Effects of Low-dose aspirin and gum diseases
-
Upload
drouganis -
Category
Health & Medicine
-
view
2.802 -
download
7
description
Transcript of Effects of Low-dose aspirin and gum diseases

Periodontal conditions in relation to
low-dose aspirin therapy
in ex- and non-smokers
by
Arthur Drouganis BDS, Grad Cert Dent
A thesis submitted for the degree ofMaster of Dental Surgery
(Periodontics)
The University of AdelaideDental School
November 1999

Dedication
This thesis is dedicated to my loving wife Helen, and
my children Vicky, Lambros and Margaret whose
support, enthusiasm and tolerance enabled me to
complete the work.

TABLE OF CONTENTS
Acknowledgments...........................................................................................................viii
Glossary of terms...............................................................................................................x
Summary...........................................................................................................................xi
Chapter 1 Introduction...................................................................................................1
Chapter 2 Review of the literature.................................................................................5
2.0 SUMMARY OF THE PRESENT UNDERSTANDING OF THE INFLAMMATORY RESPONSE.... .5
2.1 ENDOGENOUS MEDIATORS OF INFLAMMATION................................................................8
2.1.1 Histamine:.................................................................................................................8
2.1.2 Bradykinin:................................................................................................................9
2.1.3 Plasmin:....................................................................................................................9
2.1.4 Complement:..........................................................................................................10
2.1.5 Platelets:.................................................................................................................11
2.2 EICOSANOIDS....................................................................................................................12
2.2.1 General properties of eicosanoids..........................................................................13
2.3 ROLE OF EICOSANOIDS IN PERIODONTAL TISSUES.........................................................15
2.3.1 Biosynthesis of eicosanoids....................................................................................16
2.3.2 Arachidonic acid pathways: eicosanoid production.................................................17
2.3.3 Catabolism of the eicosanoids................................................................................24
2.4 THE ROLE OF CYTOKINES IN PERIODONTAL TISSUES....................................................25
2.5 CELLULAR EVENTS IN INFLAMMATION...........................................................................31
2.5.1 Macrophage phenotypes........................................................................................32
2.5.2 Alveolar bone resorption.........................................................................................33
2.6 NONSTEROIDAL ANTI-INFLAMMATORY DRUGS IN PERIODONTAL DISEASES................34
2.6.1 History of salicylates...............................................................................................34
2.6.2 Physio-chemical properties of aspirin and other salicylates ...................................35
2.6.3 Periodontal studies of the effects of NSAIDs over the last 20 years.......................39
2.7 PERIODONTAL STUDIES WITH ASPIRIN............................................................................52
2.7.1 The Waite study:.....................................................................................................52
2.7.2 The Feldman study.................................................................................................53
2.7.3 The Flemmig study.................................................................................................54
2.7.4 The Heasman study................................................................................................55
2.8 SMOKING AND PERIODONTAL DISEASES.........................................................................56
2.8.1 The periodontal effects of past smoking and smoking dose...................................57
i

2.9 PERIODONTAL MEASURES................................................................................................58
2.9.1 The experimental unit.............................................................................................59
2.9.2 Measurement of extent and severity of periodontal attachment loss......................59
2.10 NULL HYPOTHESES......................................................................................................61
Chapter 3 Materials and methods...............................................................................62
3.1. SAMPLE SELECTION.........................................................................................................62
3.2 QUESTIONNAIRE...............................................................................................................64
3.3 ORAL EXAMINATION........................................................................................................66
3.4 CLINICAL MEASUREMENTS..............................................................................................66
3.4.1 Plaque Index...........................................................................................................66
3.4.2 Calculus.................................................................................................................. 67
3.4.3 Bleeding index........................................................................................................67
3.4.3 Tooth mobility.........................................................................................................68
3.4.4 Furcation involvement.............................................................................................69
3.5 DETAILS OF THE STUDY...................................................................................................69
3.5.1 Periodontal attachment loss (PAL).........................................................................69
3.5.2 Periodontal Pocket Depths (PPD)...........................................................................70
3.5.3 Gingival Recession (GR)........................................................................................70
3.5.4 Examiner standardisation:......................................................................................70
3.5.5 Procedure...............................................................................................................70
3.6 STATISTICAL METHODOLOGY.........................................................................................71
Chapter 4 Results..........................................................................................................72
4.1 INTRA-EXAMINER ERROR................................................................................................72
4.2 PROFILE OF STUDY POPULATION:...................................................................................72
4.3 DEMOGRAPHICS...............................................................................................................73
4.3.1 Age categories of subjects......................................................................................73
4.3.2 Education status of the subjects.............................................................................74
4.3.2 Oral health behaviour..............................................................................................75
4.4 TOOTH LOSS.....................................................................................................................76
4.5 THE PERIODONTAL STATUS OF THE STUDY POPULATION..............................................76
4.6 ASSOCIATIONS OF ASPIRIN AND EX-SMOKING WITH VARIOUS MEASURES OF PAL.....78
4.6.1 The associations of aspirin and ex-smoking with mean PAL..................................78
4.6.2 The associations of aspirin and ex-smoking on the extent and severity of PAL......79
4.6.3 Associations of aspirin and ex-smoking with the most severe site of PAL
(MSS-PAL)..............................................................................................................81
4.6.4 Associations of aspirin and ex-smoking with the extreme worst site of PAL
(EWS-PAL).............................................................................................................82
ii

4.7 THE ASSOCIATIONS OF VARIOUS CLINICAL PARAMETERS ON MEAN PAL...................84
4.7.1 Site and tooth variations in recession and pocket depth by mean PAL...................85
4.7.2 Socio-economic factors and periodontal attachment..............................................86
Chapter 5 Discussion...................................................................................................115
5.1 PROFILE OF THE STUDY POPULATION...........................................................................115
5.1.1 Age groupings.......................................................................................................117
5.2 QUESTIONNAIRE.............................................................................................................119
5.2.1 Socio-economic status..........................................................................................120
5.3 PERIODONTAL ATTACHMENT LOSS..............................................................................121
5.3.1 Age associations with PAL...................................................................................121
5.4 MEASURING PAL...........................................................................................................122
5.4.1 Case definitions....................................................................................................122
5.5 OUTCOMES OF ASPIRIN AND PAST SMOKING ON PAL.................................................125
5.5.1 Mean PAL.............................................................................................................125
5.5.2 MSS-PAL..............................................................................................................127
5.5.3 EWS-PAL..............................................................................................................127
5.5.4 Plaque................................................................................................................... 128
5.5.5 Gingival bleeding..................................................................................................129
5.6 COMPARISONS WITH OTHER ASPIRIN STUDIES.............................................................130
5.7 SMOKING AND PAL........................................................................................................133
5.8 PREVALENCE OF PERIODONTAL ATTACHMENT LOSS...................................................133
5.9 FUTURE RECOMMENDATIONS........................................................................................134
Conclusions....................................................................................................................135
Appendix A....................................................................................................................137
Appendix B....................................................................................................................138
Appendix C....................................................................................................................139
Appendix D....................................................................................................................140
References......................................................................................................................144
iii

TABLES
Table 2.1 Interactions of plaque bacteria and their products in inflammation and immunity.........................................................................................................7
Table 2.2 Composition of eicosanoids..........................................................................12
Table 2.3 Cell sources and actions of prostanoids........................................................15
Table 2.4 Major tissue destructive mediators in periodontitis......................................26
Table 2.5 Neutrophil components and function ...........................................................31
Table 2.6 The types of NSAIDs (and their classification) used in periodontal studies 39
Table 2.7 Periodontal effects of NSAIDs in human studies..........................................40
Table 3.1 Inclusion and exclusion criteria.....................................................................63
Table 3.2 Aims of questionnaire...................................................................................65
Table 3.3 Plaque index..................................................................................................67
Table3.4 Modified Sulcus Bleeding Index (mSBI)......................................................67
Table 3.5 Tooth mobility index.....................................................................................68
Table 3.6 Furcation index..............................................................................................69
Table 4.1 Intra examiner reliability test using kappa statistics.....................................87
Table 4.2 The number and percentage distribution of subjects participating in the study by group........................................................................................................87
Table 4.3 Distribution of age by group.........................................................................87
Table 4.4 A Scheffés analysis of homogeneity between two groups at a time for mean age differences...............................................................................................88
Table 4.5 Scheffésa, analysis for homogeneity between subsets...................................88
Table 4.6 Demographics on pension status with group specific characteristics...........89
Table 4.7 Pension status in relation to denture use.......................................................89
Table 4.8 Demographic data on schooling of all subjects with group specific characteristics................................................................................................89
Table 4.9 A self-evaluation of English language skill..................................................90
Table 4.10 Socio-economic factors and dental behaviours.............................................90
Table 4.11 Population and percentage distribution of subjects since their last dental visit. The time range was from less than one year to never visiting the dentist............................................................................................................91
Table 4.12 Missing teeth by age and group.....................................................................92
Table 4.13 Missing teeth and smoking history...............................................................92
Table 4.14 The mean plaque index per group.................................................................92
Table 4.15a Profile of aspirin use and subject numbers....................................................93
Table 4.15b The association of age and past smoking on mean plaque scores with tests of significance....................................................................................................93
iv

Table 4.16 Distribution of mean percentage of teeth with calculus by age, aspirin and ex-smoking....................................................................................................94
Table 4.17 The association of age with mean percentage of calculus between groups. .95
Table 4.18 The association of low-dose aspirin and ex-smoking with the mean percentages of mobile teeth...........................................................................95
Table 4.19 The correlation of low-dose aspirin and past smoking with mean PAL.......96
Table 4.20 The association of aspirin dosage with mean PAL.......................................97
Table 4.21 The association of aspirin duration with mean PAL.....................................97
Table 4.22a The association of past smoking dosage and duration with mean PAL........97
Table 4.22b The correlation of the number of cigarettes smoked and duration of smoking with mean PAL with t-test of significance....................................................98
Table 4.23 Univariate analysis of variance in mean PAL at 2, 4 5 and 7mm.. .99
Table 4.24 Univariate analysis of variance on mean % PAL at 2, 4, 5 & 7mm.100
Table 4.25 The magnitude of the association of aspirin and smoking history with severity and extent of PAL at 2, 4, 5 & 7 mm PAL using the general linear model (2-way ANOVA) of analysis.................................................101
Table 4.26 The correlation of aspirin and past smoking history with MSS-PAL.........102
Table 4.27 The age class distribution of males 50+ years in metropolitan Adelaide in 1996 from census statistics and their appropriate frequency distribution.. .103
Table 4.28 The proportional weights given to each group using the percentage frequency of each class interval from census statistics for metropolitan Adelaide......................................................................................................103
Table 4.29 Descriptive statistics of EWS-PAL.............................................................103
Table 4.30 The association of aspirin and past smoking history with EWS-PAL using weighted data...............................................................................................104
Table 4.31 The ratio of aspirin to smoking on various measurements of PAL.............105
Table 4.32 Associations of plaque and age with mean PAL with tests of significance......................................................................................................................105
Table 4.33 Associations of calculus and age with mean PAL with tests of significance......................................................................................................................105
Table 4.34 Associations of gingival bleeding and age with mean PAL with tests of significance..................................................................................................106
Table 4.35 Socio-economic factors, oral hygiene patterns and mean PAL (mm).........106
Table 4.36 The statistical power values for most ANOVA analyses............................107
Table 4.37 Relative percentage of subjects with medical conditions per group...........107
Table 4.38 Outcome of age, ex-smoking and aspirin with various indices of PAL......107
v

Figures
Figure 2.1 Products and pathways of cyclo-oxygenase..................................................14
Figure 2.2 The chemical structures of PGE2 and TxB2...................................................20
Figure 2.3 Structure of aspirin........................................................................................36
Figure 2.4 Effects of aspirin on cyclo-oxygenases.........................................................38
Figure 3.1 A copy of an advertisement placed in local press media to recruit subjects.62
Figure 4.1 The mean percentage of sites with gingival bleeding (modified bleeding index)...........................................................................................................108
Figure 4.2 The mean percentage of teeth with calculus...............................................108
Figure 4.3 Cumulative distribution of MSS-PAL representing the worst score (site) per tooth per subject, averaged over all subjects...............................................109
Figure 4.4 Diagrammatic representation of PAL according to smoking and aspirin taking history, showing mean PAL, MSS-PAL and EWS-PAL.................110
Figure 4.5 Cumulative distribution of EWS-PAL. Data were weighted using age class statistics for metropolitan Adelaide population..........................................111
Figure 4.6 Variations of recession and pocket depths by tooth- and jaw type for the whole study population...............................................................................112
Figure 4.7 Variation of recession and pocket depths by tooth- and jaw type in the AXS group...........................................................................................................112
Figure 4.8 Variation of recession and pocket depths by tooth- and jaw type in the NAXS group................................................................................................113
Figure 4.9 Variation of recession and pocket depths by tooth- and jaw type in the ANS group...........................................................................................................113
Figure 4.10 Variation of recession and pocket depths by tooth- and jaw type in the NANS group................................................................................................114
vi

Signed Statement
This research report is submitted in partial fulfillment of the requirements of the Degree
of Master of Dental Surgery (Periodontics) in the University of Adelaide.
The thesis contains no material which has been accepted for the award of any other
degree or diploma in any University and that, to the best of my knowledge and belief, the
thesis contains no other material previously published or written by another person,
except where due reference is made in the text of the thesis.
I give consent to this copy of my thesis, when deposited in the University Library, being
made available for photocopying and loan if accepted for the award of the degree.
……………………………………..
Arthur Drouganis.
November 1999
vii

Acknowledgments
I wish to take this opportunity to thank those people who have assisted me in completing
my candidature. I am particularly grateful to many people but utmost to my wife, and
family for their patience and understanding throughout this challenging course.
I am truly indebted to two individuals. Robert Hirsch my supervisor, a true researcher,
for his kindness, knowledge and in particular his insight and wisdom who lent me
unconditional support, tempered at times, by considerable forbearance. To Bryon
Kardachi, for his clinical knowledge, expert guidance and for his enthusiasm. The
knowledge I have gained from both of them is, and will be invaluable.
My thanks go to the Colgate Australian Clinical Dental Research Centre for the use of its
state-of-the-art facilities and I am especially grateful to Kerrie Ryan and Jane Burns who
gave excellent support and assistance. To Colgate Australia for their generosity in
supplying the Oral Care Kits which were given to each participant in the study. A
special thank you to Professor Felix Bochner, Department of Clinical and Experimental
Pharmacology, Division of Health Sciences University of Adelaide for his initial
guidance.
I am deeply grateful to Knute Carter for his meticulous statistical analyses of the data.
To Jane Carter for her enthusiasm and ideas on the study
These people have inspired and encouraged me to ask questions, to learn to reason and
think independently. I truly believe I have been educated.
Thank you.
viii

" Do not be rash to make friends; but, when once they are made, do not drop them"
DIOGENES (412-332 B.C.)
A Greek philosopher
I can quite honestly say that I have made life time friends.
ix

Glossary of terms
ANS Aspirin Never Smoked group
AXS Aspirin eX-Smoker group
COX Cyclo-oxygenase, an enzyme that produces the prostanoid and thromboxane mediators of inflammation
Cytokines Polypeptide mediators released by cells involved in inflammation healing and homeostasis
EWS-PAL The extreme worst site of PAL per subject, then averaged across each group
Extent The proportion of tooth sites of an individual with PAL exceeding 1mm and often measured at various threshold values
GCF Gingival Crevicular Fluid
IgG Immunoglobulin-G
IL-1 Interleukin-1 an inflammatory cytokine involved in inflammation, immunity, tissue breakdown and homeostasis
IL-6 Interleukin-6 an inflammatory cytokine involved in inflammation, immunity, tissue breakdown and homeostasis
Low-dose aspirin 300mg per day
LPS Lipopolysaccharide
Mean PAL The average PAL of all sites per subject, then averaged across each group
MSS-PAL The most severe site of PAL per tooth per person then averaged across each group
NANS No Aspirin Never smoked group
NAXS No Aspirin eX-Smoker group
NSAIDs Non-steroidal anti-inflammatory drugs
PAL Periodontal attachment loss
PGE2 Prostaglandin-E2. A primary cyclo-oxygenase mediator of inflammation
Prevalence The proportion of group who have PAL (ie cases)
Severity The degree of PAL averaged per affected tooth sites
TNF- A proinflammatory cytokine with synergistic effects with other cytokines
x

Summary
In the 1970's, Vane proposed that the anti-inflammatory effects of aspirin and aspirin-like
drugs (non-steroidal anti-inflammatory drugs, NSAIDs) were due to inhibition of the enzyme
cyclo-oxygenase, which stops the production of prostanoids (prostaglandins and
thromboxanes). By the early 1980's, high doses of aspirin and other NSAIDs were shown to
significantly reduce gingivitis, periodontal attachment loss and alveolar bone loss in humans.
However, long-term use of these agents in periodontal therapy was not advocated, due to their
side effects and the inconsistent findings between studies. Often test and control groups were
not from the same sample population, results were based on concurrent use of other NSAIDs,
dosages and duration varied between groups, and there was no control for smoking effects.
Research in the 1990's showed that periodontitis is a multifactorial disease, being dependent
on genetic and environmental influences, which modify the host response to the microbial
challenge. One of the primary environmental risk factors for periodontitis is cigarette
smoking. Ex-smokers lie between non-smokers and current smokers with regard to the
severity and extent of periodontal attachment loss and alveolar bone loss; people who quit
smoking respond to periodontal therapy similarly to non-smokers.
There is no information in the literature about the periodontal effects of low-dose aspirin on
the periodontium in either non-smokers or ex-smokers. The aim of this study was to assess the
periodontal status of a self-selected sample of men (aged 50 and above), residing in
metropolitan Adelaide, South Australia, with respect to aspirin intake and smoking history.
Subjects were targeted by advertisements placed in the local press.
Demographic data were collected from information obtained from a self-administered
questionnaire and periodontal health was assessed by a periodontal examination carried out by
one operator, blind to each subject’s aspirin and smoking history. Measurements of pocket
depths and gingival recession were made at six sites of all teeth present and were used to
xi

compute periodontal attachment loss (PAL) for all subjects. Other parameters recorded were
plaque and calculus accumulation, gingival and bleeding indices and tooth mobility.
Periodontal assessments were carried out in 392 men, aged 50-85 years. Significant age
effects were found on PAL but these were of small magnitude in comparison to the significant
influences that aspirin and ex-smoking had on PAL. The subjects were divided amongst four
sub-groups:
aspirin never smoked (ANS),
aspirin ex-smokers (AXS).
no aspirin never smoked (NANS)
no aspirin ex-smokers (NAXS).
The extent and severity of PAL was evaluated against a background of age, ethnicity, socio-
economic and dentition status. The study population comprised low, middle and higher
educational levels and there were no significant distribution differences between the groups.
The study population comprised a much higher group of educated subjects when compared to
the general population of Adelaide. Higher educated subjects with good English skills
brushed more frequently and had a more recent scale and clean than the lower educated
groups. A measure of subjects’ economic level was their pension status; pensioners
representing low income. Approximately 58.9% of subjects were pensioners; there were no
significant differences in mean PAL between pensioners and non-pensioners.
In order to correlate the effects of aspirin and smoking habits on advanced PAL,
three measures of PAL were used; mean PAL, the most severe site of PAL (MSS-
PAL) and the extreme worst site of PAL (EWS-PAL). Mean PAL was the overall
mean PAL of all sites per tooth/per subject/per group. MSS-PAL was the most
severe site of PAL of the six sites per tooth/subject. This method associated the
effects of aspirin and ex smoking on advanced PAL by reducing the overwhelming
xii

effects of sites with low PAL. EWS-PAL was the extreme worst site of PAL/mouth.
The results were as follows:
Mean PAL mm se MSS-PAL mm se EWS-PAL mm se
ANS 2.5 0.01 3.7 0.13 6.2 0.22
AXS 2.8 0.09 4.1 0.11 7.0 0.18
NANS 2.7 0.08 4.0 0.10 6.8 0.17
NAXS 3.1 0.08 4.4 0.10 7.5 0.17
Prevalence was measured using different threshold levels of PAL. Significant positive effects
of aspirin for the extent of PAL were found for all threshold levels. At thresholds of 2mm
PAL, the prevalence of PAL was approximately 94%. At a moderate threshold of 4mm PAL,
28.7% of subjects exhibited PAL 4mm with a mean severity score of 4.6 0.03mm (se),
indicating that the percentage of subjects with advanced PAL was low particularly at higher
thresholds. Controlling for age, ANOVA analysis showed that the prevalence rate of PAL
was significantly lower in aspirin takers when compared to non-aspirin takers and these
effects were independent of smoking history. In addition, ex-smokers had significantly more
PAL compared to non-smokers and this effect was independent of aspirin history. The
prevalence of advanced PAL in subjects (using 7mm PAL as a threshold) was found to be
2.6% with a mean PAL of 7.7 0.05mm (se).
Epidemiological studies (including this one) attribute all PAL to the effects of destructive
periodontal diseases. No account is given to other causes of PAL such as continuous tooth
eruption, alveolar dehiscence, cervical enamel projections, cracked or split roots and
retrograde periodontitis. Taking these factors into account, the true prevalence of advanced
PAL due to periodontitis within the community must be lower than the estimated rate of 10-
15%.
xiii

My findings suggest that men aged 50 and above may benefit from taking low-doses of
aspirin daily in order to reduce their risk of PAL. With the reduced severity and extent
of PAL in ex-smokers taking aspirin, it is tempting to speculate that subjects with
periodontitis may benefit significantly by taking low-dose aspirin to reduce their
periodontal and cardiovascular risks, irrespective of their smoking history. Further
research should aim to establish whether patients with periodontitis would benefit from
taking low-dose aspirin as an adjunct to periodontal therapy and whether low-dose
aspirin modulates the effects of periodontitis in females and current smokers.
xiv

Chapter 1 Introduction
Destructive periodontal diseases are multifactorial in origin; the interplay between lifestyle
factors, the social environment and the dental biofilm determine an individual’s susceptibility
(Clarke and Hirsch 1995). Inflammatory as well as immunological responses are activated by
the many components of dental biofilm which constitutes the microbial challenge to the host
(Miyasaki 1996; Wilson and Kornman 1996; Darveau et al. 1997). The vascular and cellular
responses occurring in inflammation are controlled by the release of endogenous
inflammatory mediators (Page and Schroeder 1976; Page 1991; Genco et al. 1994;
Offenbacher 1996). There is an extensive list of endogenous inflammatory mediators known
to be involved in the regulation of the inflammatory response. In periodontal tissues, these
mediators are the link between health, tissue damage, inflammation and immunity (Page and
Schroeder 1976; Offenbacher et al. 1990; Page 1991; Offenbacher et al. 1993a; Offenbacher
et al. 1993b; Genco et al. 1994; Wilson and Kornman 1996).
One of the first and major pathways of tissue destruction in inflammatory periodontal
diseases is the synthesis and release of eicosanoids. Eicosanoids are formed from
membrane polyunsaturated fatty acids (mainly arachidonic acid), which include the
prostaglandins, prostacyclins, thromboxane A2 and the leukotrienes (Rang et al. 1996).
Eicosanoids are not found preformed in cells like histamine, but are generated de novo
from cell plasma membrane phospholipids when tissues are damaged (Salmon and Higgs
1987; Davies and MacIntyre 1992). They control many physiological and pathological
processes and are the most important mediators and modulators of the immuno-
inflammatory pathways (Rang et al. 1996). In response to microbial virulence factors,
damaged gingival tissues produce phospholipids, which become the substrate for
phospholipase. This enzyme synthesizes and releases free arachidonic acid (Howell and
1

Williams 1993) which may be synthesized into either prostanoids or leukotriene
products. These are associated with platelet aggregation, vasodilatation, chemotaxis of
neutrophils, increased vascular permeability and alveolar bone resorption. Prostanoids
are produced from arachidonic acid by cyclo-oxygenase (COX) which occurs in
neutrophils, macrophages, mast cells, fibroblast, lymphocytes keratinocytes, osteoblasts
and platelets (Howell and Williams 1993; Offenbacher 1996; Wiebe et al. 1996).
Leukotrienes are products produced by lipoxygenase and are restricted to neutrophils,
eosinophils, monocytes/macrophages and mast cells (Salmon and Higgs 1987).
The predominant prostanoid product in immuno-inflammatory responses in periodontal
diseases is prostaglandin E2 (PGE2) (Howell and Williams 1993; Offenbacher et al.
1993b). PGE2 is considered to be one of the key components in the pathogenesis of
periodontitis (Page 1991). A large portion of periodontal pathology is attributed to PGE2,
especially in association with other proinflammatory cytokines (IL-, IL-6, IL-8 and
TNF-) (Alexander and Damoulis 1994; Mathur and Michalowicz 1997; Soskolne 1997;
Ellis 1998; Okada 1998). The principal sources of PGE2 in periodontal tissues are
macrophages, monocytes and fibroblasts (Fu et al. 1990).
In the 1970's, Vane (1971) advanced the hypothesis that the anti-inflammatory effects of
aspirin-like drugs lay in their ability to inhibit prostanoid synthesis (prostaglandins and
thromboxanes). Among its actions, aspirin irreversibly inhibits COX which exists in two
forms (Smith 1992; Meade et al. 1993; Vane 1994; Sharma and Sharma 1997; Dubois et
al. 1998):
COX-1 is found in all cells as a constitutive enzyme, which produces the prostanoids
that regulate normal homeostasis (e.g. regulating vascular responses and coordinating
the actions of circulating hormones).
2

COX-2 is the inflammatory cyclo-oxygenase that is induced only by inflammatory
stimuli, releasing prostaglandin E2 (PGE2). Platelets do not contain COX-2.
In the early 1980's, the effects of aspirin and other nonsteroidal anti-inflammatory drugs
(NSAIDs) on periodontal attachment loss started to be investigated in humans. People
taking high doses of aspirin or other NSAIDs were found to have significantly lower
plaque scores, less gingival inflammation, less attachment and bone loss than the controls
(Waite et al. 1981; Feldman et al. 1983; Williams et al. 1989; Jeffcoat et al. 1991;
Heasman et al. 1993b; Howell 1993; Offenbacher et al. 1993b; Flemmig et al. 1996;
Offenbacher 1996). NSAIDs were considered to have modified the host responses by
inhibiting PGE2 production and therefore reducing bone and periodontal attachment loss.
Unfortunately many factors in these studies were not controlled, such as age, sex, poor
comparison or control groups (sampling frame error), smoking and systemic disease.
Furthermore, most human studies were retrospective and often relied on the subjects'
recollection of dosage and duration, and more than one NSAID was often used
concurrently. The majority of aspirin studies used patients suffering from rheumatoid
arthritis who were taking high daily doses (650mg->3gm/day). These confounding
factors made comparisons between studies difficult and resulted in conflicting outcomes
with respect to plaque indices, gingival indices, periodontal attachment loss and alveolar
bone loss.
Low-dose aspirin's ability to irreversibly inhibit cyclo-oxygenase over the whole lifetime
of platelets has made it a widely used anti-thrombogenic agent in middle-aged and
elderly populations to prevent coronary artery disease, stroke and peripheral vascular
diseases, with low gastro-intestinal side effects (Vane and O'Grady 1993; Underwood
1994; Lloyd and Bochner 1996; Diener 1998; Müller 1998). Low-dose aspirin has
3

decreased the incidence of heart attacks and stroke by up to 50% (Vane 1994). Low-
dose aspirin can inhibit thromboxane A2 production by platelets equipotently as can
doses > 300mg. In Australia, the maximum benefit/risk ratio dose used is 100-150mg of
aspirin per day (Lloyd and Bochner 1996).
Smoking is recognised as the most important cause of preventable death and disease in the
western world (MacGregor 1992) and there is a clear association between smoking and the
prevalence and severity of PAL (Bergström and Floderus-Myrhed 1983; Haber et al. 1993;
Bergström and Preber 1994; Zambon et al. 1996). The greater the exposure in terms of pack
years, the greater the amount of PAL and alveolar bone loss (Grossi et al. 1996; Grossi et al.
1997).
To-date, no studies have investigated the effects of long-term low-dose aspirin on PAL.
Since there is a large pool of people in the community taking low-dose aspirin daily for
many years, this study was undertaken to correlate PAL with aspirin and smoking
histories. In particular, the aim of this study was to gather descriptive epidemiological
data relating to the extent and severity of periodontal attachment loss in an adult male
population within metropolitan Adelaide specifically targeting men with and without a
history of long-term low-dose aspirin therapy, with or without a history of smoking.
Data from this study could also provide information relating to oral hygiene habits,
dental attendance, socio-economic factors, tooth loss and attachment loss patterns in an
elderly population.
4

Chapter 2 Review of the literature
2.0 Summary of the present understanding of the inflammatory response.
Periodontal diseases are mostly chronic infections characterised by a destructive
inflammatory process affecting the supporting tissues of the tooth, with subsequent
pocket formation and resorption of the alveolar bone (Offenbacher 1996). The intent of
this review is to place the current understanding of the regulatory mechanisms that
influence the inflammatory response in perspective, focussing on prostaglandins as
important elements of the inflammatory process and as major mediators of periodontal
attachment loss (PAL) and alveolar bone loss (Offenbacher 1996; Gemmell et al. 1997;
Page et al. 1997).
Inflammation is the normal response of the body to infection, tissue injury or insult; it is
rapid and provides a first line of defence. It is initially a nonspecific host response,
eliciting the same reaction irrespective of the nature of the insult. The insult may be
microbial, physical or chemical in nature, and all initiate a series of local processes to
neutralise, limit the spread and eradicate the insulting agent(s) (Lakhani et al. 1993;
Offenbacher 1996; Gemmell et al. 1997; Page et al. 1997). Inflammation is divided into
acute and chronic forms based on the duration of the response and the predominant
inflammatory cell type. Whether acute or chronic, the process may be modified by many
environmental and host factors; such as the pathogenicity and virulence of the microbial
challenge, nutritional status, host immune status, use of antibiotics, anti-inflammatory
drugs and / or surgical/non-surgical therapy (Lakhani et al. 1993; Miyasaki 1996;
Wilson and Kornman 1996; Page et al. 1997). These responses are characterised by
dilatation of the local blood vessels, increased permeability of capillaries, plasma
exudate, with the chemotactic accumulation of neutrophils, monocytes/macrophages,
5

eosinophils, basophils and mast cells to the site of injury or infection (Kay 1970;
Bienenstock et al. 1986; Faccioli et al. 1991; Page et al. 1997). The chemotactic factors
are both chemotactic and cell activating, leading to increased cell numbers and / or
affinity of adherence receptors on the surfaces of both endothelial and inflammatory cells
(Page 1991; Page et al. 1997). The expression of adhesion receptors enables the
migration of inflammatory cells from the circulation into the sites of injury (Page 1991;
Page et al. 1997), where they actively eliminate the noxious agent and participate with
resident tissue cells in wound healing and tissue remodelling (Miyasaki 1996; Wilson
and Kornman 1996; Page et al. 1997). In addition to the cellular response, plasma
constituents including complement and immunoglobulins are poured into the sites of
inflammation (medications are also transported to these sites by the plasma or
inflammatory exudate).
The host through the neutrophils and macrophages has the capacity to destroy all
biological structures (Williams et al. 1996). In the process of containing the microbial
challenge, host defences can cause bystander tissue destruction which can be more
offensive than the original insult (Page et al. 1997; Okada 1998). The damage is either
essential, such as the removal of collagen allowing room in the tissue for an
inflammatory cell infiltrate, or the damage may be bystander damage (accidental) in the
process on containing the microbial challenge. "Bystander damage" is a common feature
of chronic inflammatory diseases such as rheumatoid arthritis, tuberculosis, and
emphysema. Loss of periodontal attachment in periodontitis is caused by bystander
damage from the host response to the microbial plaque (Williams et al. 1996; Page et al.
1997).
6

Inflammatory reactions consist of two components, the inflammatory exudate (the
plasma component) and the cellular response. Both responses are activated by the many
constituents of dental plaque biofilm which constitutes the microbial challenge to the
host in periodontal diseases (Miyasaki 1996; Wilson and Kornman 1996; Darveau et al.
1997). Aerobic and anaerobic bacteria found in the gingival crevice or periodontal
pockets release a variety of products that can cause the onset of vascular changes, leading
to acute inflammation. These products include metabolic acids, extracellular enzymes,
volatile sulphur compounds, lipoteichoic acid and lipopolysaccharides.
Table 2.1 Interactions of plaque bacteria and their products in inflammation
and immunity
Any stimulus that damages host cells or other components will trigger inflammation, and the resulting inflammation helps activate an immuno-inflammatory response against foreign or antigenic material present. Conversely, humoral immune reactions will activate an inflammatory reaction at the site where the antibody binds to the antigen (Williams et al. 1996).
Bacterial products Effects
Whole bacteria activate complement activate neutrophils and macrophages are antigenic
Most peptides and proteins secreted by bacteria
chemotactic for neutrophils and macrophages
Enzymes
damage host cells degrade connective tissue matrix activate and degrade complement degrade antibodies are antigenic
Lipopolysaccharide (LPS)
activates complement damages some host cells activates neutrophils and macrophages are antigenic
Polysaccharide plaque matrix andBacterial capsule
polyclonal B-cell activator are antigenic
Other toxins, acids, reducing agents and metabolites
damage host cells are antigenic
7

