Connection of Cerebellum
-
Upload
jegaphysio -
Category
Documents
-
view
217 -
download
0
Transcript of Connection of Cerebellum

8/8/2019 Connection of Cerebellum
http://slidepdf.com/reader/full/connection-of-cerebellum 1/6
Cerebellum: Connections and Functions
Mitchell Glickstein & Karl Doron
Published online: 11 November 2008# Springer Science + Business Media, LLC 2008
Abstract In addition to its role in motor control, reflex
adaptation, and motor learning, three sorts of evidence have been put forward to support the idea that the cerebellum
may also be involved in cognition. Patients with cerebellar
lesions are reported to have deficits in performing one or
another cognitive task. The cerebellum is often seen to be
activated when normal subjects perform such tasks. There
are connections to and from areas of the prefrontal cortex
that may be involved in cognition. In this paper, we review
the anatomical evidence to support the claim. We suggest
that there are only minor connections with cognitive areas
of the cerebral cortex and that some of the imaging
evidence may reflect the cerebellum’s role in the control
of eye movements rather than cognition.
Keywords Cerebellum . Eye movements . Pontine nuclei .
DTI . Cerebral peduncle
Introduction
From the earliest experimental studies in animals, it was
apparent that cerebellar lesions cause impairment in the
control of movement. Rolando [1] described the resultant
profound motor deficit that was caused by cerebellar
lesions. Fifteen years later, Flourens [2] argued that, after
such lesions animals can still move, but their coordinationis impaired. In the following 70 years, there were many
attempts to characterize more precisely the nature of the
deficit. In the most extensive of these studies, Luciani [3]
related the deficits to a fundamental impairment of muscle
control. According to Luciani’s careful observations and
conclusions, lesions of the cerebellum cause atonia or loss
of muscle tone, asthenia, or muscular weakness and astasia,
which, according to Luciani, manifests itself as tremor,
oscillation, and lack of coordination.
Although no one denied the obvious motor symptoms
that are caused by lesions of the cerebellum, there was an
occasional claim that they may also produce cognitive
deficits.
André-Thomas [4] dismissed such claims, basing his
conclusions on a systematic description of clinical symp-
toms and postmortem evidence. He wrote:
“While it is true that in a sufficiently large number of
observations the coincidence of intellectual disturbances
with cerebellar lesions has been noted, how many times
has a relation of cause and effect been established in a
rigorously scientific manner between the two? Those
who have thought that they have found this relation have
not taken into consideration the possibility of thecoexistence of cerebral lesions, or the insufficiency of
the examination of the cerebrum.”
For normal posture and movements, inputs from the
vestibular system as well as muscle, tendon, and joint
afferents are required. Sherrington [5] interpreted the
deficits that are caused by cerebellar lesions as being due
to loss of proprioceptive input to the motor system. The
cerebellum, he said, is the head ganglion of the proprio-
ceptive system.
Cerebellum (2008) 7:589 – 594
DOI 10.1007/s12311-008-0074-4
M. Glickstein (*)
Department of Cell and Developmental Biology,
University College London,
Gower Street,
London WC1E 6BT, UK
e-mail: [email protected]
K. Doron
Department of Psychology,
University of California at Santa Barbara,
Santa Barbara, CA 93106-9660, USA

8/8/2019 Connection of Cerebellum
http://slidepdf.com/reader/full/connection-of-cerebellum 2/6
At the time that these interpretations were put forward,
there were few anatomical techniques available for estab-
lishing fiber connections in the nervous system. Ramon y
Cajal [6] would sometimes attempt to follow the course and
termination of fiber tracts using the Golgi stain, but this
method is not really suitable for tracing long pathways.
Such connections were typically studied either by gross
dissection or by using the degeneration method of Marchi.But the Marchi technique stains degenerating myelin,
hence, it is strongly biased in favor of the largest diameter
axons.
Recent Suggestions of a Cognitive Function
for the Cerebellum
The idea that the cerebellum may be involved in cognition
has reappeared from time to time, and recently seems to
have become popular again [7]. Following the suggestions
in Leiner ’s paper, there are now hundreds of reports whichargue for a cognitive role for the cerebellum. The original
syllogism seems to be that the cerebellum, and particularly
the cerebellar hemispheres, is particularly large in humans,
monkeys, and apes; humans, apes, and monkeys are clever,
so the cerebellum is a likely brain structure for cleverness.
(An alternative syllogism might be that humans, apes, and
monkeys are most skillful in the use of their fingers…).
