Effect of environment on enzyme activity Substrate concentration pH Temperature.
-
Upload
loraine-phillips -
Category
Documents
-
view
215 -
download
1
Transcript of Effect of environment on enzyme activity Substrate concentration pH Temperature.

Effect of environment on enzyme activity
Substrate concentration
pH
Temperature

Substrate concentration
Enzyme activity increases with increasing substrate concentrations
At a certain concentration the enzyme will be saturated and operate at maximum velocity = Vmax

Substrate concentration
Plot of velocity (activity) vs. substrate concentration results in a hyperbola

Michaelis constant (Km)
Used to measure the affinity of an enzyme for its substrate
Km = substrate concentration required to achieve half maximal velocity

Effect of pH and temperature on enzyme activity
Enzymes are most active at optimum pH and temperatures
Deviations from the optima can slow activity and damage the enzyme

Effect of pH and temperature on enzyme activity
Loss of activity due to extreme pH, temperature or other factors is known to as denaturation
Temperature and pH optima of microorganism’s enzymes usually reflect the microorganism’s environment

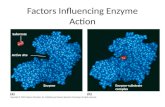
Enzyme inhibition
Many poisons and antimicrobial agents are enzyme inhibitors
Can be accomplished by competitive or noncompetitive inhibitors
Competitive inhibitors - compete with substrate for the active site
Noncompetitive inhibitors - bind at another location

Competitive inhibitors
Usually resemble the substrate but cannot be converted to products

Noncompetitive inhibitors
Bind to the enzyme at some location other than the active site
Do not compete with substrate for the active site
Binding alters enzyme shape and slows or inactivates the enzyme
Heavy metals often act as noncompetitive inhibitors (e.g. Mercury)

Metabolic regulation
Important to conserve energy and resources
Cell must be able to respond to changes in the environment
Changes in available nutrients will result in changes in metabolic pathways

Metabolic regulation
Metabolic channeling
Stimulation or inhibition of enzyme activity
Transcriptional regulation of enzyme production

Allosteric enzymes
Activity of enzymes are altered by small molecules known as effectors or modulators
Effectors bind reversibly and noncovalently to the regulatory site
Binding alters the conformation of the enzyme

Allosteric enzymes
Positive effectors increase activity
Negative effectors decrease activity

ACTase regulation
Regulation of aspartate carbamyltransferase is a well studied example of allosteric regulation
CTP inhibits activity and ATP stimulates activity

ACTase regulation
Binding of effectors cause conformational changes that result in more or less active forms of the enzyme

ACTase regulation
Binding of substrate also increases enzyme activity (more than one active site)
Velocity vs. substrate curve is sigmoid

Covalent modification of enzymes
Attachment of group to enzyme can result in stimulation or inhibition of activity
Attachment is covalent and reversible

Covalent modification of enzymes
Attachment of phosphate groups often used to regulate enzyme activity
Other groups can also be used to regulate enzyme activity

Feedback inhibition
Metabolic pathways contain at least one pacemaker enzyme
Usually catalyzes the first reaction in the pathway
Activity of the enzyme determines the activity of the entire pathway

Feedback inhibition
Feedback inhibition occurs when the end product interacts with the pacemaker enzyme to inhibit its activity
Branching pathways regulate enzymes at branch points

Overview of metabolism
Metabolism = the total of all chemical reactions occurring within the cell
Catabolism = the breaking down of complex molecules into simple molecules with the release of energy
Anabolism = the synthesis of complex molecules from simple molecules with the use of energy

Sources of energy
Microorganisms use one of three sources of energy
Phototrophs - radiant energy of the sun
Chemoorganotrophs - oxidation of organic molecules
Chemolithotrophs - oxidation of inorganic molecules

Electron acceptors
Chemotrophs vary regarding their final electron acceptors
Fermentation - no exogenous electron acceptor is required
Aerobic respiration - oxygen is the final electron acceptor
Anaerobic respiration - another inorganic molecule is acceptor

Electron acceptors
Chemolithotrophs can use oxygen or another inorganic molecule as the final electron acceptor

The three stages of catabolism
Catabolism can be broken down into three stages
Stage 1
Larger molecules (proteins, polysaccharides, lipids) are broken down into their constituents
Little or no energy is generated

The three stages of catabolism
Stage 2
Amino acids, monosaccharides, fatty acids, glycerol and other products degraded to a few simpler products
Can operate aerobically or anaerobically
Generates some ATP and NADH or FADH

The three stages of catabolism
Stage 3
Nutrient carbon is fed into the tricarboxylic pathway and oxidized to CO2
ATP, NADH and FADH produced
ATP generated from oxidation of NADH and FADH in electron transport chain

Amphibolic pathways
Pathways that can function both catabolically and anabolically
Glycolysis and the tricarboxylic acid cycle are two of the most important amphibolic pathways
Most reactions are reversible

The glycolytic pathway/glycolysis
Also known as the Embden-Meyerhof pathway
Most common pathway of degradation of glucose to pyruvate
Found in all major groups of microorganisms
Can function aerobically or anaerobically

The glycolytic pathway/glycolysis
Occurs in 2 stages
The six-carbon stage
Glucose is phosphorylated 2x and converted to fructose-1,6-bisphosphate
Other sugars converted to glucose-6-phosphate or fructose-6-phosphate and fed into pathway

The glycolytic pathway/glycolysis
The six-carbon stage
Does not yield energy
Uses 2 ATPs
Serves to “prime the pump”

The glycolytic pathway/glycolysis
The three-carbon stage
Fructose-1,6-bisphosphate split in half by fructose-1,6-bisphosphate aldolase
Yields glyceraldehyde-3-phosphate and dihydroxyacetone phosphate

The glycolytic pathway/glycolysis
The three-carbon stage
Dihydroxyacetone phosphate readily converts to glyceraldhyde-3-phosphate
Fructose-1,6-bisphosphate
2 glyceraldehyde-3-phosphate

The glycolytic pathway/glycolysis
The three-carbon stage
Glyceraldehyde-3-phosphate converted into pyruvate in 5 steps
Oxidized by NAD+ and a phosphate is added 1,3-bisphosphoglycerate
Phosphate on carbon 1 donated to ADP to form ATP
Substrate-level phosphorylation

The glycolytic pathway/glycolysis
The three-carbon stage
3-phosphoglycerate shifted to carbon 2 2-phosphoglycerate
Dehydration results in high energy phosphate bond in phosphoenolpyruvate
Phosphate transferred to ADP to form ATP (substrate-level phosphorylation)

The glycolytic pathway/glycolysis
Glucose 2 pyruvates + ATP +NADH
2 ATP used in six-carbon stage
4 ATP + 2 NADH formed in three-carbon stage

The glycolytic pathway/glycolysis
Glucose + 2ADP + Pi + 2NAD+ 2 pyruvate + 2ATP + 2NADH

![Enzyme Concentration- [Lactase] and Lactose](https://static.fdocuments.in/doc/165x107/5515cbbc497959f81d8b4f2d/enzyme-concentration-lactase-and-lactose.jpg)