Table 2.1 summarises the interactions of plaque products and their effects on inflammation
and immunity. These factors can directly or indirectly damage sulcular epithelium and
underlying connective tissue, disrupt microvasculature and initiate an inflammatory response
(Darveau et al. 1997). Some aspects of the inflammatory response are clearly distinct but the
precise role played by many of the mediators has not been completely clarified (Page and
Schroeder 1976; Page 1991; Genco et al. 1994; Offenbacher 1996).
2.1 Endogenous mediators of inflammation
The inflammatory exudate flowing from the gingival tissues into the gingival crevice or
periodontal pocket consists of blood components and host defence mediators which can
contain the microbial challenge, or they themselves act as a source of nutrients for the
microbes. The rate of gingival crevicular fluid flow generally reflects the severity of the
inflammation, the increased volume of inflamed tissue and the greater surface area of
pockets (Williams et al. 1996). The initial host response to the bacterial challenge is
characterised by the release of a number of vasoactive and antimicrobial factors:
2.1.1 Histamine:
This mediator of acute inflammation is present in mast cells. Histamine may be released
directly either by:
(a) bacterial mediators such as lipopolysaccharide and enzymes (trypsin like
or proteases) which activate the complement pathway (alternate pathway)
eventually releasing C3a and C5a or
(b) direct complement activation (C3a and C5a), or interleukin-1 and other
factors from endothelial cells, neutrophils and lymphocytes. In addition,
8

antibody-antigen complexes can activate complement (through the classic
pathway) releasing C3a and C5a.
These mediators activate the release of mast cell granules, which increase vascular
permeability (in capillaries and venules), and characteristically are the major mediator of
acute short-lived inflammatory responses.
2.1.2 Bradykinin:
With tissue and vascular injury, serum Hageman factor (Factor XII of the coagulation
cascade) activates the release of bradykinin, a nonapeptide (a long-lived vasodilator)
(Rang et al. 1996; Wilson and Kornman 1996). Bradykinin often follows the release of
histamine and is capable of increasing vascular permeability (Nisengard and Newman
1996). Bradykinin induces:
continued exudation and crevicular fluid flow
bone resorption in organ cultures via the prostaglandin cyclo-oxygenase pathway
(Newman et al. 1976; Nisengard and Newman 1996).
2.1.3 Plasmin:
Plasminogen enzyme is a normal constituent of plasma proteins. It is converted to
plasmin by the action of plasminogen activator (also called kallikrein). When the
intrinsic coagulation system is activated, the fibrinolytic system is activated through the
action of plasminogen activators. Activation of Hageman factor (XII) begins a cascade
of reactions in which it catalyses the reaction of circulating plasminogen to plasmin.
Plasmin is a multi-functional protease enzyme that digests fibrin and fibrinogen
(fibrinolysis) and other plasma proteins namely clotting factors II, V, VII and many other
tissue proteins. Plasmin is also an activator of several matrix metalloproteinases (Okada
9

1998). Plasmin derived lysis of the fibrin clot generates fibrin degradation products that
induce vascular permeability and trigger the complement system with the formation of
C3a and C5a components causing the release of histamine from mast cells. These fibrin
products are chemotactic to other inflammatory cells (Walter and Grudy 1993).
2.1.4 Complement:
Specific antibody and complement are two very important antimicrobial factors in GCF.
Activation of complement is one of the first host defences after injury, with these effects:
vasodilation and increased blood flow (by C2, C3a and C5a)
activate mast cells to release histamine (by C3a, C5a)
augment opsonisation (C3b) of bacteria by antibodies and allow some antibodies to
kill bacteria or by phagocytosis
chemoattractant to neutrophils and macrophages (C3a, C5a) and trigger the release of
prostaglandins, leukotrienes and enzymes into the tissue
cause pores to open in the membranes of pathogens causing cell lysis (C5-9)
(Dennison and Van Dyke 1997).
Bacteria in the gingival sulcus can activate the complement system via two major
pathways (Page 1991; Offenbacher et al. 1993a):
The classical pathway:
Activation of this system occurs rapidly. This pathway is activated by antigen-
antibody complexes (Dennison and Van Dyke 1997). Complement C1qrs binds
to the Fc component of IgG or IgM antibodies. This is fixed to the bacterial
10

receptor via the Fab region of immunoglobulin activating a cascade of enzymic
reactions to release C3 the precursor of C3A, C3b, C5a, C5b-9 which cause lysis
of cell membranes or functional alterations to promote phagocytosis (Lakhani et
al. 1993; Offenbacher 1996).
The alternative pathway:
Activation of this cascade does not involve immunoglobulin. Activation occurs directly
by bacterial surface lipopolysaccharide (LPS) and endotoxin (from gram-negative
anaerobes). This pathway also involves a series of reactions to release the precursor
complement protein C3 and produce the cleavage products C3a and C5a to C9 (Lakhani
et al. 1993; Offenbacher 1996).
The central event in both pathways is activation or splitting of C3 to C3b which becomes
attached to the activating stimulus (usually bacterial surfaces or antigen-antibody
complexes). Whichever pathway is activated, large amounts of C3a and C3b component
are released, fixing to the inflammatory stimulus and resulting in increased histamine
release, vascular permeability, chemotaxis to phagocytes (promoting phagocytosis), and
promotion of blood clotting. Complement can cause bystander damage since a small
amount may bind to host cells causing lysis or triggering neutrophils to attack.
2.1.5 Platelets:
Platelet adhesion and granule release plays an important role in the early development of the
vascular and cellular aspects of the inflammatory process (Walter and Grudy 1993). Release
of granules from platelets can also help initiate vascular permeability. Mediators released
from platelets include serotonin, a number of coagulation factors and thromboxane A2
(TxA2), all of which are pro-inflammatory. Platelet-derived growth factor (PDGF) is derived
11

from the platelet granules that contribute to the repair process (anabolic) following inflam-
matory responses or damaged blood vessels (Walter and Grudy 1993). Other anabolic effects
of PDGF are down regulation of alkaline phosphatase and promotion of proliferation of fi-
broblasts and periodontal regeneration (Okada 1998).
2.2 Eicosanoids
Cellular disturbances (e.g. from cell damage, LPS, complement, thrombin, bradykinin and
antigen-antibody complexes) cause enzymes known as phospholipases to generate
arachidonic acid from the cell membrane phospholipids. Arachidonic acid metabolites are a
small group of lipids known collectively as eicosanoids. Eicosanoids are not found pre-
formed in cells like histamine, they are generated de novo from cell membrane phospholipids.
They control many physiological processes and are the most important mediators and
modulators of the inflammatory reaction (Campbell and Halushka 1996). The prostanoids,
and in particular prostaglandins, are produced from arachidonic acid by cyclo-oxygenase that
occurs in neutrophils, macrophages, mast cells, fibroblasts, lymphocytes, keratinocytes,
osteoblasts and platelets (Offenbacher 1996). Prostanoids encompass all cyclo-oxygenase
products (Table 2.2). The predominant prostanoid product of the inflammatory response in
destructive periodontal diseases is thought to be PGE2 (Howell and Williams 1993).
Table 2.2 Composition of eicosanoids
EicosanoidsProstanoids
All cyclo-oxygenase products
prostaglandins
thromboxane
prostacyclins
Leukotrienes
All lipoxygenase products
12

2.2.1 General properties of eicosanoids
Eicosanoids are found almost in every tissue and body fluid and have the following
properties:
they are mediators derived from membrane phospholipids
they are effector molecules which are formed from polyunsaturated fatty acids
(lipids), mainly arachidonic acid. These include the prostaglandins,
prostacyclins, thromboxane A2 and the leukotrienes.
their production increases in response to diverse stimuli and they produce a broad
spectrum of biological effects.
these lipids contribute to a number of physiological and pathological processes
including inflammation, smooth muscle tone, haemostasis, thrombosis,
parturition, and gastrointestinal secretion.
several classes of drugs, most notably the nonsteroidal anti-inflammatory drugs (and
in particular aspirin), are therapeutically active because they block the formation of
eicosanoids.
(Salmon and Higgs 1987; Davies and MacIntyre 1992; Campbell and Halushka 1996;
Rang et al. 1996).
General effects of prostanoids vary and the type of response elicited is related to specific
target cell receptors (Table 2.2). Their effects are:
i. production of fever, pain and inflammation (Campbell and Halushka 1996).
ii. bone resorption by PGE's (Davies and MacIntyre 1992)
13

iii. PGE2 stimulates cAMP formation in cells, phospholipase C, and calcium influx in
osteoblasts
iv. PGE's also have insulin-like effects on carbohydrate metabolism and exert
parathyroid hormone-like effects that result in mobilisation of calcium ions from
bone (Campbell and Halushka 1996).
v. stimulation of the release of adrenal steroids (ACTH & growth hormone), and of
erythropoietin from the kidney (Davies and MacIntyre 1992; Campbell and
Halushka 1996).
vi. prostaglandins (PGE2, PGD2, PGA2) and prostacyclins (PGI2) are potent
vasodilators, while PGG2, PGH2 and TXA2 are powerful vasoconstrictors (Campbell
and Halushka 1996).
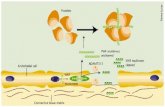
Figure 2.1 shows the products and pathways of cyclo-oxygenase.
Figure 2.1 Products and pathways of cyclo-oxygenase
(Salmon and Higgs 1987).
14

Table 2.3 Cell sources and actions of prostanoids
(Davies and MacIntyre 1992; Campbell and Halushka 1996).
ProstanoidReceptor
typeEffect Derived from
PGD2 DP
vasodilatationinhibition of platelet aggregationrelaxation of gastrointestinal muscleuterine contraction
mast cells
PGF2a FP
myometrial contractionincrease in cytoplasmic calcium ionsvasoconstrictor of pulmonary arteries and veins
Corpus luteum
Prostacyclin IP
vasodilatationinhibition of platelet aggregationrenin releasetubular reabsorption of sodium ionsincrease cAMPvasoconstrictionplatelet aggregationbronchial-constrictionincrease of cytoplasmic calcium ions
vascular epithelium
PGE2
EP1,EP2EP3
bone resorptionincrease in cAMP increases vasodilationincreases vascular permeabilitycontraction of bronchial and smooth musclebronchial-dilationstimulation of intestinal fluid secretionsrelaxation of gastrointestinal smooth musclecontraction of intestinal muscleinhibition of gastric acid secretioninhibition of lipolysisinhibition of autonomic neurotransmitter releasecontraction of uterusdecrease of cAMP in adipose cells
most nucleated cellsespecially monocytesand macrophages
2.3 Role of eicosanoids in periodontal tissues
Prostanoid products in the periodontal tissue are primarily mediators of inflammation
and tissue destruction (Offenbacher et al. 1993b; Offenbacher 1996). In view of the
large number of compounds that belong to the eicosanoid family, this section focuses on
15

the main mediator of periodontal inflammation and tissue destruction, i.e. the
prostaglandins and in particular PGE2. In periodontal tissues the actions of PGE2 induce
(Birkedal-Hansen 1993; Offenbacher et al. 1993b):
vasodilation and increased vascular permeability in the gingival plexus.
matrix-metalloproteinases (MMP) secretion from macrophages, monocytes and
fibroblasts stimulating connective tissue breakdown.
increases cAMP in macrophages
interacts with IL-1 and TNF- to enhance their effects.
modulation of platelet and leucocyte reactivity
inhibition of T cell proliferation
lysosomal enzymes release from neutrophils
generation of toxic oxygen radicals from neutrophils
histamine release from mast cells.
inhibition of macrophage/monocyte and lymphocyte activation
generation and secretion of other cytokines.
osteoclastic bone resorption i.e. increased severity of periodontal diseases (PGE2 has a
major role in periodontitis as a long-lived potent mediator of bone resorption interfer-
ing with the bone remodelling coupling mechanism between osteoblasts and osteo-
clasts) (Offenbacher 1996; Wiebe et al. 1996; Gemmell et al. 1997; Page et al. 1997;
Schwartz et al. 1997; Ueda et al. 1998).
2.3.1 Biosynthesis of eicosanoids.
The main source of the eicosanoids is arachidonic acid, a 20-carbon polyunsaturated fatty
acid found in the phospholipids of cell membranes and to a lesser extent, in the
glycerides of cell membranes (Davies and MacIntyre 1992). The initial and rate-limiting
step to eicosanoid production is the liberation of arachidonate from the membranes
16

(Rang et al. 1996), either by a one-step process involving phospholipase A2 (PLA2)
directly or the indirect two step process involving either phospholipase C and
diacylglycerol lipase or phospholipase D.
Phospholipase D is an important signal transducer that induces phagocytosis by
phagocytic cells. There are intracellular and extracellular forms of phospholipase A2. It
is mainly the intracellular form that is implicated in the generation of inflammatory
mediators; it generates arachidonic acid and platelet activating factor (PAF), another
powerful mediator of inflammation (Campbell and Halushka 1996; Rang et al. 1996).
The anti-inflammatory action of the glucocorticoids (adrenal hormones e.g. steroids) is
mainly due to the fact that they inhibit the formation of PLA2, inhibiting the induction of
cyclo-oxygenase within the cell and thus reducing free arachidonic acid (Campbell and
Halushka 1996; Rang et al. 1996).
2.3.2 Arachidonic acid pathways: eicosanoid production
There are three pathways for synthesis of eicosanoids from arachidonic acid (Campbell
and Halushka 1996; Sharma and Sharma 1997).
Pathway 1
This involves COX (also known as prostaglandin synthetase). Many stimuli acting on
different cell types can liberate arachidonic acid, for example:
thrombin on platelets
C5a on neutrophils
bradykinin on fibroblasts
antigen-antibody reactions on mast cells F
17

Free arachidonic acid is metabolised by COX to generate the endoperoxide products
(PGG2/PGH2) which are unstable at normal physiological pH and temperature and are
pivotal in the formation of other products (Salmon and Higgs 1987; Davies and
MacIntyre 1992). These products are either:
enzymatically converted into either prostaglandins, prostacyclins or
thromboxanes (collectively called prostanoids).
converted to hydroxy fatty acid (HHT) and malondialdehyde (MDA) by
enzymatic or non-enzymatic pathways (Salmon and Higgs 1987; Davies
and MacIntyre 1992).
COX is bound to the endoplasmic reticulum and primarily has two functions:
to produce cyclic endoperoxide PGG2
to convert PGG2 to another cyclic endoperoxide PGH2.
The next steps in arachidonate metabolism vary according to the cell-type secreting
various mediators, each eliciting different physiological functions (Fu et al. 1990; Rang
et al. 1996):
platelets only produce thromboxane A2 mediator
vascular endothelium produces prostacyclin mediator
macrophages/monocytes, fibroblasts produce PGE2
mast cells produce PGD2
The principal source of PGE2 in periodontal tissues is from macrophages/monocytes and
fibroblasts (although most nucleated cells can produce PGE2) (Fu et al. 1990). There are
two mechanisms whereby PGE2 is produced by macrophages/monocytes.
(a) Bacterial LPS induced PGE2 release
LPS will bind to LBP (a LPS binding protein found in serum) forming a complex
which binds to the high affinity CD14 receptor of macrophages/monocytes,
triggering high intracellular cAMP levels (with very low levels of LPS). This
18

stimulates the release of PGE2, TNF- and IL-1. Bacterial antigen-antibody
(IgG) or C3b can elicit the same reaction (Offenbacher et al. 1993b; Offenbacher
1996).
(b) Host induced PGE2
TNF- and IL- have an autocrine effect on the secretory
macrophage/monocyte and a paracrine effect on the residential fibroblast cells
which elicit PGE2, perpetuating the inflammatory response and activating an
immune response.
COX exists in two forms:
COX-1, a constitutive enzyme found in all cells i.e. it is always present at a constant
concentration in cells but may increase by 2-4 fold upon physiological stimulation,
producing low levels of mediators that are necessary for the maintenance of normal
tissue integrity and function. COX-1 produces the prostanoids (PGI2/6-keto-PGF1 &
TXB2) that regulate normal homeostasis (Sharma and Sharma 1997; Dubois et al.
1998).
COX-2 is the pro-inflammatory enzyme that is induced by inflammatory stimuli
only; its activity increases 10-80 times following injury or insult. Inflammatory
stimuli (eg LPS) or ligands (eg cytokines) bind to inflammatory cells and eventually
induce the prostanoid mediators of inflammation (Seibert and Masferrer 1994;
Seibert et al. 1994; Gierse et al. 1995; Sharma and Sharma 1997; Dubois et al. 1998).
There are approximately 10 prostaglandins; all have a cyclopentane ring (five carbon
ring) between carbon 8-12 (Figure 2.2). Prostaglandins are named alphabetically from A-
J, with three members in each group (except PGI). These are numbered 1, 2 or 3
19

(representing the double bonds on the prostaglandin molecule). For example PGE2 has
two double bonds between carbon 5-6, and 13-14. The prostacyclins have only two
members, (PGI2 and PGI3).
The thromboxanes (Tx) are closely related to the prostaglandins and are synthesised from
PGH2 (Figure 2.2). These molecules contain an oxane ring (a six carbon ring with an
oxygen atom) instead of a cyclopentane ring. Thromboxane A2 (TxB2) is a potent
vasoconstrictor and triggers platelet aggregation, causing thrombus formation (Sharma
and Sharma 1997; Dubois et al. 1998).
Figure 2.2 The chemical structures of PGE2 and TxB2
(Davies and MacIntyre 1992).
20

Pathway 2
The leukotrienes (LOX)
The second pathway for arachidonic acid metabolism is via the lipoxygenase (LOX)
pathway (Figure 2.1) to provide the parent molecule hydoperoxyeicosatetraenoic acid
(HPETE). The HPETEs are then further metabolised to leukotrienes, hepoxilins,
trioxilins and lipoxins (Sharma et al. 1997). To date there are six HPETEs (5,8,9,11,12
and 15-HPETEs) and each is formed by its corresponding enzyme (Sharma 1997).
Lipoxygenases are soluble enzymes found in the cytosol of cells of lung, platelets, mast
cells and leucocytes. The main enzyme in this group is 5-lipoxygenase; it converts 5-
HPETE to leukotrienes, 12-LOX converts 12-HPETE to hepoxilins and trioxilins, 15-
LOX converts 15-HPETE to lipoxins. Lipoxygenases differ in their specificity according
to the hydroperoxy group (-OOH) on arachidonic acid, and tissues differ in the
lipoxygenase(s) that they contain. For example, platelets have only 12-lipoxygenase and
synthesise 12-(HPETE) whereas leucocytes contain both 5-LOX and 12-LOX and
produce both 5-HPETE and 12-HPETE (Rang et al. 1996) (see Figure 2.1). Arachidonic
acid is enzymatically reduced to hydroxy acids (HPETEs).
The HPETEs are unstable intermediate metabolites (like PGG2 or PGH2) and are further
metabolised by a variety of enzymes. In the leukotriene pathway 5-HPETE is
enzymatically to converted leukotriene-A4 (LTA4) which is unstable, but pivotal in the
formation of other leukotrienes. LTA4 is enzymatically hydrolysed to LTB4 or non-
enzymatically to di-hydroxy acids (di-HETEs). Additionally, LTA4 can be converted
directly to the precursor of cysteinyl-leukotriene LTC4, which is further metabolised to
LTD4, LTE4, and LTF4. LTB4, LTC4, and LTD4 are also known as the "slow reacting
21

substance of anaphylaxis" (SRS-A) (Lewis et al. 1990; McMillan et al. 1992; Salmon et
al. 1987; Snyder et al. 1989).
LTB4, LTC4, and LTD4 are the most potent leukotrienes. LTB4 is a powerful chemotactic
agent for neutrophils and macrophages (acting in picogram amounts) and are important
in the early stages of inflammation. It causes up-regulation of membrane adhesion
molecules of neutrophils, increasing the production of toxic oxygen products and the
release of granule enzymes. It can stimulate proliferation and/or cytokine release from
macrophages (Abramson et al. 1989; Lewis 1990; Rang 1996; Samuelsson 1983).
Arachidonic acid or other polyunsaturated fatty acids may be further metabolised by
lipoxygenases to other oxygenated derivatives of polyunsaturated fatty acids (Rang
1996). A recent addition to these compounds is the lipoxins which were first isolated
in 1984 (Serhan et al. 1984) and generated from within various cells or during cell-cell
interactions. Lipoxins are generated from one of three pathways which can operate
independently or simultaneously (Serhan 1997).
(a) A 15-LOX initiated pathway:
This enzyme is found in eosinophils, macrophages, monocytes and epithelial
cells, under cytokine (IL-1, TNF-) and LPS control and regulated by IL-4
and IL-13 (two anti-inflammatory cytokines) (Levy et al. 1993; Nassar et al.
1994; Serhan 1997). Once these cells are stimulated, arachidonic acid is
converted to 15-HPETE or 15-HETE in the donor cell which serve as a
substrate for 5-LOX in the recipient cell (generally neutrophils) which is
converted to lipoxins (by transcellular metabolism) causing vasodilation,
leucocyte regulation and blocking leukotriene metabolism (Serhan 1997).
22

(b) 5-LOX initiated pathway:
This is generally a platelet-neutrophil interaction. This pathway involves 5-LOX
within neutrophils which converts arachidonic acid to 5-HPETE to LTA4 and
platelet 12-LOX induces lipoxin biosynthesis (Romano et al. 1993; Romano et al.
1992; Serhan 1997).
(c) Aspirin-triggered lipoxins (ATLs).
Aspirin has the ability to irreversibly inhibit COX-1 and COX-2 by acetylating an
essential serine residue site in both enzymes. The acetylated COX enzymes
cannot produce prostaglandins, however more recent medical research shows that
acetylated COX-2 in endothelial or epithelial cells converts arachidonic acid to
15-HETE (Claria et al. 1995; Claria et al. 1996). The 15-HETEs are released
from these cells by cell to cell adherence i.e. to leucocytes (especially
neutrophils) and further metabolised via transcellular pathways by 5-LOX of
leucocytes to form 15-epimeric-lipoxin (15-epi-LX) metabolites (Claria 1995).
The 15-epi-LX metabolites are also termed aspirin triggered lipoxins (ATLs).
Endothelial and epithelial cell COX-2 when induced by pro-inflammatory
cytokines (IL-1, TNF-) and LPS in the presence of aspirin can shunt
arachidonic metabolism to synthesise 15-epi-LX molecules. The 15-epi-LX
molecules serve as “stop signals” (i.e. to evoke anti-inflammatory effects)
causing vasodilation, inhibiting neutrophil adhesion to endothelial cells
(diapedesis), chemotaxis and cell proliferation (Claria 1996; Claria 1995; Clish et
al. 1999; Serhan 1997; Takano et al. 1997).
From these recent findings it may be that lipoxin production may be an important
anti-inflammatory event, especially by ATLs. However most studies on lipoxins
23

use in vitro or in vivo models. However further research is needed to understand
the biofeedback regulatory mechanisms involved in converting from a pro-
inflammatory eicosanoid phenotype to an anti-inflammatory phenotype.
Currently there is no research on lipoxin (especially ATL) involvement in the
gingival or periodontal inflammatory process. Nevertheless ATLs may be a
further anti-inflammatory (beneficial) pathway provided by aspirin.
Pathway 3
This pathway involves the cytochrome P450 group of enzymes (present in endoplasmic
reticulum) breaking down arachidonic acid to HETEs and DiHETEs.
The predominant pathways in eicosanoid production are pathways I and 2 (Sharma and
Sharma 1997).
2.3.3 Catabolism of the eicosanoids
A number of intra-cellular enzymes are involved in the catabolism and inactivation of
most eicosanoids. There are prostaglandin-specific enzymes that rapidly inactivate the
prostaglandins and their metabolites are excreted in the urine. About 95% of PGE2,
PGE1 and PGF2a are inactivated during their first passage through the pulmonary
circulation. The half-life of most prostaglandins is less than a minute in the circulation
(Campbell and Halushka 1996; Rang et al. 1996). Leukotriene products are inactivated
by oxidative pathways or degraded in the kidneys, lungs and liver (Campbell and
Halushka 1996).
2.4 The role of cytokines in periodontal tissues
Table 2.4 shows the major mediators thought to be involved in the pathogenesis of
periodontitis (Schenkein 1999). Arachidonic acid metabolites have been discussed in
24

section 2.3, the next section only discusses the interleukins and Tumour Necrosis Factor
alpha (TNF-). All these mediators primarily interact with each other and have impact
on the pathogenesis of periodontal diseases, but prostaglandins are the major mediators
involved in tissue destruction in conjunction with the following cytokines.
The term cytokine means “cell protein”. Cytokines direct and regulate inflammation and
wound healing (Page et al. 1997; Okada 1998). Subgroups of cytokines are:
the interleukins which carry complex and detailed messages between leucocytes,
the growth factors which trigger myelopoesis, leucocyte mitosis and cell
differentiation
chemokines which trigger cell recruitment
interferons, lymphocyte activating molecules (Birkedal-Hansen 1993; Offenbacher
1996).
Cytokines, lymphokines and monokines have autocrine (self-regulate the cells producing
the cytokine), paracrine (modulate distant cells not producing the cytokine) and
intracrine (actions within a cell) effects on target cells (Okada 1998). All cytokines
activate target cells by binding to specific receptors on their cell membranes. This
receptor-ligand coupling triggers cellular activation of the target cell, modifying the cell's
activity (Birkedal-Hansen 1993; Offenbacher 1996), e.g. IL-1 binds to fibroblasts to
trigger the release of collagenase to degrade collagen in the immediate environs. Often
inflammatory cytokines trigger the secretion of specific enzymes, lipids, bioactive
amines and reactive oxygen metabolites that serve as effector molecules (Alexander and
Damoulis 1994). Periodontal tissue destruction is via mobilisation and activation of
25

macrophage/monocytes, lymphocytes and fibroblasts. The modulation of these events is
via catabolic cytokines and inflammatory mediators.
Table 2.4 Major tissue destructive mediators in periodontitis
Interleukins (1, 6, 8)
Tumour Necrosis Factor alpha (TNF-)
Arachidonic Acid Metabolites (PGE2)
Interleukin-1 (IL-1)
Interleukin-1 is a polypeptide, which has diverse roles in immunity, inflammation, tissue
breakdown and homeostasis. It is synthesised by macrophages, monocytes,
lymphocytes, endothelial cells, fibroblasts, keratinocytes and brain cells. In the
periodontal tissues, macrophages predominantly secrete IL-1, Il-1 and Il-1 Both
forms bind to the same cell receptors of many cell types and in various densities
(Alexander and Damoulis 1994; Offenbacher 1996; Mathur and Michalowicz 1997;
Soskolne 1997; Ellis 1998; Okada 1998; Schenkein 1999)
Properties of IL-1:
increases adhesion molecules on fibroblasts, immunocytes (stimulates
proliferation of keratinocytes and endothelial cells)
enhances fibroblast synthesis of collagenase, fibronectin and PGE2
induces the production of matrix metalloproteinases (MMPs) in
periodontitis.
elevates the levels of pro-collagenase in gingival and periodontal ligament
fibroblasts.
stimulates plasminogen activator in gingival fibroblasts, resulting in the
generation of plasmin which is a naturally occurring activator of several
matrix metalloproteinases.
26

activates both T- and B-cells. It also promotes B-cell activation,
proliferation, clonal expansion and antibody secretion. It “primes”
macrophages and neutrophils by up-regulating receptors for complement
and immunoglobulins.
T-cells regulate the immune response by increasing or decreasing IL-1
secretion. T-cells release gamma interferon (IFN-) which enhances
secretion of IL-1 and PGE2 from LPS-stimulated macrophages. Therefore
IFN- serves to up-regulate the inflammatory response. (Page 1991;
Birkedal-Hansen 1993; Alexander and Damoulis 1994; Mathur and
Michalowicz 1997; Soskolne 1997; Ellis 1998; Okada 1998; Page 1998)
IL-1and IL-are potent connective tissue catabolic stimulators. They
directly stimulate bone resorption, and trigger the release of PGE2 from
fibroblasts and macrophage/monocytes. PGE2 (Seymour et al. 1993;
Tatakis 1993).
Interleukin-6 (IL-6)
IL-6 influences immune and inflammatory responses and the main sources are from
stimulated fibroblasts, endothelial cells, macrophages, T and B-cells and keratinocytes.
IL-6 shares many biological properties with IL-1 and has been found to:
be in higher concentrations levels in inflamed sites than healthy sites
stimulate eicosanoid production
stimulate MMP production
stimulate B-cells into Ig-secreting plasma cells.
be a potent stimulator of IgG1.
27

plays a major role in regulating bone turnover and is essential for bone loss
caused by oestrogen deficiency (menopause)
act as a paracrine and/or autocrine factor in bone resorption in pathologic
states, by stimulating osteoclasts and activating bone resorption (Page 1991;
Alexander and Damoulis 1994; Offenbacher et al. 1996; Mathur and
Michalowicz 1997; Soskolne 1997; Ellis 1998; Okada 1998; Schenkein
1999).
Interleukin-8 (IL-8)
Produced by leucocytes and keratinocytes in response to LPS, IL-1 or TNF- with the
following properties:
proinflammatory
strong chemoattractant to neutrophils.
selectively stimulate MMP in macrophages keratinocytes
Local tissue destruction in periodontitis or inflamed gingiva may be due to the
continuous and excessive IL-8 levels that in turn mediate chemotactic and activation
effects on neutrophils and production of MMPs. IL-8 may also attract and induce T-cell
proliferation (Alexander and Damoulis 1994; Mathur and Michalowicz 1997; Soskolne
1997; Ellis 1998; Okada 1998).
Tumour necrosis factor (TNF-)
This proinflammatory cytokine is mainly secreted by monocytes and macrophages; it has
the following properties:
induces secretion of collagenase by fibroblasts
induces resorption of cartilage and bone
induces periodontal tissue breakdown in periodontitis
28

in resting macrophages it induces synthesis of IL-1 and PGE2
activates osteoclasts and thus induces bone resorption although both forms of
IL-1 are at least 10 times more potent on a molar level than TNF- in the
induction of bone demineralisation
has synergistic effects with IL-1 in bone resorption actions
lipopolysaccharide (LPS) from gram negative bacteria can initiate the
production of TNF- from macrophages/monocytes (Alexander and
Damoulis 1994).
Periodontal homeostasis represents a delicate balance between anabolic and catabolic
activities (Offenbacher 1996). Myriads of cytokines are involved in tissue turnover and
the maintenance of the integrity of the periodontium; of interest are the interleukins,
prostaglandins, interferons and colony stimulating factors which mediate inflammatory
and immune responses (Williams et al. 1996). Cytokines in association with PGE2 are
thought to lead to alveolar bone resorption, inhibition of bone formation and synthesis of
collagenase by gingival fibroblasts which degrades matrix collagen (Page 1991;
Alexander and Damoulis 1994; Offenbacher et al. 1996; Mathur and Michalowicz 1997;
Ellis 1998; Okada 1998; Ueda et al. 1998).
One of the significant advances in periodontal research in the last 20 years has been the
finding that normal residential cells of the periodontium can be induced to a catabolic
state by exposure to LPS, IL-1, TNF- and PGE2 and participate in tissue destruction
(Reynolds and Meikle 1997; Schwartz et al. 1997). In periodontal health, fibroblast
genes for collagen synthesis and TIMPs are turned on while the genes for MMPs are
turned off. During periodontitis, the reverse applies, with fibroblasts producing also IL-
1This cytokine may cause autocrine stimulation with more IL-1being secreted or
affect other target cells such as monocytes/macrophages, epithelial and endothelial cells
29

(paracrine stimulation) to further enhance the production of the MMPs and PGE2
(Reynolds and Meikle 1997; Schwartz et al. 1997; Schenkein 1999).
Clinically healthy gingival and periodontal tissues express a number of anabolic growth
factors:
epidermal growth factor (EGF)
platelet-derived growth factor (PDGF)
transforming growth factor (TGF-- this is a superfamily of proteins and
contains a number of bone morphogenic proteins (BMPs)
insulin-like growth factor (IGF)
cementum-derived growth factor (CGF)
inflammatory cytokines such as (IL-1, IL-6, and TNF-) are in low
concentrations compared to inflamed sites
These anabolic molecules are involved in the rebuilding of the extracellular matrix by:
chemoattracting fibroblasts, periodontal ligament cells and bone
generating cells
stimulating cells from a stable (nondividing) cycle to undergo mitosis and
thus increasing the number of stromal cells
inducing cell differentiation of connective tissue mesenchymal cells to
matrix secreting cells (Bartold et al.1998).
Many of these molecules are incorporated into newly formed extracellular matrices,
which they induce. During wound healing or repair, macrophages are drawn to these
sites by clotting factors. These macrophages respond differently than the pro-
inflammatory macrophages which are chemo-attracted to sites by bacterial products or
complement products (Bartold et al.1998).
2.5 Cellular events in inflammation
The acute inflammatory response is characterised by the presence of neutrophils
(Miyasaki 1991; Miyasaki et al. 1994; Miyasaki 1996) which constitute approximately
30

