Debate: Neoadjuvant Chemotherapy versus Primary ... · Carboplatin AUC2 & Paclitaxel 80 mg/m. 2....
Transcript of Debate: Neoadjuvant Chemotherapy versus Primary ... · Carboplatin AUC2 & Paclitaxel 80 mg/m. 2....

Debate:Neoadjuvant Chemotherapy
versus Primary Radiochemotherapyin FIGO IB2 Cervical Cancer
Neoadjuvant chemotherapy
Giovanni Aletti, MDEuropean Institute of Oncology
Milan, Italy

Stage IB2 Cervical CancerRound 1
Chemoradiation
NACT surgery

Locally Advanced Cervical Cancer
Different approaches:
- Chemo/radiation ± chemotherapy
- NACT ⇒ surgery ± chemo/radiation
- Surgery ± chemo/radiation
ALL TREATMENTS REQUIRE A MULTIMODALITY APPROACH

Locally Advanced Cervical CancerWORLDWIDE…
- CT/RT is considered standard of care
- 1999 NCI announcement:
“The results of 5 large studies have shown that women with invasive cervical cancer have better survival when they receive chemotherapy which includes the drug cisplatin along with radiation therapy.”

Chemoradiotherapy for Cervical Cancer Meta-Analysis Collaboration (CCCMAC). Cochrane Database Syst Rev. 2010;(1):CD008285.
Locally Advanced Cervical Cancer

• HR correlated withstage
• HR similar for CDDP and non CDDP-basedchemo-RT
• HR best for two trials with concurrent and adjuvant CT
Neoadjuvant Chemotherapy for Cervical Cancer Meta-Analysis Collaboration (NACCCMA) Collaboration. Cochrane Database Syst Rev. 2004;(2):CD001774.
Locally Advanced Cervical Cancer

Neoadjuvant Chemotherapy for Cervical Cancer Meta-Analysis Collaboration (NACCCMA) Collaboration. Cochrane Database Syst Rev. 2004;(2):CD001774.
Locally Advanced Cervical Cancer

Locally Advanced Cervical CancerNACT & S vs RT
Favors NACT-S Favors RT
Neoadjuvant Chemotherapy for Cervical Cancer Meta-Analysis Collaboration (NACCCMA) Collaboration. Cochrane Database Syst Rev. 2004;(2):CD001774.

• Stage Ib2-IIb• Stage IIIb
Randomized RT: 214 patients
NACT+S: 227 patients
Benedetti-Panici P, et al. J Clin Oncol. 2002;20(1):179-188.
• 210/227 (NACT) and 199/214 (RT) patients eligible• Stage IB2-IIA (174 pt); IIB (148 pt); III (87 pt) well balanced in the 2 ARMS• 152 (NACT) and 144 (RT) received treatment according to protocol
Neoadjuvant Chemotherapy and Radical Surgery Versus Exclusive Radiotherapy in Locally Advanced Squamous Cell Cervical Cancer:
Results From the Italian Multicenter Randomized Study

Neoadjuvant Chemotherapy and Radical Surgery Versus Exclusive Radiotherapy in Locally Advanced Squamous Cell Cervical Cancer:
Results From the Italian Multicenter Randomized Study
0
10
20
30
40
50
60
70
80
90
100
6 12 18 24 30 36 42 48 54 60
Prob
abili
ty
Months
NACT + RSRT
Pts Events210 84 199 100
P value: .001
100
60
10
20
30
40
50
60
70
80
90
0 6 12 18 24 30 36 42 48 54
NACT + RSRT
Pts Events210 84 199 100
P value: .01
Prob
abili
ty
Months
Benedetti-Panici P, et al. J Clin Oncol. 2002;20(1):179-188.

Criticisms to the Italian RCT
ARM 1: RADIOTHERAPY 27% of patients had external radiotherapy only
ERT+BRT total dose was less than 70 Gy
82% of patients received RT not on time (>55 days)
OTHER ISSUES
38/164 patients received RT after RS (3 modalities)
No benefit for stage III
Neoadjuvant Chemotherapy and Radical Surgery Versus Exclusive Radiotherapy in Locally Advanced Squamous Cell Cervical Cancer: Results From the Italian Multicenter Randomized Study

1. Chemoradiation is better than radiation alone in the treatment of LACC
2. NACT-radical surgery is better than radiation alone in the treatment of LACC
3. In stage III NACT-radical surgery is not helpful
SO…CHEMORADIATION OR NACT-RADICAL SURGERY?
What Have We Learned So Far?Take-Home Messages (Round 1)

IT LOOKS LIKE A TIED GAME…

EORTC - 55994RCT Comparing NACT+RS Versus CT/RT
RANDOMIZED
NACT + SURGERY
ExclusiveChemoradiation
Cervical CancerIB2, IIA>4 cm, IIB


While Awaiting the Results of EORTC 55994…
Use your weapons in different ways!!!

