Chem Notes
What is wavelength
What is the speed of light?

Chem Notes
What is wavelength
What is the speed of light?
The distance from crest to crest in a wave.

Chem Notes
What is wavelength
What is the speed of light?
The distance from crest to crest in a wave.
3x108 m/s, and it is constant!

Wave Diagram

Chem Notes
How do we calculate frequency using wavelength?

Chem Notes
How are wavelength and energy related?

Chem Notes
How are wavelength and energy related?
They are inversely related!
As E increases, the wavelength gets smaller!

Chem Notes
What are photons?
How can we calculate frequency from energy?

Chem Notes
What are photons?
How can we calculate frequency from energy?
Fixed packets of energy with a specific wavelength.

Chem Notes
What are photons?
How can we calculate frequency from energy?
Fixed packets of energy with a specific wavelength.
Where h is plancks constant: 6.63x10-
34 Js

Chem Notes
What is the photoelectric effect?

Chem Notes
What is the photoelectric effect?
When light collides with an atom, it interacts with an electron, causing it to be thrown off of the atom due to the fixed amount of energy of the photon.

Chem Notes
What were the weaknesses of the Bohr Model?

Chem Notes
What were the weaknesses of the Bohr Model?
1. It could only account for the light transitions of the Hydrogen atom
2. Could not explain why some transitions were allowed while others were not.

Practice Problem
What is the frequency of light that has a wavelength of 432.4nm?

Practice Problem
Using Planck’s constant, what is the energy of red light?

Practice ProblemIn hydrogen the energy change of an electron jumping from E3 to E2 is 3.03 x 10-19 J
What is the frequency?
What is the wavelength? (and therefore color)

Chem Talk
Complete the checking up questions.

Summary
Essential Questions, p. 42--How do you know?--Why do you believe--Why should you care
LO and SC reflection