90% of total circulating leucocytes and are the body's first line of defence against
microbes (Van Dyke and Hoop 1990). Neutrophils have at least three types of
cytoplasmic membrane-enclosed granules: primary granules or azurophil granules,
secondary granules or specific granules and tertiary or secretory granules (Table 2.5).
These granules can release a large variety of enzymes that can degrade host tissue
(collagenase, elastase, -glucuronidase); this is a part of normal tissue homeostasis
resulting in remodelling or healing. In addition these granules have a large number of
antimicrobial substances which can kill ingested microorganisms once phagocytosed.
Table 2.5 Neutrophil components and function (Williams et al. 1996).
Granule Function (all function under anaerobic and aerobic conditions)Granule component Effect
Primary granules(azurophil)
Cellular myeloperoxidase Microbial killingLysosomeCationic proteins Histamine release +
enhances phagocytosisAcid hydrolases AntibacterialGlucoronidaseannoxidaseNeutral proteaseElastase Exacerbates and mediates in
inflammationCathepsin
Secondary (specific) granules
Lysosome Hydrolysis of cell wall proteoglycansAlkaline phosphatase
Collagenase Collagen degradationVitamin B12 binding proteinsLactoferrin Bactericidal
Tertiary (secretory) granules
GelatinaseAlkaline phosphatase
Replenish cell surface receptor expression and adhesion
2.5.1 Macrophage phenotypes
Since the host defences cannot inactivate biofilm completely, the inflammatory response
in periodontitis is longstanding and chronic. The presence of the macrophage signals
chronicity; the neutrophil/macrophage characterises chronic inflammation while the
31

presence of macrophage/lymphocytes characterises the immune response (Williams et al.
1996). The distinction between chronic inflammation and immunity is not that clear-cut.
Macrophages have an important role in antigen processing as parts of the development of
an immune response and a subset of these cells have phagocytic capacity (Page et al.
1997). Macrophages arise from bone marrow in a functional, immature condition, but
eventually differentiate in the tissues. Macrophage phenotypes may phagocytose
bacteria, modulate the clearance of damaged tissue debris during inflammation, modulate
tissue remodelling, and trap and present antigens to lymphocytes (helper T-cells) to
induce the immune response. (Miyasaki 1996; Page et al. 1997).
Macrophages are capable of synthesizing cytokines that contribute to healing and repair
(anabolic) but can also have pro-inflammatory effects (catabolic) in the presence of a
chronic microbial challenge. (Page 1991; Seymour 1991; Birkedal-Hansen 1993;
Offenbacher et al. 1993b; Genco et al. 1994). Macrophages represent 5-39% of
infiltrating cells in inflamed periodontal tissues (Toppal et al. 1989; Zappa et al. 1991;
Okada 1998).
The macrophage is the key cell in directing whether anabolic or catabolic changes occur
within the periodontal tissues. The catabolic changes occurring in the gingival and
periodontal tissues are due to the presence of the proinflammatory macrophages which
have distinctive properties:
reactive oxygen metabolites, including the superoxide anion(O2-), hydrogen
peroxide (H2O2), the hydroxyl ion (OH-), and hypochlorous acid (HOCl-). All
are bactericidal but can also be toxic to host cells (Klebanoff 1992; Alexander
and Damoulis 1994).
32

arachidonic acid metabolites such as PGE2 and LTB4 and these can be
produced in large amounts to create an inflammatory reaction (Samuelsson
1983; Salmon and Higgs 1987; Davidson 1992; Offenbacher et al. 1993b).
secreting the proinflammatory cytokines IL-1 IL-6, and TNF- (Offenbacher
1996)
In the periodontium, macrophage PGE2 has many regulatory effects:
decrease adherence and migration of macrophages,
under the influence of IL-1 and TNF-inhibitsthe genes controlling the
synthesis of collagen and non-collagenous matrix proteins and tissue inhibitors
of metalloproteinases (TIMPs) in fibroblasts
stimulates the synthesis and release of matrix metalloproteinases (e.g.
collagenase)
it is the major mediator of pathological bone resorption
suppresses leucocyte function
along with cytokines IL-1, TNF- and interferon- (INF- it can regulate IgG
production (where high concentrations of PGE2 inhibit antibody production
and low concentrations act synergistically with IL-4 and enhance IgG
production (Miyasaki 1996; Page et al. 1997; Reynolds and Meikle 1997).
2.5.2 Alveolar bone resorption
Knowledge about alveolar bone resorption has lagged behind the understanding and
research on connective tissue breakdown. There are, however, some well established
facts about bone remodelling and resorption (Schwartz et al. 1997):
bone resorption-formation is a tightly coupled process and PGE2, IL-1, TNF-
are known mediators of bone loss.
IL-6 mediates the formation osteoclasts to resorb bone
activities between osteoblasts, osteoclasts and osteocytes are highly
integrated and coordinated with one another.
33

there are biofeedback loops between these cells, where osteoblasts produce
local factors that induce osteoclastic activity and vice versa.
cell control is regulated by circulating factors such as steroid hormones,
parathyroid hormone, calcitonin and vitamin D.
More information is needed to fully understand this area of periodontal pathogenesis.
This overview of the inflammatory response is brief; the reaction of the inflammatory
response is very complex, and is integrated with the immune system. The aim of this
review was to show the major inflammatory pathways and their roles in the overall
pathologic process.
2.6 Nonsteroidal anti-inflammatory drugs in periodontal diseases.
The main anti-inflammatory agents are the glucocorticoids and the non-steroidal anti-
inflammatory drugs (NSAIDs). Most of these drugs have anti-inflammatory, analgesic
effects and antipyretic effects which are related to their inhibition of the actions of COX
(Vane 1971) and thus the inhibition of prostaglandins and thromboxanes (Heasman and
Seymour 1989; Heasman et al. 1989; Williams et al. 1989; Heasman et al. 1990; Czuszak
et al. 1996).
2.6.1 History of salicylates
Aspirin (acetylsalicylic acid) is the most widely used medicinal agent in the Western world
(Vane et al 1992; Rainsford 1994). Natural products that contain precursors of salicylic acid,
such as willow bark (which contains the glycoside salicin) and oil of wintergreen (which con-
tains methylsalicylate) have long been used in the treatment of rheumatism. (Walton et al.
1994). Since the early 1800’s, salicin was hydrolysed to glucose and salicylic alcohol, which
was then converted to salicylic acid. Sodium salicylate was first used in 1875 in the treatment
of rheumatic fever and as an anti-pyretic. The success of this drug prompted Hoffman, a
34

chemist employed by Bayer, to prepare acetylsalicylic acid based on the earlier, but forgotten,
work of Gerhardt in 1853. The name aspirin is derived from Spiraea, the plant species from
which salicylic acid was once prepared (Campbell and Halushka 1996). According to Rains-
ford (Rainsford 1984) the Bayer Company enjoyed immense profitability from aspirin by pro-
tecting its patents until the beginning of World War I. This was when other companies started
to process aspirin and challenged the Bayer monopoly. In USA the patent office cancelled
Bayer’s registered rights to the name of aspirin, in 1918 and the US Supreme Court ruled that
there was no infringement of tradename rights by US companies because Bayer’s aspirin had
been over-advertised to such an extent that it had become a common name. At about the
same time in Australia, the Federal government also suspended the Bayer’s patent rights. A
pharmacist George Nicholas with a chemist called Smith produced the first Australian aspirin
by 1915. Smith later withdrew from the partnership. George Nicholas joined with his brother
and formed the Nicholas Proprietary Ltd. They registered their aspirin under the trademark of
“Aspro” (Rainsford 1984).
2.6.2 Physio-chemical properties of aspirin and other salicylates .
The structure of aspirin is shown in Figure 2.3 (Campbell and Halushka 1996). Aspirin is
rapidly absorbed from the gastrointestinal tract, partly from the stomach and mainly from the
upper small intestine where it is quickly hydrolysed to salicylate (salicylic acid) by esterase
enzymes in the gut wall, blood and liver.
Figure 2.3 Structure of aspirin
(Rang et al. 1996).
35

C O O H
O C O C H 3
The rate of absorption of aspirin and salicylic acid is governed by:
their physio-chemical properties - aspirin and salicylate are weak acids and
have high lipid solubility
the surface area of the gastric and intestinal tract available for absorption
the pH of the gastrointestinal lumen. Absorption of salicylic acid is
facilitated at the low pH existing in the stomach. The absorption of aspirin is
half to one-third the rate of absorption of salicylic acid because of their
different partition coefficients (lipophilicity). Also, the delayed absorption
rate of aspirin is governed by the enzymatic hydrolysis by aspirin esterases in
the mucosa, ie the spontaneous hydrolysis of aspirin. This is a rate-limiting
step.
the rates of gastric emptying and intestinal transit times (which are dependent
on the osmolality and acidity of the gastric contents).
the presence of food in the gastrointestinal tract
the physical form of the drug (tablet, capsule or soluble forms).
(Rainsford 1994; Campbell and Halushka 1996; Rang et al. 1996).
The half-life of aspirin in humans is 20-30 minutes and the half-life of salicylic acid is 2-3
hours (Walton et al. 1994). Metabolism of aspirin to salicylic acid occurs by:
hydroxylation by liver cytochrome P450 system to gentisic acid or non-
enzymatically by Fe(II)-catalysed hydroxyl radical (OH)
36

conjugation to phenolic or acyl-glucuronides by the enzyme UDP-
glucuronosyltransferase. These glucuronides are synthesised in the small
intestine, liver, bladder, kidney, lung, gastrointestinal mucosa and spleen.
conjugation of the carboxylate hydroxyl groups or gentisic acids to form
salicylurate or gentisturate. These reactions activate acetyl-Co A in
mitochondria of the liver and kidney.
Various derivatives of salicylic acid have been synthesised for systemic use because
salicylic acid is irritating to gastric mucosa. These comprise esters of salicylic acid (of
which aspirin is one) or salts of salicylic acid. Sodium salicylate is a salt of salicylic acid
and has two thirds of the potency of aspirin. Aspirin itself is relatively insoluble, the
drug is nearly always taken orally, and soluble formulations are more efficacious than
tablet formations (Seymour et al. 1986; Holland et al. 1988). Aspirin and salicylate
covalently modifies COX-1 and COX-2 by acetylating the active sites of both enzymes,
and irreversibly inhibiting COX activity. Aspirin is 10-100 times as potent against COX-
1 than against COX-2 (Figure 2.4) (Bing et al. 1999).
Platelets cannot bio-synthesise cyclo-oxygenase (COX-1) and are therefore susceptible to
the action of aspirin. Aspirin and its derivative acetylsalicylic acid have the same
inhibitory effect on platelet cyclo-oxygenase at low and high doses (Lloyd and Bochner
1996; Diener 1998; Müller 1998). A single low-dose of aspirin inhibits platelet cyclo-
oxygenase for the life of the platelet (10 days) (Meade et al. 1993). Since aspirin and
other NSAIDs are organic acids, they accumulate at sites of infection, an attractive
pharmacokinetic property (Campbell and Halushka 1996).
Figure 2.4 Effects of aspirin on cyclo-oxygenases
(Bing et al. 1999).
37

As low-dose aspirin irreversibly inhibits cyclo-oxygenase over the whole lifetime of
platelets, it is widely used in middle-aged and elderly populations to prevent coronary
artery disease, stroke and peripheral vascular diseases. Additionally the lower doses
have lower gastro-intestinal side effects than high doses (Vane and O'Grady 1993;
Underwood 1994; Lloyd and Bochner 1996; Diener 1998; Müller 1998). Other NSAIDs
are either organic acids or (once absorbed) are converted to organic acids to cause
reversible COX inhibition. Aspirin and other NSAIDs do not affect the leukotriene
pathway. The majority of NSAIDs are non-selective inhibitors of the COX enzymes
(Brooks et al. 1991).
Most side effects of NSAIDs (gastric/duodenal ulcers, bleeding, cardiovascular and renal
failure) are due to the non-selective inhibition of COX-1 (Sharma and Sharma 1997;
Dubois et al. 1998). Longitudinal studies show that 75mg can cause small but significant
increase in gastrointestinal bleeding, the effect doubles with 300mg and there is a 5 fold
increase with 1.5-2.4 g per day (Lloyd and Bochner 1996). New agents have been
developed that selectively inhibit COX-2, minimising the systemic side effects.
However low-dose, slow release or enterically coated aspirins and its low cost of
38

production, make aspirin the highly used non-steroidal anti-inflammatory drug it is today
(Vane and Botting 1992; Lloyd and Bochner 1996).
2.6.3 Periodontal studies of the effects of NSAIDs over the last 20 years
There have been two reviews of NSAIDs and their effects on the gingival and periodon-
tal diseases (Howell and Williams 1993; Drisko 1996). Most studies prior to 1990 were
retrospective, making comparisons difficult because of variable drug regimes, poor con-
trol of specific medications, dosage or systemic health status of test patients. Overall,
subjects taking NSAIDs had lower plaque indices, gingival indices, probing pocket
depths, attachment loss and less bone loss (Howell and Williams 1993). Table 2.6 lists
the current NSAIDs used in periodontal studies.
The current review shows that prostanoids and leukotrienes are implicated in a wide range of
events that are associated with disease, such as platelet aggregation, vasodilation and vasocon-
striction, chemotaxis of neutrophils, increased vascular permeability and bone resorption. In
periodontal diseases, these metabolites seem to be closely associated with connective tissue
destruction and alveolar bone resorption.
Table 2.6 The types of NSAIDs (and their classification) used in periodontal
studies
Acetylated salicylates Aspirin
Acetic acid Indomethacin, Sulindac, Ketorlac
Propionic acids Ibuprofen, Naproxen, Flurbiprofen, Ketoprofen,
Fenamic acid Mefenamic acid,
Enolic acids Piroxicam
The physiochemical properties determine their distribution in the body and differences in these properties may lead to variable therapeutic efficacies (Brooks and Day 1991).
39

The following review discusses the effect of NSAIDs (as inhibitors of cyclooxygenase) on the
clinical course of experimental and naturally occurring gingivitis and periodontitis in humans.
Table 2.7 details the periodontal effects of NSAIDs in human studies.
Table 2.7 Periodontal effects of NSAIDs in human studies
Flurbiprofen
Study Observations and comments
(Williams et al. 1989) A double-blind placebo controlled study using 44 individuals of 24 months duration (pre-and treatment phase) with periodontal disease. Gingival inflammation was assessed every 2 months, standardised radio-graphs every 6 months determined the rate of al-veolar bone loss. The 2-year treatment group was compared to the 6-month baseline.
Individuals taking flurbiprofen had a sig-nificant lower rate of alveolar bone loss compared to baseline and control up to 18 months but not at 24 months ie returned to pre-treatment baseline values. There was no significance in the long-term use of this drug.
(Heasman et al. 1989) A double-blind split mouth experimental gingivitis study with con-tralateral sides as controls. The study used 24 healthy subjects for 17 days. Each subject cleaned one upper quadrant with 10mM-flurbi-profen solution every second day. A follow-up investigation of 6 subjects was performed only using placebo irrigation.
There was a serious flaw in the study's de-sign. Flurbiprofen is absorbed by the whole oral mucosa and this could have systemic effects affecting the control sites in split mouth studies. Flurbiprofen solu-tion reduced gingival inflammation in ex-perimental gingivitis in both test and con-trols. Due to this flaw, the experiment was modified so that an extra 6 subjects irrigated the whole mouth with a placebo solution and data compared to the flur-biprofen groups; there were significant differences in gingivitis and probing depths.
40

Flurbiprofen
Study Observations and comments
(Heasman and Seymour 1989) A single blind controlled parallel study of 27 days duration looking at sys-temic effects of flurbiprofen on ex-perimental gingivitis. There were 25 healthy volunteers who stopped tooth brushing for 21 days. On day 22 each subject was randomly placed into one of three groups. Then assessed for plaque, gingival index and pocket depths.
There were no changes in probing pocket depths for all three groups. Systemic flurbiprofen re-duced gingivitis over a 21-day period. But there are problems justifying long term use of 100mg/day flurbiprofen due to possible gastric side effects. Therefore topical application may be of more value (as shown by a previous study) (Heasman et al. 1989) bypassing the gastric side effects.
(Heasman et al. 1990) A follow-up prospective 28-day study on 5 indi-viduals ingesting flurbiprofen and then measuring flurbiprofen levels in GCF by high performance liquid chromatography.
Flurbiprofen levels in GCF were found to be higher than plasma levels. This led to the as-sumption that flurbiprofen concentrates in the periodontal tissues.
41

Flurbiprofen
Study Observations and comments
(Heasman and Seymour 1990) A controlled cross sectional study of 92 patients age and sex matched. Fifty hospital rheumatoid arthritis test patients were on a range of NSAIDs with a 2-30 year duration. The controls were 42 non-rheumat-oid hospital patients with no history of nonsteroidal therapy. The clinical parameters measured were 6 Ramf-jord teeth for plaque index, gingival index, pocket depth, loss of attach-ment, gingival recession, gingival crevicular flow and non-standard-ised radiographic analysis of bone loss.
There were no differences for any clinical pa-rameters when compared to the control group, these results were contrary findings to other studies (Waite et al. 1981; Feldman et al. 1983). One should question the results of this study since there were a number of serious flaws in the design and selection of the sample.
(a) One of the main problems in selecting rheumatoid arthritis patients is that some studies have indicated that these individuals may have higher severity and extent of peri-odontal disease (Tolo and Jorkend 1990; Kasser et al. 1997).
(b) With a mean age of 50, females may be en-tering menopause (Payne et al. 1997) these changes may affect the outcome in non-steroidal anti-inflammatory periodontal pro-gression studies.
(c) No control of smoking (smoking is a risk factor for periodontitis and osteoporosis) (Jeffcoat 1998)
Individuals were taking a number of different NSAIDs and no attempt was made to differenti-ate between them; different NSAIDs have dif-ferent effects on cyclo-oxygenase
(Abramson et al. 1992) Double blind parallel randomised controlled study consisting of 21 subjects of 57-day duration. The main aim to determine the effect of systemic flurbiprofen administration on GCF-PGE2 and TxB2 levels
Systemic flurbiprofen decreased GCF-PGE2 and TxB2 during treatment; these returned to normal levels 7 days after cessation of drug. These correlated well to beagle dog findings (Williams et al. 1988; Offenbacher et al. 1989) and similar findings to topical application of flurbiprofen (Heasman et al. 1989). One of the problems acknowledged with this study was that the individuals had a low prevalence of severe gingivitis and periodontitis and it is not known whether the effects of flurbiprofen would be the same in severe cases of disease.
42

Flurbiprofen
Study Observations and comments
(Heasman et al. 1993a) A placebo controlled clinical trial of 12 months duration. Assessing the efficacy of flurbiprofen toothpaste as an adjunc-tive measure in non-surgical manage-ment chronic adult periodontitis. There were 25 test subjects and 24 control subjects and both groups were age and sex matched. Plaque scores, bleeding scores, crevicular fluid flow, probing pocket depths and attach-ment levels were measured at baseline, 3, 6, 9 and 12 months. Ra-diographs were taken at baseline and 12 months.
Radiographically, a significantly number of sites (8%) had more bone gain compared to controls (3.3%) but there were no significant differences between the test and control groups in plaque, bleeding scores, probing depths or attachment loss. The conclusion was that flurbiprofen-con-taining toothpaste had a small significant effect on bone metabolism. Possible reason discussed for these findings was the Hawthorne effect.
(Heasman et al. 1994) A double-blind parallel controlled study of 27 days duration with 47 (male and fe-male) subjects. Looking at the sys-temic effects of 100mg flurbiprofen in conjunction with tooth brushing on experimental gingivitis.
There were no significant differences between groups for plaque, and gingival crevicular flow. There was borderline significance in the resolu-tion of gingivitis compared to placebo controls. This reinforced the findings of other studies (Waite, et al. 1981; Offenbacher et al. 1987; Of-fenbacher et al.1989) that not all inflammation is due to cyclo-oxygenase derivatives.
43

Flurbiprofen
Study Observations and comments
(Brägger et al. 1997). A double blind split mouth placebo controlled clinical trial of 6 months duration, using flurbiprofen follow-ing flap surgery on 19 subjects with moder-ate to severe adult periodontitis. At baseline all patients underwent surgical periodontal therapy at 2 sites with >5mm pocket depths and two sites with >5mm pocket depths with no surgical treatment. Ten subjects received 150mg/day flurbipro-fen postoperatively for 30 days with 9 sub-jects using a placebo during healing phase. Measurements taken were of plaque index, gingival index, probing pocket depths, sub-traction digital image radiography analysing alveolar bone density, recession, probing at-tachment levels and bleeding on probing.
After 6 months there were no statistically significant differences between 4 groups in bone density or bone remodelling. A significant difference in probing depths and clinical attachment gain was seen in all groups irrespective if flurbiprofen or placebo, indicating there was no influence of the NSAID on bone metabolism post-surgically. The authors stated that a possi-ble explanation for these results was that 150mg flurbiprofen/day was too low or that there may be completely different bi-ological mechanism or pathways involved in pathological alveolar bone resorption of periodontitis compared to bone healing (repair) following surgery. The non-sig-nificant results could have been influ-enced by independent variables such as age and gender (not stated in this study), poor control for smoking and diabetes. These risk factors could have influenced the outcome.
44

Ketorolac
Study Observations and comments
(Jeffcoat et al. 1995) A random, parallel-placebo, double-blind positive control design of 6-month duration on 55 patients with un-treated adult periodontitis. The main aim was to measure the effects of topical ketoro-lac on alveolar bone height, bone mass, peri-odontal bone mass, probing attachment lev-els, plaque, gingival index. The groups were balanced for gender. To determine and iden-tify active alveolar disease sites each patient was given a radioactive agent orally and a bone scan detector identified these sites. At baseline, 3-months and 6-months plaque scores, probing depth, attachment levels, blood, urine, GCF-PGE2 and IL-1 were col-lected. Digital subtraction radiograph analy-sis was alsoused. Each patient had 2x/day ketorolac mouth rinsing. Three groups were developed, a placebo rinse group (negative control), a placebo rinse plus 100mg-flur-biprofen tablet group (positive control) and a 10% ketorolac rinse and a placebo tablet.
The flurbiprofen and ketorolac groups had significantly less bone loss with sig-nificant reductions in GCF-PGE2 levels compared to the placebo group. In addi-tion, ketorolac, preserved more bone than systemic flurbiprofen. Studies (Jef-fcoat et al. 1988; Heasman et al. 1989; Williams et al. 1989; Jeffcoat et al. 1991) on NSAIDs and their effects on gingivitis had mixed results . Ibuprofen, naproxen, ketoprofen, meclofenamate effects on gingivitis have been minimal but appear to affect the progression of bone loss. Topical oxazlopyridine inhib-ited gingivitis and systemic flurbiprofen inhibited experimental gingivitis in hu-mans. There was a significant decrease in gingivitis in this study. This study did not control for the regular prophylaxis, oral hygiene and Hawthorne effects on gingivitis, which could have accounted for the results.
45

Ibuprofen
Study Observations and comments
(Taiyeb and Waite 1993) A random, split-mouth, controlled prospective study of 8 weeks duration with 17 indi-viduals. Investigated the systemic ef-fects of ibuprofen on mild to severe generalised chronic periodontitis. Plaque, gingival inflammation, BoP and pocket depths were measured. Two groups were developed both given whole mouth oral hygiene in-structions. Scaling on one side only randomly selected, making each pa-tient their own control. The test group was given 200mg 4x/d of ibuprofen for 14 days. The 2 week regime avoided placing excessive demands on patient compliance and minimised GIT side effects. The controls received placebo tablets.
Ibuprofen significantly reduced gingival bleed-ing, colour change and pocketing in the test group augmenting periodontal treatment. This effect diminished rapidly after the drug regime ceased with no significant differences by 8 weeks. The authors stated that clinical applica-tions of ibuprofen were not justifiable and at-tribute this to the inability of ibuprofen to block lipoxygenase products which they believed may be the more prominent pathway of the two in early inflammatory reactions, (different NSAIDs do have different pharmacokinetic and pharmocodymamic properties (Brooks and Day 1991). Further factors which could have had an impact on the outcome was that the sample se-lected was not controlled for smoking and dis-ease severity may have varied between groups (at baseline). The authors recognised that the duration was too short and that the mode of de-livery may have affected the level of the drug in the gingival tissue, therefore affecting its effi-cacy. Ibuprofen is of moderate potency on cy-clo-oxygenase, explaining the moderate resolu-tion of inflammation. A further hypothesis was that NSAIDs were only effective in active dis-ease sites in the presence of acute inflammation (since increased cyclo-oxygenase and lipoxyge-nase are present in these sites) but not in quies-cent sites.
46

Ibuprofen
Study Observations and comments
(Haffajee et al. 1995) Double blind, longi-tudinal comparative study with 98 subjects aged range 14-71 with evidence of attach-ment loss. All subjects had a minimum of 20 teeth with at least 4 pockets >4mm and 4 sites with LOA > 3mm. The duration of study was 12 months with 2 month moni-toring intervals measuring six sites/tooth of all teeth present. The aim was to assess the effects of periodontal surgery and 4 system-ically administered agents (Augmentin, tetracycline, ibuprofen and a placebo) on clinical and microbiological parameters of periodontal disease. Two months into study individuals with LOA >2.5mm and pockets >4mm at one or more sites during longitu-dinal monitoring (this was taken as sign of disease progression) were treated by modi-fied Widman flap, scale and cleaning, 0.12% chlorhexidine and during the treat-ment phase they were randomly placed on an agent for 30 days, blind for the whole surgical period. Plaque, gingivitis, BoP, suppuration, PPD and PAL were measured. The clinician performing the surgery was different to clinician measuring all clinical parameters in a blind fashion.
Overall, the whole group gained attachment of 0.34mm with a pocket depth reduction of 0.6mm. However a few subjects in each treatment group had a poor response. The antibiotic groups had significant greater "gain" in attachment than ibuprofen or placebo groups with no statistically signifi-cant difference between Ibuprofen and placebo. Shallow pockets (< 4mm) lost at-tachment, intermediate pockets (4-6mm) showed moderate gain, and deep pockets (>6mm) showed the most gain. Adjunctive use of antibiotics during "active" disease pro-duced more effective outcomes than mechan-ical instrumentation alone. From this study one cannot discern if individuals without "ac-tive" disease would respond in the same way. The results of this study are contrary to past studies on Ibuprofen (Offenbacher et al. 1992; Taiyeb and Waite 1993). The main problems in this study were that there was a small sample size (6 subjects) in the Ibupro-fen group compared to the antibiotic groups (13 and 10 subjects), therefore affecting the statistical power and it is difficult doing mul-tiple comparisons (scaling, root planing, chlorhexidine rinses, oral hygiene and test agents) and drawing conclusions on inflam-mation, bleeding and attachment status. No history of smoking was given for subjects.
47

Ibuprofen
Study Observations and comments
(Ng and Bissada 1998) The aim was to investigate the efficacy of a systemic an-tibiotic (doxycycline) and ibuprofen, ad-ministered either separately or as adjunc-tive treatment to SRP.A placebo, con-trolled split-mouth study on 32 subjects (18 males and 14 females) aged 32-72 years with generalised moderate adult pe-riodontitis of 24 weeks duration. At weeks 3, 6, 12, 24 plaque, probing depths, (CAL) clinical attachment levels (with stents) were taken. Intraoral photographs and pe-riapical radiographs were taken 24 weeks following baseline scaling.
There were statistically significant gains in CAL for both doxycycline (0.4mm) and doxycycline/ibuprofen (0.5mm) groups and significant reduction of pocket depths (0.7mm) and gingival index scores. The control group with SRP had CAL gain of 0.5mm and reduction of 0.4mm pocket depths and gingival scores. Although there was improvement in the doxycycline and ibuprofen/doxycycline groups it was only modest in comparison to the improvement achieved by SRP. The ibuprofen only group had short-lived effects with only a 12-week gain in CAL, and only 6 weeks for reduction of pocket depths. These effects are similar to other findings (Feldman et al. 1983; Williams et al. 1989). Doxycycline achieves high levels in gingival tissues and possesses anti-collagenase activity, while the findings for ibuprofen could have been due to the low dosage.
48

Naproxen
Study Observations and comments
(Johnson et al. 1990) Double blind placebo controlled study where 102 pa-tients were given 500mg naproxen for 30 days. Investigating the effect of sys-temic naproxen on experimental gin-givitis.
This was the first human study on the gin-gival effects of naproxen. There was no ef-fect on plaque, gingival inflammation or bleeding indices at day 20, but on day 28 after plaque removal there was statistically significant improved resolution of gingival inflammation in the naproxen group by day 30. An explanation for these results is that the inflammatory pathway involves many different endogenous mediators that com-pensate for prostaglandin inhibition, namely leukotrienes, complement, cy-tokines and direct stimulation of inflamma-tion by lipopolysaccharides. In addition the authors stated that as long as plaque is present NSAIDs are not efficacious in sup-pressing inflammation due to other prod-ucts of the arachidonic acid metabolites. Naproxen may have a greater effect in in-hibiting the cyclo-oxygenase pathway in bone loss than preventing soft tissue in-flammation.
49

Naproxen
Study Observations and comments
(Jeffcoat et al. 1991) A double blind placebo controlled study with 15 patients having refrac-tory periodontitis. All patients were treated with scaling and root planing. Seven patients were given 500mg naproxen b.i.d. adjunct ther-apy for 3 months. Bone loss or gain was mea-sured with digital subtraction radiography, alveolar bone metabolism changes and alveolar bone height was measured radiographically us-ing intravenous radioactive methylene diphos-phate, which is an alveolar bone-seeking radio-pharmaceutical.
Naproxen decreased the rate of alve-olar bone loss in conjunction with periodontal therapy. Digital subtrac-tion radiography showed there was significantly less bone mass and height loss in patients taking the naproxen compared to placebo group. This sup-ported the Johnson et al., (1990) find-ings that naproxen is more efficacious in preventing alveolar bone loss than gingivitis.
(Offenbacher et al. 1990) An in vitro study on human pooled gingival tissue samples from 10 patients with severe periodontitis who had not received prior treatment with drugs. The aim was to see which agents (ketoprofen, in-domethacin, naproxen, ibuprofen, a-tocopherol, docosahexaenoic acid eicosapentaenoic acid, flurbiprofen, meclofenamate, sulindac and di-flunisal) were efficacious inhibitors of arachi-donic acid metabolism at different concentra-tions. By measuring the concentration of drug that caused 50% inhibition of maximum PGE2 synthesis in gingival epithelial and fibroblast cells. The reference agent was indomethacin since it is a moderately potent cyclo-oxygenase inhibitor.
This study found the following drugs to be inhibitors of cyclo-oxygenase from the most to the least potent; -to-copherol, ketoprofen, indomethacin, flurbiprofen, meclofenamate, naproxen, docosahexaenoic acid, eicosapentaenoic acid, ibuprofen, di-flunisal and sulindac. Naproxen was found to be a moderate inhibitor of PGE2 synthesis in vitro. The authors recognised the fact that in vitro tests have inherent problems and may not predict in vivo effects. Drugs that may be efficacious in preventing inflamma-tory changes in arthritis may not be ef-fective inhibitors of periodontal dis-ease.
50

Meclofenamate
Study Observations and comments
(Reddy et al. 1993) Double blind controlled clinical trial of 6-month duration. 22 subjects (7 male 15 females) with a mean age of 36.5 years. The aim was to determine the efficacy of meclofenamate as an adjunct to scaling and root planing in rapidly progressive periodonti-tis and disease active sites.
The meclofenamate groups had significantly less bone loss and more gain in attachment but the major improvement in the clinical pa-rameters was due to mechanical therapy; oral hygiene and or the Hawthorne effect may have contributed as well. The bone gains were similar to those seen in the naproxen study (Jeffcoat et al. 1991) The authors questioned the use of high dose meclofena-mate since there were gastrointestinal side effects.
Sulindac
Study Observations and comments
(Vogel et al. 1983) A randomised double blind study of 7 weeks duration on 18 male dental students with no pe-riodontal disease. Investigating the ef-fect of systemic sulindac on experi-mental gingivitis. All were taken to state of optimal gingival health. At day 0 of the treatment period the GCF flow, gingival inflammation and gingi-val bleeding were measured on se-lected teeth in the maxillary right quadrant, then all subjects randomly placed into 3 groups. One group re-ceived a placebo gel and capsule, an-other group received placebo gel plus 150mg sulindac capsule 2x/day. The third group received a placebo capsule, and 0.05% flucinonide gel (a steroid). All oral hygiene ceased for 22 days on the maxillary right quadrant. A biopsy of free gingiva on the mid-buccal sur-face of the maxillary right molar was taken for histological assessment
As plaque increased, all three groups showed an in-crease in GCF flow and inflammation. The topical steroid significantly inhibited gingival inflammation and systemic sulindac had no effects on GCF flow or inflammation clinically and histologically. Sulin-dac is a cyclo-oxygenase inhibitor, flucinonide in-hibits phospholipase limiting the availability of arachidonic acid metabolites from the cyclo-oxyge-nase and lipoxygenase pathway and in addition in-hibits immunologic responses. The authors gave three reasons for the findings:
(i) topical steroids may affect the plaque di-rectly
(ii) the treatment period of 22 days was too short for sulindac to have reached a gingival concentration level to inhibit inflammation
(iii) topical steroids have a greater concentration locally than systemic administration of an agent.
Inflammatory mediators peak at 2-3 months from onset and the response of alveolar bone to NSAIDs is delayed, with effects seen between 3-6 months.
51