Three sorts of evidence have been put forward to support
the idea of a role for the cerebellum in cognitive functions;
neuropsychological deficits in patients with cerebellar
lesions, activation of the cerebellum in normal subjects as
they perform a cognitive task, and anatomical connections
showing links to and from the cerebellum of structures in
the cerebral cortex that are known or thought to be involved
in cognition. Some of this evidence is summarized in a
paper by Schmahmann [8].
In the past hundred years, there have been major advances
in the techniques available for studying connections within
the brain, and this evidence should help to clarify the
functions of the cerebellum. We can ask: what are the inputs
to the cerebellum and where does it project? In this paper, we
restrict ourselves largely to the anatomical evidence, al-
though we share with André-Thomas a skeptical view of the
alleged clinical evidence for a role for the cerebellum in
cognition and the associated claims based on imaging
techniques.
The cerebellum is concerned with the direct ongoing
regulation of movement, planning of movements, and motor
learning. Actual and planned movements may masquerade
as cognition. For example, much of what we do is preceded
by an eye movement [9], and the frontal cortex has several
regions that are involved in the control of eye movements
[10 – 12]. What we think we may do may be preceded by
activity in those same structures. Just as functional magnetic
resonance imaging (fMRI) activity may be associated with
planned as well as actual movement, so planned eye
movement may be associated with the activation of the
same brain structures that control eye movement. Some
of the inputs from the cerebral cortex to the cerebellum
that appear to be cognitive in function may be primarily
involved in eye movement control.
Gross Connections of the Cerebellum
The cerebellum is connected by three prominent paired
stalks, the cerebellar peduncles, which link it to the rest of
the brain. The inferior peduncle is largely afferent from
the inferior olivary nucleus as well as spinocerebellar and
vestibular systems. Few would argue that the inferior
peduncle is a link to cognitive structures. The alleged input
from cognitive areas of the cerebral cortex would reach the
cerebellum by way of a relay in the pontine nuclei. The axonsof pontine cells project to the cerebellar cortex by way of
the middle cerebellar peduncle, which is by far the largest
of the three peduncles in the human brain.
The output from the cerebellum is by way of the axons
of the cerebellar nuclei, most of which travel in the superior
cerebellar peduncle to the red nucleus and the thalamus.
The thalamus, in turn, relays that input to the cerebral
cortex. In this paper, we examine the nature of the input to
the pontine nuclei from the cerebral cortex and the output
targets of the cerebellar nuclei to the cerebral cortex.
Input to the Cerebellum
In humans and the higher primates, by far the largest source
of input to the pontine nuclei is from the cerebral cortex.
Two obvious questions can be asked about that input:
which cells in the cerebral cortex project to the pontine
nuclei and which areas of the cortex project there? The
second questions bears directly on the issue of a possible
role of the cerebellum in cognition. If an area of the
cerebral cortex that is known to function in cognitive tasks
projects to the pons or receives an input from the cerebellar
nuclei, that would be evidence for a role for the cerebellum
in such tasks. Both questions, which cells and how they are
distributed, can be addressed by filling the pontine nuclei
with a retrograde tracer and identifying the location and
distribution of retrogradely labeled cells in the cerebral
cortex. The answer to the first question is easy. All input
from the cerebral cortex to the pontine nuclei arises from
layer V pyramidal cells. Pontine-projecting cells often form
a continuous sublamina within layer V of the cerebral
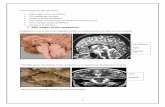
cortex [13, 16] (Fig. 1).
590 Cerebellum (2008) 7:589 – 594

8/8/2019 Connection of Cerebellum
http://slidepdf.com/reader/full/connection-of-cerebellum 3/6
Corticopontine fibers enter the internal capsule and
proceed ventrally to join the cerebral peduncle at the base of
the midbrain. In rats, the projection from the cerebral cortex
through the cerebral peduncle is spatially ordered [14]. Fibers
arising from cells in the temporal and occipital cortex travel
in the dorsolateral region of the cerebral peduncle. Fibers
originating from the frontal cortex travel in the ventromedial
region of the peduncle. Parietal lobe-originating fibers are
between these two. In the rat barrel field, two sublaminae of
layer V can be distinguished. Cells in the superficial
sublamina Va project to the basal ganglia. Cells in the deeper
layer Vb project to the pontine nuclei [13]. The cells in layer
Vb appear darker in cytochrome oxidase preparations, and
unlike Va, Vb receives a direct projection from the thalamus.
