Electron Arrangement Another link for review S/28Elec-config.pdf 1.
-
Upload
roger-holmes -
Category
Documents
-
view
216 -
download
1
Transcript of Electron Arrangement Another link for review S/28Elec-config.pdf 1.

1
Electron Arrangement
Another link for reviewhttp://www.mrcoulter.com/LECTURES/28Elec-config.pdf

2
Quantum Numbers
• Quantum numbers specify the properties of atomic orbitals and the properties of electrons in orbitals. This is also known as probable location of electrons about the nucleus of an atom.

3
Principal Quantum Number, n
Indicates the main energy level occupied by an electron(n=1,2,3,…7)
http://library.thinkquest.org/C006669/

4
Angular momentum quantum number has 4 basic shapes
• The values and shapes are • l = 0 s shape• l = 1 p• l = 2 d• l = 3 f

5
Angular Momentum Quantum Number, l
• s shape
sparknotes.com

6
p shape

7
d shaped orbitals
Courtesy of bluelight.ru

8
f orbitals
f orbitals are very complex and you will not be responsible for drawing them. The site below has more informtion.
http://www.d.umn.edu/~pkiprof/ChemWebV2/AOs/ao4.html

9
Spin quantum number, s, has only two possible values
• Has only two possible values: +1/2, -1/2.
• See table 2 on page 110 to get a better idea of the number of orbitals and electrons per energy level.

10
Electron Configurations
I. Rules Governing Electron ConfigurationsII. Orbital NotationIII. Electron-Configuration Nottion

11
Rules Governing Electron Configurations
I. Aufbrau Principle – an electron occupies the lowest-energy orbital that can receive it.
Source: https://chemistry.twu.edu/tutorial/AufbauSum.html
This is really a thought process in which we think about building up an atom from the one that preceeds it in atomic number, by adding a proton and neutrons to the nucleus and one electron to the appropriate atomic orbital.

12
II. Pauli Exclusion Principle – no two electrons in the same atom can have the same set of four quantum numbers.
Rules Governing Electron Configurations, cont.
The box at left shows the different spin states of two electrons in the same orbital. This drawing is the electron configuration of a helium atom’s 1 s orbital.

13
Rules Governing Electron Configurations, cont.
III. Hund’s Rule – orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron, and all electrons in singly occupied orbitals must have the same spin state.
en.citizendium.org

14Source: chem.wisc.edu
Periodic Table: Orbitals

15
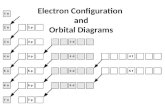
Orbital Notation
Source: naturalphilosophers.org
Pair of electrons in an orbital
Unpaired electron
Orbital name
What element is this showing?Fluorine

16
Sample problems
Write the electron configuration notation for Silicon and Scandium (use the Aufbrau diagram)Si has 14 electrons2 in 1s 2 in 2s6 in 2p 2 in 3s 2 in 3p written as:1s2 2s2 2p6 3s2 3p2

17
Scandium Electron Configuration Notation
Sc has 21 electrons
1s2 2s2 2p6 3s2 3p6 4s2 3d1

18
Scandium Notations
Electron Configuration Notation1s2 2s2 2p6 3s2 3p6 4s2 3d1
Orbital Notation__ __ __ __ __ __ __ __ __ __ __ __ __ __ ___
1s 2s 2p 3s 3p 4s 3d

19
Notations,cont.Noble gas notation – back up to the last noble gas prior to the element
and add orbitals:[Ar}4s23d1
Dot notation - shows only the outer shell electrons for an element’s atom (maximum number of e’s in an outer shell is 8) for Sc: 4s2 for F: 2s22p5 for Ne: 2s22p6
.. . . Sc : : F : : Ne : . . .
Other info: atoms are more stable when they have a half-filled or completely
filled outer shell

20
Remaining Ch5 asgns. From book
• 137/1-3; 149/1-5; 165/1-3• 166/1-15 (use periodic chart)• 166/17a-c,18a-c,19c-e,20,21a-c• 167/24-29,32,34• 167/35-40