Generally NSAIDs have been shown to reduce alveolar bone loss in humans, but their effect
on gingivitis is not clear, perhaps because different host response pathways operate in gingivi-
tis and in bone resorption. Since inhibition of COX may cause shunting towards other path-
ways eg pathway-2 or 3 (Sharma and Sharma 1997), drugs blocking one biochemical path-
way may have a variable effects in gingival tissue (e.g. ATLs), whereas the bone destruction
pathway is a more tightly coupled mechanism on which modulation of PGE2 could have more
profound and significant effects.
The ability of NSAIDs to reduce or inhibit bone and periodontal attachment loss are strongly
linked to PGE2 as the major mediator of bone resorption, in association with IL-1IL-6 and
TNF-(Offenbacher et al. 1993b). The NSAIDs studies show that the inhibition of COX
is advantageous since it may directly limit the bone resorption activity of PGE2 and modulate
the activity of the other synergistic agonists (Offenbacher et al. 1993b; Page et al. 1997).
2.7 Periodontal studies with aspirin
Few studies have investigated the effects of aspirin intake on periodontal status (Waite et al.
1981; Feldman et al. 1981; Heasman et al. 1990; Flemmig et al. 1996).
2.7.1 The Waite study:
(Waite et al. 1981)
There were 44 subjects participating, with an age range 22–68 years (mean age 45.8 years).
The test group consisted of 22 rheumatology patients from a London hospital (13 females and
9 males). One test patient was on aspirin, the rest were on indomethacin and/or
phenylbutazone. The duration of drug therapy ranged from 1-20 years, no dosage or drug
regime data were given. The control group had 22 subjects (14 females and 8 males) who
were office workers. All participants had to have all six Ramfjord teeth present. Subjects
were cross-matched for age and plaque levels. There were no significant differences in
52

periodontal attachment loss but gingival indices and pocket depths differed significantly.
Subjects taking aspirin, indomethacin and / or phenylbutazone had a lower gingival index
(GI), shallower pocket depths, with a pattern for less loss of attachment but not significantly
less. The author proposed that these effects were due to PGE2 inhibition principally based on
findings from past studies. Problems in this study were that the control subjects were from a
different source, the sample sizes were small, there was no control for age, sex and smoking
influences and the study relied on the subjects' memories of dosage and duration. In addition,
only one subject was on aspirin, making it of little value in understanding the effects of aspirin
on the periodontium.
2.7.2 The Feldman study
(Feldman et al. 1983)
This was a retrospective study investigating the effects of long term aspirin with and without
indomethacin therapy on alveolar bone levels in men only. The test group was composed of
75 subjects (mean age of 57 years) who had been taking high dose aspirin for more than 5
consecutive years for rheumatic problems. The control group consisted of 75 male volunteers
(mean age of 58 years) from an ongoing dental longitudinal study. Alveolar bone loss on the
mesial and distal surface of every tooth was assessed radiographically. Both groups were
matched for age and remaining dentition and denture use. The findings from this study were
that:
the test group had significantly fewer interproximal sites with 10% or more
alveolar bone loss (33.2) compared to the controls (37.2 sites)
the mean percentage bone loss per individual, although lower in the test group
was not statistically significantly different from the control group.
53

The authors concluded that the better periodontal conditions were probably due to the
inhibition of PGE2. However, it is not possible to differentiate between the effects of aspirin
and indomethacin, since some subjects used more than one prescribed medication over the 5
year period. Furthermore, the radiographs were not standardised (measurement error may
have had an effect on the results) and the dosages of NSAIDs were not constant over the five
years, (patients took from 1-12 tablets per day). The number of patients taking both aspirin
and indomethacin was not reported.
2.7.3 The Flemmig study
(Flemmig et al. 1996)
This was a double blind, placebo controlled, split-mouth study of 30 males and females, aged
30-65 with untreated moderate to severe adult periodontitis. Inclusion criteria was a mini-
mum of 18 teeth and each quadrant having one 6mm pocket or 4mm PAL. The primary aim
was to investigate the efficacy of 500mg (q.i.d) systemic acetylsalicylic acid as an adjunct to
scaling, using GCF-elastase levels as a measure of disease activity. A placebo was given for 6
weeks (q.i.d) to all subjects, establishing a baseline. Then a 6-week treatment phase of acetyl
salicylic acid was given to the test subjects and the placebo continued for the controls. Mono-
therapy of 2g/day of acetyl salicylic acid had no significant effects except in reducing GCF-
elastase. These findings were contrary to the Waite et al., (1981) findings for gingival in-
dex and probing pocket depths. On the other hand, the combination of acetyl salicylic acid
(inhibiting inflammatory response) and scaling (reduction in bacterial plaque) resulted in a
significant therapeutic efficacy, approximately equivalent to the sum of each individual ther-
apy in reducing gingival inflammation, pocket depth and probing attachment loss (p<.001) a
highly significant result. Combined therapy did not significantly increase the efficacy of
acetyl salicylic acid to reduce GCF-elastase. These findings indicated a decreased risk of
54

periodontal disease progression in subjects taking aspirin since GCF-elastase has been posi-
tively correlated with connective tissue and alveolar bone destruction (Zafiriopoulos et al.
1991).
2.7.4 The Heasman study
(Heasman and Seymour 1990):
This was a retrospective study investigating the effect of long-term use of NSAIDs on the
severity of periodontal disease. The test group comprised 50 rheumatology patients selected
from a Newcastle hospital and from general dental practitioners. There were 21 male and 29
female patients, with a minimum 2-year duration of NSAID therapy. The control group (16
males 26 females) were selected from subjects attending a Dental Hospital for a routine dental
check up. Both groups were matched for age, remaining teeth (20- 25) and plaque indices and
periodontal conditions around only the six Ramfjord teeth. No statistically significant
differences were found between the test and the control groups for plaque index, gingival
index, probing depth, loss of attachment, recession and alveolar bone loss. There was a
significant difference for gingival fluid flow. Some of the problems with this study were that
the test group had 72% of its subjects referred from general dental practitioners and the
controls were recruited from a dental hospital as well as dental practices, creating serious
sample selection error or bias, since the majority of subjects in both groups were referred from
dental practitioners. Less than 10% of test and control subjects had greater than 3mm probing
depths i.e. low prevalence of periodontitis. The partial recording of six Ramfjord teeth would
have influenced the findings; this study was poorly designed and the results should be
interpreted with caution.
55

2.8 Smoking and periodontal diseases
Smoking is now recognised as the most important cause of preventable death and disease
in the western world (MacGregor 1992). There are over 4000 chemical toxins present in
cigarette smoke, these include products such as carbon monoxide, oxidating radicals,
nitrosamines (carcinogens) and nicotine; over 50% of smokers will die of smoking-
related disease. On the other hand, smoking cessation has been associated with
significant improvement in life expectancy and decreased morbidity (U.S. 1990).
Cigarette smoking has been found to be an important risk factor for periodontitis (U.S.
Department of Health, 1990). Extensive studies and reviews on the association of
smoking and dental health demonstrate a clear association between smoking and the
prevalence and severity of periodontitis (Haber et al. 1993; Bergström and Preber 1994;
Zambon et al. 1996). Smokers have more tooth loss, deeper probing sites, more
attachment loss and more furcation involvement than non-smokers (Ismail et al. 1983;
Goultschin et al. 1990; Grossi et al. 1995). The relative risk of developing periodontitis
between smokers and non-smokers varies from 2.5-14 (Haber et al.1993; Schenkein et
al.1995). Generally, 50% of individuals who smoke have an odds ratio of 6-8 in favour
developing periodontitis (Salvi et al. 1997).
Heavy cigarette smoking (> 30 pack/year history of smoking) is one of the highest risk indica-
tors for periodontitis; the greater the exposure in terms of pack years, the greater the amount
of alveolar bone loss (Grossi et al. 1996). Smoking is a risk factor for rapidly progressive pe-
riodontitis as well as adult periodontitis (Haber and Kent 1992). The exact mechanism of in-
creased smoking-related susceptibility to periodontitis is unknown (Johnson 1998). Studies
show that smoking exerts both local and systemic effects on the oral tissues, vasoconstriction,
by nicotine, depression of various arms of the immune system and affects gingival fibroblast
function and bone metabolism (Preber and Kant 1973; Preber and Bergström 1986; McGuire
56

et al. 1989; Ah et al. 1994; Preber et al. 1995; Tipton and Dabbous 1995; Salvi et al. 1997;
Bando et al. 1998; Johnson 1998; Ryder et al. 1998).
2.8.1 The periodontal effects of past smoking and smoking dose
The degree of periodontitis in ex-smokers lies in between current smokers and non-
smokers and is dependent on the dosage (pack years) of smoking (Tonetti 1998). Past
smokers tend to have similar response to treatment as non-smokers (Kaldahl et al. 1996).
Current smokers have less probing depth reduction after periodontal treatment than do
ex- and non-smokers. The response in pocket depth reduction and CAL were similar in
former smokers and non-smokers (Grossi et al. 1997). There was no correlation between
the number of years of smoking cessation and therapy response, suggesting that there are
immediate periodontal benefits from quitting smoking. A similar finding has been
reported in relation to the success rate of dental implants (Bain and Moy 1993). Smokers
who quit one week prior to implant placement and started smoking again 8 weeks after
implant placement had failure rates similar to non-smokers. Heavy smokers quitting
smoking in the first 2 weeks following thick free gingival grafting had a comparable
response in terms of root coverage to non-smokers (Miller 1987); it was also noted that
patients who smoked < 5 cigarettes/day did not have a negative impact in response to this
type of therapy. Smoking history also affects tooth loss - for a given age, ex-smokers lie
between non-smokers (least tooth loss) and smokers (most tooth loss) (Österberg and
Mellström 1986). Quitting smoking greatly reduces risk to general health; once a patient
quits, one can also expect a normal response to periodontal therapy (although we cannot
reverse the past effects of smoking). Unfortunately, smoking behaviour is not easily
changed since nicotine is an addictive drug (Johnson 1998).
57

2.9 Periodontal measures
Lindhe et al.(1986) stated that any trial to study periodontal disease to assess the effect of
a certain treatment procedure on human periodontal disease must take into account:
the nature of periodontal disease
the progression of periodontal disease
the measurement of efficacy of therapy
the experimental unit
the goals of periodontal therapy.
Plaque and gingival indices are traditional measures used to measure the presence and
severity of gingivitis and plaque (Loe et al.1965; Haffajee et al. 1983; Mombelli et
al.1987; Ainamo 1988; Listgarten 1988; Claffey et al. 1990; Haffajee et al. 1991;
Armitage 1996; Newbrun 1996). Bleeding on probing, pocket depth and attachment loss
are important measures of periodontal diseases (Lindhe et al. 1986; Armitage 1996).
They provide useful information regarding the location, presence or absence of diseased
tissues and the absence of these conventional signs is strongly indicative of a healthy,
stable periodontium (Lang et al.1986). These commonly used diagnostic procedures
have significant weaknesses. Although inflammation may be indicative of disease, it
gives no indication of severity, morbidity, or eventual outcome of periodontal infections.
The clinical signs of inflammation are unreliable in differentiating between non-
destructive and destructive forms of periodontal disease, current disease activity and the
cause of the attachment loss. Loss of periodontal attachment only measures past
episodes of disease. The parameters measured in this study were gingival bleeding on
probing (indicative of inflammation), gingival recession and periodontal pocketing.
58

2.9.1 The experimental unit
In medical research, the individual or subject forms the unit of examination whereas in
periodontology, recording units may be site specific (Lindhe et al. 1986; Ainamo 1988). The
indications for using periodontal sites as the unit are that different sites:
in the same individual show different patterns of disease severity, progression
and lesion morphology
often respond differently to periodontal therapy.
2.9.2 Measurement of extent and severity of periodontal attachment loss
Epidemiology is the science concerned with the factors that influence the distribution and
occurrence of health, disease and mortality among groups of individuals (ranging in size
from small populations to large sub-national aggregates to entire countries). A frequent
measure of disease used in epidemiology is prevalence - the proportion of individuals in
a group who exhibit the condition at a given point in time (Fletcher et al. 1988). The
total number of individuals with a given condition is also frequently referred to as the
prevalence. Prevalence is estimated from cross-sectional studies.
The periodontal status of an individual, let alone a group, is difficult to describe
concisely. To thoroughly characterise periodontal status, epidemiological studies assess
numerous sites in both dental arches for each individual. Epidemiological assessment of
a group of individuals requires a summary of the periodontal status of the individual sites
in the mouth. Development of summaries must deal with the difficult issue of case
definition as well as the number and types of sites to assess. The effect of fallible
measurement techniques is also an important issue and will be discussed later. Before
the prevalence of a "periodontal disease" can be described, the disease must be defined.
This requires consideration of severity (such as the amount of inflammation or
59

periodontal attachment loss) as well as the extent (the percentage or number of affected
sites). Although all epidemiological studies use some combination of the concepts of
prevalence, extent and severity to describe the periodontal status of a population, various
studies have assessed these concepts differently. It is difficult to compare the results
from studies that use different measurement methods or case definitions and comparisons
can therefore be misleading.
Epidemiological studies have used a variety of indicators and indices as a measure of
periodontitis. Earlier indices included the Periodontal Index (Russell 1959) and the
Periodontal Disease Index (Ramfjord 1959). These indices were not satisfactory or
completely accepted; both present a single mean index score for an individual or group,
which used an estimate of disease severity, but ignored the extent or distribution of the
disease. The World Health Organisation proposed the use of the Community Periodontal
Index of Treatment Needs (CPITN) (Ainamo et al. 1982; Cutress et al. 1987; Ainamo
1988) as an index of rapid assessment of periodontal treatment needs in a population.
However, this index only records the "worst score" for each of the six segments of the
mouth, and ignores much information obtained from a clinical examination. The CPITN
does not quantify loss of attachment (Slade and Spencer 1995). Direct measurement of
periodontal attachment loss has only recently been used in periodontal research, despite
having been proposed two to three decades ago as an important measure of disease
(Ramfjord, 1959; Glavind et al.1967). The most appropriate epidemiological indicator of
periodontal destruction is based on the measurement of loss of attachment (through its
components, gingival recession and pocket depth) (Brown and Garcia 1994; Slade and
Spencer 1995).
60

2.10 Null hypotheses
1. There is no difference in periodontal attachment loss between ex-smokers and
non-smokers irrespective of their aspirin status.
2. There is no difference in periodontal attachment loss between low-dose aspirin
takers and non-aspirin takers irrespective of their smoking history.
Level of significance
Rejection of the null hypothesis for all tests of significance was set at a probability value of alpha = 0.05.
Aims of the study
Specific aims:
1. The primary aim of this study was to assess the severity and extent of periodontal
attachment loss in males over 50 years of age with a history of long-term low-
dose aspirin therapy and compare to non-aspirin takers using a cross-sectional
design.
2. The associated aim of this study was to assess the severity and extent of
periodontal attachment loss in males over 50, with a past history of smoking and
on long-term low-dose aspirin therapy and compare them to non-smokers using a
cross-sectional design.
Secondary aims:
(i) To assess the case definition of severe attachment loss within the study
population.
(ii) To extrapolate PAL from the above defined study population to the general male
population of the metropolitan Adelaide area
(ii) To evaluate the association between education, pension status, and oral hygiene
habits with the above primary aims.
61

Chapter 3 Materials and methods
The University of Adelaide's Human Research Ethics Committee gave ethical approval for this
study. The approval number was: H/35/97.
3.1. Sample selection
A self-selected sample of males aged 50 years and over from the general population
living in Adelaide (state capital) of South Australia made up the study population.
Subjects were recruited through local press media advertising (Figure 3.1).
Figure 3.1 A copy of an advertisement placed in local press media to recruit
subjects
MEN !
Are you over 50?
Would you like a free Colgate oral care package?
Do you have at least 6 teeth of your own?
Do you take aspirin regularly?
Are you an ex-smoker or a non-smoker?
If you answered "YES" to any of these questions, we want you to take part in a research project looking into the dental health of MEN ONLY.
All you have to do is answer a short questionnaire about your health and have an examination.
HOW? -Ring Thursday, or Friday 8303 3436 for further information and to make an appointment.
WHERE? - Colgate Australia Clinical Dental Research Centre, Frome Road, Adelaide.
Unfortunately we can't include you if you are a current smoker, or have Rheumatic fever, a heart valve defect, a pace-maker, joint replacement, severe arthritis, cancer, liver or kidney disease.
62

Advertisements were placed in the following newspapers: The Advertiser,
Messenger.and the Mature Times. Table 3.1 lists the study's inclusion and
exclusion criteria.
Table 3.1 Inclusion and exclusion criteria
INCLUSION CRITERIA: 2.6.3 EXCLUSION CRITERIAMales onlyAll participants must be aged 50 years
and overMinimum of 6 or more natural teeth. All participants had to complete and
sign an ethical consent form. Anyone on aspirin therapy for longer
than 2 years and never smokedAnyone on Aspirin therapy for longer
than 2 years and an ex-smoker for longer than 2 years.
Anyone not on aspirin therapy and never smoked.
Anyone not on aspirin therapy and ex-smoker for longer than 2 years
Current smokersPeople requiring antibiotic cover (artificial
heart valves, congenital heart defects, rheumatic fever, and joint prosthesis) for dental examinations.
Drug exclusion: People on long term:- Steroids- Non-steroidal anti-
inflammatory drugs (other than aspirin),
- Dilantin Immunosuppressants
- Anti-coagulantsHistory of extensive antibiotic therapy in the last three months- Chlorhexidine mouth washes
Systemic diseases: People suffering from
- Diabetes - Rheumatoid arthritis- Hepatic or renal diseases- Cancer
People with pace makersPeriodontal surgery within the last five
yearsLess than 6 natural teeth
Subjects matching the inclusion criteria for both test (aspirin takers ex- and non-
smokers) and control groups (non-aspirin takers ex- and non-smokers) were offered
the opportunity to participate by telephoning the Colgate Australian Clinical Dental
Research Centre (CACDRC) located in the Adelaide Dental Hospital. Reception
staff conducted a further screening (of inclusion and exclusion criteria) giving a
brief description of the study over the phone, prior making appointments. The staff
63

underwent a continuous education program and discussions about the study and
given strategies to encourage subject participation.
When subjects attended their appointment, the reception staff handed them an
information sheet (Appendix A), an ethics consent form (Appendix B) and a
questionnaire. After the subjects read the information leaflet they signed a consent
form which was later signed by the investigator and an independent witness. On
completion of the periodontal examination, participants received a "Findings form"
(Appendix C), a Colgate Oral Care Kit (containing a toothbrush, toothpaste, dental
floss and pamphlet on oral care), a copy of the signed consent form and the
information leaflet.
3.2 Questionnaire
The questionnaire collected demographic information, medical and dental histories. To
simplify the subjects' task and to reduce time, a multiple-choice format was used for all
questions (Appendix D). This format standardised the data and facilitated statistical
analysis. The questionnaire was self-administered in the CACDRC waiting room to
avoid examiner's / interviewer's bias or influence (Abramson 1974; Bourque and Fielder
1995; Fink 1995a). When requested by the subjects to clarify questions, concepts or
where there was a language/reading difficulty, CACDRC staff gave assistance. The
questionnaire sought a variety of information (Table 3.2).
64

Table 3.2 Aims of questionnaire.
demographics
socioeconomic status
medical history
aspirin history (type, dosage and duration)
smoking history (dosage, duration and term of cessation) denture use
the subjects oral hygiene habits and oral care
past experiences of periodontal treatment
Question format and wordings were selected from the W.H.O. Oral Health Surveys Basic
Methods (W.H.O. 1997), Australian Bureau of Statistics (Census) (McLennan 1996) and the
South Australian Dental Longitudinal Study Five year follow-up Questionnaire (Spencer
1997).
Questions 5 and 6 (see Appendix D) were designed as a final cross-check to exclude subjects
who fell outside the study's selection criteria as well as collecting subjects' medication history.
History of aspirin use was assessed in questions 7-11. Question 7 elicited if subjects were on
aspirin and the type or generic brand of aspirin. Questions 8 & 9 recorded the frequency of
intake and daily intake. Question 10 recorded the dosage in milligrams and question 11
focussed on the duration of aspirin therapy, (relying on the subjects' recollection). No
information was obtained as to whether aspirin intake was by prescription or over the
counter. Smoking history was assessed in questions 12-16 in a similar format, again relying
on the subjects' recollection of the time since cessation, age of smoking debut and number of
cigarettes that they used to smoke per day. Dental habits were elicited by question 17-24.
Oral hygiene habits were recorded in answers to questions 20-22. Question 23 recorded the
65

subjects' actual regularity of dental visits. Question 24 measured the subjects' regularity of
periodontal care. Question 25 elicited which subjects had had periodontal surgery.
3.3 Oral Examination
The objectives of the oral examination were to determine the teeth present and the
gingival and periodontal status of the subjects in a descriptive epidemiological form. The
oral examinations were conducted by one dentist, blind to the subjects' aspirin and
smoking histories and were based on the methodology used in Slade et al., (1995) and
Flemmig et al., (1996) with some modifications to the periodontal assessment as follows:
(i) all erupted teeth (including the third molars) were assessed
(ii) measurements of gingival recession and periodontal pocket depth were
made at six sites around each tooth i.e. mesio-buccal, mid-buccal and disto-
buccal, mesio-lingual, mid-lingual and disto-lingual.
FDI notation was used during the examination. A custom written computer program was
set up in the CACDRC for nursing staff to directly input all measurements as called out
by the examiner during the examination. All survey details were entered at the
completion of the dental examination by the nursing staff.
3.4 Clinical measurements
The following clinical variables were assessed:
3.4.1 Plaque Index
Using the Silness and Löe plaque index (Silness and Löe 1964), plaque scores of the
"Ramfjord teeth" ie 16, 21, 24, 36, 41, and 44 were entered directly into the computer
program and an overall Plaque Index (PI) score was computed for each subject (Silness
66

and Löe 1964). The PI was an ordinal scale from 0-3 and each score quantified
accordingly (Newbrun 1996):
Table 3.3 Plaque index
(Silness and Löe 1964)
0 Gingival area of tooth free of plaque; the surface is tested by running a probe across the tooth surface, if no soft material adheres, then its consid-ered plaque free.
1 No plaque observed in situ by the unaided eye, but plaque is made visible on the point of a probe after the probe has been moved over the tooth surface at the entrance of the gingival crevice.
2 Gingival area is covered by thin to moderate thick layer of plaque visible to the naked eye.
3 Heavy accumulation of soft matter, the thickness of which fills the crevice produced by gingival margin and tooth surface.
3.4.2 Calculus
A dichotomous index of calculus was recorded per tooth for every tooth present. No
distinction was made between supra- and sub-gingival calculus. Calculus detected
visually or by probing was given a value of "1" and if absent, was scored "0", giving a
reading by tooth, rather than by tooth surface or specific site.
3.4.3 Bleeding index.
The modified Sulcular Bleeding Index (mSBI) was used as a quantitative measurement
of gingivitis see Table 3.4.3 (Mombelli et al.1987)
Table3.4 Modified Sulcus Bleeding Index (mSBI).
Grade Description ((Mombelli et al.1987; Newbrun 1996)
0
1
2
3
No bleeding when a periodontal probe is passed gently along the gingival margin
Isolated bleeding spots visible
Blood forms a confluent red line on margin
Heavy or profuse bleeding
67

This index gives a recording per tooth rather than by a subject's gingival sites. The
surfaces recorded per tooth were the lingual surfaces of the first and third quadrants and
the buccal surfaces of the second and fourth quadrants. The mSBI entails a gentle
probing action at the gingival crevice/sulcus with a probe angulation of 600 to the long
axis of the tooth (Newbrun 1996). This is a more sensitive indicator of gingival
inflammation and less likely to elicit false-positive bleeding than does probing to the
bottom of the pocket (van der Weijden et al. 1994a; van der Weijden et al. 1994b).
Gingival bleeding was considered to be a better estimator of the efficacy of oral hygiene
than plaque scores, which only indicate the amount of plaque present. Bleeding scores
better reflect a subject's day-to-day level of plaque control. The use of the mSBI index
necessitated the need to standardise the lighting to have a consistent visual assessment of
the gingiva.
3.4.3 Tooth mobility
Tooth mobility was recorded for every tooth using the Miller notation (Miller 1950)
(Table 3.5). The method used was by placing a mirror handle on the tooth and buccal,
lingual and occlusal force applied and recording its movement.
Table 3.5 Tooth mobility index
Score Description ( Miller 1950; Grant et al. 1988a)
0 No detectable movement when force is applied
1 Barely distinguishable movement when a force is applied.
2 The crown of tooth moves 1mm in any direction
3 Movements of more than 1mm in any direction
68

3.4.4 Furcation involvement
Furcation involvement was measured using an ordinal scale (Table 3.6) measuring
attachment loss based on horizontal probing characteristics (Hamp et al. 1975; Grant et
al. 1988b).
Table 3.6 Furcation index
Class 1 The degree of attachment loss involves only the furcation entrance by the periodontal probe or explorer.
Class II The degree of attachment loss extends under the roof of the furcation but does not penetrate through and through.
Class III The degree of penetration by the probe or explorer is through and through
3.5 Details of the study
3.5.1 Periodontal attachment loss (PAL)
Sterile, non-pressure sensitive NIDR periodontal probes (American Dental, Chicago Il.)
were used, to measure gingival recession and periodontal pocket depth at each site. The
NIDR periodontal probe is calibrated into 2mm bands, which can create examiner error
when truncating fractional band measurements. Fractional band measurements were
truncated to the lower whole millimetre, including to odd numbers. PAL was calculated
by adding the measurements for pocket depth and recession. Negative measurements of
recession (like hypertrophy or pseudopockets) were subtracted from probing pocket
depths.
Measurements were excluded from the study when the cemento-enamel junction (CEJ)
could not be visualised or when pockets could not be probed (for example when large
calculus deposits were present).
69

3.5.2 Periodontal Pocket Depths (PPD)
Periodontal pockets depths were measured from the gingival margin crest to the pocket
base. Measurements of gingival recession and periodontal pocket depth were made at six
sites per tooth.
3.5.3 Gingival Recession (GR)
Recession was measured from cemento-enamel junction to the gingival crest for all 6
sites. If the CEJ was not evident these measurements were excluded.
3.5.4 Examiner standardisation:
Prior to starting the trial, the examiner carried out repeat examinations on six individuals
(2-days apart) to establish intra-examiner error, to ensure that the recordings were made
with uniform interpretation and consistency (W.H.O. 1997).
3.5.5 Procedure
The examinations were carried out in the CACDRC in the Adelaide Dental Hospital.
After the questionnaire had been completed, the subject was escorted to the dental
surgery by the nursing staff, introduced to the examiner, and seated on a dental chair.
The patient was given a brief explanation of the oral examination procedure with a
pictorial description of the probing procedure (Berns 1993). The dental chair was
reclined. A mouth mirror and probe were set out for every patient, oral examinations
were performed under overhead illumination. The dental assistant recorded all readings
directly onto a computerised dental chart. First, all teeth missing were charted, followed
by plaque scores. The next procedure was to record the mSBI on all teeth. Then six sites
per tooth were measured for GR and PPD in the following order: distal-buccal site,
direct-buccal site, mesial-buccal site, mesial-lingual site, direct lingual site and the distal-
70

lingual site. These recordings were for all maxillary and mandibular teeth present. The
next recordings were of teeth with supra and subgingival calculus. Recordings of
furcation involvement and tooth mobility completed the examination.
3.6 Statistical methodology
The data were analysed using SPSS for Windows® 8.0.0 (SPSS Inc.), using the General
Linear Model General Factorial Procedure, incorporating Scheffe's test and Pearsons
Chi-Square tests where appropriate. A statistical model was used to measure the effects
of aspirin and ex-smoking on PAL. The general linear model used was a two-way
ANOVA including the interaction term, with age (in years) as a covariate included. If a
main effect was found to be not significant, the model was further reduced by removal of
that term/variable. Tables only present the reduced models. The level of significance
was calculated at alpha= 0.05. This model was applied to all measures of severity and
extent of PAL and on two new measures of severity of PAL, the mean of the most severe
site (MSS-PAL) per person and the extreme worst site (EWS-PAL) per person.
71

Chapter 4 Results
All tables appear at the end of this chapter (pages 87 to107).
4.1 Intra-examiner error.
Repeat examinations were carried out in 6 males 50 years of age measuring PAL at 630
sites (2 days between examinations) to establish intra-examiner error using the same
recruiting and examination procedures that were used in the subsequent study. The
accuracy of reproducibility of measurements was analysed using kappa statistics. Table
4.1 indicates that there was a very high agreement with a corresponding overall kappa
coefficient of 0.94 0.01(se) (values > 0.80 are classified as high (Hunt 1986)).
No prior sample size or power calculation planning was done. However, one long-term
study (Feldman et al.1983) investigated the effects of high dose aspirin on the
periodontium of 75 males. It was decided that an estimated sample size of 100 subjects
per group would probably be sufficient to investigate the independent variables of aspirin
and past smoking on PAL. The study commenced in June 1998 and examinations were
completed by early February 1999.
4.2 Profile of study population:
A total of 392 subjects participated in the study: they were classified into the following
four groups according to aspirin and smoking history:
Aspirin Never Smoked (ANS)
Aspirin eX-Smokers (AXS)
No Aspirin Never Smoked (NANS)
No Aspirin eX-Smokers (NAXS)
72

Table 4.2 shows the number and percentage distribution of subjects within each group.
The non-aspirin groups had the highest number of subjects (239). The aspirin groups had
153 subjects participating and of these, the ANS group had the lowest number of subjects
(51). Demographics and oral health behaviours (dental hygiene and maintenance) were
used to sub-claasify the distribution of subjects within each group.
4.3 Demographics
4.3.1 Age categories of subjects
The mean age for all subjects was 63.4 years ages with a range from 50-85 years
(Table 4.3). In addition 55.9% (219) were under the age of 65 years and 44.1% (173)
were 65 years. The ANS and AXS groups had the highest mean ages when compared
to NANS and NAXS groups (Table 4.3). The mean age of the ANS group was 68.2
years, representing the group with the highest proportion of elderly subjects 65 years.
The non-aspirin groups had more subjects 64 than 65 in approximately a 2:1 ratio.
One way ANOVA indicated that there were significant differences between the four
groups for mean age i.e. there was an uneven age distribution between groups. Multiple
comparisons for age differences between groups were analysed using a Scheffé's test
which found significant mean age differences between groups, the NANS being (6.1
1.06 years) younger than the AXS group (p=<0.001). Additionally, the NANS group
was 8.3 1.31 years younger than the ANS group (p <0.001). The mean difference
between the ANS and the NAXS groups was 5.6 1.32 years, with NAXS being the
younger group (p=0.001) (Table 4.4). A Scheffé's analysis on the homogeneity between
two groups at a time found that there was no significant difference in mean age between
the groups of subjects taking aspirin (ANS AXS) (p=0.324), no significant difference
73

between the non-smoking groups (NANS NAXS) (p=0.157) and no significant
difference between the ex-smoking groups (NAXS AXS) (p=0.052) (Table 4.5).
As the study targeted males over 50, it was assumed that many participants would be of
or into retirement age. The minimum age qualifying for an aged pension for males in
Australia is 65 years. Pension status was taken as a gross indicator of income, as
pensioners are generally of a lower income than non-pensioners. Table 4.6 presents the
distribution of all subjects based on pension status; of all subjects 58.9% were
pensioners. The two aspirin groups had the highest number of pensioners (108)
compared to non-pensioners (45). Table 4.7 shows denture use in pensioners and non-
pensioners: 79.4% of non-pensioners did not wear a denture compared to 58.9% of
pensioners.
4.3.2 Education status of the subjects.
Of the 392 subjects, 80.9% had at least secondary or higher education. The highest
educated group (tertiary) was the NANS group (41.0%) with the NAXS group having the
lowest percentage of tertiary educated subjects (20.5%) (Table 4.8). The ANS group
had the highest percentage of subjects with the lowest education level (no schooling plus
primary levels) (23.5%).
English language skill was a further measure of education. Table 4.9 shows that 94.6%
of the study population stated that they spoke English well to very well. Data from
Tables 4.8 and 4.9 show that a large proportion of subjects had high education levels and
had good English language skills. These attributes have been shown to help in the
socialisation of subjects and correlate positively with good oral health (Srikandi 1982).
74