Fig. 1. Distribution of labeled
cells in the cerebral cortex of a
monkey after an injection of a
retrograde tracer (W.G.A.H.R.P.)
into the pontine nuclei. a Plane
of sections illustrated in b. b
Extent of primary injection site
in pontine nuclei. c Location of
retrogradely labeled cells in the
cerebral cortex; each dot repre-sents 25 cells. d Representative
cross sections of cortex to show
location of labeled cells (from
Glickstein et al. [16])
Cerebellum (2008) 7:589 – 594 591591

8/8/2019 Connection of Cerebellum
http://slidepdf.com/reader/full/connection-of-cerebellum 4/6
Which Cortical Areas Project to the Pons in Monkeys?
Layer V cells provides the input to the pontine nuclei in
all mammals that have been studied, but there are major
species differences among mammals in the number of
cortical areas that give rise to corticopontine fibers. In rats,
all of the cerebral cortex projects to the pontine nuclei [15];
in monkey, only about half of the cerebral cortex projects tothe pontine nuclei [16].
The differential projection from cortical visual areas
provides a clue to the function of the corticopontocerebellar
system. In monkeys, there is a dense projection to the pons
from the dorsal visual stream of extrastriate visual areas, a
region in which most of the neurons are motion-sensitive.
There are few or no inputs from areas in the ventral stream
of cortical visual areas whose cells are involved in higher
visual processes, such as face recognition and form
discrimination. Lesions of the dorsal stream of visual areas
impair skilled visually guided use of the hand and fingers.
Control lesions of the ventral stream visual areas do not [17].
The kind of visual information that is sent to the
cerebellum in cats is similar to that which is sent in monkeys.
Corticopontine [18] and pontine visual cells of cats [19] are
sensitive to the direction and velocity of moving targets; they
are relatively uninfluenced by the precise shape or orienta-
tion of those targets.
Disconnection of Sensory Areas of the Cerebral Cortex
from the Cerebellum Impairs the Sensory Guidance
of Movement
A patient in Professor Hans Joachim Freund’s group [20]
suffered a stroke in the caudal limb of the internal capsule
without damage either to the cerebral cortex or to cortico-
cortical fiber systems. The lesion led to a profound deficit
in visuomotor control, most marked when the patient
attempted to use the hand opposite the side of the stroke.
Recently, we used diffusion tensor imaging (DTI) in a set
of 20 normal brains to confirm the origin and course of
the fibers in this region of the internal capsule. We
replicated the lesion site from the Classen et al. case in
stereotaxic space and demonstrated a subcortical circuit
whose interruptions at the site of the lesion blocked dorsal
stream visual input to the pontine nuclei and cerebellum.
Fibers in this region of the internal capsule receive their
input from the dorsal stream of visual areas. In the case of
this DTI study, we found that these fibers arise from the
superior parietal lobule. The fibers from this area trace
ventrally and caudally to the lateral edge of the cerebral
peduncle, which is consistent with monkey and human
neuropathology evidence.
In rats, all of the cerebral cortex projects to the pons. The
projection is orderly. It is as if there is a miniature
representation of the cerebral cortex in the cerebral peduncle
[14]. Because of that arrangement, it is possible to selectively
cut the fibers from different cortical areas. Rats were trained
to jump across a 16 cm gap between two platforms; a
distance that they could just manage to reach with their
whiskers [21]. If the second platform was 1 cm beyond thereach of the whiskers, the rat would refuse to jump. We cut
the fibers within the cerebral peduncle on one side,
disconnecting the barrel field connection to the pontine
nuclei, thus leaving the rat with only one set of whiskers
connected to the pons. When the whiskers connected to the
pons were cut, the rat refused to jump. In contrast, cutting the
whiskers that were disconnected from the pons had no effect
on jumping. When the whiskers connected to the pons were
allowed to regrow, the rat would jump again. In all cases,
jumping in the light was unimpaired. The sensory informa-
tion reaching the cerebellum from the rat barrel cortex plays
an important role for judging distance for jumping. It seemslikely that other cortical projections play a similar role in
motor control.
In addition to direct sensory control of movement, the
anatomical study of the corticopontine system suggest
another important role for this system. Ramon y Cajal and
later Ugolini and Kuypers [22] showed that pyramidal tract
fibers, as they descend through the pons, give off collaterals
that connect to cells in the pontine nuclei. These collaterals
are an obvious candidate to serve as an efference copy of a
given movement.
