Cycles of Matter Chapter 3 The Biosphere Section 3-3; pages 74-80.
-
Upload
annabel-oneal -
Category
Documents
-
view
266 -
download
0
Transcript of Cycles of Matter Chapter 3 The Biosphere Section 3-3; pages 74-80.

Cycles of Matter
Chapter 3 The BiosphereSection 3-3; pages 74-80

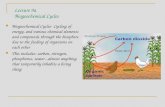
BIOGEOCHEMICAL CYCLES-connecting the biological, geological, and chemical aspects
of the biosphere
Matter within ecosystems is recycled within
and between ecosystems
Matter Statement:

BIOGEOCHEMICAL CYCLES
Cycle - water, carbon, oxygen, nitrogen & other elements cycle from the abiotic (“geo” nonliving environment) to biotic (“bio” living organisms) & then back to the environment.
Simply put, biogeochemical cycles pass the same molecules around again and again within the biosphere.
bioticabiotic

BIOGEOCHEMICAL CYCLES
Most element cycles have an atmospheric “bank” where the element is found in large amounts.
atmosphere
“bank”

BIOGEOCHEMICAL CYCLES
Decomposers (or combustion or erosion) break down organic matter.
What is the result of their actions?
RIP
atmosphere“bank”
Organisms release elements in daily activities or after death.
Elements move from the
“bank” into organisms.

BIOGEOCHEMICAL CYCLES
Three example cycles:
– Water
– Carbon (carbon-oxygen)
– Nitrogen
H2O C
N O

WATER CYCLE evaporation – heating of water from
liquid to atmospheric gas. condensation – cooling of water from
vapor to tiny droplets that form clouds. precipitation – droplets too large to be
contained in clouds and water returns to earth as rain, snow, or hail.
transpiration – evaporation of water from leaves of plants.
runoff – excess water from precipitation that comes from streams and rivers and carried to oceans and lakes.
accumulation – collection of water on earth.

http://ga.water.usgs.gov/edu/watercyclesummary.html
Accumulation
Refer to Figure 3-11, page 75

CARBON CYCLEThe exchange of gases during
photosynthesis and respiration is a major example of the living-nonliving cycle of carbon-oxygen.
respirationphotosynthesis
O2
CO2

CARBON CYCLE
How does carbon enter the living part of the cycle?
CO2 + H2O ----> C6H12O6 + O2
CO2CO2
CO2
CO2 CO2
CO2
CO2CO2

CARBON CYCLE
Carbon is returned to the atmosphere environment by:
– cellular respiration
– erosion
– combustion
– decomposition

CARBON CYCLE respiration – process where organisms
obtain energy from breakdown of glucose.
photosynthesis – plants use light energy (sun) to make glucose.
decomposition – breakdown of dead organic matter.
combustion – burning of fossil fuels (gas, petroleum, coal) which release CO2 into atmosphere.
erosion – runoff of soil into body of water

Carbon Cycle
Refer to Figure 3-13, page 77
http://www.windows.ucar.edu/earth/climate/images/carboncycle.jpg

NITROGEN CYCLE
79% of the atmosphere is nitrogen gas but it is in a form most living things cannot use.
N2free
nitrogen

NITROGEN CYCLE
If we can’t take in free nitrogen, how do we acquire it so we can use it in our bodies?
Why do we need nitrogen in our bodies?

NITROGEN CYCLEHow do we acquire usable
nitrogen?Nitrogen-fixing bacteria convert
nitrogen into nitrates.Plants absorb nitrates.Animals eat plants.
N2
in airnitrogen-fixing
bacteria
NITRATES

NITROGEN CYCLEHow does the nitrogen return to
the atmosphere?Denitrifying bacteria convert
the nitrates back into nitrogen.
N2
in airnitrogen-fixing
bacteria
NITRATES
denitrifyingbacteria

NITROGEN CYCLE
Why do we need nitrogen?
Nitrogen
protein
?

NITROGEN CYCLESimplified
Free N2 in Atmosphere
nitrogen-fixingbacteria
NITRATES
denitrifyingbacteria
RIP
Organicmaterial

NITROGEN CYCLESimplified
free N2 bank – atmosphere nitrogen fixation – conversion of atmospheric
nitrogen to ammonia nitrates – converted form of nitrogen from
atmosphere. organisms – living things organic material – components of living things
made of C, H, O, N denitrification – ammonia and nitrates converted
to nitrogen and released back into atmosphere

N2 in Atmosphere
NH3
NO3- & NO2-
Refer to Figure 3-14, page 78

All Cycles Are Related