4.3.2 Oral health behaviour
Table 4.10 shows the distribution of all subjects based on demographics and dental
behaviours. Pensioners tended to brush more frequently (> 1/day), than non-pensioners.
The more highly educated subjects (93.7%) brushed more frequently than once per day
compared to lower educated subjects (6.7%). Subjects who had visited the dentist more
frequently tended to brush more than once per day (92.5%); while 57.9% of subjects who
had not visited the dentist in the last 5 years claimed that they brushed more than once
per day.
A measure of the subjects' oral health awareness was the time interval since their last
dental visit. Approximately 56.7% of the study population had visited the dentist in the
last 12 months and 33.6% between 1-5 years previously (Table 4.11). Only 9.8% had not
seen a dentist for more than 5 years, indicating that this group was reasonably dentally
aware. The group with the lowest rate of dental attendance rate NAXS, with 31% having
not attended for at least 3 years. The majority of the study population reported that they
brushed their teeth at least once per day, hardly ever used a mouth rinse, flossed less than
once a week and most had a scale and clean within the last two years.
Chi-Square analysis (Pearsons Chi-Square) between pensioners and non-pensioners
found no significant differences between groups in the distribution of tooth brushing,
mouth rinses and flossing (p>0.05). There was a significant difference between a
subject’s last scale and clean visit with pension status. Overall 60% of subjects had a
scale and clean in the last two years, of these, significantly more pensioners (53.7%) had
a scale and clean in the last 2 years compared to non-pensioners (45.0%) (p=0.043).
There were no significant differences in mouth rinsing habits based on education, but
there were significant differences in tooth brushing (p<0.001), flossing (p=0.04) and last
75

scale and clean visit (p<0.001). Subjects who had visited the dentist within the last 12
months were significantly more likely to brush once or more per day (p<0.001), floss
more often (p<0.001) and have had a more recent scale and clean than subjects who
visited the dentist less frequently (p<0.001). There were no significant differences
between 64 and 65 year-olds for all the above parameters (Table 4.10).
4.4 Tooth loss
All 392 persons examined were dentate. The association between age and tooth loss is
shown in Table 4.12. The mean number of missing teeth for all subjects was 9.5 0.31
(se); subjects aged 65 had a significantly higher mean number of missing teeth (11.7
0.50 se) compared to subjects aged 64 (7.8 0.37 se) p=0.001. Both ex-smoking
groups had more missing teeth (10.2 0.43 se) than the non-smoking groups (8.6
0.45 se) (p=0.010) (Table 4.13). Aspirin had no significant association with the mean
number of missing teeth.
4.5 The periodontal status of the study population
Table 4.14 is a descriptive table of the mean plaque scores for each group and by age.
The overall mean plaque index as shown in Table 4.14 was 1.4 0.69 (sd), with the 64
year olds having a mean plaque score of 1.3 0.70 (sd) and 1.6 0.67 (sd) for 65
year olds. The younger NANS group had the lowest plaque score (1.2 0.61 sd)
compared to the older NAXS group (1.7 0.69 sd).
Table 4.15a shows the range and generic names of aspirin use in the current study. The
two primary compositions used were aspirin and acetyl salicylic acid. Thirty four
subjects used acetyl salicylic acid and 119 were on aspirin. Only one subject used the
generic brand of Ecotrin, which is supplied as a 650mg tablet (this subject took a half a
tablet per day). The majority of subject (95) were on 300mg of aspirin per day compared
76

to 57 subjects on 100mg of aspirin per day. The most popular brands were Solprin (63
subjects) and Cartia (33 subjects). No information was gathered regarding whether
brands of aspirin had been switched during the period of intake.
The hypothesis that there was no difference in plaque scores between the different age,
aspirin and smoking history groups was tested using ANOVA analysis. A two way
ANOVA with mean age as the covariate showed there were no significant differences in
plaque scores between aspirin takers and non-aspirin takers and so aspirin was removed
from the model. A further one way ANOVA of smoking history with age as the
covariate showed a significant age association with mean plaque scores, with every one-
year increase in age having a significant increase of 0.02 in the mean plaque score
(p<0.001) (Table 4.15b). Ex-smoking subjects had significantly higher plaque scores by
an average value of 0.14 0.07 (se) than non-smoking subjects (p<0.043) (Table 4.15b).
Figure 4.1 shows the distribution of gingival bleeding. Overall, 57.7% of teeth had
associated gingival bleeding; the ANS subjects aged 64 had the lowest percentage of
sites with bleeding 49.7%. However two-way ANOVA analysis found no significant
associations for aspirin (p=0.310) or ex-smoking history (p=0.424) with gingival
bleeding between the four groups.
The descriptive Table 4.16 presents the mean percentage of teeth with calculus among
groups, aspirin takers and the ex-smokers. The inferential analysis (using two-way
ANOVA) found no significant associations of aspirin and ex-smoking history with the
mean percentage of teeth with calculus, other than for age associations. An ordinary
least square regression was conducted, showing that age was significantly correlated with
calculus, with older subjects having more teeth with calculus across all groups (p=0.042)
77

(Table 4.17). Older subjects had significantly more teeth with calculus than younger
subjects but these correlations were minor (0.78% per year increase) (Table 4.17).
Two-way ANOVA found no significant associations of aspirin with tooth mobility
(p=0.139); no table is shown since there was no statistical association. A further analysis
(one-way ANOVA) found that ex-smokers overall had a statistically significantly higher
mean percentage of teeth with mobility compared to non-smokers (indicating worse
periodontal status compared to non-smokers), independent of age associations (Table
4.18).
ANOVA analysis showed that there were no significant associations of age (p=0.168),
aspirin (p=0.550) and smoking history (p=0.910) on furcation involvement between
groups.
4.6 Associations of aspirin and ex-smoking with various measures of PAL
4.6.1 The associations of aspirin and ex-smoking with mean PAL
The hypotheses that there were no differences in periodontal attachment loss between
aspirin takers-non-aspirin takers and ex-smokers-non-smokers was evaluated using mean
PAL and ANOVA analysis. The estimated means and test of significance were analysed
using two-way ANOVA evaluated at the mean age covariate (63.4 years). There were
significant associations of low-dose aspirin and ex-smoking on mean PAL. Ex-smokers
had significantly more PAL (2.9 0.07mm) compared to non-smokers (2.6 0.08mm),
independent of aspirin history. In addition, low-dose aspirin takers had significantly less
PAL (2.6 0.08mm) compared to non-aspirin takers (2.9 0.06mm) and this
association was independent of past smoking status (Table 4.19). These results reject the
null hypotheses that there were no statistical differences in PAL between the groups.
Controlling for age, this model showed that ex-smoking had an association with mean
78

PAL with a magnitude of 0.4mm. At a given point in time, ex-smokers had 0.4mm more
PAL than non-smokers while aspirin takers had approximately 0.2mm less PAL than
non-aspirin takers (Table 4.19). The same analysis was performed on mean PAL by
dosage and duration of low-dose aspirin; no differences were found (Table 4.20 and
4.21).
A further analysis of the association of dosage and duration of past smoking on mean
PAL was carried out using ANOVA. Tables 4.22a & b show that there were significant
associations between mean PAL and the number of cigarettes smoked and past duration
of smoking years. There was significantly more mean PAL in subjects who smoked
more than 10-30 cigarettes per day compared to those smoking <10 cigarettes per day
(p=0.013). In subjects who smoked >30 cigarettes per day the association was highly
significant (p<0.001) compared with subjects who smoked <10 cigarettes per day.
Subjects who smoked 10-19/day had more mean PAL by 0.5mm than non-smokers while
those who had smoked 20/day had 0.74mm more mean PAL than non-smokers.
Subjects who had smoked 5 years had significantly more mean PAL (0.42 mm) than
subjects who had smoked for <5 years (0.35mm) (p=0.036). The majority of ex-smokers
(196) had quit smoking for 5 years; 17 had quit smoking in the last 5 years. There was
no association between mean PAL and the time since quitting smoking (p=0.077) (Table
4.22b).
4.6.2 The associations of aspirin and ex-smoking on the extent and severity of PAL
The general linear model of two-way ANOVA with one covariate was applied to
measure the associations of low-dose aspirin and ex-smoking at various thresholds of
severity (2 to 7mm) of PAL (Table 4.23). Aspirin and ex-smoking had significant
associations at threshold levels, 2 to 4 mm mean PAL. Table 4.23 shows that as
79

severity increased, the number of subjects with severe PAL decreased. At 5 mean
PAL, there was no significant association of aspirin with PAL (p>0.05) while there was
borderline significance (p=0.044) for ex-smoking history.
The threshold of 5mm and 6mm PAL showed no statistically significant correlation
between aspirin and mean PAL. The values for 6mm PAL were not included in the
table since no significant statistical associations were seen. The threshold 7mm mean
PAL had no significant statistical associations but was included since this measure is
indicative of severe attachment loss and is often used in studies of mean PAL. Only 191
subjects had mean 7mm PAL, probably explaining the lack of significant associations
of aspirin and ex-smoking on severe PAL.
The same analysis was performed for the extent of disease. Table 4.24 summarises the
associations of aspirin and ex-smoking using the statistical analysis of the estimated
mean percentages of PAL at 2, 4 and 7mm PAL and the corresponding p-values.
Aspirin and ex smoking had significant statistical associations with the extent of PAL
from threshold levels 2 to 7mm mean percentage PAL. At the low mean percentage
of 2mm PAL, there was an interaction association of aspirin on ex-smokers and non-
smokers. Analysed separately, there was no significant aspirin association with PAL
2mm in ex-smokers, however there was a significant aspirin association with PAL
2mm in non-smokers.
Table 4.25 summarises the magnitude of the significant statistical associations of aspirin
and ex-smoking for severity and extent of PAL. The negative scores for aspirin reflect
less PAL at various levels of PAL for severity and extent, independent of smoking
history. Ex-smokers had a greater magnitude of PAL loss for the same PAL levels than
non-smokers. For example, ex-smokers at 4mm PAL had more overall mean loss of
80

PAL (0.2mm) and an extra mean 9.1% of sites with 4 mm PAL than non-smokers.
Aspirin takers overall had less mean loss of PAL (0.1 mm) and less sites (5.1%) at mean
4mm PAL than subjects who had not taken aspirin.
Further to this, the mean number of teeth present per subject was 22.5 with a mean total
of 135 sites per person (22.5 x 6 sites). At mean PAL 4mm, each ex-smoker had a
mean of 45 sites (33.3%) with a mean PAL of 4.7mm in severity (Tables 4.23). Aspirin
takers had an average of 35 sites (26.2%) of mean PAL 4mm (Table 4.24), a
difference of 10 sites compared to ex-smokers. Since 6 sites/tooth were measured, this is
equivalent to aspirin takers having approximately 2 fewer teeth with 4mm PAL than
ex-smokers. That is, aspirin takers had significantly less attachment loss compared to
non-aspirin takers. Using the same formulation non-aspirin takers had 39 sites involved,
or approximately 1 tooth less with 4mm PAL compared to ex-smokers.
4.6.3 Associations of aspirin and ex-smoking with the most severe site of PAL
(MSS-PAL)
In a separate analysis of the data, the most severe site of PAL (MSS-PAL) for each tooth
was measured and then averaged per subject. This was used as another observation of
the severity of PAL (Figure 4.3). These results highlighted a reduced MSS-PAL
observed in the AXS subjects compared with NAXS subjects in the range 4.5-6.5mm
MSS-PAL. In addition, Figure 4.3 shows that ex-smokers had more MSS-PAL than non-
smokers. Figure 4.3 also shows that the ANS and NANS groups had a similar pattern of
MSS-PAL distribution indicating that there were no differences between both groups at
various levels of MSS-PAL, with cross-over occurring at 4mm MSS-PAL and at 5.5mm
MSS-PAL.
81

A two-way ANOVA analysis of MSS-PAL using age as a covariate found significant
statistical associations of aspirin and smoking histories (Table 4.26). Subjects on low-
dose aspirin had significantly less MSS-PAL (0.33mm) (p=0.013) than non-aspirin takers
and ex-smokers had significantly more MSS-PAL (0.44mm) (p<0.001). Figure 4.4
represents the magnitude and direction of PAL using mean PAL and MSS-PAL.
4.6.4 Associations of aspirin and ex-smoking with the extreme worst site of PAL
(EWS-PAL)
The distribution of subjects by numbers and age between each group was uneven (Table
4.3). The ANS group had 51 subjects; who were older than subjects in the other three
groups. In order to have a better estimate of prevalence of severe periodontal disease and
to enable extrapolation to the population at large, the data were weighted. The age class
interval distribution of each group was matched to the age class intervals of Australian
males aged 50 years or more as estimated by the Australian Bureau of Statistics (Census
1996) in the 50-59, 60-69, 70-85 age groups. These weighted age class intervals of the
study population were analysed separately for PAL. The data were analysed on the
extreme worst site of PAL (EWS-PAL) per subject.
Table 4.27 shows the age class intervals and corresponding percentage distributions of
males in the general population of Adelaide. These data were used to calculate the
corresponding sample class intervals, weighted for all groups (Table 4.28). Low
numbered age class intervals had heavier weighting; for example in the age class interval
of 50-59 in the ANS group (7 subjects) each had a weight of approximately 2.91
compared to the same age group of NANS subjects (63 subjects) who each had a weight
of 0.77. That is, each subject aged 50-59 years in the ANS group counted as nearly
three. The 70+ age ANS group had a weight factor of 0.70 counting as ¾ of an
82

individual; the class interval of 60-69 had the least weight fluctuation for all four groups.
Table 4.29 presents descriptive data for EWS-PAL. Weighted data was used in a two-
way ANOVA with age as the covariate on EWS-PAL. This showed that there was a
significant protective association of low-dose aspirin with EWS-PAL in ex-smokers and
non-smokers (Table 4.30). Figure 4.5 shows the cumulative percentages of EWS-PAL of
all groups. The differences between the ANS and NANS groups were exaggerated when
the data were weighted (compared with Figure 4.3 MSS-PAL).
Figure 4.5 shows that the percentage of subjects with severe attachment loss decreased as
PAL increased, and that the association of aspirin with EWS-PAL was positive in
subjects who had smoked (AXS) when compared to NAXS subjects, but not for low
MSS-PAL (6mm). In addition, both non-smoking groups had less severe EWS-PAL.
All subjects had at least one worst site of 4mm EWS-PAL. The NAXS group had 58.2%
of subjects with a maximum 7mm EWS-PAL, while AXS had 49.5% of subjects with a
worst site of 7mm EWS-PAL. The major differences were seen in the range of 7-11mm
PAL. As a whole group, 48.7% subjects had at least one site with 7mm EWS-PAL
compared to 6.1% of subjects with a worst score of 11mm EWS-PAL.
Ex-smokers had an EWS-PAL averaging 0.75mm more compared to non-smokers while
aspirin takers had 0.54mm lower mean EWS-PAL compared to non-aspirin takers (Table
4.31). This could be interpreted as indicating that aspirin takers were approximately 0.21
in magnitude better off in EWS-PAL than non-aspirin takers (Table 4.30).
The ratio of beneficial associations of aspirin to the detrimental associations of ex-
smoking were similar in trend for all measures of severity of PAL, ranging from 59% to
75% (Table 4.31). That is, aspirin-takers had a lower PAL compared to ex-smokers by a
magnitude of approximately half to three-quarters, depending on the severity index used.
83

4.7 The associations of various clinical parameters on mean PAL.
One-way ANOVA with one covariate was used to assess the associations of plaque,
calculus, gingival bleeding, mobility and furcation with PAL. Mean PAL (6
sites/tooth/mouth) was used as the dependent variable. The ordinal scales of the plaque
score were dichotomised as being present (1, 2 or 3 score) or not present (0 score). There
was an association between age, plaque and mean PAL for all four groups. The overall
mean PAL was 2.8 1.00 (sd) mm. The higher the mean plaque scores the higher the
mean PAL (0.48mm), with a 0.01 increase in mean PAL due to age, indicating that older
subjects and those with plaque present had higher mean PAL (Table 4.32).
A one-way ANOVA using the dichotomous scale of calculus showed there was an
association between calculus with mean PAL for all four groups. The calculus
correlation with mean PAL was significant but very minor (approximately 0.004),
indicating that subjects with calculus had slightly higher mean PAL (Table 4.33).
Gingival bleeding (using mBSI) and mobility were assessed as either being present (1, 2
& 3) or absent (0). Figure 4.1 shows that the mean percentage of bleeding sites per
group varied from 56.5% for the 65 and 59.3% for >65 year old subjects. The ANS
had 54.2% of sites with bleeding, the AXS had 57.4%, the NANS had 57.3% and NAXS
had 60.0% of sites with gingival bleeding. In all four groups, mean PAL significantly
increased with increasing age and with the increasing percentage of teeth with gingival
bleeding (p < .001). For every one-year increase in age, there was an increase in mean
PAL by 0.02 mm. For every percent increase in the percentage of teeth with gingival
bleeding, there was an extra 0.01mm increase in mean PAL (Table 4.34). The same
trends were seen with furcation involvement and mobility.
84

4.7.1 Site and tooth variations in recession and pocket depth by mean PAL
The components of mean PAL (gingival recession and pocket depth) were investigated
for tooth and jaw variations. Figure 4.6 represents the mean (mm) of gingival recession
and periodontal probing pocket depths for all groups using the mean of 6 sites per tooth.
The vertical dimensions of each bar represent the mean PAL per tooth. Maxillary teeth
are represented with the positive PAL scores; the negative PAL scores represent the
mandibular teeth. Each tooth type the mean represents the overall mean PAL for that
tooth type for both left and right sides of each jaw type. Figures 4.7-4.10 represent the
intra-group jaw and tooth variation in mean PAL.
Different patterns of PAL were seen within and between groups for tooth and jaw type.
In the maxilla, most PAL was seen in 1st, 2nd and 3rd molars with maxillary 1st molars
having the largest amount of mean PAL (3.2mm) with intra-group variation ranging from
3mm for ANS and NANS to 3.4 mm for NAXS and 3.5 mm AXS. The pattern of
attachment loss was different in the lower arch, with the largest mean PAL occurring
around the mandibular incisors. The pattern of mean PAL varied between groups,
ranging from a mean PAL of 2.4 mm for AXS to a mean PAL of 3.4 mm for NAXS.
The main contributor to mean PAL in maxillary molars was pocket depth (average of
2.6mm for pocket depth and 0.5mm recession). Recession contributed to the larger
component of attachment loss in the mandibular incisors as compared to the maxillary
molars (an average of 2.0mm per pocket depth and 1.1 mm recession).
In the AXS group, the principal component of PAL was pocket depth (Figure 4.7).
Recession mainly occurred around maxillary and mandibular molars and lower anterior
teeth. The NAXS group showed a similar pattern as in the AXS group but had a larger
component of PAL in the lower anteriors (Figure 4.8). The ANS group had a pattern
85

similar to both smoking groups but the pocket depth component of PAL was less in this
group (Figure 4.9). The NANS group had a similar pattern to the ANS group (Figure
4.10).
4.7.2 Socio-economic factors and periodontal attachment
Table 4.35 summarises the demographic and socio-economic associations with mean
PAL. All analyses were performed under the same statistical model (two-way ANOVA
with age as a covariate on the dependant variable of mean PAL). Education had
significant statistical associations (p=0.008) on the mean PAL between groups. The
lower educated subjects had more periodontal attachment loss. A further analysis using
the ANOVA model with education as the covariate found no associations with mean
PAL (data not presented).
Controlling for age and using the ANOVA model, there were no statistically significant
associations with mean PAL in pensioners and non-pensioners. There was a statistically
significant association between mean PAL and the time interval of the last dental visit.
There were no statistically significant associations between mean PAL and subjects that
brushed > 1/day and subjects that brushed < 1/day. There were no statistically significant
associations between mean PAL and mouth rinsing. There was a statistically significant
association between mean PAL and subjects who practiced interproximal cleaning;
subjects who flossed once or more per week had statistically significantly less mean PAL
compared to those who flossed less than once per week or never flossed. Education level
and floss use were the two independent factors that were most significantly correlated
with PAL.
86

Table 4.1 Intra examiner reliability test using kappa statistics
Number of sites Kappa value Std errorRecession 630 0.922 0.02Pocket depths 630 0.914 0.02(combined pocket and recession measurements)
1260 0.941 0.01
Table 4.2 The number and percentage distribution of subjects participating in
the study by group
Frequency PercentAspirin never smoked 51 13.0Aspirin ex-smoker 102 26.0Aspirin takers total. 153 39.0No aspirin never smoked 122 31.1No aspirin ex-smoker 117 29.8Non-aspirin takers total 239 60.9Overall Total 392 100
Table 4.3 Distribution of age by group
Age distribution of males
Numbers in group
Age rangeStd
Deviation 64 yrs
65 yrs Mean Minimum Maximum
ANS 19 32 51 68.2* 53 85 7.71
NANS 85 37 122 59.9* 50 84 7.67
AXS 45 57 102 66.0* 50 85 7.93
NAXS 70 47 117 62.6* 50 83 8.12
Total 219 173 392 63.4 50 85 8.39One-way ANOVA. SSQ 3427.55, df =3, F =18.40, (*) Significant mean age differences between groups, p < 0.001
87

Table 4.4 A Scheffés analysis of homogeneity between two groups at a time for
mean age differences
(I) (J) Difference (I-J) in means (years)
Std. Error Sig(p-value)
ANS ANSAXS 2.2 1.35 0.435
NANS 8.3 1.31 < 0.001NAXS 5.6 1.32 0.001
AXS
ANS -2.2 1.35 0.435AXS
NANS 6.1 1.06 < 0.001NAXS 3.3 1.07 0.021
NANS ANS -8.3 1.31 < 0.001AXS -6.1 1.06 < 0.001
NANSNAXS -2.7 1.02 0.067
NAXS ANS -5.6 1.32 0.001AXS -3.3 1.07 0.021
NANS 2.7 1.02 0.067NAXS
Table 4.5 Scheffésa, analysis for homogeneity between subsets
Subsets for alpha = 0.05Group N 1 2 3NANS 122 59.8NAXS 117 62.6 62.6AXS 102 66.0 66.0ANS 51 68.2Significance value 0.157 0.052 0.324Means for all groups in homogeneous subsets are displayed.(a) Uses Harmonic Mean Samples Size = 86.7.
88

Table 4.6 Demographics on pension status with group specific characteristics.
Pension or Health Card? ANS AXS NANS NAXSGROUP TOTAL
Yes N 37 71 61 62 231% 72.0% 69.6% 50.4% 53.00% 58.9%
No N 14 31 61 55 161% 28.0% 30.40% 49.6% 47.0% 41.1%
Total N 51 102 122 117 392% 100.0% 100.0% 100.0% 100.0% 100.0%
N = Number of subjects
Table 4.7 Pension status in relation to denture use.
Pension or health card?
Have a dentureyes no Total
yes 41.1% 20.6% 33.2%no 58.9% 79.4% 66.8%
Total 100% 100.0% 100.0%
Table 4.8 Demographic data on schooling of all subjects with group specific
characteristics.
Education ANS AXS NANS NAXS TOTAL
N % N % N % N % N %No schooling 1 3.9 1 1.0 2 0.8 6 1.7 10 1.5Primary School
10 19.6 18 17.6 19 15.6 22 18.8 69 17.6
Secondary School
21 41.2 56 54.9 52 42.6 69 59.0 198 50.5
Tertiary School
18 35.3 27 26.5 50 41.0 24 20.5 119 30.4
Total 51 100.0 102 100.0 122 100.0 117 100.0 392 100.0N = number of subjects per group% = percentage of subjects per group
89

Table 4.9 A self-evaluation of English language skill.
How well do you speak English?
ANS AXS NANS NAXSGROUP TOTAL
N % N % N % N % N %Very well 37 74.0 83 81.4 84 68.3 76 65.5 281 71.6Well 10 20.0 14 13.7 35 28.5 31 26.7 90 23.0Not well 2 4.0 5 4.9 3 2.5 9 7.8 19 5.10Not at all 1 2.0 1 0.3Total 50 100.0 102 100.0 122 100.0 116 100.0 391 100.0N = number of subjects per group% = percentage of subjects per group
Table 4.10 Socio-economic factors and dental behaviours.
Tooth brush >1/day
Tooth brush < 1/day
Use a mouth rinse
Hardly ever use a mouth rinse
Floss more than 1/week
Floss less than 1/week
Scale and clean 2 years
Scale and clean 3 years
Pension status
Pension 201 28 55 229 68 161 123 106
non-pension 146 14 31 129 59 102 103 58
Education level
< primary school 54 21 17 58 14 61 25 50
> secondary school 293 21 69 245 113 202 201 114How long since last dental visit< 1 yr 211 9 56 164 92 129 180 41
1yr-5yr 114 17 23 108 32 99 45 86
> 5 years 22 16 7 38 3 35 1 37
Age
64 189 29 52 166 75 144 130 89
65 158 13 34 137 52 119 96 75
Groups
ANS 44 5 10 39 14 35 23 26
AXS 92 10 23 79 37 65 64 38
NANS 112 9 25 96 47 75 77 45
NAXS 99 18 28 89 29 88 62 55
90

Table 4.11 Population and percentage distribution of subjects since their last
dental visit. The time range was from less than one year to never
visiting the dentist.
How long since your last dental visit?
ANS AXS NANS NAXS Total
< 6 months
Number of subjects
11 42 41 46 140
% within group
22.4% 41.2% 33.6% 39.3% 35.9%
6-11 months
Number of subjects
14 18 34 15 81
% within group
28.6% 17.6% 27.9% 12.8% 20.8%
1-2 years
Number of subjects
11 19 23 20 73
% within group
22.4% 18.6% 18.9% 17.1% 18.7%
3-5 years
Number of subjects
6 16 13 23 58
% within group
12.2% 15.7% 10.7% 19.7% 14.9%
6-10 years
Number of subjects
3 4 6 4 17
% within group
6.1% 3.9% 4.9% 3.4% 4.4%
> 10 years
Number of subjects
3 3 5 7 18
% within group
6.1% 2.9% 4.1% 6.0% 4.6%
Never
Number of subjects
1 2 3
% within group
2.0% 0.0% 0.0% 1.7% 0.8%
Total
Number of subjects
49 102 122 117 390
% within group
100.0% 100.0% 100.0% 100.0% 100.0%
91

Table 4.12 Missing teeth by age and group.
Age 64 yrs 65 yrs Group Total
MeanStd error
N MeanStd error
N MeanStd error
N
ANS 6.7 0.91 19 11.3 1.10 32 9.6 0.82 51
NANS 7.0 0.56 85 11.1 1.11 37 8.2 0.54 122
AXS 8.0 0.79 45 12.5 0.87 57 10.5 0.63 102
NAXS 9.0 0.71 70 11.5 0.98 47 10.0 0.59 117
Group Total
7.8* 0.37 219 11.7* 0.50 173 9.5 0.31 392
* significantly different (2-tailed t-test) p < 0.001N = The number of subjectsComputed at alpha = 0.05 level
Table 4.13 Missing teeth and smoking history
Number of subjects
Mean number of missing
teeth
Std error of mean
t-test
Ex-smokers (AXS and NAXS)
219 10.2 0.43p = 0.01
Non-smokers (ANS and NANS
173 8.6 0.45
Table 4.14 The mean plaque index per group.
64 yrs 65 yrs Group Total
mean SD N Mean SD N mean SD NANS 1.3 0.52 19 1.5 0.81 32 1.4 0.72 51NANS 1.2 0.61 85 1.5 0.56 37 1.3 0.61 122AXS 1.4 0.80 45 1.5 0.62 57 1.4 0.70 102NAXS 1.5 0.76 70 1.7 0.69 47 1.6 0.74 117Group Total 1.3 0.70 219 1.6 0.67 173 1.4 0.69 392SD = Standard deviationN = Number of subjects
92

Table 4.15a Profile of aspirin use and subject numbers
Brand Name Composition Concentration Number of subjectsSOLPRIN Aspirin 300 mg 63CARTIA Acetyl Salicylic acid 100 mg 33ASTRIX Aspirin 100 mg 18SPREN Aspirin 300 mg 16ASPIRIN Aspirin 300 mg 10CARDIPRIN Aspirin 100 mg 6DISPRIN Aspirin 300 mg 4ASPRO Aspirin 300 mg 2ECOTRIN Acetyl Salicylic acid 650 mg 1 (1/2 tab/day)
Total 153
Table 4.15b The association of age and past smoking on mean plaque scores with
tests of significance.
ANOVA (one-way) of mean plaque
scores
Sum of Squares
dfF
statisticSig
Outcomea on
means plaque score.
Std error
95% confidence interval
Lower limit
Upper limit
Intercept 1.28 1 10.09 0.096 0.37 0.26 -0.15 0.88Age association 6.50 1 2.79 < 0.001 0.02* 0.004 0.007 0.02
Ex-smoking association
1.90 1 14.13 0.043 0.14* 0.07 0.005 0.28
Error 178.90 389Total 985.15 392
a = magnitude of association on mean plaque scores.
Estimated mean plaque scores from ANOVA
(score se)Ex-smoking history Total
Yes NoTotal 1.5 0.05 1.3 0.05 1.4 0.03*
Number of subjects is calculated from descriptive Table 4.14.Estimated means from model evaluated at the mean age covariate.Age: p<0.001Ex-smoking: p =0.043(*) Age and ex-smoking had significant differences with mean plaque scores
93

Table 4.16 Distribution of mean percentage of teeth with calculus by age,
aspirin and ex-smoking.
Mean % of teeth with calculus
(% sd) 64 years 65 years Group total
ANS26.0 38.01
(19)64.2 64.91
(32)50.0 59.0
(51)
AXS49.3 59.76
(45)55.0 58.5
(57)52.5 58.85
(102)
NANS44.5 54.24
(85)63.7 64.05
(37)50.3 57.80
(122)
NAXS54.4 76.90
(70)64.9 74.75
(47)58.6 75.89
(117)
Group total47.0 62.49
(219)61.2 65.17
(173)53.3 64.00
(392)
Ex-smokersyes no Total
Aspirin takers
yes52.5 58.85
(102)50.0 59.00
(51)
51.6 58.71 (153)
no58.6 75.89
(117)50.3 57.80
(122)
54.4 67.26 (239)
Total55.7 68.40
(219)50.2 57.99
(173)
53.3 64.00 (392)
94

Table 4.17 The association of age with mean percentage of calculus between
groups
Ordinary least squares
regression of mean % of teeth
with calculus(% se)
Sum of Squares
dfF
statistic
SigOutcome
a on estimates
Std error
95% confidence
interval
Lower limit
Upper limit
Intercept 89.27 1 0.022 0.882 3.64 24.56 -44.64 51.92Age association 16902.92 1 4.16 0.042 0.78 0.38 0.028 1.54
Error 1584392.60 390Total 2714822.42 392
Computed using level of significance at alpha = 0.05.a = magnitude of association on mean plaque scores.Number of subjects as in Table 4.16.Age: p=0.042
Age was significantly associated with mean percentage of calculus.
Table 4.18 The association of low-dose aspirin and ex-smoking with the mean
percentages of mobile teeth.
Mean % of mobile teeth
Std. deviation
N p- value
Aspirin takers 3.6 10.52 153 p > 0.05F = 2.20df = 1Non-aspirin takers 4.2 13.15 239
Ex-smokers 5.5 14.89 219 p = 0.004F= 8.30df = 1Non-smokers 2.0 7.00 173
ANOVA (2-way) using mean age covariate. Computed at alpha = 0.05.Number of subjects as in Table 4.16
95

Table 4.19 The correlation of low-dose aspirin and past smoking with mean
PAL.
2-way ANOVA of mean PAL(mm se)
Sum of Squares
dfF
statisticSig
Outcomea
on mean PAL (mm)
Std error
95% confidence
intervalLower limit
Upper limit
Intercept 8.8 1 9.39 0.002 1.10 0.38 0.39 1.9Age associations
14.47 1 15.44 < 0.001 0.02* 0.01 0.01 0.04
Ex-smoking associations
12.87 1 13.73 < 0.001 0.40* 0.1 0.17 0.57
Aspirin associations
3.94 1 4.20 0.041 -0.20* 0.11 -0.43 -0.09
Error 363.67 388Total 3464.59 392Computed using level of significance at alpha = 0.05.a = magnitude of association on mean PAL from age, ex-smoking & low-dose aspirin.(-) Magnitude of reduced mean PAL
Estimated mean PAL (mm) from ANOVA
Ex-smokers
Yes No Total
Aspirin takers
Yes 2.8 0.09(102)
2.5 0.01(51)
2.6 0.08(153)
No 3.1 0.08(117)
2.7 0.08(122)
2.9 0.06(239)
Total 2.9 0.07(219)
2.6 0.08(173)
2.8 0.05(392)
Estimated means from model evaluated at the mean age covariate.( ) number of subjectsMean PAL (mm se)The hypotheses 1 and 2 were rejected for all independent variables. There were significant associated differences (*) for age, ex-smoking and aspirin with mean PAL.Age: p<0.001Ex-smoking p<0.001Aspirin: p=0.041
96

Table 4.20 The association of aspirin dosage with mean PAL.
Mean PAL (mm)
Std error Number of subjects
p-value
Dosage 150 mg 2.8 0.08 131*
0.184 300 mg 3.1 0.16 19*No subjects took aspirin between 151-299mg.The maximum dosage was 325mg by one subject.* Three subjects did not enter their dosage on questionnaire.
Table 4.21 The association of aspirin duration with mean PAL.
Duration Mean PAL (mm)
Std error Number of subjects
p-value
2-5 years 2.7 0.10 810.8176-10 years 2.8 0.11 53
11 years 2.9 0.31 19
Table 4.22a The association of past smoking dosage and duration with mean
PAL.
Mean PAL (mm)
Std errorNumber of
subjectsp-value
Time since quit smoking
Quit < 5 yrs 3.4 0.24 170.077*
Quit 5yrs 2.9 0.08 196
Number of cigarettes /
day
< 10 / day 3.1 0.16 40 0.164*
10 - 19 / day 2.9 0.12 79 0.013*
20 - 30 / day 2.7 0.14 58 0.013*
> 30 / day 3.4 0.16 42 0.001*
Years was smoker
< 5 yrs 2.7 0.30 13 0.257*
5 yrs 2.7 0.20 36 0.036*
(*) Compared to non-smokers
97