Stage IB2 Cervical CancerRound 2
Stage IB2 cervical cancer
NACT…how?

Locally Advanced Cervical Cancer
Neoadjuvant Chemotherapy for Cervical Cancer Meta-Analysis Collaboration (NACCCMA) Collaboration. Cochrane Database Syst Rev. 2004;(2):CD001774.

Locally Advanced Cervical CancerNACT & RT vs RT
Neoadjuvant Chemotherapy for Cervical Cancer Meta-Analysis Collaboration (NACCCMA) Collaboration. Cochrane Database Syst Rev. 2004;(2):CD001774.

Locally Advanced Cervical CancerNACT & RT vs RT
Neoadjuvant Chemotherapy for Cervical Cancer Meta-Analysis Collaboration (NACCCMA) Collaboration. Cochrane Database Syst Rev. 2004;(2):CD001774.

The Cochrane Review Indicated That:
Trials using cycle lengths shorter than 14 days or the cisplatin dose greater than 25mg/m2 per week exhibited an advantage of NACT on the survival of patients with LACC. It indicates that the timing and dose intensity may greatly impact the curative effect of NACT.

Round 2: NACT…how?When you do it…do it right!!!
Be fast…and intense!!! Use DOSE-DENSE

Stage IB2 Cervical CancerRound 3 – potential advantages
NACT
Stage IB2 cervical cancer

Rydzewska L, et al. Cochrane Database Syst Rev. 2010;(1):CD007406.
Locally Advanced Cervical Cancer

Locally Advanced Cervical Cancer• Better OS and PFS for
NACT/RS vs RS• Fewer local recurrences• Fewer distant
recurrences• More radical resections• Less N +• Less parametrial
involvement
Rydzewska L, et al. Cochrane Database Syst Rev. 2010;(1):CD007406.

POTENTIAL ADVANTAGES OF NACT 1. Reduction of tumor burden Biologic marker of
aggressiveness
2. Potential decrease of distant metastases
3. Potential eradication of micrometastases
4. Potential for better prognosis…
5. Identification of patients requiring multimodality approach
What Have We Learned So Far?Take Home Messages (Round 3)

Round 3: Potential advantages
Understand your rival first…Then we use the right strategy!!!

Stage IB2 Cervical CancerRound 4 – integration of strategies
Stage IB2 cervical cancer
NACT Chemoradiation

INduction ChemoThERapy in Locally Advanced CErvical Cancer
INTERLACE
Mary McCormack for the NCRI Gynaecological
Clinical Studies Group

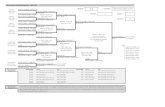
INTERLACERandomize (IB2-IVA)
Carboplatin AUC2 & Paclitaxel 80 mg/m2
Weeks 1-6
Weeks 7-13Standard CRT
Standard CRT
Follow-up3 monthly for 2 years; 6 monthly for 3 years
Standard CRT : 40 Gy - 50.4 Gy in 20-28 fractions plus Intracavitary brachytherapy to give total EQD2 dose of 78-86Gy to point A/volume.Weekly cisplatin 40mg/m2 x 5 weeks

Induction Chemotherapy
Week 1 2 3 4 5 6
CarboplatinAUC2 • • • • • •
Paclitaxel 80mg/m2 • • • • • •
Paclitaxel and carboplatin—weekly treatment for six weeks

Chemoradiation
Cisplatin weekly treatment for five weeks
Week 7 8 9 10 11 12
Days 1-5 8-12 15-19 22-26 29-33 36-40
Radiotherapy:40-50.4 Gy in20-28 fractions
• • • • • • • • • • • • • • • • • • • • • • • • • • • •
Cisplatin 40 mg/m2 • • • • •

Stratification
• FIGO stage• Node status—positive/negative• Tumor volume• Squamous vs nonsquamous• IMRT vs no IMRT• Age• Recruiting site

Outcome MeasuresPrimary endpoint: OS at 5 years
Secondary endpoints: PFS Toxicity QOL (UK & Eire only) Pattern of relapse (local and/or systemic)

Stage IB2: Role of NACT
Dose-dense chemo
No response
Chemo-RT
Response
Lymphadenectomy(staging or curative?)
Positive nodes
Negative nodes
Radical surgery

Round 4: NACT different strategies
Ok use your weapons right!We know, your rival is tired now!

• Is NACT a reasonable approach for patients with stage IB2 cervical cancer? – YES but…do it right (DOSE DENSE)
• Is NACTsurgery superior to definitive chemoradiation? – Waiting the EORTC results
• Is NACTCRT superior to definitive chemoradiation?– Waiting for the INTERLACE trial
• Can prognosis of patients with stage IB2 be improved?– Better selection strategies– Better biology/definition– Multimodality approach– Adjuvant chemotherapy– New modalities (targeted therapies, immunotherapy,...)– Extended field irradiation
Conclusions—Stage IB2

THANKS!