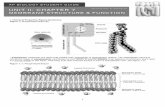
Diffusion Protein Mediated Diffusion integral membrane proteins transport proteins (transporters)...
-
Upload
rachel-atkins -
Category
Documents
-
view
214 -
download
1
Transcript of Diffusion Protein Mediated Diffusion integral membrane proteins transport proteins (transporters)...

Diffusion
Protein Mediated Diffusion
• integral membrane proteins
• transport proteins
(transporters)– diffusion through hydrophilic channels– facilitated diffusion

HydrophilicChannels
Channels have specificity; they are (more or less) specific for certain ions, e.g., Na+ channel, K+ channel, Ca++ channel, Cl- channel.
Leak channels: Some are always open.Gated channels: usually closed, opened by a specific stimulus
Fig. 3.8

Osmosis• Water transport
– diffusion across the phospholipid bilayer– diffusion through hydrophilic channels (aquaporins)
• Osmosis– diffusion of water across a semipermeable
membrane
Fig. 3.15
Water moves down its concentration gradient,from an area of higher water concentrationto an area of lower water concentration.
Therefore, the net movement of water is from an area of lower solute concentrationto an area of higher solute concentration.

Fig. 3.15
units of measurement for osmotic pressure: mm Hg or osmolality
osmolality = osmoles / (Kg solvent)
for a dilute solution: osmolality osmolarity = osmoles / (L solution)Osmolality is more accurate than osmolarity, because volume varies with temperature, but weight
does not. But, you are more familiar with calculating molarities than molalities. Therefore,
know how to calculate osmolarity.
Osmosis
No net movement becausehydrostatic pressure = osmotic pressure

• Osmolality/osmolarity is a measure of the number of particles in one liter of solution.[For the purposes of this course assume complete
dissociation of any solutes.]– e.g., 1 mole of glucose 1 osmole of glucose– e.g., 1 mole of NaCl 2 osmoles NaCl
(1 mole Na+ + 1 mole Cl-)
Osmolarity

• Osmolarity is a colligative property.
• Therefore, osmolarity can be measured by freezing point depression or boiling point elevation.
• freezing point of plasma = - 0.54 °Ctherefore, osmolality = 290
mosmolal
number to memorize: body osmolarity = 300 mosmolar

• What is the osmolarity of “normal” saline?
normal saline = 0.9% NaClmolecular weights: Na = 23, Cl = 35.5: NaCl = 58.5 g
mole NaCl
0.9% NaCl = 0.9g NaCl x 1000 mL x 1 mole NaCl x 2 osmoles100 mL sol’n 1 L sol’n 58.5g NaCl mole
NaCl
= 0.308 osmoles NaCl x 1000 mosmoles NaCl 1 L sol’n 1 osmole NaCl
= 308 mosmoles NaCl L sol’n
= 308 mosM

Behavior of Animal Cells in Solutions of Different Osmolarities
same osmolarity as plasmaosmolarity lower than plasma osmolarity higher than plasma
assumptions: Solute is impermeant and is not metabolized.
Fig. 3.16