Table 4.22b The correlation of the number of cigarettes smoked and duration of
smoking with mean PAL with t-test of significance.
Number of cigarettes smoked
B Std
errort statistic Sig
95% confidence interval
Outcomea
on mean PAL (mm)
Lower limit
Upper limit
Intercept 3.40 0.16 21.05 < 0.001 3.08 3.72Association of smoking<10 cigarettes/day
-0.32 0.23 -1.40 0.164 -0.78 0.13
Association of smoking10-19 cigarettes/day
-0.50 0.20 -2.50 0.013 -0.90 -0.11
Association of smoking > 20 cigarettes/day
-0.74 0.21 -3.50 0.001 -1.16 -0.33
Duration of smokingIntercept 3.07 0.08 36.58 < 0.001 2.90 3.23Association of smoking < 5 years
-0.35 0.031 -1.14 0.257 -0.96 0.26
Association of smoking 5 years
-0.42 0.20 -2.106 0.036 -0.81 -0.027
a = magnitude of association on mean PAL by years of smoking.(-) Magnitude of lost mean PAL
98

Table 4.23 Univariate analysis of variance in mean PAL at 2, 4 5 and
7mm.
SEVERITYMean PAL (mm se)
Smoking historyAspirin p-
valueyes no Total
2mmAspirin takers
yes3.0 0.71
(102)2.7 0.08
(51)2.9 0.07
(153)
0.019no3.2 0.07
(117)2.9 0.07
(122)3.1 0.05
(239)
Total3.1 0.05
(219)2.8 0.06
(173)3.0 0.04
(392) p-value of smoking 0.001
4mmAspirin takers
yes4.6 0.05
(102)4.4 0.06
(51)4.5 0.04
(153)
0.027no4.8 0.04
(117)4.5 0.04
(122)4.7 0.34
(239)
Total4.7 0.04
(219)4.5 0.04
(173)4.6 0.03
(392)p-value of smoking < 0.001
5 mmAspirin takers
yes5.7 0.04
(99)5.6 0.05
(46)5.6 0.03
(145)
0.972no5.7 0.04
(111)5.6 0.05
(106)5.6 0.03
(217)
Total5.7 0.04
(210)5.6 0.05
(152)5.6 0.3
(362)p-value of smoking 0.044
7mm
Aspirin takers
yes7.8 0.08
(53)7.8 0.11
(24)7.8 0.08
(77)
0.053no7.6 0.08
(68)7.6 0.09
(46)7.6 0.07
(114)
Total7.7 0.06
(121)7.7 0.08
(70)7.7 0.05
(191)
p-value of smoking 0.916
ANOVA (2-way).Estimated means from model evaluated at the mean age covariate.( ) number of subjects
99

Table 4.24 Univariate analysis of variance on mean % PAL at 2, 4, 5 &
7mm.
Extent2-way ANOVA of mean % PAL (% se)
Ex-smokersyes no p-value of
aspirin
2mma Aspirin takers
yes95.9 1.00
(102)N/A
0.196no94.1 0.92
(117)N/A
Total95.0 0.67
(219)N/A
2mmb Aspirin takers
yes N/A89.4 0.42
(51)
0.006no N/A95.3 1.07
(122)
Total N/A92.4 0.97
(173)
Ex-smokersp-value of
aspirinyes no Total
4mmAspirin takers
yes30.7 1.98
(102)21.7 2.34
(51)26.2 1.85
(153)
0.035no
35.8 1.83(117)
26.7 1.82(122)
31.3 1.43(239)
Total33.3 1.48
(219)24.2 1.72
(173)28.7 1.14
(392)p-value of ex-smoking < 0.001
5 mmAspirin takers
yes13.1 1.55
(102)6.4 1.84
(51)9.7 1.45
(153)
0.003no
18.7 1.44 (117)
12.0 1.43 (122)
15.4 1.13 (239)
Total15.9 1.16
(219)9.2 1.35
(173)12.6 0.90
(392)p-value of ex-smoking < 0.001
7mmAspirin takers
yes2.9 0.55
(102)0.8 0.69
(51)1.87 0.55
(153)
0.043no
4.4 0.54 (117)
2.2 0.54(122)
3.3 0.43 (239)
Total3.7 0.44
(219)1.5 0.51
(173)2.6 0.34
(392)p-value of ex-smoking < 0.001
( ) = number of subjects.Estimated means from model evaluated at the mean value of the age covariate.(a) Associations of aspirin on subjects who are ex-smokers (b) Associations of aspirin on subjects who have never smokedN/A not applicable due to interaction associations.
100

Table 4.25 The magnitude of the association of aspirin and smoking history
with severity and extent of PAL at 2, 4, 5 & 7 mm PAL using
the general linear model (2-way ANOVA) of analysis.
Severity Overall Mean PAL (c) (mm)
Aspirin association on
Mean PAL (mm)
Ex-smoking association on
Mean PAL (mm)Mean PAL at sites with PAL 2mm
3.0 -0.2 0.3
Mean PAL at sites with PAL 4mm
4.6 -0.1 0.2
Mean PAL at sites with PAL 5mm
5.6 N/S 0.1
Mean PAL at sites with PAL 7mm
7.7 N/S N/S
Extent Overall Mean % PAL
Aspirin Association (mean %)
Ex-smoking association(mean %)
Mean % of all sites with PAL 2mm (a) 92.4 -5.9 I/E
Mean % of all sites with PAL 2mm (b) 95.0 N/S I/E
Mean % of all sites with PAL 4mm
28.7 -5.1 9.1
Mean % of all sites with PAL 5mm
12.6 -5.6 6.7
Mean % of all sites with PAL 7mm
2.6 -1.5 2.2
(a) Non-smokers only (b) Ex-smokers only. (c) From Table 4.23.N/S Not significant. N/A not applicable. I/E interaction associationSee Tables 4.19, 4.23 & 4.24 for p-values.
101

Table 4.26 The correlation of aspirin and past smoking history with MSS-PAL.
2-way ANOVA of mean MSS-
PAL (mm se)
Sum of Squares
dfF
statistic
Sig
B
Std error
95% confidence
intervalMean
outcomea
(mm)
Lower limit
Upper limit
Intercept 24.61 117.2
9< 0.001 1.98 0.47 1.05 2.91
Age association 24.45 117.1
7< 0.001 0.03* 0.01 0.02 0.05
Ex-smoking association
18.26 112.8
3< 0.001 0.44* 0.12 0.20 0.69
Aspirin association
8.81 1 6.19 0.013 -0.33* 0.13 -0.59 -0.07
Error 552.43 388Total 7171.03 392
Computed using level of significance at alpha = 0.05.a = magnitude of association on mean PAL from age, ex-smoking & low-dose aspirin.(-) magnitude of reduced MSS-PAL
Estimated mean MSS-PAL (mm) from ANOVA (mm se)
Ex-smokers
Aspirin takers
Yes No TotalYes 4.1 0.11
(102)3.7 0.13
(51)3.9 0.10
(153)No 4.4 0.10
(117)4.0 0.10
(122)4.2 0.08
(239)Total 4.3 0.08
(219)3.8 0.09
(173)4.0 0.06
(392)Computed using level of significance at alpha = 0.05.Estimated means from model evaluated at the mean age covariate.( ) number of subjects
The hypotheses 1 and 2 were rejected for all independent variables. Age, ex-smoking and aspirin were significantly associated (*) with MSS-PAL.
102

Table 4.27 The age class distribution of males 50+ years in metropolitan
Adelaide in 1996 from census statistics and their appropriate
frequency distribution.
Australian Bureau of Statistics age class interval
Number of males in metropolitan Adelaide [Census, 1996 #290]
Percentage frequency
Relative frequency
50-59 51,418 39.87% 0.4060-69 39,741 30.83% 0.3170-85 37,749 29.84% 0.30Total 128,908 100% 1.00
Table 4.28 The proportional weights given to each group using the percentage
frequency of each class interval from census statistics for
metropolitan Adelaide
Age interval 50-59 years 60-69 years 70+ years Group TotalRelative class interval
weight .40 .31 .30 1
ANS51 x 0.40 / 7*
= 2.9151 x 0.31 / 22* = 0.72
51 x 0.30 / 22* = 0.70
N=51
AXS102 x 0.40 / 23* = 1.77
102 x 0.31 / 44* = 0.72
102 x 0.30 / 35* = 0.87
N=102
NANS122 x 0.40 / 63* = 0.77
122 x 0.31 / 48* = 0.79
122 x 0.30 / 11* = 3.33
N=122
NAXS117 x 0.40 / 46* = 1.01
117 x 0.31 / 47* = 0.77
117 x 0.30 / 24* = 1.46
N=117
Group Total 139 161 92 N=392N = The number of subjects per group* = The number of subjects in each class interval
Table 4.29 Descriptive statistics of EWS-PAL
Means have weighted age class intervals values
Number of subjects
EWS-PAL (mm)
Standard deviation
AXS 102 7.00 1.80ANS 51 6.30 1.91Total of aspirin takers 153 6.74 1.86NAXS 117 7.50 2.22NANS 122 6.70 2.24Total of non-aspirin takers 239 7.10 2.26Total of ex-smokers 219 7.20 2.05Total of non-smokers 173 6.60 2.16Group Total 392 7.00 2.12
103

Table 4.30 The association of aspirin and past smoking history with EWS-PAL
using weighted data.
2-way ANOVA
EWS-PAL(mm se)
Sum of Squares
dfF
statisticSig
Mean outcomea
(mm)
Std error
95% confidence
intervalLower limit
Upper limit
Intercept 125.21 1 29.75 <0.001 4.02 0.76 2.52 5.51Age
associations56.82 1 13.50 <0.001 0.04 0.01 0.02 0.07
Ex-smoking associations
26.31 1 6.25 <0.001 0.75 0.22 -0.96 -0.12
Aspirin associations
52.33 1 12.44 0.013 -0.54 0.21 0.33 1.16
Error 1632.73 388Total 20765.4
1392
Computed using level of significance at alpha = 0.05.a = magnitude of association on mean PAL from age, ex-smoking & low-dose aspirin.(-) magnitude of reduced EWS-PALAll data were weighted using metropolitan Adelaide population age class statistics (see Table 4.28)
Estimated weighted EWS-PAL from ANOVA (mm se)
Ex-smokers
Aspirin takers
Yes No Total
Yes7.0 0.18
(102)6.2 0.22
(51)6.6 0.17
(153)
No7.5 0.17
(117)6.8 0.17
(122)7.1 0.13
(239)
Total7.2 0.14
(219)6.4 0.16
(173)6.9 0.11
(392)
Computed using level of significance at alpha = 0.05.Estimated means from model evaluated at the mean age covariate.( ) number of subjects
104

Table 4.31 The ratio of aspirin to smoking on various measurements of PAL.
Severity (mm)
4 mm mean PAL
Mean PAL MSS-PALEWS-PAL
(weighted data)Aspirin -0.13 -0.22 -0.33 -0.54
Smoking 0.22 0.37 0.44 0.75Ratio 0.59 0.59 0.75 0.72
7mm mean PAL not included since there were no significant associationsRatio = aspirin PAL/ex-smoking PAL
Table 4.32 Associations of plaque and age with mean PAL with tests of
significance.
ANOVA (one-way) of mean PAL(mm se)
Sum of Squares
dfF
statisticSig
Outcomea
on estimates
Std error
95% confidence interval
Lower limit
Upper limit
Intercept 9.41 1 10.8 0.001 1.18 0.36 0.48 1.89Age
associations5.78 1 6.66 0.100 0.01 0.01 0.004 0.026
Plaque associations
40.84 1 47.02 <0.001 0.48 0.07 0.34 0.61
Error 337.87 389Total 3464.59 392
Computed using level of significance at alpha = 0.05.a = magnitude of association on scoresN= 392 subjects.
Table 4.33 Associations of calculus and age with mean PAL with tests of
significance.
ANOVA (one-way) of mean PAL(mm se)
Sum of Squares
dfF
statisticSig
Outcomea
on estimates
Std error
95% confidence interval
Lower limit
Upper limit
Intercept 12.36 1 13.64 <0.001 1.34 0.37 0.63 2.08Age
associations10.24 1 11.29 0.001 0.02 0.01 0.008 0.03
Calculus associations
26.00 1 28.68 <0.001 0.004 .001 0.003 0.01
Error 352.71 389Total 3464.59 392
Computed using level of significance at alpha = 0.05.a = magnitude of association on scoresN= 392 subjects.
105

Table 4.34 Associations of gingival bleeding and age with mean PAL with tests
of significance.
ANOVA (one-way) of mean PAL(mm se)
Sum of Squares
dfF
statisticSig
Outcomea
on estimates
Std error
95% confidence interval
Lower limit
Upper limit
Intercept 6.43 1 7.03 0.008 1.00 0.38Age associations 12.95 1 14.16 < 0.001 0.02 0.01 0.01 0.03Gingival bleeding associations
23.01 1 25.16 < 0.001 0.01 0.001 0.004 0.01
Error 355.70 389Total 3464.59 392
Computed using level of significance at alpha = 0.05.a = magnitude of association on scoresN= 392 subjects.
Table 4.35 Socio-economic factors, oral hygiene patterns and mean PAL (mm).
ANOVA (2-WAY) Numbers Means Std error P value
EducationNo school / Primary 75 3.1 0.11 >0.05Secondary / Tertiary 317 2.7 0.06 0.008
Pension statusYes 231 2.9 0.07
>0.05No 161 2.7 0.08
Time since last dental visit
< 12 months 221 2.7 0.07>0.05
> 12 months 169 2.9 0.08
Brushing1 + / day 347 2.8 0.05
>0.05< 1 / day 42 2.9 0.20
Time since scale and clean
< 12 months 148 2.7 0.07
>0.051-2 years 78 2.7 0.113-5 years 87 3.0 0.106-10 years 30 2.8 0.24> 11 years 47 2.8 0.19
Frequency of mouth rinse
Never 227 2.8 0.07>0.05< 1/ wk 95 2.6 0.09
1 + / week 67 2.9 0.12
Frequency of flossingNever 144 3.0 0.10 >0.05
< 1/ week 119 2.7 0.08 >0.051+ /week 127 2.6 0.70 0.002
106

Table 4.36 The statistical power values for most ANOVA analyses
Levels of power from ANOVA analyses (a)Ex-smoking Aspirin
Severity2mm PAL 0.986 0.6534mm PAL 0.984 0.6027mm PAL 0.051 0.491
Extent2mm PAL 0.794 0.3634mm PAL 0.981 0.5507mm PAL 0.910 0.526
Mean PAL 0.959 0.533MSS-PAL 0.947 0.699EWS-PAL* 0.940 0.703(*) Weighted values(a) Observed power from ANOVA computed using = 0.05 Low to moderate power of association
Table 4.37 Relative percentage of subjects with medical conditions per group
Group Percentage
ANS 57.0%
AXS 63.7%
NANS 18.0%
NAXS 30.7%
Table 4.38 Outcome of age, ex-smoking and aspirin with various indices of PAL
PAL unit of measure
Outcome of age per year per group
Outcome of ex-smoking per year per group
Outcome of aspirin year per group
Mean PAL. 0.02 mm 0.40 mm -0.20 mm
MSS-PAL 0.03 mm 0.44 mm -0.33 mm
EWS-PAL 0.04 mm 0.75 mm -0.54 mm
107

Figure 4.1 The mean percentage of sites with gingival bleeding (modified
bleeding index).
Figure 4.2 The mean percentage of teeth with calculus
108

Figure 4.3 Cumulative distribution of MSS-PAL representing the worst score
(site) per tooth per subject, averaged over all subjects.
109

Figure 4.4 Diagrammatic representation of PAL according to smoking and
aspirin taking history, showing mean PAL, MSS-PAL and EWS-
PAL.
110

Figure 4.5 Cumulative distribution of EWS-PAL. Data were weighted using age
class statistics for metropolitan Adelaide population.
0
10
20
30
40
50
60
70
80
90
100
4 5 6 7 8 9 10 11 12
PAL
Per
cen
t
NAXS
AXS
NANS
ANS
111

Figure 4.6 Variations of recession and pocket depths by tooth- and jaw type for
the whole study population.
Figure 4.7 Variation of recession and pocket depths by tooth- and jaw type in
the AXS group.
112

Figure 4.8 Variation of recession and pocket depths by tooth- and jaw type in
the NAXS group.
Figure 4.9 Variation of recession and pocket depths by tooth- and jaw type in
the ANS group.
113

Figure 4.10 Variation of recession and pocket depths by tooth- and jaw type in
the NANS group.
114

Chapter 5 Discussion
5.1 Profile of the study population
The total number of males residing in metropolitan Adelaide in 1996 (excluding overseas
visitors) was 506,351 of whom 132,382 males were over 50 years of age (Census 1996).
As this study targeted older males through media advertising, the subjects who volunteered
were self-selected (a convenience sample). Therefore the population sample was not
representative of Adelaide's male population aged 50 and above. It would have been useful
to have had a random sample but resources and time did not allow this to occur. Females
were excluded as participants because hormonal/osteoporosis factors could have had
confounding effects on the periodontium (Genco and Löe 1993; Beck 1996; Bando et al.
1998; Jeffcoat 1998).
It was considered that there was a high chance of finding the appropriate cohorts, by
targeting older males by advertisement. Sampling frame error was minimised because both
exposed and non-exposed subjects were targeted from the same sampling frame. This
selection method facilitated the creation of exposed groups and non-exposed groups that
were similar for age, ethnicity and pension status. Every attempt was made to define the
study population with regards to variables relevant to PAL. Provided that the "convenience
sample" did not differ too greatly from the general population in terms of those variables
and because PAL is usually prevalent, it can be argued that some degree of extrapolation
should be possible with regard to the periodontal status of the two populations. Based on
this argument, one analysis was performed by applying weight to the data (EWS-PAL)
(Tables 4.27, 4.28 & Figure 4.5). This enabled a better separation of groups, especially
since there was some crossover between the ANS and NANS groups (Figure 4.3), and an
115

adjustment for the differing age structures between the groups. Otherwise all other
analyses were carried out on unweighted data.
A large sample frame, confidentiality, gifts and eligibility criteria all help in minimising
response bias and promote responses, especially from non-random samples (Fink 1995c). In
the current study, efforts were made to promote a response from as many males over 50 from
the general population as possible, by offering a free dental examination and a free Colgate
Oral Care Kit. Considerable efforts were made to reduce sources of population bias (by
advertising in the two major newspapers with the largest amount of coverage of Metropolitan
Adelaide). This selection method only allowed the selection of subjects who could read
English (creating a biased sample); this may have accounted for the high percentage of
subjects with good English skills in the study population compared to the general population.
The present study population contained less people with primary or no schooling (19.1%),
(Table 4.8) than the figure of 35% from Adelaide's general community (Census 1996);
30.4% of subjects occupied the upper education level (tertiary education), compared to 13%
(Census 1996) for Adelaide's general population. The percentage of subjects with a
secondary education (50.5%) compared favourably with the 52% for Adelaide's general
population.
The ANS group had the lowest percentage (13%) of subjects participating in the study
compared to the whole sample (Table 4.8). The subject groupings were dissimilar in
education and age class distribution. On the other hand, subjects in a higher educated group
could be expected to have "better" health behaviour, as other studies have shown (Srikandi
1982; Mullally and Linden 1992).
116

Due to limited time and financial resources, this study relied on press advertisements for
recruitment of subjects; mailed flyers to households in metropolitan Adelaide may have
helped in recruiting a more representative study sample from the general population.
5.1.1 Age groupings
In comparison to Adelaide's male population, this study population had lower, and in some
cases, higher numbers in each age class interval (50-59, 60-69 and 70+), using census data as
a basis for comparison (Tables 4.27 & 4.28). The relative number of subjects in the 50-59
year aged class interval for the ANS group was 14% compared to 51% for the NANS group
and 40% from the general population of Adelaide. The distribution of subjects in the class
interval of 60-69 years had the least fluctuation between groups, with a relative frequency of
approximately 40% compared to 30% for the general population.
The mean age of the ANS group was 68.2 years, representing the group with the highest
proportion of elderly subjects (Table 4.3). The two groups of aspirin takers had the highest
mean ages when compared to non-aspirin takers. Low numbers in a sample biases the
distribution and lowers the power (sensitivity) of the statistical analyses. Low power makes
it more difficult to assess the real association of the independent variable(s) (e.g. aspirin and
ex-smoking), by statistically increasing the probability of Type II errors, making it more
difficult to reject the null hypothesis (Fletcher et al. 1988). The relatively low numbers in
the ANS group could have contributed to the low power in the present study (see Table
4.36).
This is evident from Table 4.36 that all power values were lower for the aspirin associations
compared to ex-smoking associations, (153 aspirin takers compared to 239 non-aspirin
takers) (Table 4.2). The relationship that low numbers could have contributed to the low
117

power statistic is also evident for ex-smoking at a severity level of 7mmPAL, (121 ex-
smokers to 70 non-smokers).
At a threshold of 5mm mean PAL, there were no statistically significant differences
between aspirin takers and non-aspirin takers (Table 4.23). This may have been due to the
low number of aspirin takers (145) i.e. a statistical result rather than a non-physiological
association as there was insufficient statistical power to reject the null hypothesis. However
210 ex-smokers did give enough power to give a significant finding at this level of PAL. At
higher thresholds with lower numbers of subjects, both aspirin and ex-smoking, did not have
statistically significant associations with severity of PAL.
In assessing the extent of PAL, all thresholds had statistically significant associations with
both aspirin and smoking histories because the ‘N’ values were higher for extent of PAL
than for severity values, since it measured the dispersion of PAL (Table 4.25). However
the low to moderate power values highlight the fact that the study population was a
convenience sample and indicates that caution is warranted in extrapolating the findings to
the general population (Table 4.36).
There may be more males in the community (with poorer health due to past smoking) who
are taking aspirin giving a relatively larger pool of subjects with these characteristics.
Individuals on aspirin with a past history of smoking generally had more systemic illnesses
(Table 4.37). This could have accounted for the higher numbers in the AXS group and lower
numbers in the AXS group (less disease experience).
There are many reasons for people to volunteer and participate in a study. The AXS subjects
may have been more informed about the protective associations of aspirin and may have
been interested to participate. A shortcoming of this study was that no information was
118

obtained as to why the subjects participated, why they were on aspirin, how they obtained
the aspirin and whether they knew about aspirin's effects.
Alternatively there may be fewer people in the community on aspirin who have never
smoked. This is probably reflected by the fact the NANS group had the highest number of
subjects (Table 4.2). Yet another explanation for the lower number of subjects in the ANS
group is that people who have never smoked but who take aspirin may consider that they are
healthier than others and that they do not need to see a health professional. Better incentives
to participate (other than a free dental check up and a Colgate Oral Care Kit) may have been
needed to target this group of people.
The advertising targeted subjects for all four groups with emphasis on aspirin takers; more
specific advertisements' placed for a longer period of time may have resulted in more ANS
subjects volunteering.
5.2 Questionnaire
Questions were worded as in the W.H.O. Oral Health Surveys (W.H.O. 1997) and Australian
of Bureau Statistics (McLennan 1996); a multiple-choice format was used (Appendix D).
The questionnaire was self administered and unsupervised to avoid interviewer bias and
influence (Abramson 1974). This format minimised the number of staff required, made it
more likely that all the subjects answered the same questions and sensitive topics like
pension status and medical conditions were answered truthfully (Fink 1995b). However, this
method relied on individuals being able to read and comprehend the questions, eliminating
subjects who were illiterate, had poor English language skills or who were mentally
handicapped. Since this study had subjects with high levels of English language skills (Table
4.9) the results obtained from the questionnaire could be assumed to be relatively valid.
119

Due to financial constraints, questionnaires were not mailed to subjects prior to their
examinations. This could have avoided errors relating to the subject’s recollection of aspirin
dosage and duration, and smoking history, which relied on subjects' unaided memories; it
may have given the subjects time to seek information from other members of their family or
their medical practitioner.
5.2.1 Socio-economic status
Socioeconomic status has often been associated with the prevalence of periodontal diseases.
In general, the higher the socioeconomic status, the greater the awareness of oral health
practices (less tooth loss and less PAL) (Srikandi 1982; Papapanou 1996). In this study the
indicators used for socio-economic status were education and pension status (as the factor
indicating income).
Education appears to be more closely related to oral health than occupational income level
(Richards and Barmes 1971; Srikandi 1982). Since most people feel uneasy or sensitive
about releasing information about their income, the main indicator used in this study was
pension card status. Classification of educational level was based on that used by the
Australian Bureau of Statistics (McLennan 1996) and a similar format for pension card
status as used by the South Australian Dental Longitudinal Study (Spencer 1997). It is
assumed that the higher the education level, the better the socialisation process since
socialisation is associated with "better" health behaviour (Blaikie 1979; Hicks and Newcomb
1981). The study population comprised subjects with middle and higher educational levels
and there were no significant distribution differences among the 4 groups. Higher educated
subjects brushed more frequently, and had a more recent scale and clean than the lower
educated groups (Tables 4.10, 4.11). The number of subjects with higher education and
good English skills was high compared to the general population of Adelaide. These
attributes have been shown to help in the socialisation of subjects and correlate well with
120

good oral health (Srikandi 1982). This could mean that this study population might have
had less PAL than the general population of males aged 50 and over in Adelaide. This
would need to be confirmed in a wider study.
The economic variable taken into account was the pension status; pensioners representing
low income. Approximately 58.9% of subjects were pensioners with the two aspirin groups
having the highest number of pensioners (Table 4.6). In this context, the aspirin groups
could be interpreted as the lower income groups compared to non-aspirin groups. Compared
with non-pensioners, pensioners were less likely to use a mouth rinse, floss and have a scale
and clean. Surprisingly, more pensioners brushed more than once per day compared to non-
pensioners (Table 4.10).
5.3 Periodontal attachment loss
5.3.1 Age associations with PAL
PAL increases with increasing age, reflecting cumulative episodes of active PAL over
time (Beck et al. 1990; Brown and Löe 1993; Brown and Garcia 1994; Mullally and
Linden 1994; Slade and Spencer 1995; Locker et al. 1998). In the current study,
ANOVA analyses were used to assess the statistical associations of age with different
indices of PAL (Table 4.38). The associations of age with PAL were statistically
significant, but minor in comparison to the associations with smoking and aspirin history.
This finding is in agreement with the epidemiological literature which shows that age is
associated with increasing PAL and alveolar bone loss among older age groups, but that
this association was clinically insignificant (Burt 1994). Burt concluded that the
associations of age and low level of PAL:
"is not sufficient alone to cause tooth loss which
is the most clinical important endpoint of periodontal disease".
121

PAL measurements are retrospective in cross-sectional studies and therefore cannot
determine if PAL in the elderly is a function of time or aging (Burt 1994). Many studies
have shown similar results to the current study (Papapanou et al. 1989; (Ismail et al.
1990; Haffajee et al. 1991; Mullally and Linden 1992; Locker and Leake 1993b).
Other studies have found no association of increasing age and increasing PAL (Albandar
1990; Gribic et al. 1991; Brown et al. 1994; Beck et al. 1995; Ship and Beck 1996).
ANOVA analyses showed statistically significant associations between age and PAL; the
magnitude of age on PAL was small, giving further validity to the independent
associations of aspirin and ex-smoking.
In summary, the age relationship with PAL was:
(i) an association with PAL rather than a cause of PAL
(ii) minor in magnitude, indicating that age was a poor predictor of PAL and
not a clinically significant risk factor for PAL.
5.4 Measuring PAL
5.4.1 Case definitions
One of the fundamental principles in measuring PAL is to capture the degree of
destruction of periodontal support. Two methodological characteristics required to
measure the degree of destruction are concise definitions of "cases" and units of
analysis. The selection of periodontal sites around the dentition with which to
calculate summary measures is an important methodological issue. Since the number
of sites that could be measured is large, a careful selection of sites that minimises loss
of information from partial recording is often necessary. Although a number of studies
investigated various combinations of sites to indicate overall periodontal status,
additional research is needed to better assess information lost from partial recordings.
Prevalence of PAL is potentially underestimated with partial recording; the degree of
122

under estimation varies widely, depending on factors such as the severity level at
which the disease is assessed and its true distribution (extent) within and between
subjects. Compared with prevalence, the estimates of extent or severity of PAL are
less influenced by partial recording (Carlos et al. 1986; Papapanou et al. 1993;
Papapanou 1996).
Past studies (Alabandar et al. 1999; Anagnou-Vareltzides et al. 1996; Slade and
Spencer 1995; Bagramian et al. 1993; Gilbert and Heft 1992; Horning et al. 1990;
Brown et al. 1989) have measured the extent and severity of periodontitis using
different baseline measures of PAL. Beck (1990) considered that a severity score of
4mm loss of PAL would be serious in young subjects while the same score in older
subjects would have a different severity value. To identify the subjects with severe
attachment loss, the same author used a threshold measure of 4 sites or more with a
loss of attachment 5mm and one of those sites had to have a pocket depth of 3mm.
These threshold definitions can hardly be described as severe in the everyday clinical
context. Partial recordings reduce the number of sites recorded and with few people
expressing most of the PAL, these methods underestimate the prevalence and severity
of PAL.
Other studies (Bagramian et al. 1993; Beck 1990; Gilbert and Heft 1992) have used
different definitions for severity of PAL, some looking at the worst site score per
mouth, others use means set at 7 mm PAL as the measure of severe periodontal
disease in older subjects (Gilbert and Heft 1992; Slade and Spencer 1995). A threshold
is set to measure the severity and extent of disease using the mean values of PAL as the
unit of measure. Partial and full-mouth recordings are used and it is therefore often
difficult to compare and analyse the results of different studies. In addition, taking
123

mean values of all sites or setting threshold values is not an accurate representation of
the true attachment loss, since 80-99% of sites have PAL between threshold values of
1mm-4mm PAL (Baelum et al. 1986; Beck et al. 1990; Gilbert and Heft 1992;
Mullally and Linden 1992; Slade and Spencer 1995). The very large number of
shallow sites lowers the mean, which lowers the severity and prevalence rates. Data
for deeper sites are swamped by the shallow sites, underestimating the true values of
severity and extent. In addition there are some major shortcomings when setting high
thresholds (7mm PAL):
as the number of deeper sites decreases dramatically the power of the
statistical analysis is reduced. Therefore, large sample sizes are required.
if one subject had numerous sites of 6mm PAL, they would not be classified
as having severe PAL, whereas a subject with one site 7mm PAL would be.
Is the presence of one severe site more indicative of severity than or the
presence of multiple moderate sites? There is no satisfactory answer to this
question.
These arguments highlight that epidemiological studies of PAL have no standard case
definitions for severity of PAL and most are arbitrary and inconsistent (Locker and
Leake 1993a; Papapanou 1996).
In order to overcome the problems of low subject numbers, the numerous sites with
low mean PAL and to identify subjects with multiple sites with moderate PAL, the
most severe site per tooth (MSS-PAL) was developed as a different measure of PAL.
This method may be a more relatively valid measure of site specific severe attachment
loss than mean PAL. Other studies have used a subject-specific measure of attachment
loss; the worst site of attachment loss per mouth as a measure of severity, bypassing
124

the overwhelming lower value site scores (Beck et al. 1990; Gilbert and Heft 1992;
Slade and Spencer 1995). However the limitation of this method is that a small
number of people in a given population are affected by the most severe PAL
(Papapanou 1996; Locker et al. 1998) while the majority of subjects would have low
values of worst scores. Therefore, the number of affected sites are low and the
variation or standard deviation of the values increases making it more difficult to reject
the null hypothesis. This measure may be more useful in epidemiological studies with
very large samples.
5.5 Outcomes of aspirin and past smoking on PAL
Long term, low-dose aspirin provided a significant protective association by lowering
the severity and extent of PAL in both ex- and non-smokers. Controlling for age,
aspirin takers had significantly less PAL than non-aspirin takers in both ex- and non-
smokers. Long term, low-dose aspirin use had statistically significant positive
correlations with different measures of PAL (Tables 4.19, 4.23-25) which were evident
at low and high levels of attachment loss. Figure 4.4 illustrates the study’s findings in
a diagrammatic form, highlighting the differences in subjects according to aspirin and
smoking histories using three different measures of PAL. This is the first time these
outcomes have been reported; the findings could have interesting ramifications in
terms of the management of periodontal diseases and may give further insights into
their pathogenesis.
5.5.1 Mean PAL
The most intriguing finding was that a lower mean PAL was consistently observed in
those subjects who took aspirin, compared with those who did not. The findings were
similar to those of past NSAID studies (Williams et al. 1989; Jeffcoat et al. 1991;
125