The densest area of cells projecting to the pontine nuclei
from the cerebral cortex is from the primary motor and
premotor cortex. There is also a strong projection from area
8, the frontal eye field. There is a much weaker projection
from more rostral prefrontal cortical areas [16]. Some of
these prefrontal regions are cognitive in function, but some
may be more closely related to the control of eye move-
ments [12].
Afferent Connections, Thalamic targets of the Cerebellar
Nuclei, on the Thalamic Input to Frontal Eye Fields
All links from the cerebellum to the cerebral cortex
originate from the cerebellar nuclei and relay to the cerebral
cortex by way of the thalamus. Middleton and Strick [23]
injected the prefrontal cortex of monkeys, areas 46 and 9,
with a virus-based label and showed that there were
retrograde transneuronal labeled cells in the ventral dentate
nucleus. Their data are unequivocal in demonstrating that
there is a link between the cerebellar hemispheres, which
project to the dentate nucleus and prefrontal cortex. We
suggest that this connection may be part of an eye
592 Cerebellum (2008) 7:589 – 594

8/8/2019 Connection of Cerebellum
http://slidepdf.com/reader/full/connection-of-cerebellum 5/6
movements’ circuit. May et al. [24] injected a retrograde
tracer into the superior colliculus and showed that this area
receives a direct projection from the cerebellar dentate
nucleus. See, for example, their Fig. 1 sections K and L and
Fig. 3 sections H, I, and J.
An alternative way to study the connections would be to
fill all of the cerebellar deep nuclei with an orthograde
tracer and map the distribution of the orthogradely labeledfibers in the thalamus. This evidence could then be
compared with the pattern of retrograde label in the
thalamus nuclei after an injection is made in the frontal
cortex. Sakai et al. [25] injected all three cerebellar nuclei
and traced the fibers to their termination in the thalamus.
Her interest was in comparing the distribution of cerebellar
input to the thalamus with that from the basal ganglia, but
her data serve to outline the thalamic targets of the cerebellar
nuclei. The great majority of the terminations that she saw
were in the ventral thalamus. There was a small extension
into the nucleus medialis dorsalis which was restricted to its
far lateral region, adjacent to the external medullary lamina.The cortical target of this thalamic area is the frontal eye
fields. She did not show a projection to the more central
parvocellular region of medialis dorsalis.
Middleton and Strick injected area 46 in some cases and
area 9 in others. Lynch [12] showed that area 46 in the
monkey and a corresponding cortical area in the human
brain is part of an eye movement controlling area.
If prefrontal cortical areas known to be involved in
cognitive functioning project to the pontine nuclei, then the
case might be made that the cerebellum is involved in
cognition. Ramnani and his colleagues [26] used DTI to
study efferent pathways from the cortex. They found that
nearly half of the cerebral peduncle contains efferent fibers
from the human prefrontal areas. But the areas that they
studied are known to be involved in eye movement control
as well as cognition. Their study of the efferent pathway
would not have distinguished between the two functions. In
an ongoing study, we divided the prefrontal cortical areas in
stereotaxic space into six regions. We then used DTI
tractography to confirm that, in humans, the origin of the
prefrontal projections to pons arise primarily from the
premotor regions and dorsal precentral sulcus. Our findings
to date suggest that the prefrontal areas that project to the
pons are similar to those that are seen in fMRI studies of
eye movements [11].
Conclusion
The cerebellum is involved in the regulation of movement
of the limbs, the eyes, and the fingers [27]. Anatomical
evidence at best reveals only a weak connection between the
cerebellum and cerebral cortical areas involved in cognition.
Many of the papers demonstrating cognitive deficits follow-
ing cerebellar damage may be due to concomitant damage to
other brain structures. Neural activity in the cerebellum
during cognitive tasks may be associated with actual or
planned eye movements.
References
1. Rolando L (1968) Saggio sopra la vera struttura del cervello
dell’uomo e degl’animali e sopra le funzionei del sistema nervosa:
Sassari: 1809. English translation in Clarke E, O’Malley C. The
human brain and spinal cord. California University Press,
Berkeley and Los Angeles, pp 653 – 656
2. Flourens P (1968) Recherche experimentales sur les proprietes et
les fonctions du systeme nerveux dans les animaux vertebres.
Paris: Crevot 1824. English translation in Clark E, O’Malley C.
