COVID19 - njmgma.memberclicks.net - 1.4 presntation.pdf · •COVID19 is the disease associated...
Transcript of COVID19 - njmgma.memberclicks.net - 1.4 presntation.pdf · •COVID19 is the disease associated...


COVID19 Testing for SARS -CoV-2
& Evolving Guidelines

Speakers• Dan Fabius D.O. – Chief Medical Informatics Officer
• Dr. Fabius provides clinical oversight of population analysis and strategy; serves as a clinical liaison to the care coordination team; and acts as an advisor for clinical operation.
• In addition to his role at Continuum (Consensus), Dr. Fabius is currently a practicing physician at OptumCare NJ Heights Primary Care in Haddon Heights, NJ. Dr. Fabius is board certified in internal medicine and clinical informatics.
• Dr. Fabius was selected to SJ Biz’s “Who’s Who in Healthcare” for 2019.
• Nicholas Melchiorre IV- General Manager – Accurate Diagnostic Labs• Nick has greater than twenty years experience leading High Complex Commercial Laboratory
Operations in Clinical, Pathology, Virology, Molecular and Genetics Medicine.
• In addition, Nick is a private Corporate Consultant on Billing, Coding, and Diagnostics Reimbursement in Managed Care Coverage Policy, and Molecular Policy and Laboratory Developed Testing.
• Nick is member or former member of the National Business Group on Health, Disease Management Association of America, Founding Board member of the Long Island Healthcare Task Force, Founding member of the National Association of Quality Ancillary Service Providers.

Coronavirus
• Coronaviruses (CoV) are a large family of enveloped viruses with a single-stranded RNA.
• This family of virus cause illness ranging from the common cold to more severe diseases such as Middle East Respiratory Syndrome (MERS-CoV) and Severe Acute Respiratory Syndrome (SARS-CoV).
• Coronaviruses are zoonotic, meaning they are transmitted between animals and people.
• COVID19
• Common signs of infection include respiratory symptoms, fever, cough, shortness of breath and breathing difficulties.

Clinical operation overview
Screening
• Symptoms + risk factor
Telemedicine
• Place of service 02
• Modifier GT and 95
Infection Control / Disaster Recovery Plan
• Mitigation
Testing Criteria
• Call local Department of Health
• Determine if testing and/or self quarantine is best

FFCRAFAMILIES FIRST CORONAVIRUS RESPONSE ACT
•What is the Families First Coronavirus Response Act?
Notes:
FFCA was signed into law March 14th, 2020 “Making Testing Free” for everyone –
Nancy Pelosi” Three most important parts of this Bill – “TESTING, TESTING, TESTING”
A—SECOND CORONAVIRUS PREPAREDNESS AND RESPONSE SUPPLEMENTAL APPROPRIATIONS ACT, 202B—NUTRITION WAIVERS C—EMERGENCY FAMILY AND MEDICAL LEAVE EXPANSION ACTD—EMERGENCY UNEMPLOYMENT INSURANCE STABILIZATION AND ACCESS ACT OF 2020E—EMERGENCY PAID SICK LEAVE ACT F—HEALTH PROVISIONS DIVISION G—TAX CREDITS FOR PAID SICK AND PAID FAMILY AND MEDICAL LEAVEH—BUDGETARY EFFECT
“We cannot fight coronavirus effectively unless everyone in our country who needs to be tested knows they can get their test free of charge.“Nancy Pelosi

Coronavirus&COVID19
•The Coronavirus test IS NOT for SARS – CoV-2- (COVID19) –
• COVID19 is the disease associated with the Virus SARS – CoV -2.
• Three SARS CoV2 Options.
• (China CDC) • (US CDC)• Non CDC/Non China Protocol

Specimen Collection
•How do I collect a COVID19 Test?
• Preferred Specimen:• One (1) Nasopharyngeal and/or Oropharyngeal
swab.
• M4, VTM, UTM
• Refrigerated 72hrs – acceptable
• Preferred FROZEN –

CMS, CDC, Guidance
•What do I tell patients?
“If you are a close contact of someone with COVID-19 or you are a resident in a community where there is ongoing spread of COVID-19 and develop symptoms of COVID19, call your healthcare provider and tell them about your symptoms and your exposure. They will decide whether you need to be tested, but keep in mind that there is no treatment for COVID-19 and people who are mildly ill may be able to isolate and care for themselves at home.”
• > 65 years of age
• Immune Compromised - (e.g., diabetes, heart disease, receiving immunosuppressive medications, chronic lung disease, chronic kidney disease)
• Underlying Respiratory Conditions

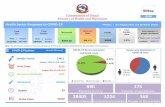
TestingAvailability
Is Testing Widely Available?
>50,000 TESTS PER DAY & INCREASINGManufacturersAWAITING EUA APPROVAL:
LUMINEX,GENMARK,BIOFIRE, HOLOGIC, DIASORIN,THERMO,CEPHIED,MANY MORE…
EUA LaboratoriesAdventHealthARUP LaboratoriesBaylor Scott and White Medical Center BioReference LaboratoriesThe Children's Hospital of PhiladelphiaHMH Hackensack University MedicalHospital of the University of PennsylvaniaJohns HopkinsLaboratory Corporation of AmericaMontefiore Medical CenterNew York Presbyterian HospitalNYU Langone Medical CenterQuest Diagnostics Infectious Disease, Inc.Stanford Health Care Clinical LaboratoryTexas Children's HospitalUCSF-HealthUniversity of WashingtonViracor Eurofins Clinical Diagnostics

March 13th, April 2020
COVID19Test Options
CDC1/5/2020
DOH2/17/2020
Labcorp3000 per day3/5/2020
IVD automation3/13/2020
Introduction of Automation

Performance
&
Characteristics
Is this a good Test?

Coding & Coverage Policy:
What ICD10 – Coding Should I use?
There is no specific code for COVID-19 in the U.S. at this time
U07.01 – DO NOT USE – Not Effective October 1ST, 2020.
B34.2 – DO NOT USE – The site would not be “unspecified”
USE THE CODES THAT ARE AVAILABLE
• Z03.818 - (Encounter for observation for suspected exposure to other biological agents ruled out). Used for cases where there is a concern about a possible exposure to COVID-19, but this is ruled out after evaluation.
• Z20.828 - (Contact with and (suspected) exposure to other viral communicable diseases). Used for cases where there is an actual exposure to someone who is confirmed to have COVID-19.

Coding for COVID19 Testing
What will the test cost?87635 – Commercial Insurance
U0001 – Medicare CDC Test
U0002 – Medicare LDT (Non-CDC Test)

FAQ
• Who should be Tested - Testing for Everyone? –• Follow CDC Guidelines, Contact local Department of Health
• Will PSC or Phlebotomist Collect COVID19 Test –• NO- Only healthcare professionals can collect.
• A Patient Called w/ Symptoms, Should I collect?• Continue to follow CDC Guidance and contact your local
health department.
• How do I Access Testing –• Contact your local Health Department, notify you have a
Person Under Investigation. They can best direct you.
• What is expected TAT –• 3-4 days TAT, we are still unsure.
• Will this be State Reported –• YES, all positives and tracking must be state reported.
• If Negative / Positive – Is this truly a Pos/Neg patient –• Not Necessarily.

Additional Resources
• World Health Organization
• Centers for Disease Control and Prevention (CDC)
• National Institutes of Health
• The NJ Department of Health or call the 24-hour public hotline:1-800-222-1222

Q&AThank you
Questions?
Feel Free to Email Me:
Text/Call:
609-513-4654

Q&A