Reddy et al. 1993). The data could be interpreted as indicating that aspirin reduced the
rate of attachment loss. In addition non-periodontal in vitro and in vivo models show
that ATLs reduce pro-inflammatory cytokines (LTB4, IL-8, TNF- & IL-1) from
epithelial, endothelial and neutrophil cells and may increase INF-, IL-4 and IL-13
(Claria 1996; Claria 1995; Serhan 1997; Takano et al. 1997 Chiang et al. 1998;
Gronert et al. 1998; Clish et al. 1999;). Therefore ATLs and lipoxins are anti-
inflammatory and could have modulating effects on PAL, alveolar bone loss and
immune responses (there are no periodontal studies of the effects of ATLs on PAL to
date). These hypotheses needs to be confirmed by prospective human periodontal
studies. Although the significant association between the subject groupings could have
been related to selection bias, the statistically significant differences and the internal
consistency of the findings indicate that this explanation is unlikely to be valid.
Furthermore, the data showed that ex-smokers had higher levels of PAL than non-
smokers, in line with the well-documented detrimental association of smoking on PAL
(Bergström et al.1983; Bergström et al.1986; Haber et al.1992; Haber et al.1993;
Grossi et al.1997) and further supporting the validity of the data.
Severity analyses (Table 4.23) showed significantly greater amounts of mean PAL in
the older aged subjects and ex-smokers, with less PAL in subjects who did not smoke.
Extent analysis showed significantly greater amounts of mean PAL among ex-smokers
than those who did not smoke. The magnitude of severity and extent on mean PAL
was consistently less in aspirin takers than in ex-smokers as shown in Table 4.25 with
the ratio of aspirin to ex-smoking being 3:4 (Table 4.31).
126

5.5.2 MSS-PAL
Often severity and extent scores are expressed as mean values. The arithmetic mean is
the sum of values divided by the number of events, making the mean a measure of
central tendency and not very representative in skewed samples. In the current study,
the majority of subjects expressed a large number of sites with low mean PAL values
(Table 4.24), skewing the sample towards low mean PAL values. When the data were
analysed using the most severe site of PAL (MSS-PAL) for each tooth, averaged per
subject, the same pattern of PAL was observed in relation to the subjects’ aspirin and
smoking histories. This measure of PAL was chosen in addition to mean PAL to
reduce the data from shallow sites, which predominated in this study. The MSS-PAL
measure selected sites with the most attachment loss; the differences between the
measures of PAL are apparent in Fig 4.4. Low-dose aspirin and smoking had highly
significant associations with the severity of MSS-PAL.
5.5.3 EWS-PAL
When analysing the age distribution of subjects within groups the older age distribution
of the ANS group had major implications on the extent and severity of disease. There
was a mean age difference of 8-9 years between ANS and NANS (Tables 4.4 and 4.5)
and in addition the ANS group had the lowest number of subjects which may have
influenced the statistical analysis. Since the current study population differed from the
general population in terms of age of subjects in the different age class intervals, data
weighting was used in another assessment of severe PAL (EWS-PAL) (Tables 4.28
and 4.30). This method highlighted the aspirin and smoking differences that were also
seen in mean and MSS-PAL. The cumulative percentage scores from Figure 4.5
showed that all subjects had at least one site of maximum 4 mm EWS-PAL. At 7 mm
EWS-PAL, all groups showed a similar trend. As the severity of PAL increased, the
127

number of subjects with sites 7mm EWS-PAL decreased. These data showed that
subjects taking low-dose aspirin with or without a history of smoking had significantly
less severe EWS-PAL than non-aspirin takers with or without a history of smoking.
Some age class intervals had more subjects assigned to them by the weighting process than
other age class intervals (Table 4.28). For example, the 50-59 age class in the ANS group had
7 subjects; when weighted, the final ‘number’ was 21. The same age class interval in the
NANS group had 63 subjects and when weighted, the final ‘number’ was 48. These
highlights the shortcomings of the weighting process, which on one hand, extrapolates data
obtained from a few subjects and, on the other, disregards data from others. It is the former
that is most disturbing, because it is assumed that the weighted subjects had identical patterns
of PAL as the subjects who had been examined. One must take particular caution in
extrapolating current EWS-PAL data to the general population, (because of data weighting)
nevertheless the findings followed the pattern of the other analyses (mean and MSS-PAL).
5.5.4 Plaque
There were no significant differences in plaque accumulation between the groups
(Tables 4.14 & 4.15). Older subjects had significantly more plaque than younger
subjects in all four groups, as has been reported by others (Holm-Pedersen et al. 1975;
Locker and Leake 1993a; Grossi et al. 1995). One study found the reverse
relationship, with younger subjects having higher plaque scores and gingival bleeding,
although the age differences were not large (van der Velden et al. 1985). A similar
study using a broader age range and experimental gingivitis found no association of
plaque and gingival bleeding with age (Winkel et al. 1987). Findings from this study
indicate that plaque levels and PAL were not associated, as has been shown in other
studies (Baelum et al. 1986; Bagramian et al. 1993).
128

5.5.5 Gingival bleeding
Using the mBSI, 57.7% of all the subjects' sites had gingival bleeding, with younger subjects
having less gingival bleeding (Figure 4.1). ANOVA showed that low-dose aspirin and ex-
smoking history had no association with gingival bleeding. Smoking in some studies has
been shown to inhibit the inflammatory response with less gingival bleeding, less redness and
less gingival fluid flow (Preber and Bergström 1986; Danielsen et al. 1990; Grossi et al.
1997). Of more significance, there were no differences in gingival bleeding between aspirin
takers and non-aspirin takers. This is a surprising finding since the anti-platelet and anti-in-
flammatory effects of aspirin could have been expected to reduce gingival inflammation, with
increased gingival bleeding. This result is contrary to some past findings that subjects on sys-
temic NSAIDs had reduced gingival indices (Waite et al. 1981; Heasman and Seymour 1989;
Johnson et al. 1990). Other studies of NSAIDs (of less than 8 weeks duration) showed no
differences in gingival inflammation (Vogel et al. 1983; Heasman et al. 1989; Flemmig et al.
1996). In longer-term studies of NSAIDs, there was reduced gingival bleeding and colour
change by 2-3 months followed by a gradual return to normal, indicating that the inflamma-
tory response has compensating mechanisms (Taiyeb and Waite 1993; Ng and Bissada 1998).
It is assumed that by 2-3 months, the leukotriene mediators (LTB4) may compensate for the
inhibited COX mediators (PGE2 and TxB2), restoring a normal inflammatory response. Al-
ternatively, other factors such cytokines IL-1neutrophil/platelet adhesion may affect the
cellular and vascular elements of inflammation . Recent studies (Claria 1995; Claria 1996;
Clish et al. 1999; Serhan 1997; Takano et al. 1997) investigating the effects of ATLs
could account for the non-significant differences in gingival bleeding between aspirin takers
and non-aspirin takers. However to date there are no studies investigating the effects of ALTs
in the periodontal inflammatory process.
129

The lack of significant differences in gingival bleeding between groups may support these as-
sumptions. This current study may have benefited in measuring GCF-mediators as more ac-
curate measurements of gingival inflammation.
5.6 Comparisons with other aspirin studies
It is not possible to compare the present study with past studies on the association of
aspirin on the periodontium because no other studies have investigated low-dose
aspirin. In addition, previous studies have a wide disparity in drug combinations, drug
doses, difficulty in controlling for specific medications or the physical health status of
the subject, subject selection and, in some studies, the very low numbers of subjects.
Subjects in previous studies that have investigated the associations of aspirin on
periodontal conditions using doses that ranged from 650mg to >3 gm per day (Waite et
al. 1981; Feldman et al. 1983; Flemmig et al. 1996).
Waite et al., (1981) found a significant decrease for gingival index and pocket depth in
subjects (suffering from rheumatoid arthritis) receiving NSAID therapy. Although there
was a trend towards less PAL in aspirin takers, it was not significant. Possible reasons for
the non-statistical difference on loss of attachment in the Waite et al. study were that the
number of test subjects was low (22), and that the study was biased as the controls were
not from the same population. The uneven distribution of the duration of drug therapy
(this was not controlled for) was another factor to account for. Furthermore, only one
subject took aspirin while the rest were on different NSAIDs. The dosage regime varied,
some subjects took the drugs as required for pain (or continuously) others took different
NSAIDs simultaneously. In addition, no account of smoking history was provided;
smoking effects could have overwhelmed the aspirin associations.
130

The present study did find that low-dose aspirin had significant associations with PAL.
The differences in attachment loss findings between studies was possibly due to the
stringent inclusion criterion of long-term duration of aspirin therapy (minimum of 2
years) in the present study, while the subjects in the Waite study had been on NSAID
therapy for a minimum of one year, and this study's exclusion of subjects suffering from
rheumatoid arthritis. Some authors believe that there is an increased prevalence and
severity of periodontal disease in rheumatoid patients which may have influenced the
Waite (and other) results (Tolo and Jorkend 1990; Kasser et al. 1997). Furthermore,
females were included by Waite et al.(1981) which may have added the confounding
effect of gender. The large age range (22 – 68) of the test group in the Waite study may
have affected the prevalence of PAL (less PAL).
Feldman et al. (1983) carried out a cross-sectional periodontal radiographic study of 75
males (from a rheumatology clinic) taking aspirin (650mg-3.9g per day) for at least 5
years. Controls were 75 males from an ongoing longitudinal study. The aspirin group
was matched to the control group for age, dentition similarity and similarity of remaining
teeth (10 teeth minimum). Aspirin takers had significantly fewer sites, which exhibited
10% or more interproximal bone loss than the controls. The average percentage of
bone loss per dentition was lower in the group taking aspirin, although the difference
was not statistically significant. It is difficult to compare these findings with those
from the present study because of the different methods used to measure PAL, and the
high doses of aspirin that subjects had been taking in the Feldman study (650mg-3.9g
per day). The current study did not investigate radiographic changes in alveolar bone. A
longitudinal study using radiographs on subjects taking low-dose aspirin with or without
periodontal treatment would be an important extension of the present study.
131

Flemmig et al.(1996) found that acetylsalicylic acid (2g per day) in conjunction with
scaling in 30 patients with moderate-severe periodontitis resulted in a synergistic
therapeutic efficacy approximately equivalent to the sum of each therapy, significantly
reducing gingival inflammation, pocket depth and probing attachment loss. However,
this study did not control for age and sex, it used high doses of aspirin over only 6
weeks and there was no control for smoking.
According to Flemmig et al.(1996):
“The clinical effects of systemically administered NSAID (aspirin) do not appear to extend beyond the termination of the medication, either when administered alone, or in combination with scaling. Thus, significant benefits of systemically administered NSAID (aspirin) on periodontal health can only be expected from a long term regimen”.
Although these comments apply particularly to the high doses of aspirin taken over a
short time, they are pertinent in relation to the present findings.
The findings from this current study contradict the findings of Heasman et al.(1990) who
evaluated the collective associations of various NSAIDs in a group of hospital
rheumatology patients with non-rheumatology patients used as controls. Males and
females were studied and no statistically significant differences were found between the
exposed and non-exposed groups for plaque index, gingival index, probing depth, loss of
attachment, recession and alveolar bone loss. The non-statistical differences may have
been due to the fact that the exposed and non-exposed subjects came from different
sample frames and less than 10% of test and control subjects had probing depths greater
than 3mm.
132

5.7 Smoking and PAL
Smoking is strongly associated with PAL (Bergström and Floderus-Myrhed 1983; Haber
and Kent 1992; Haber et al. 1993). The amount of periodontal damage is associated with
the number of pack years and duration of smoking (Ismail et al. 1983; Goultschin et al.
1990; Grossi et al. 1995; Grossi et al. 1997; Salvi et al. 1997). People who quit smoking
lie between non-smokers and current smokers in terms of periodontal conditions (Grossi
et al. 1997).
Current smokers were excluded from the present study because of the known
differences in the dynamics of attachment loss between ex- and current smokers
(Grossi et al. 1997). In addition, the inclusion of current smokers in the study would
have necessitated the recruitment of an extra 2 groups of subjects (current smokers
taking and not taking aspirin) making the study too large to undertake. In the present
study, ex-smokers had more PAL than non-smokers, confirming previous studies.
Even though 89% of the ex-smokers had quit for at least 5 years, the adverse effects of
smoking on PAL were apparent, reflecting the increased susceptibility of smokers to
PAL and the cumulative nature of attachment loss. Ex-smokers taking low-dose
aspirin had significantly less PAL than ex-smokers who did not take aspirin. The
powerful association of low-dose aspirin, with reduced PAL was emphasised by this
finding since it occurred in both non- and ex-smokers. The findings give credence to
the hypothesis that aspirin lowered PAL.
5.8 Prevalence of periodontal attachment loss.
In the NAXS group, 38% of subjects had mean MSS-PAL of 5mm, while AXS had
26% of subjects with a MSS-PAL of 5mm. The percentage of subjects with severe
PAL was low. These findings correspond with other reports (Committee of Research
133

Science and Therapy 1996; Locker et al. 1998). For example in a study by Ismail et al.
(1987), 34% of subjects had PAL 7 mm but only 7% of sites had 7mm PAL.
However, it is erroneous to assume that the attachment loss observed in this and other
epidemiological studies was solely due to the effects of destructive periodontal
diseases (periodontitis). Epidemiological studies do not discriminate between the
varied causes of PAL, which may masquerade as periodontitis, or be present in
addition to periodontitis. These causes of PAL include continuous tooth eruption
(Clarke et al. 1986; Clarke and Hirsch 1992; Newman 1999), dehiscence, cervical
enamel projections, cracked or split teeth and retrograde periodontitis (Bergenholtz and
Hasselgren 1998) and result in over-estimation of the prevalence of periodontitis in all
epidemiological studies (Clarke and Hirsch 1992). The prevalence of severe
periodontal diseases may be less than the general accepted prevalence rate of 10-15%
within populations.
5.9 Future recommendations
With the reduced severity and extent of PAL in ex-smokers taking aspirin, it is
tempting to speculate that current smokers who are unable to quit may also benefit
from taking low-dose aspirin to reduce their periodontal and cardiovascular risks.
Further work needs to be done to establish whether patients with periodontitis would
benefit from taking low-dose aspirin as an adjunct to periodontal therapy and whether
low-dose aspirin modulates the effects of periodontitis in susceptible, untreated
populations. It would be of interest to investigate the associations of aspirin with PAL in
a prospective study. Studies of similarly aged females and younger subjects would also
be beneficial.
134

It would be of considerable benefit to follow up the same subjects (by converting this
study into a longitudinal study), which would yield further epidemiological data in
relation to aspirin and ex-smoking associations with PAL. Prospective studies on low-
dose aspirin would be useful to investigate:
its use as an adjunct to conventional periodontal treatment
the associations of low-dose aspirin with the periodontal status of current
smokers
the associations in women of similar age groups
the associations on young subjects
the its associations in implant cases
Conclusions
Long term (2 years), low-dose (350mg) aspirin may infer a significant
protective benefit on the periodontium, as both ex- and non-smokers had
reduced severity and extent of PAL.
Controlling for age, both ex- and non-smoking aspirin takers had
significantly less PAL than non-aspirin takers.
Subjects on long term, low-dose aspirin had significant correlations with
different measures of PAL, which were evident at both low and high levels
of attachment loss.
The present study population was highly dentally aware and this may have
been due to the higher than normal education levels.
This is the first time these associations have been reported; the findings could have
interesting ramifications in terms of the management of periodontal diseases and may
give further insights into their pathogenesis.
135

The most intriguing finding was that a significantly lower mean PAL was consistently
observed in those subjects who took aspirin, compared with those who did not. One
interpretation of the data is that aspirin reduced the rate of attachment loss. This
hypothesis needs to be confirmed by a prospective study. Although the significant
differences between the subject groupings could have been related to selection bias, the
significant statistical outcomes and the consistency of the findings indicate that this
explanation is unlikely. Furthermore, the data showed that ex-smokers had higher
levels of PAL than non-smokers, in line with the well-documented detrimental
association of smoking on PAL and further supporting the validity of the data.
With the lower levels of PAL in ex-smokers taking aspirin, it is tempting to speculate
that current smokers who are unable to quit may benefit from taking low-dose aspirin
to reduce both their periodontal and cardiovascular risks. Enterically coated aspirin
has few side effects (Axon and Huskisson 1992), has proven benefits in protecting
against vascular diseases and is inexpensive, giving extra weight to the advice that
patients take aspirin to reduce their risk of PAL. Further work needs to be done to
establish whether patients with periodontitis can benefit from taking low-dose aspirin
as an adjunct to periodontal therapy and whether low-dose aspirin modulates the
effects of periodontitis in susceptible, untreated populations.
136

Appendix A
The University of AdelaideDepartment of Dentistry
Information sheet for participants in the research project"Periodontal conditions in smokers and non-smokers
on long term, low-dose aspirin therapy.”
Purpose of the study Healthy gums mean that your teeth are well supported in your jaw. Although we know that gum diseases can start when people do not clean their teeth properly and when they have risk factors (for example, smoking), there is still much to learn about why some people are more likely to develop gum diseases than others. This research project aims to find out whether people who are taking small amounts of aspirin every day have less gum disease than people who are not taking aspirin regularly. In order to find this out, we need to look at the health of the gums in people who are both smokers and non-smokers and aspirin takers or not.
What is involved? Details about your past and present medical and dental history will be recorded. Of
importance it will be necessary for you to tell us whether you are on aspirin medication, what type and how much you are taking.
Your smoking history - whether you are a smoker and if so how many cigarettes are smoked per day and for how long you have been smoking.
A single-visit check up of the health of your gums. This will include a measure of the amount of inflammation (redness, bleeding) of the gums and measuring any areas of more severe gum disease using an instrument (periodontal probe) which slides down into the space between the tooth and gum.
This examination will take about 10 minutes.
What are the benefits to me? You will be informed if there are any conditions which need attention. The information obtained in this study of several hundred people may be useful in
helping us work out what makes some people more likely to develop gum diseases than others and whether aspirin treatment can help prevent gum disease.
Are there any risks?The risks of being part of this study are very low. The clinical examination involves the same steps used in a thorough dental check-up; there is minimal discomfort during periodontal probing. You may withdraw from the study at any time. All information you give us will be treated with the utmost confidentiality.
Please contact the following person if you have any questions:Dr Arthur Drouganis
137

Appendix B
THE UNIVERSITY OF ADELAIDE
CONSENT FORM
See also Information Sheet attached.
1. I (please print) hereby consent to take part in the research project entitled:Periodontal conditions in ex-smokers and non-smokers on long term, low-dose aspirin therapy.
2. I acknowledge that I have read the Information Sheet entitled:“Periodontal conditions in ex-smokers and non-smokers on long term, low-dose aspirin therapy.”
3. I have had the project, so far as it affects me, fully explained to my satisfaction by the research worker. My consent is given freely.
4. Although I understand that the purpose of this research project is to improve the quality of medical care, it has also been explained that my involvement may not be of any benefit to me.
5. I have been given the opportunity to have a member of my family or a friend present while the project was explained to me.
6. I have been informed that, while information gained during the study may be published, I will not be identified and my personal results will not be divulged.
7. I understand that I am free to withdraw from the project at any time and that this will not affect medical advice in the management of my health, now or in the future.
8. I am aware that I should retain a copy of this Consent Form, when completed, and the relevant Information Sheet.
SIGNED DATE _________
NAME OF WITNESS (Please print)
SIGNED DATE _________
I, have described to (Please print)
the nature of the procedures to be carried out. In my opinion she/he understood the explanation.
SIGNED DATE _________
STATUS IN PROJECT Examiner
138

Appendix C
Oral Health Report Form
University of AdelaideDepartment of Dentistry.
NAME……………………………….. DATE…………….I Dr A Drouganis, conducting a periodontal study from the Department of Dentistry at the University of Adelaide, have examined the above named participant.The following conditions were noted:
Dental Caries ……………………………………………………………..Periodontal Pocketing (4mm or more) ……………………………..Mobility (greater than class III) ……………………………………..Calculus / Gingivitis ……………………………………………………..Inflamed / abnormal oral mucosa ……………………………………..Other……………………………………………None of the above conditions
From the findings of this examination I have advised the patient that in my opinion, he/she should seek dental advice/care:
ImmediatelyIn the near future
During a regular recall visit in the future.
Examiner: Dr. A Drouganis. B.D.S., MDS Postgraduate student.
Signature: ________________________________________ Date: _________
139

Appendix D
QuestionnaireID. Number
Surname: Dr/Mr/Mrs/Miss/Ms __________________________________________________
Preferred Name:__________________________________________________
Address:__________________________________________________
Suburb: _____________________
Post code _____________________
Age: _____________________Is there a phone number we could contact you on for further information in relation to this project, if needed?
Ph:_____________________________________Please tick your answer to each question. It should take 10 - 15 minutes to complete. Q1. In which country were you born?
1 - Australia2 - England3 - Scotland4 - Italy5 - Greece6 - New Zealand7 - Viet Nam8 - Other – please specify?
______________________Q2. Education: What level of education did you achieve?
No schooling 1 - Primary 2 - Secondary 3 - Tertiary 4 -
Q3. How well do you speak English?1- Very well2 - Well3 - Not well4 - Not at all
Q4. Are you on a Pension or on Health a Concession Card?1 - Yes Go to Q 52 - No Go to Q 4
MEDICAL HISTORY
140

Medical History:Q5. Do you have or have suffered from any of the following conditions:
Please tick: Yes NoHeart murmurHeart / vascular disordersRheumatic FeverRheumatoid ArthritisDiabetesLiver or Kidney diseaseCancer therapyHepatitis / HIV / AIDSExcessive bleedingAntibiotic therapyQ6. Are you taking any medications or drugs other than aspirin:
1 – Yes 2 - No
If yes, please specify name of condition(s). ________________________________________________________________________________________Name of Drug(s). _________________________________________Dosage: _________________________________________
ASPIRIN HISTORYQ7. Do you take prescribed aspirin?
1 - Yes 2 - No Go to Q 12
If yes, what type or brand do you use?_________________________________Q8. How often do you take aspirin? Please tick appropriate box.
1 - 1/day 2 - 2/day 3 - 3/day
Q9. How many tablets of aspirin do you take each time?1 - ½ /tab2 - 1 tab3 - 2 tabs4 - Greater than 2 tabs
Q10. How much aspirin is in each tablet ____________ mg/tab.Q11. How long have you been taking aspirin? Please tick.
1 - 2-5 yrs 2 - 6-10 yrs 3 - 11-20 yrs 4 - greater 20 yrs
SMOKING HISTORYQ12. Have you ever smoked cigarettes regularly? 1 - Yes
2 - No Go to Q 17
Q13. Do you currently smoke cigarettes? 1 - Yes Go to Q152 - No
Q14. If you have quit smoking when did you quit?1 - Quit 2-5 yrs ago
141

2 - Quit 5 or more yrs agoQ15. How many cigarettes did you or are you currently smoking in a day? Please tick.
1 - Less than 10 / day2 - 10-19 / day3 - 20 –30 / day4 - More than 30 / day
Q16. How many years had you been a smoker?1 - Less than 5 years2 - 5-10 years3 - Greater than 10 years
DENTAL HISTORYQ17. Do you have more than 10 natural teeth?
1 Yes 2 - No
Q18. Do you have a denture or false teeth for your upper jaw?1 - Yes 2 - No
Q19. Do you have a denture or false teeth for your lower jaw?1 - Yes 2 - No
Q20. How often do you brush your natural teeth?1 - Never
2 - Twice a day or more 3 - Once a day4 - 4-6 times/ week5 - 1-3 times / week6 - less once / week7 - Intermittently or hardly ever
Q21. How often do you use a commercial mouth rinse?1 - Never2 - Twice a day or more3 - Once a day4 - 4-6 times a week5 - 1-3 times / week6 - less than once / week7 - Intermittently or hardly ever
Q22. How often do you use dental floss or a special brush to clean the spaces between your teeth?
1 - Twice a day or more2 - Once a day3 - 4-6 times a week4 - 1-3 times / week5 - less than once a week6 - Intermittently or hardly ever7 - Never
Q23. How long has it been since you have seen your dentist, hygienist, about your teeth and gums?
1 - Less than 6 month2 - Last 6-12 months3 - 1-2 years
142

4 - 3-5 years5 - greater than 5 years6 - greater than 10 years7 - Never
Q24. Have you had your teeth scaled or cleaned?1 - Never2 - In the last 6mths3 - In last 6-12 months4 - In last 1-2 years5 - In last 3-5 years6 - Greater than 6-10 years7 - Greater than 11 years.
Q25. Have you ever had gum treatment or surgery?1 - Yes 2 - No
Q26, How long ago?Write the number of years ago. …………………..
143

References
Abramson JH. Survey methods in community medicine. New York: Churchill Livingstone 106-116, 1974.
Abramson MM, Wolff LF, Offenbacher S, Aeppli DM, Hardie ND, Friedman HM. Flubiprofen effect on gingival crevicular fluid prostaglandin and thromboxane levels in humans. Journal of Periodontal Research 27:539-543;1992.
Abramson SR, Weissmann G. The mechanism of action of non-steroidal anti-inflammatory drugs. Arthritis Rheumatology. 32:1-9.;1989.
Ah MKB, Johnson GK, Kaldahl WB, Patil KD, Kalkwarf KL. The effect of smoking on the response to periodontal therapy. Journal of Clinical Periodontology 21:91-97;1994.
Ainamo J. What are the problems in defining periodontal disease. In: B. Guggenheim, ed. Periodontology Today. München: Karger, 50-58. 1988.
Ainamo JB, Beagrie G, Cutress T, Martin J, Sardo-Infirri J. Develpoment of the World Health Organisation (WHO) Community Periodontal Index of Treatment Needs (CPITN). International Dental Journal 32:281-291;1982.
Albandar JM, Brunelle JA, Kingman A. Destructive periodontal disease in adults 30 years of age and older in the United States, 1988-1994. Journal of Periodontology 70:13-29;1999.
Albandar JM. A six year study on the pattern of periodontal disease progression. Journal of Clinical Periodontology 17:467-471;1990.
Alexander MB, Damoulis PD. The role of cytokines in the pathogenesis of periodontal disease. Current Opinion in Periodontology 1: 39-53;1994.
Anagnou-Vareltzides A, Diamanti-Kipioti A, Afentoulidis N. A clinical survey of periodontal disease. Journal of Clinical Periodontology 23:758-763;1996.
Armitage GC. Periodontal diseases: Diagnosis. Annals of Periodontology 1:37-215;1996.
Axon JMC, Huskisson EC. Use of aspirin in inflammatory diseases. In: Vane JR, Botting RM, ed. Aspirin and other salicylates. London: Chapman and Hall Medical, 295-319. 1992.
Baelum V, Fejerskov O, Karring T. Oral hygiene, gingivitis and periodontal breakdown in adult Tanzanians. Journal of Periodontal Research 21:221-232;1986.
Bagramian RA, Farghaly MM, Lopatin D, Sowers M, Syed SA, Pomerville JL. Periodontal disease in an Amish population. Journal of Clinical Periodontology 20:269-272;1993.
144

Bain CA, Moy PK. The association between the failure of dental implants and cigarette smoking. International Journal of Oral Maxillofacial Implants 8:609-615;1993.
Bando K, Nitta H, Matsubara M, Ishikawa I. Bone mineral density in periodontally healthy and edentulous postmenopausal women. Annals of Periodontology 3:322-326;1998.
Bartold PM, Narayanan AS. Biology of the periodontal connective tissue. Chicago: Quintessence Publishing Company . Quintessence books 28-29, 47-69, 1998.
Beck JD. Periodontal implications: Older adults. Annals of Periodontology :323-357;1996.
Beck JD, Koch GG, Offenbacher S. Incidence of attachment loss over 3 years in older adults - new and progressing lesions. Community Dentistry and Oral Epidemiology 23:291-296;1995.
Beck JD, Koch GG, Rozier RG, Tudor GE. Prevalence and risk indicators for periodontal attachment loss in a population of older community dwelling blacks and whites. Journal of Periodontology 61:521-528;1990.
Bergenholtz G, Hasselgren G. Endodontics and periodontics. (3rd ed.) Lindhe J, Karring T, Lang NP, ed. Clinical Periodontology and Implant Dentistry. Copenhagen: Munksgaard 296-331, 1998.
Bergström J, Floderus-Myrhed B. Co-twin control study of the relationship between smoking and some periodontal disease factors. Community Dentistry and Oral Epidemiology 21:113-116;1983.
Bergström J, Preber H. Tobacco use as risk factor. Journal of Periodontology 65:545-550;1994.
Bergström J, Preber H. The influence of cigarette smoking on the development experimental gingivitis. Journal of Periodontal Research 21:668-676;1986.
Berns JM. Understanding periodontal diseases. (2nd ed.) Carol Stream Illinois: Quintessence Publishing Co. Inc. Holstom BE, ed. 1-73, 1993.
Bienenstock J, Befus AD, Denburg JA. Mast cell heterogeneity: basic questions and clinical implications. In: Befus AD, Bienenstock J, Denburg JA, ed. Mast Cell Differentiation and Heterogeneity. New York: Raven Press Inc., 379-402. 1986.
Bing RJ, Yamamoto T, Yamamoto M, Kakar R, Cohen A. New look at myocardial infarction: toward a better aspirin. Cardiovascular Research 43:25-31;1999.
Birkedal-Hansen H. Role of cytokines and inflammatory mediators in tissue destruction. Journal of Periodontal Research 28:500-510;1993.
145

Blaikie DC. Cultural barrier to preventative dentistry. Australian Dental Journal 24:398-401;1979.
Bourque LB, Fielder EP. How to conduct self-administered and mail surveys. London: SAGE Publications The Survey Kit; 3: 1-223, 1995.
Brägger U, Mühle T, Fourmousis I, Lang NP, Mombelli A. Effect of the NSAID flurbiprofen on remodelling after periodontal surgery. Journal of Periodontal Research 32:575-582;1997.
Brooks MD, Day RO. Nonsteroidal anti-inflammatory drugs: Differences and similarities. New England Journal of Medicine 324:1716-1725;1991.
Brown LF, Beck JD, Rozier RG. Incidence of attachment loss in community-dwelling older adults. Journal of Periodontology 65:316-323;1994.
Brown LJ, Garcia R. Utilization of dental services as a risk factor for periodontitis. Journal of Periodontology 65:551-563;1994.
Brown LJ, Löe H. Prevalence, extent, severity and progression of periodontal disease. Periodontology 2000 2:57-71;1993.
Brown LJ, Oliver RC, Löe H. Periodontal diseases in the U.S. in 1981: Prevalence, severity, extent and role in tooth mortality. Journal of Periodontology 60:363-370;1989.
Burt BA. Periodontitis and aging: reviewing recent evidence. Journal of The American Dental Association 125:273-279;1994.
Campbell WB, Halushka PV. Lipid-derived autocoids. Eicosanoids and Platelet-Activating Factor. In: Hardman JG, Limbird LE, Molinoff PB, Ruddon MD, Goodman Gilman A, ed. Goodman and Gilman's The pharmacological basis of therapeutics. Ninth edition ed. New York: McGraw-Hill, 601-657. 1996.
Carlos JP, Wolfe MD, Kingman A. The extent and severity index: A simple method for use in epidemiological studies of periodontal disease. Journal of Clinical Periodontology 13:500-505;1986.
Census. Basic community profile. Catalogue number 2020.0. In: Adelaide: Australian Bureau of Statistics, 1996.
Chiang N, Takano T, Clish CB, Petasis NA, Tai HH, Serhan CN. Aspirin-triggered 15-epi-lipoxin A4 (ATL) generation by human leukocytes and murine peritonitis exudates: development of a specific 15-epi-LXA4 ELISA. The Journal of Pharmacology and Experimental Therapeutics 287:779-90;1998.
Claffey N, Nylaund K, Kiger R, Garrett S, Egelberg J. Diagnostic predictability of scores of plaque, bleeding, suppuration and probing depth for probing attachment loss.3 1/2 years of observation following initial periodontal therapy. Journal of Clinical Periodontology 17:108-114;1990.
146

Claria J, Lee MH, Serhan CN. Aspirin-triggered lipoxins (15-epi-LX) are generated by the human lung adenocarcinoma cell line (A549)-neutrophil interactions and are potent inhibitors of cell proliferation. Molecular Medicine 2:583-96;1996.
Claria J, Serhan CN. Aspirin triggers previously undescribed bioactive eicosanoids by human endothelial cell-leukocyte interactions. Proceedings of the National Academy of Sciences of the United States of America 92:9475-9;1995.
Clarke NG, Carey SE, Srikandi W, Hirsch RS. Periodontal disease in ancient populations. American Journal of Physiology and Anthropology 71:173-183;1986.
Clarke NG, Hirsch RS. Personal risk factors for generalized periodontitis. Journal of Periodontology 22:136-145;1995.
Clarke NG, Hirsch RS. Two critical confounding factors in periodontal epidemiology. Community Dental Health 9:133-141;1992.
Clish CB, O'Brien JA, Gronert K, Stahl GL, Petasis NA, Serhan CN. Local and systemic delivery of a stable aspirin-triggered lipoxin prevents neutrophil recruitment in vivo. Proc Natl Acad Sci U S A 96:8247-52;1999.
Committee of Research Science and Therapy. Epidemiology of periodontal disease. Journal of Periodontology 67:935-945;1996.
Cutress TW, Ainamo J, Sardo-Infirri J. The community periodontal index of treatment needs (CPITN) procedure for population groups and individuals. International Dental Journal. 37:222-233;1987.
Czuszak CA, Sutherland DE, Billman MA, Stein SH. Prostaglandin, E2 potentiates
interleukin-induced interleukin-6 production by human gingival fibroblasts. Journal of Clinical Periodontology 23:635-640;1996.
Danielsen B, Manji F, Nagelkerke N, Fererskov O, Baelum V. Effects of cigarette smoking on the transition of dynamics in experimental gingivitis. Journal of Clinical Periodontology 17:159-164;1990.
Darveau RP, Tanner A, Page RC. The microbial challenge. Periodontology 2000 14:12-32;1997.
Davidson JM. Wound repair. In: Gallin JI, Goldstein IM, Snyderman, ed. Inflammation: Basic Principles and Clinical Correlates. New York: Raven Press, 809-820. 1992.
Davies P, MacIntyre ED. Prostaglandins and Inflammation. In: Gallin JI, Goldstein IM, Snydermam R, ed. Inflammation Basic Principles and Clinical Correlates. New York: Raven Press, 123-138. 1992.
147