The human brain and spinal cord. California University Press,
Berkeley and Los Angeles
3. Luciani L (1891) Il cereveletto. Successori Le Monniere, Firenze
4. André-Thomas J (1912) Cerebellar functions. Journal of Nervous
and Mental Disease Publishing Company, New York (Nervous
and Mental Disease Monograph Series No. 12. Translated from
the French by W. Conyers Herring)
5. Sherrington C (1906) The integrative action of the nervous
system. Yale, New Haven
6. Ramon y Cajal S (1955) Histologie du Nysteme Nerveux. CSIC,
Madrid
7. Leiner H, Leiner A, Dow R (1989) Reappraising the cerebellum:
what does the hindbrain contribute to the forebrain? Behav
Neurosci 103:989 – 1008
8. Schmahmann J (1991) An emerging concept. The cerebellar
contribution to higher function. Arch Neurol 48:1178 – 1187
9. Land M (2006) Eye movements and the control of actions in
everyday life. Prog Retin Eye Res 25:296 – 324
10. Moschovakis A et al (2004) Oculomotor areas of the primatefrontal lobes: a transneuronal transfer of rabies virus and (14C)-2-
deoxyglucose functional imaging study. J Neurosci 24:5726 – 5740
11. Rosano C et al (2002) Pursuit and saccadic eye movement
subregions in human frontal eye field: a high-resolution fMRI
investigation. Cereb Cortex 12:107 – 115
12. Lynch JC, Tian JR (2005) Cortico-cortical networks and cortico-
subcortical loops for the higher control of eye movements. Prog
Brain Res 151:461 – 501
13. Mercier B, Legg C, Glickstein M (1990) Basal ganglia and
cerebellum receive different somatosensory information in rats.
Proc Natl Acad Sci U S A 87:4388 – 4392
14. Glickstein M, Kralj-Hans I, Legg C, Mercier B, Ramna-Rayan M,
Vaudano E (1992) The organisation of fibres within the rat basis
pedunculi. Neurosci Lett 135:75 – 79
15. Legg C, Mercier B, Glickstein M (1989) Corticopontine projec-tion in the rat: the distribution of labelled cortical cells after large
injections of horseradish peroxidase in the pontine nuclei. J Comp
Neurol 286:427 – 441
16. Glickstein M, May J, Mercier B (1985) Corticopontine projection
in the macaque: the distribution of labelled cortical cells after
large injections of horseradish peroxidase in the pontine nuclei.
J Comp Neurol 235:343 – 359
17. Glickstein M, May J, Buchbinder S (1997) Visual control of the
arm, the wrist, and the fingers; Pathways through the brain.
Neuropsychologia 36:981 – 1001
18. Gibson A, Baker J, Mower G, Glickstein M (1978) Corticopontine
cells in area 18 of the cat. J Neurophysiol 41:484 – 495
Cerebellum (2008) 7:589 – 594 593593

8/8/2019 Connection of Cerebellum
http://slidepdf.com/reader/full/connection-of-cerebellum 6/6
19. Baker J, Gibson A, Glickstein M, Stein J (1976) Visual cells in the
pontine nuclei of the cat. J Physiol 255:415 – 433
20. Classen J et al (1995) Subcortical origin of visuo-motor apraxia.
Brain 118:1365 – 1374
21. Jenkinson E, Glickstein M (2000) Whiskers, barrels, and cortical
efferent pathways in gap-crossing by rats. J. Neurophysiol
84:1781 – 1789
22. Ugolini G, Kuypers HG (1986) Collaterals of corticospinal and
pyramidal fibres to the pontine grey demonstrated by a new
application of the fluorescent fibre labelling technique. Brain Res365:211 – 227
23. Middleton F, Strick P (2001) Cerebellar projections to prefrontal
cortex of the primate. J Neurosci 21:700 – 712
24. May P et al (1990) Cerebellotectal pathways in the macaque:
implications for collicular generation of saccades. Neurosci
36:305 – 324
25. Sakai S, Inase M, Tanji J (1996) Comparison of cerebellothalamic
and pallidothalamic projections in the monkey ( Macaca fuscata):
a double anterograde labelling study. J Comp Neurol 368:215 –
228
26. Ramnani N et al (2006) The evolution of prefrontal inputs to the
cortico-pontine system: diffusion imaging evidence from macaque
monkeys and human. Cereb Cortex 16:811 – 81827. Glickstein M, Waller J, Baizer J, Brown B, Timmann D
(2005) Cerebellum lesions and finger use. Cerebellum 4:189 –
197
594 Cerebellum (2008) 7:589 – 594