Dennison DK, Van Dyke TE. The acute inflammatory response and the role of phagocytic cells in periodontal health and disease. Periodontology 2000 14:54-78;1997.
Diener HC. Use of acetylsalicylic acid in secondary prevention of stroke. International Journal of Clinical Practice :52: 12-15;1998.
Drisko CH. Non-surgical pocket therapy: Pharmacotherapeutics. Annals of Periodontology 1:491-566;1996.
Dubois RN, Abramson SB, Crofford L, Gupta RA, Simon SL, Van De Putte LBA, Lipsky PE. Cyclooxygenase in biology and disease. FASEB Journal 12:1063-1073;1998.
Ellis SD. Factors for progression of periodontal disease. Journal of Oral Pathology and Medicine 27:101-105;1998.
Faccioli LH, Nourshagh S, Moqbel R, Williams FM, Sehmi R, Kay AB, Williams TJ. The accumulation of 111 In-eosinophils induced by inflammatory mediators. Immunology 73:222-227;1991.
Feldman RS, Szeto B, Chauncey HH, Goldhaber P. Non-Steroidal anti-inflammatory drugs in the reduction of human alveolar bone loss. Journal of Clinical Periodontology 10:131-136;1983.
Fink A. How to ask survey questions. London: SAGE Publishers 1-105, 1995a.
Fink A. How to conduct self-administered and mail surveys. London: SAGE Publications 1-223, 1995b.
Fink A. The survey handbook: Learning objectives. London: SAGE Publishers 23-38, 1995c.
Flemmig TF, Rumetsch M, Klaiber B. Efficacy of systemically administered acetylsalicylic acid plus scaling on periodontal health and elastase-a-proteinase inhibitor in gingival crevicular fluid. Journal of Clinical Periodontology 23:153-159;1996.
Fletcher RH, Fletcher SW, Wagner EH. Clinical Epidemiology: the essentials. (2nd ed) Baltimore: Williams & Wilkins . 77, 178-182, 1988.
Fu JY, Masferrer JL, Seibert K, Raz A, Needleman P. The induction and suppression of prostaglandin H2 synthetase (cyclooxygenase) in human monocytes. Journal of Biological Chemistry 1990:16737-16740;1990.
Gemmell E, Marshall RI, Seymour GJ. Cytokines and prostaglandins in immune homeostasis and tissue destruction in periodontal disease. Periodontology 2000 14:112-143;1997.
148

Genco R, Hamada S, Lehner T, McGhee J, Mergenhagen S. Section II. Host factors : cytokines and other effector molecules. In: Genco R, Hamada S, Lehner T, McGhee J, Mergenhagen S, ed. Molecular pathogenesis of periodontal disease. Washington: ASM Press, 1994: 135-387.
Genco RJ, Löe H. The role of systemic conditions and disorders in periodontal disease. In: Löe H, Jackson Brown L, ed. Classification and epidemiology of periodontal diseases. Copenhagen: Munksgaard, 98-116. 1993.
Gierse JK, Hauser SD, Creely DP, Rangwala SH, Isakson PC. Expression and selective inhibition of the constitutive and inducible forms of human cyclo-oxygenase. Biochemistry 305:479-484;1995.
Gilbert GH, Heft MW. Periodontal status of older Floridians attending senior activity centers. Journal of Clinical Periodontology. 19:249-255;1992.
Glavind L, Löe H. Errors in clinical assessment of periodontal destruction. Journal of Periodontal Research 2:180-184;1967.
Goultschin J, Sgan-Cohen HD, Donchin M, Brayer L, Soskolne WB. Associations of smoking with periodontal treatment needs. Journal of Periodontology 61:364-367;1990.
Grant DA, Stern IB, Listgarten MA. Periodontics in the tradition of Gottlied and Orban. (6th ed.) St Louis: The C.V. Mosby Company 248, 1988a.
Grant DA, Stern IB, Listgarten MA. Periodontics in the tradition of Gottlied and Orban (6th ed.). St Louis: The C.V. Mosby Company 551, 931, 1988b.
Gribic JT, Lamster IB, Celenti RS, Fine JB. Risk indicators for future clinical attachment loss in adult periodontitis. Patient variables. Journal of Periodontology 62:322-329;1991.
Grossi SA, Skrepcinski FB, DeCaro T, Zambon JJ, Cummins D, Genco RJ. Response to periodontal therapy in diabetics and smokers. Journal of Periodontology 67:1094-1102;1996.
Grossi SG, Genco RJ, Machtei EE. Assessment of risk for periodontal disease II. Risk indicators for alveolar bone loss. Journal of Periodontology 66:23-29;1995.
Grossi SG, Zambon J, Machtei EE, Schifferle R, Andreana S, Genco RJ, Cummins D, Harrap G. Effects of smoking and smoking cessation on healing after mechanical periodontal therapy. Journal of the American Dental Association 128:599-607;1997.
Haber J, Kent RL. Cigarette smoking in a periodontal practice. Journal of Periodontology 63:100-106;1992.
149

Haber J, Wattles J, Crowby MRM, Joshipura K, Kent RL. Evidence for cigarette smoking as a major risk factor for periodontitis. Journal of Periodontology 64:16-23;1993.
Haffajee AD, Dibart S, Kent JRL, Socransky SS. Clinical and microbiological changes associated with the use of 4 adjunctive systemically administered agents in the treatment of periodontal infections. Journal of Clinical Periodontology 22:618-627;1995.
Haffajee AD, Socransky SS, Goodson JM. Clinical parameters as predictors of destructive periodontal disease activity. Journal of Clinical Periodontolgy 10:257-265;1983.
Haffajee AD, Socransky SS, Lindhe J, Kent RL, Okamoto H, Yoneyama T. Clinical risk indicators for periodontal attachment loss. Journal of Clinical Periodontology 18:117-125;1991.
Hamp SE, Nyman S, Lindhe J. Periodontal treatment of multi-rooted teeth: results after 5 years. Journal of Clinical Periodontology 2:126;1975.
Heasman PA, Benn DK, Kelly PJ, Seymour RA. The use of topical flurbiprofen, as an adjunct to non-surgical management of periodontal disease. Journal of Clinical Periodontology 20:457-464;1993a.
Heasman PA, Offenbacher S, Collins JG, Edwards G, Seymour RA. Flurbiprofen, in the prevention and treatment of experimental gingivitis. Journal of Clinical Periodontology 20:732-738;1993b.
Heasman PA, Seymour RA. The effect of a systemically administered non-steroidal anti-inflammatory drug (flurbiprofen) on experimental gingivitis in humans. Journal of Clinical Periodontology 16:551-556;1989.
Heasman PA, Seymour RA. An association between long term non-steroidal anti-inflammatory drug therapy and the severity of periodontal disease. Journal of Clinical Periodontology 17:654-658;1990.
Heasman PA, Seymour RA, Boston PF. The effect of a topical non-steroidal anti-inflammatory drug on the development of experimental gingivitis in man. Journal of Clinical Periodontology 16:353-358;1989.
Heasman PA, Seymour RA, Kelly PJ. The effect of systemically-administered flurbiprofen as an adjunct to tooth brushing on the resolution of experimental gingivitis. Journal of Clinical Periodontology 21:166-170;1994.
Heasman PA, Ward A, Barrett AW, Seymour RA, Edwards G. Flurbiprofen in human crevicular fluid analyzed by high-performance liquid chromatography. Journal of Periodontal Research 25:88-92;1990.
Hicks N, Newcomb GM. A sociological analysis of the differential use of dental services. Community Health Studies V:117-125;1981.
150

Holland IS, Seymour RA, Ward-Booth RP, Ord RA, Lim KLM. An evaluation of different doses of soluble aspirin and aspirin tablets in postoperative dental pain. British Journal of Clinical Pharmacology. 26:463-468;1988.
Holm-Pedersen P, Agerbaek P, Theilade E. Experimental gingivitis in relation to susceptibility to periodontal disease. Journal of Clinical Periodontology 2:14-24;1975.
Horning GM, Hatch CL, Lutskus J. The prevalence of periodontitis in a military treatment population. Journal of the American Dental Association 121:616-622;1990.
Howell TH. Blocking periodontal disease with anti-inflammatory agents. Journal of Periodontology 64:828-833;1993.
Howell TH, Williams RC. Nonsteroidal anti-inflammatory drugs as inhibitors of periodontal disease progression. Critical Review in Oral Biology and Medicine. 4:177-196;1993.
Hunt RJ. Percent agreement, Pearson's correlation, and kappa as measures of inter-examiner reliability. Journal of Dental Research 65:128-130;1986.
Ismail AI, Morrison EC, Burt BA, Caffesse RG, Kavanagh MT. Natural history of periodontal disease in adults: findings from the Tecumseh Periodontal Disease Study, 1959-87. Journal of Dental Research 69:430-435;1990.
Ismail M, Burt BA, Eklund SA. Epidemiologic patterns of smoking and periodontal disease in the United States. Journal of the American Dental Association 106:617-621;1983.
Jeffcoat MK. Osteoporosis: A possible modifying factor in oral bone loss. Annals of Periodontology 3:321-321;1998.
Jeffcoat MK, Page R, Reddy M, Wannawissute A, Waite P, Palcanis K, Cogen R, Williams RC, Basch C. Use of digital radiography to demonstrate the potential of Naproxen as an adjunct in the treatment of rapidly progressive periodontitis. Journal of Periodontal Research 26:415-421;1991.
Jeffcoat MK, Reddy MS, Haigh S, Buchanan W, Doyle MJ, Meredith MP, Nelson SL, Goodale MB, Wehmeyer KR. A comparison of topical ketorolac, systemic flurbiprofen, and placebo for the inhibition of bone loss in adult periodontitis. Journal of Periodontology 66:329-338;1995.
Johnson G. Clinical effect of tobacco use: Biologic impact on periodontium, risk factor, effect on disease management. American Academy of Periodontology. Audio tape;1998.
151

Johnson RH, Armitage GC, Francisco C, Page RC. Assessment of the efficacy of a nonsteroidal antiinflammatory drug Naprosyn the treatment of gingivitis. Journal of Periodontal Research 25:230-235;1990.
Kaldahl WB, Johnson GK, Patil KD, Kalkwarf KL. Levels of cigarette consumption and response to periodontal therapy. Journal of Periodontology 67:675-681;1996.
Kasser UR, Gleissener C, Dehne F, Mitchell A, Willerhausen-Zönchen B, Bolten WW. Risk for periodontal disease in patients with longstanding rheumatoid arthritis. Arthritis and Rheumatology 40:2248-2251;1997.
Kay AB. Studies on eosinophil leucocyte migration. I. Eosinophil and neutrophil accumulation following antigen-antibody reactions in guinea pig skin. Clinical and Experimental Immunology 6:75-86;1970.
Klebanoff S. Phagocytic cells: Products of oxygen metabolism. In: Gallin J, Goldstein I, Snyderman R, ed. Inflammation: Basic Principles and Clinical Correlates. New York: Raven Press, 541-588. 1992.
Lakhani SR, Dilly SA, Finlayson CJ. Basic Pathology an introduction to the mechanisms of disease. London: E. Arnold 1-58. 1993.
Lang N, Joss A, Orsanic T, Gusberti F, Siegrist B. Bleeding on probing. A predictor for the progression of periodontal disease. Journal of Clinical Periodontology 13:590-596;1986.
Levy BD, Romano M, Chapman HA, Reilly JJ, Drazen J, Serhan CN. Human alveolar macrophages have 15-lipoxygenase and generate 15(S)- hydroxy-5,8,11-cis-13-trans-eicosatetraenoic acid and lipoxins. Journal of Clinical Investigation 92:1572-9;1993.
Lewis RA, Austen KF, Soberman RJ. Leukotrienes and other products of the 5-lipoxygenase pathway. Biochemistry and relation to pathobiology in human diseases. New England Journal of Medicine. 323:645-655;1990.
Lindhe J, Socransky S, Wennström J. Design of clinical trials of traditional therapies of periodontitis. Journal of Clinical Periodontology 13:488-497;1986.
Listgarten MA. Why do epidemiologic data have no diagnostic value? In: Guggenheim B, ed. Periodontology today. Basel: Karger, 1988: 59-67.
Lloyd J, Bochner F. Aspirin: how low is low-dose? Department of Clinical and Experimental Pharmacology. Adelaide: University of Adelaide, 79-81, 1996.
Locker D, Leake JL. Periodontal attachment loss in independently living older adults in Ontario, Canada. Journal of Public Health and Dentistry 53:6-11;1993a.
Locker D, Leake JL. Risk indicators and risk markers for periodontal older adults living independently in Ontario, Canada. Journal of Dental Research 72:9-17;1993b.
152

Locker D, Slade G, Murray H. Epidemiology of periodontal disease among older adults: a review. Periodontology 2000 16:16-32;1998.
MacGregor IDM. Smoking and periodontal diseases. In: Seymour RA, Heasman PA, MacGregor IDM, ed. Drugs, diseases, and the periodontium. Oxford: Oxford University Press, 117-134. 1992.
Mathur S, Michalowicz BS. Cell-mediated immune system regulation in periodontal diseases. Critical Reviews in Oral Biology and Medicine 8:76-89;1997.
McGuire JR, McQuade MJ, Rossman JA, Garnik JJ, Sutherland DE, Scheidt MJ, Van Dyke TE. Cotinine in saliva and gingival crevicular fluid of smokers with periodontal disease. Journal of Periodontology 60:176-181;1989.
McLennan W. How Australia takes a Census. Australian Bureau of Statistics Census Australian Government Publishing. 1-60 1996
McMillan RM, Walker ER. Designing therapeutically effective 5-lipoxygenase inhibitors. Trends in Pharmacological Science 13:323-330;1992.
Meade EA, Smith WL, DeWitt DL. Differential inhibition of prostaglandin endoperoxide synthetase (cyclooxygenase) isoenzymes by aspirin and other non-steroidal anti-inflammatory drugs. Journal of Biological Chemistry. 268:6610-6614;1993.
Miller PD. Root coverage with the free gingival graft. Factors associated with incomplete coverage. Journal of Periodontology 58:674-681;1987.
Miller SC. Textbook of periodontia. (3rd ed.) Philadelphia: The Blakeston Co. 1950.
Miyasaki KT. The neutrophil: mechanisms of controlling periodontal bacteria. Journal of Periodontology. 62:761-774;1991.
Miyasaki KT. Altered leukocyte function and periodontal disease. In: Carranza FA, Newman MG, ed. Clinical periodontology. 8th ed. Philadelphia: W.B. Saunders, 132-150. 1996.
Miyasaki KT, Bodeau AL, Shafer WM, Pohl J, Murthy RK, Lehrer RI. New ideas about neutrophil antimicrobial mechanisms: antibiotic peptides, postphagocytic protein processing, cytosolic defence factors. In: Genco R, Shigeyuki H, Lehner T, McGhee J, Mergenhagen S, ed. Molecular Pathogenesis of Periodontal Disease. Washington: ASM Press, 321-336. 1994.
Mombelli A, van Oosten MAC, Schürch E, Lang NP. The microbioata associated with successful or failing implants. Oral Microbiology and Immunology 2:145-151;1987.
Mullally BH, Linden GJ. The periodontal condition of regular dental attenders in Northern Ireland. Journal of Clinical Periodontology 19:174-181;1992.
153

Mullally BH, Linden GJ. The periodontal status of irregular dental attenders. Journal of Clinical Periodontology 21:544-548;1994.
Müller TH. Pharmacological reasons to combine low-dose acetylsalicylic acid with dipyridamole. International Journal of Clinical Practice 52:7-11;1998.
Nassar GM, Morrow JD, Roberts LJd, Lakkis FG, Badr KF. Induction of 15-lipoxygenase by interleukin-13 in human blood monocytes. Journal of Biological Chemistry 269:27631-4;1994.
Newbrun E. Indices to measure gingival bleeding. Journal of Periodontology 67:555-561;1996.
Newman HN. Attrition, eruption and the periodontium. Journal of Dental Research 78:730-734;1999.
Newman MG, Sockransky SS, Savitt ED, Propas DA, Crawford A. Studies of the microbiology of periodontosis. Journal of Periodontology 47:373-379;1976.
Ng VW, Bissada NF. Clinical evaluation of systemic doxycycline and ibuprofen administration as an adjunctive treatment for adult periodontitis. Journal of Periodontology 69:772-776;1998.
NHMRC. Oral health care for adults. Adelaide: NHMRC . 1-51, 1993.
Nisengard RC, Newman MG. Host response: Basic concepts. In: Carranza FA, Newman MG, ed. Clinical periodontology. Philadelphia: W.B. Saunders Company, 111-120. 1996.
Offenbacher S. Periodontal diseases: pathogenesis. Annals of Periodontology 1:821-878;1996.
Offenbacher S, Braswell LD, Loos AS, Johnson HG, Hall CM, McClure H, Orkin JL, Strobert EA, Green MD, Odle BM. Effects of flurbiprofen on the progression of periodontitis in Macaca mulatta. Journal of Periodontal Research 22:473-481;1987.
Offenbacher S, Collins JG, Arnold RR. New clinical diagnostic strategies based on pathogenesis of disease. Journal of Periodontol Research 28:523-535;1993a.
Offenbacher S, Heasman PA, Collins JG. Modulation of host PGE2 secretion as a determinant of periodontal disease expression. Journal of Periodontology 64:432-444;1993b.
Offenbacher S, Katz V, Fertik G, Collins J, Boyd D, Maynor G, McKaig R, Beck J. Periodontal infection as possible risk factor for preterm low birth weight. Journal of Periodontology. 67:1103-1113;1996.
154

Offenbacher S, Olde BM, Braswell LD, Johnson HG, Hall CM, McClure H, Orkin JL, Strobert EA, Green MD. Changes in cyclooxygenase metabolites in experimental periodontitis in Macaca mulatta. Journal of Periodontal Research 24:63-74;1989.
Offenbacher S, Olde BM, Green MD, Mayambala CS, Smith MA, Fritz ME, Van Dyke TE, Yeh KC, Sen FJ. Inhibition of human periodontal Prostaglandin E2 synthesis with selected agents. Agent Actions 29:232-238;1990.
Offenbacher S, Williams RC, Jeffcoat MK, Olde BM, Smith MA, Johnson HG, Goldhaber P. Effects of NSAIDs on beagle crevicular cyclooxygenase metabolites and periodontal bone loss. Journal of Periodontal Research 27:207-213;1992.
Okada H. Cytokine expression in periodontal health and disease. Acta Odontologica Scandinavica 9:248-261;1998.
Oringer RJ, Fiorellini JP, Reasner DS, THH. The effect of different diagnostic thresholds on incidence of disease progression. Journal of Periodontology 69:872-878;1998.
Österberg T, Mellström D. Tobacco smoking: a major risk factor for loss of teeth in three 70-year-old cohorts. Community Dentistry and Oral Epidemiology 14:367-70;1986.
Page R. The pathology of periodontal diseases may affect systemic diseases: Inversion of a paradigm. Annals of Periodontology 3:108-120;1998.
Page RC. The role of inflammatory mediators in the pathogenesis of periodontal disease. Journal of Periodontal Research 26:230-242;1991.
Page RC, Offenbacher S, Schroeder HE, Seymour GJ, Kornman KS. Advances in the pathogenesis of periodontitis: summary of developments, clinical implications and future directions. Periodontology 2000 14:216-248;1997.
Page RC, Schroeder HE. Pathogenesis of chronic inflammatory periodontal disease. A summary of current work. Laboratory Investigations 33:235-249;1976.
Papapanou PN. Periodontal diseases: Epidemiology. Annals of Periodontology 1:1-36;1996.
Papapanou PN, Wennström JL, Grondahl KA. A 10 year retrospective study of periodontal disease progression. Journal of Clinical Periodontology 16:403-411;1989.
Papapanou PN, Wennström JL, Johnsson T. Extent and severity of periodontal destruction based on partial clinical assessments. Community Dentistry and Oral Epidemiology 21:181-184;1993.
Payne JB, Zachs NR, Reinhardt RA, Patil K. The association between estrogen status and alveolar bone density changes in post menopausal women with a history of periodontitis. Journal of Periodontology 68:24-31;1997.
155

Preber H, Bergström J. Occurrence of gingival bleeding in smokers and non-smokers. Acta Odontologica Scandinavia 43:315-320;1986.
Preber H, Kant T. Effect of tobacco smoking on periodontal tissue of 15 year old school children. Journal of Periodontal Research 8:278-283;1973.
Preber H, Linder L, Bergström J. Periodontal healing and periopathogenic microflora in smokers and non-smokers. Journal of Clinical Periodontology 22:946-952;1995.
Ramfjord S. Indices for prevalence and incidence of periodontal disease. Journal of Periodontology. 30:51-59;1959.
Rainsford KD. Aspirin and the salicylates. London.: Butterworth 1984.
Rainsford KD. Non-steroidal anti-inflammatory drugs. In: Textbook of immunopharmacology, 3rd edition. Oxford: Blackwell Scientific Publications Dale MM, Foreman J, Fan TPD, ed..1994.
Rang HP, Dale MM, Ritter JM. Pharmacology. Third edition. New York: Churchill Livingstone 1996.
Ramfjord S. Indices for prevalence and incidence of periodontal disease. Journal of Periodontology. 30:51-59;1959.
Reddy MS, Palcanis KG, Haigh ML, Charles S, Jeffcoat MK. Efficacy of
meclofenamate sodium (Meclomen®) in the treatment of rapidly progressive periodontitis. Journal of Clinical Periodontology 20:635-640;1993.
Reynolds JJ, Meikle MC. Mechanisms of connective tissue matrix destruction in periodontitis. Periodontology 2000 14:144-157;1997.
Richards ND, Barmes DE. Social factors in dental epidemiology. In: In Richards NDC, L.K., ed. Social sciences and dentistry. A critical bibliography. The Hague: Fed. Dent. Int., 308-325. 1971.
Romano M, Chen XS, Takahashi Y, Yamamoto S, Funk CD, Serhan CN. Lipoxin synthase activity of human platelet 12-lipoxygenase. Biochemical Journal 296:127-33;1993.
Romano M, Serhan CN. Lipoxin generation by permeabilized human platelets. Biochemistry 31:8269-77;1992.
Ryder MI, R. F, Johnson G, Hyun W. Alterations of neutrophil oxidative burst by in vitro smoke exposure: Implications for oral and systemic diseases. Annals of Periodontology 3:76-87;1998.
Salmon JA, Higgs G, A. Prostaglandins and leukotrienes as inflammatory mediators. British Medical Journal 43:285-296;1987.
156

Salvi GE, Lawrence HP, Offenbacher S, Beck JD. Influence of risk factors on the pathogenesis of periodontitis. Periodontology 2000 14:173-201;1997.
Samuelsson B. Leukotrienes: mediators of immediate hypersensitivity reactions and inflammation. Science 220:568-575;1983.
Schenkein H. The pathogenesis of periodontal diseases. Journal of Periodontology 70:457-470;1999.
Schenkein HA, Gunsolley JC, Koertge TE, Schenkein JG, Tew JG. Smoking and its effects on early-onset periodontitis. Journal of the American Dental Association 126:1107-1113;1995.
Schwartz Z, Goultschin J, Dean DD, Boyan BD. Mechanisms of alveolar bone destruction in periodontitis. Periodontology 2000 14:159-169;1997.
Seibert K, Masferrer JL. Role of inducible cyclooxygenase (COX-2) in inflammation. Receptor 4:17-23;1994.
Seibert K, Zhang Y, Leahy KM, Hauser S, Masferrer J, Perkins W, Lee L, Isakson P. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proceedings of the National Academy of Science USA 91:12013-12017;1994.
Serhan CN. Lipoxins and novel aspirin-triggered 15-epi-lipoxins (ATL): a jungle of cell-cell interactions or a therapeutic opportunity? Prostaglandins 53:107-37;1997.
Serhan CN, Hamberg M, Samuelsson B. Lipoxins: novel series of biologically active compounds formed from arachidonic acid in human leukocytes. Proceedings of the National Academy of Sciences of the United States of America 81:5335-9;1984.
Seymour GJ. Importance of the host response in the periodontium. Journal of Clinical Periodontology 18:421-426;1991.
Seymour GJ, Gemmell E, Reinhardt RA, Eastcott J, Taubman MA. Immunopathogenesis of chronic inflammatory periodontal disease: cellular and molecular mechanisms. Journal of Periodontal Research 28:478-486;1993.
Seymour RA, Williams FM, Luyk N, Boyle MA, Whitfield PM, Nicholson E, Ward-Booth P, Rawlins MD. Comparative efficacy of soluble aspirin and aspirin tablets in postoperative dental pain. European Journal of Clinical Pharmacology 30:495-498;1986.
Sharma S, Sharma SC. An update on eicosanoids and inhibitors of cyclooxygenase enzyme systems. Indian Journal of Experimental Biology 35:1025-1031;1997.
Ship JA, Beck JD. Ten-year longitudinal study of periodontal attachment loss in healthy adults. Oral Surgery Oral Medicine Oral Pathology Oral Radiology and Endodontics 81:281-290;1996.
157

Silness J, Löe H. Periodontal disease in pregnancy II. Correlation between oral hygiene and periodontal condition. Acta Odontologica Scandinavia 24:747-759;1964.
Slade GD, Spencer AJ. Periodontal attachment loss among adults aged 60 + in South Australia. Community Dentistry and Oral Epidemiology 23:237-242;1995.
Smith WL. Prostanoid biosynthesis and mechanism of action. American Journal of Physiology 268:F181-F191;1992.
Snyder DW, Fleisch JH. Leukotriene receptor antagonists as potential therapeutic agents. Annual Review of Pharmacological Toxicology 29:123-43;1989.
Soskolne WA. Subgingival delivery of therapeutic agents in the treatment of periodontal disease. Critical Reviews in Oral Biology and Medicine 8:164-174;1997.
Spencer AJ. South Australian dental longitudinal study: Five year follow-up questionnaire. In: Adelaide: University of Adelaide, 1997.
Srikandi W. Epidemiology of periodontal disease, industrial population, Adelaide, South Australia. University of Adelaide, 1982.
Taiyeb ATB, Waite IM. The effect of systemic ibuprofen on gingival inflammation in humans. Journal of Clinical Periodontology 20:723-728;1993.
Takano T, Fiore S, Maddox JF, Brady HR, Petasis NA, Serhan CN. Aspirin-triggered 15-epi-lipoxin A4 (LXA4) and LXA4 stable analogues are potent inhibitors of acute inflammation: evidence for anti- inflammatory receptors. Journal of Experimental Medicine 185:1693-704;1997.
Tatakis DN. Interleukin -1 and bone metabolism: A review. Journal of Periodontology 64:416-431;1993.
Tipton DA, Dabbous MK. Effects of nicotine on proliferation and extracellular matrix production of human gingival fibroblasts in vitro. Journal of Periodontology 66:1056-1064;1995.
Tolo K, Jorkend L. Serum antibodies and loss of periodontal bone in patients with rheumatoid arthritis. Journal of Clinical Periodontology 17:288-291;1990.
Tonetti MS. Cigarette smoking and periodontal diseases: etiology and management of disease. Annals of Periodontology 3:88-101;1998.
Toppal H, Zwadlo G, Lange D, Sorg G. Phenotypic dynamics of macrophage subpopulations during human experimental gingivitis,. Journal of Periodontal Research 24:106-112;1989.
Ueda N, Koide M, Ohguchi M, Ishihara Y, Noguchi T, Nishihara T. Involvement of prostaglandin E2 and interleukin-1in the differentiation and survival of
osteoclasts induced by lipopolysaccharide from Actinobacillus actinomycetemcomitans Y4. Journal of Periodontal Research 33:509-516;1998.
158

Underwood MJ. The aspirin papers: aspirin benefits patients with vascular disease and those undergoing revascularisation. British Medical Journal 308:71-72;1994.
U.S. Department of Health and Human Services. The health benefits of smoking cessation: Cardiovascular disease. A report of the surgeon general. Rockville: MD Center for chronic disease prevention and health promotion, 1990.
van der Velden U, Abbas F, Hart AAM. Experimental gingivitis in relation to susceptibility to periodontal disease. Journal of Clinical Periodontology 12:61-68;1985.
van der Weijden GA, Timmerman MF, Nijboer A, Rejerse E, van der Velden U. Comparison of different approaches to asses bleeding on probing as indicators of gingivitis. Journal of Clinical Periodontology 21:589-594;1994a.
van der Weijden GA, Timmerman MF, Saxton CA, Russell JL, Huntington E, van der Velden U. Intra/inter-examiner reproducibility study of gingival bleeding. Journal of Periodontal Research 29:236-241;1994b.
Van Dyke T, Hoop G. Neutrophil function and oral disease. Critical Reviews in Oral Biology and Medicine 1:117-133;1990.
Vane J. Towards a better aspirin. Nature 367:215-216;1994.
Vane J, O'Grady J. Therapeutic applications of prostaglandins. London: Edward Arnold 1993.
Vane JR. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nature New Biology 231:232-239;1971.
Vane JR, Botting RM. Aspirin and other salicylates. London: Chapman and Hall Medical . 1-165, 295-446, 1992.
Vogel RI, Copper SA, Schneider LG, Goteiner D. The effects of topical steroidal and systemic nonsteroidal anti-inflammatory drugs on experimental gingivitis in man. Journal of Periodontology 55:247-251;1983.
W.H.O. Oral Health Surveys Basic Methods. (4th ed.) Geneva: WHO 67, 1997.
Waite IM, Saxton CA, Young A, Wagg BJ, Corbet M. The periodontal status of subjects receiving non-steroidal anti-inflammatory drugs. Journal of Periodontal Research 16:100-108;1981.
Walter JB, Grudy MC. Walter, Hamilton and Israel's Principles of pathology for dental students. (5th ed.) Edinburgh: Churchill Livingstone Dental Series 51-65, 1993.
Walton JG, Thompson JW, Seymour RA. Textbook of Dental Pharmacology and Therapeutics (3rd ed.) Oxford: Oxford University Press, 66-71. 1994.
159

Wiebe S, Hafezi M, Sandhu HS, Sims SM, Dixon SJ. Osteoclast activation in inflammatory periodontal diseases. Oral Diseases. 2:167-180;1996.
Williams DM, Hughes FJ, Odell EW, Farthing PM. Pathology of periodontal disease. Oxford: Oxford University Press, 71-97. 1996.
Williams RC, Jeffcoat MK, Howell H, Rölla A, Stubbs D, Teoh KO, Reddy MS, Goldhaber P. Altering the progression of human alveolar bone loss with the non-steroidal anti-inflammatory drug flurbiprofen. Journal of Periodontology. 60:485-490;1989.
Williams RC, Offenbacher S, Jeffcoat MK, Howell TH, Hall CM, Wechter HG, Goldhaber P. Indomethacin, or Flurbiprofen treatment of periodontitis in Beagles: Effects on crevicular fluid arachidonic acid metabolites compared with effect on alveolar bone loss. Journal of Periodontal Research 23:134-138;1988.
Wilson TGJ, Kornman KS. Fundamentals of Periodontics. Chicago: Quintessence Publishing Co, Inc. . Quintessence books 159-167, 1996.
Winkel EG, Abbas F, Van derVelden U, Vroom TM, Scholte G, Hart AAM. Experimental gingivitis in relation to age in individuals not susceptible to periodontal destruction. Journal of Clinical Periodontology 14:499-507;1987.
Zafiriopoulos GGK, Flores-de-Jacob L, Todt G, Klob G, Havemann K, Taktakis DN. Gingival crevicular fluid elastase-inhibitor complex: correlation with clinical indices and subgingival flora. Journal of Periodontal Research 26:24-32;1991.
Zambon JJ, Grossi SG, Machtei EE, Ho AW, Dunford R, Genco RJ. Cigarette smoking increases the risk for subgingival infection with periodontal pathogens. Journal of Periodontology 67:1050-1054;1996.
Zappa U, Reinking-Zappa M, Graf H, Espland M. Cell population and episodic periodontal attachment loss in humans. Journal of Clinical Periodontology 18:508-515;1991.
160







![· Formula Tablets Arthritis Strength Bufferin Tablets Arthropan Liquid Arthrotec Ascodeen Ascriptin, All products Asperbuf Tablets Aspergum [chewing gum] Aspirin Asprimox Tablets](https://static.fdocuments.in/doc/165x107/5e76e2c5b6b4706c14128b70/formula-tablets-arthritis-strength-bufferin-tablets-arthropan-liquid-arthrotec-ascodeen.jpg)